Advanced Amperometric Biosensors for Real-Time Glucose Monitoring in Yeast Fermentation: From Fundamentals to Bioprocess Applications
Real-time, online monitoring of glucose is critical for optimizing yeast fermentation processes in biomedical and pharmaceutical production, yet it remains a significant challenge outside controlled laboratory settings.
Advanced Amperometric Biosensors for Real-Time Glucose Monitoring in Yeast Fermentation: From Fundamentals to Bioprocess Applications
Abstract
Real-time, online monitoring of glucose is critical for optimizing yeast fermentation processes in biomedical and pharmaceutical production, yet it remains a significant challenge outside controlled laboratory settings. This article comprehensively explores the development and application of amperometric biosensors for continuous glucose tracking. It covers the foundational principles of electrochemical glucose sensing, detailing recent methodological breakthroughs in sensor design, including nanostructured electrodes and integrated multi-sensor arrays. The scope extends to practical strategies for overcoming common operational hurdles such as oxygen limitation, interference, and fouling in complex fermentation broths. Finally, the article provides a critical evaluation of sensor performance through validation against standard analytical methods and a comparative analysis of commercial systems, offering researchers a clear roadmap for implementing robust online monitoring solutions to enhance bioprocess control and productivity.
The Science of Amperometric Glucose Biosensors: Principles and Bioprocess Relevance
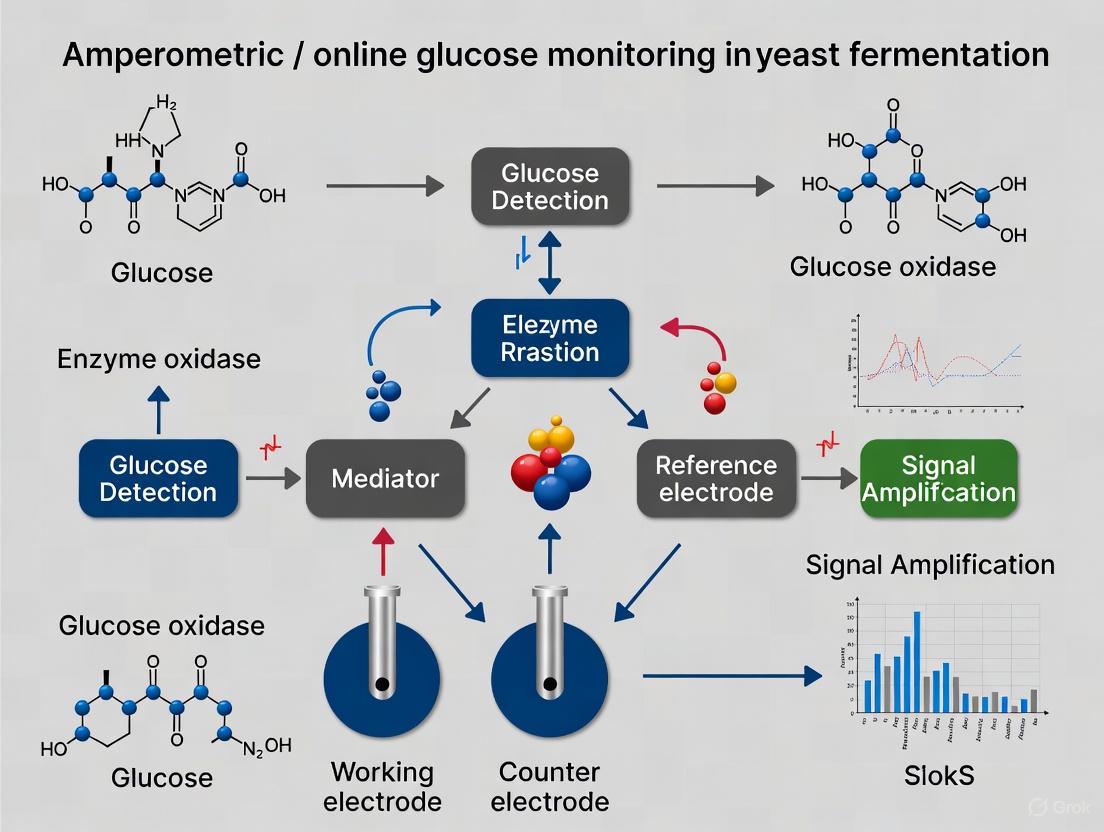
The enzyme-based electrochemical detection of glucose is a cornerstone of modern analytical biotechnology, particularly for monitoring bioprocesses such as yeast fermentation. This detection principle primarily relies on the specific catalytic activity of the enzyme glucose oxidase (GOx) [1] [2]. GOx catalyzes the oxidation of β-D-glucose to D-glucono-1,5-lactone, which subsequently hydrolyzes to gluconic acid [1]. During this reaction, the enzyme's flavin adenine dinucleotide (FAD) cofactor is reduced to FADH₂. For the catalytic cycle to continue, the enzyme must be re-oxidized. This is achieved by molecular oxygen (O₂), which is reduced to hydrogen peroxide (H₂O₂) [2]. The general reaction scheme is as follows: Glucose + GOx(FAD) → Gluconolactone + GOx(FADH₂) GOx(FADH₂) + O₂ → GOx(FAD) + H₂O₂
In amperometric biosensors, this biochemical reaction is transduced into a measurable electrical current. The consumption of oxygen or the production of hydrogen peroxide can be measured electrochemically at a specific applied potential [3] [4]. The magnitude of the resulting current is directly proportional to the glucose concentration in the solution, enabling quantitative analysis. This forms the basis for so-called first-generation biosensors, where the natural cosubstrate (oxygen) is involved in the electron transfer chain [2]. For online monitoring in fermentation processes, where oxygen levels can fluctuate, this dependency can be a limitation. Advances have led to second-generation biosensors that use synthetic redox mediators to shuttle electrons from the enzyme to the electrode, and third-generation biosensors that facilitate direct electron transfer [2]. The integration of these biosensors into flow-through cells allows for continuous, real-time monitoring of glucose levels in fermenters, providing critical data for optimizing feeding strategies and maximizing productivity [3].
Experimental Protocols for Biosensor Construction and Application
This section provides detailed methodologies for fabricating a glucose biosensor and applying it to monitor fermentation processes.
Protocol: Fabrication of a Glucose Oxidase-Based Biosensor
This protocol details the immobilization of Glucose Oxidase (GOx) on an electrode surface via a two-step electrochemical polymerization and chemical attachment method, adapted for robustness in fermentation environments [1].
Key Materials:
- Working Electrode (e.g., Platinum disk, Graphite Rod, or Screen-printed electrode)
- Counter Electrode (e.g., Graphite bar)
- Reference Electrode (e.g., Ag/AgCl or Saturated Calomel Electrode (SCE))
- Monomer solution (e.g., 0.01 M 2,2′-bithiophene (2,2′-BT) in acetonitrile)
- Supporting electrolyte (e.g., 0.1 M Tetraethylammonium hexafluorophosphate (TEAFP₆) in acetonitrile)
- Glucose Oxidase (GOx) from Aspergillus niger
- Condensing agent (e.g., N-cyclohexyl-N′-(2-morpholinoethyl)carbodiimide metho-p-toluenesulfonate (CMC))
- Phosphate Buffered Saline (PBS, 0.1 M, pH 7.4)
Procedure:
- Electrode Pretreatment: Polish the working electrode with alumina slurry (1.0 and 0.3 µm sequentially) on a microcloth. Rinse thoroughly with deionized water and acetone in an ultrasonic bath for 5 minutes to remove any residual polishing material [1].
- Electrochemical Polymerization:
- Place the cleaned working electrode, counter electrode, and reference electrode into a cell containing the monomer solution (e.g., 2,2′-BT) and supporting electrolyte.
- Purge the solution with an inert gas (e.g., Argon) for 20 minutes to remove dissolved oxygen.
- Using a potentiostat, apply a constant potential of +1.33 V (vs. SCE) until a total charge of 100 mC is passed. This forms a conductive polymer film (e.g., poly(2,2′-BT)) on the working electrode surface. The charge density directly influences the film thickness [1].
- Enzyme Immobilization:
- Prepare a solution containing GOx (e.g., 360 U/mg) and the condensing agent CMC in PBS.
- Apply a precise volume (e.g., 5 µL) of this solution onto the surface of the polymer-modified electrode.
- Allow the enzyme to covalently attach to the functionalized polymer surface under controlled humidity for a specified period (e.g., 2 hours at 4°C) [1].
- Biosensor Storage: After immobilization, rinse the biosensor gently with PBS to remove any unbound enzyme. The biosensor can be stored dry at 4°C when not in use.
Protocol: Online Monitoring of Glucose in a Yeast Fermentation Broth
This protocol describes the setup for at-line or on-line glucose monitoring during a yeast fed-batch fermentation using a commercial flow-through biosensor platform [3].
Key Materials:
- Commercial biosensor platform with integrated electrochemical flow-through cell (e.g., B.LV5 chip).
- Peristaltic or syringe pump with appropriate tubing.
- Potentiostat.
- Fermentation bioreactor.
- Automated sampling system (optional, for cell separation if needed).
Procedure:
- System Calibration:
- Prior to fermentation, calibrate the biosensor using standard glucose solutions in a relevant buffer (e.g., PBS, pH 7.4) or a simulated fermentation medium.
- Connect the biosensor to the pump and potentiostat. Flush the system with the calibration standards.
- Measure the amperometric response (e.g., by applying a potential of +0.7 V vs. Ag/AgCl for H₂O₂ oxidation) at each concentration.
- Construct a calibration curve of current response versus glucose concentration.
- Biosensor Integration:
- For on-line monitoring, integrate the biosensor's flow-through cell directly into a bypass loop from the fermentation broth. A cell-separation unit (e.g., microfilter) may be installed upstream to prevent clogging and fouling [3].
- For at-line monitoring, connect the biosensor to an automated sampler that periodically draws samples from the bioreactor.
- Fermentation Monitoring:
- Initiate the yeast fermentation process. The pump continuously draws broth (or cell-free permeate) through the biosensor flow cell.
- The potentiostat applies a constant potential and records the current generated in real-time.
- The software converts the current signal into a glucose concentration based on the pre-established calibration curve.
- Monitor the glucose concentration throughout the fermentation, which can be used to implement feedback control of nutrient feed pumps to maintain glucose at a desired set-point [3].
- System Calibration:
Performance Data and Analysis
The performance of enzyme-based electrochemical glucose biosensors is characterized by several key analytical parameters. The following tables summarize typical performance metrics and operational characteristics relevant to fermentation monitoring.
Table 1: Analytical Performance Characteristics of Representative Glucose Biosensors
| Sensor Type / Configuration | Linear Detection Range (mM) | Sensitivity | Detection Limit (mM) | Stability / Reference |
|---|---|---|---|---|
| GOx/PtCo Nanozyme on Graphite | 0.04 – 2.18 | 19.38 μA mM⁻¹ cm⁻² | 0.021 | 95.33% after 14 days [4] |
| Commercial Flow-Cell (B.LV5) | Up to 150 | Not specified | Not specified | Stable in fermentation broth [3] |
| GOx immobilized on Poly(4,4'-bBT) | Tested in fruit juices | Not specified | Not specified | Suitable for real samples [1] |
Table 2: Key Operational Parameters for Fermentation Monitoring
| Parameter | Consideration | Impact on Measurement |
|---|---|---|
| pH Range | Operational range typically 5–9 [3] | Affects enzyme activity; must be compatible with fermentation broth pH. |
| Temperature | Often operated at 30-37°C | Impacts reaction kinetics and enzyme stability; requires thermostatting. |
| Response Time | Typically < 5 minutes for platform [3] | Crucial for real-time process control and feeding strategies. |
| Anti-Interference | Test against species like ascorbic acid, uric acid, dopamine [4] | Ensures specificity in the complex matrix of fermentation broth. |
| Oxygen Dependency | Critical for 1st generation biosensors [2] | Fluctuating dissolved O₂ in fermenters can affect signal accuracy. |
Schematic Diagrams of Working Principles and Workflows
Glucose Biosensor Working Principle
Online Fermentation Monitoring Workflow
The Scientist's Toolkit: Research Reagent Solutions
Table 3: Essential Materials and Reagents for Glucose Biosensor Development
| Item Name | Function / Role | Example / Specification |
|---|---|---|
| Glucose Oxidase (GOx) | Biological recognition element; catalyzes glucose oxidation. | From Aspergillus niger, lyophilized powder, ~360 U/mg [1] [4]. |
| Conducting Polymer | Electrode modification; provides a matrix for enzyme immobilization. | Poly(2,2'-bithiophene) or derivatives, electrochemically polymerized [1]. |
| Nanozymes | Artificial enzymes; enhance electron transfer and catalyze H₂O₂ reduction. | Bimetallic PtCo nanoparticles with peroxidase-like activity [4]. |
| Redox Mediator | Shuttles electrons between enzyme and electrode (2nd Gen sensors). | p-Benzoquinone (BQ), Ferrocene derivatives [1] [2]. |
| Nafion Membrane | Permselective coating; blocks interference from anions (e.g., urate, ascorbate). | 5% wt. solution in aliphatic alcohols [4]. |
| Cross-linking Agent | Facilitates covalent attachment of enzymes to the sensor surface. | N-cyclohexyl-N′-(2-morpholinoethyl)carbodiimide (CMC) [1]. |
Online glucose monitoring is a critical requirement in yeast fermentation research, enabling precise control over bioprocesses for applications in biofuel production, pharmaceuticals, and food sciences. Amperometric biosensors provide an effective solution for this analytical challenge by combining the specificity of enzyme-based biological recognition with the sensitivity of electrochemical transduction. This document details the core components, experimental protocols, and performance characteristics of amperometric biosensors, with specific application to glucose monitoring in yeast fermentation systems. The content is structured to provide researchers with practical methodologies for biosensor construction, integration, and validation within flow injection analysis (FIA) systems commonly used for offline fermentation monitoring.
Core Biosensor Components and Materials
Research Reagent Solutions and Essential Materials
Table 1: Essential Research Reagents and Materials for Glucose Biosensor Construction
| Component Category | Specific Item/Example | Function/Purpose |
|---|---|---|
| Biological Recognition Element | Glucose Oxidase (GOx) from Aspergillus niger | Catalyzes glucose oxidation, producing H₂O₂ as a measurable electroactive product [5]. |
| Biological Recognition Element | Pyranose Oxidase (PyOx) | An alternative oxidase enzyme for glucose detection; offers different substrate specificity and stability profiles [5]. |
| Transducer Material | Gold working electrode (e.g., polyamidoamine/cysteamine-modified) | Serves as the physical interface for electrical signal transduction; modifications enhance enzyme immobilization and electron transfer [5]. |
| Transducer Material | Graphite-Epoxy Biocomposite (bulk-modified with Au-Pd) | Functions as both the enzyme support matrix and the electrocatalytic surface for H₂O₂ oxidation [6]. |
| Electrocatalytic Elements | Gold (Au) and Palladium (Pd) nanoparticles | Incorporated into the biocomposite to electrocatalytically oxidize H₂O₂, lowering the operational potential and reducing interference [6]. |
| Reference Electrode | Ag/AgCl (Silver/Silver Chloride) | Provides a stable, known reference potential for the amperometric measurement in a three-electrode system [6]. |
| Carrier Solution | Phosphate Buffer (e.g., 0.1 M, pH 7.0 with 0.1 M KCl) | Serves as the supporting electrolyte in FIA systems, maintaining stable pH and ionic strength [6]. |
System Architecture and Operational Workflow
The following diagram illustrates the typical workflow for offline monitoring of yeast fermentation using a flow-injection integrated amperometric biosensor.
Experimental Protocols and Methodologies
Protocol 1: Construction of a PyOx/GOx-Modified Gold Electrode
This protocol describes the assembly of an enzyme-based biosensor for offline glucose monitoring in yeast culture, adapted from established methods [5].
3.1.1 Objectives: To immobilize Pyranose Oxidase (PyOx) or Glucose Oxidase (GOx) onto a modified gold electrode for the amperometric detection of glucose via measurement of oxygen consumption.
3.1.2 Materials:
- Gold working electrode (e.g., 2 mm diameter)
- Polyamidoamine (PAMAM) dendrimers and cysteamine
- Pyranose Oxidase (PyOx) or Glucose Oxidase (GOx) enzyme
- Phosphate buffer (50 mM, pH 7.0 for PyOx; or 50 mM citrate buffer, pH 4.0 for GOx)
- Glutaraldehyde (2.5% v/v) or EDC/NHS crosslinking chemistry
- Flow Injection Analysis (FIA) system with peristaltic pump and injection valve
3.1.3 Step-by-Step Procedure:
- Electrode Pretreatment: Clean the gold electrode surface by sequential polishing with alumina slurries (1.0, 0.3, and 0.05 µm) and sonicate in distilled water. Electrochemically clean via cyclic voltammetry in 0.5 M H₂SO₄.
- Surface Modification: Incubate the clean electrode in an aqueous solution of cysteamine (e.g., 20 mM) for 1 hour to form a self-assembled monolayer (SAM). Rinse thoroughly with distilled water.
- Polymer/Enzyme Immobilization: Layer PAMAM dendrimers onto the cysteamine-modified surface. Subsequently, immobilize the enzyme (PyOx or GOx) using a crosslinker such as glutaraldehyde. Optimize the enzyme loading for maximum sensitivity and stability (e.g., 1-2 U per electrode).
- Biosensor Conditioning: Prior to use, condition the biosensor by incubating it in the respective buffer (phosphate for PyOx, citrate for GOx) for 12-24 hours to stabilize the enzymatic activity.
3.1.4 Critical Parameters:
- pH: The optimal activity for PyOx-based sensors is at pH 7.0, while for GOx-based sensors, it is at pH 4.0 [5]. Use the appropriate buffer system.
- Applied Potential: The amperometric detection is typically performed at -0.7 V (vs. Ag/AgCl) to monitor the reduction in oxygen concentration [5].
- Interference: Assess the impact of common interferents (e.g., ascorbic acid, uric acid) in the fermentation matrix. The use of a permselective membrane (e.g., Nafion) may be required.
Protocol 2: Fabrication of a Bulk-Modified Graphite-Epoxy Biocomposite Biosensor
This protocol outlines the preparation of a robust, mediator-free glucose biosensor where the enzyme and electrocatalytic metals are incorporated directly into the transducer material [6].
3.2.1 Objectives: To fabricate a graphite-epoxy biocomposite electrode bulk-modified with Glucose Oxidase (GOD), gold, and palladium for the electrocatalytic detection of H₂O₂ in a flow system.
3.2.2 Materials:
- Graphite powder (e.g., 50 µm particle size)
- Epoxy resin (e.g., Epo-Tek H77) and hardener
- Glucose Oxidase (GOD) enzyme
- Gold (Au) and Palladium (Pd) powders (micro-particle size)
- Phosphate buffer saline (PBS: 0.1 M phosphate, 0.1 M KCl, pH 7.0)
3.2.3 Step-by-Step Procedure:
- Biocomposite Preparation: Mix graphite powder, Au powder, Pd powder, and GOD enzyme in a defined mass ratio (e.g., 40:15:15:30 w/w) thoroughly.
- Epoxy Integration: Add the epoxy resin and hardener to the solid mixture. Blend rigorously until a homogeneous, sticky paste is formed.
- Electrode Curing: Pack the composite paste into the cavity of an electrode body (e.g., a Teflon sleeve). Insert a electrical contact wire. Cure the assembly at room temperature for at least one week.
- Surface Renewal: Before use, gently polish the electrode surface on fine grit sandpaper to expose fresh, active composite material. Rinse with buffer.
- FIA Integration: Integrate the biosensor as the working electrode in a flow cell, positioned downstream of the sample injection valve.
3.2.4 Critical Parameters:
- Curing Time: Incomplete curing can lead to leaching of components and signal instability. Ensure a full one-week cure.
- Applied Potential: For H₂O₂ oxidation on Au-Pd surfaces, apply a potential of +0.9 V (vs. Ag/AgCl) [6].
- Sampling Frequency: The FIA system allows for a high throughput of approximately 20 samples per hour [6].
Performance Characterization and Data Analysis
Analytical Performance Metrics
The performance of the described biosensor configurations must be rigorously validated. The table below summarizes key quantitative data from the literature for easy comparison.
Table 2: Performance Comparison of Amperometric Glucose Biosensors for Fermentation Monitoring
| Biosensor Configuration | Linear Range | Sensitivity | Optimal pH | Applied Potential | Key Advantages |
|---|---|---|---|---|---|
| PyOx-based (Au Electrode) [5] | 0.025 – 0.5 mM | Not specified (Calibration: y = 3.358x + 0.028) | 7.0 (Phosphate Buffer) | -0.7 V (vs. Ag/AgCl) | High sensitivity in neutral pH; suitable for physiological and fermentation conditions. |
| GOx-based (Au Electrode) [5] | 0.01 – 1.0 mM | Not specified (Calibration: y = 1.539x + 0.181) | 4.0 (Citrate Buffer) | -0.7 V (vs. Ag/AgCl) | Wider linear range; optimal for acidic fermentation broths. |
| GOD-based (Graphite-Epoxy-Au-Pd Biocomposite) [6] | 1 – 10 g L⁻¹ (≈ 5.6 – 56 mM) | Not explicitly stated | 7.0 (Phosphate Buffer) | +0.9 V (vs. Ag/AgCl) | Extremely robust; integrated electrocatalysis reduces interference; adapted for high glucose levels in bioprocesses. |
| Enzyme-free (Nanostructured Composite) [7] | Not fully specified | 95.12 ± 2.54 µA mM⁻¹ cm⁻² | Not specified | Not specified | High stability; avoids limitations of enzymatic sensors (e.g., denaturation). |
Data Validation and Correlation with Reference Methods
A critical step in the application of any biosensor is the validation of its results against established standard methods.
- HPLC Correlation: For offline monitoring of yeast fermentation, the glucose concentrations determined by the FIA-combined biosensor (e.g., the PyOx/GOx-based sensor) must show a strong correlation with concentrations measured using High-Performance Liquid Chromatography (HPLC) [5]. A successful validation yields a high correlation coefficient (R² > 0.99), confirming the accuracy and reliability of the biosensor for complex fermentation matrices [5] [6].
- Commercial Sensor Correlation: Similarly, results from the bulk-modified graphite-epoxy biocomposite sensor should be compared with those from a commercially available glucose sensor to ensure parity and industrial relevance [6].
System Integration and Schematic
The integration of the biosensor into a flow system is paramount for automated, offline monitoring. The following diagram details the components and logical flow of a typical FIA setup.
The Critical Need for Online Monitoring in Yeast Fermentation Bioprocesses
Effective monitoring and control of yeast fermentation bioprocesses is paramount for optimizing productivity, ensuring product quality, and maximizing yield in both laboratory and industrial settings. Traditional monitoring methods often rely on infrequent manual sampling and offline analysis, which are resource-intensive, time-consuming, and provide only a retrospective view of the process state. This data lag prevents real-time intervention, leading to suboptimal process control and inconsistent outcomes. The transition to online monitoring, particularly for critical parameters like glucose, represents a significant leap forward. Framed within the broader context of developing amperometric biosensors for online glucose monitoring, this article details the pivotal parameters, advanced sensing platforms, and practical protocols that enable researchers to achieve unprecedented visibility and control over their fermentation processes.
Key Parameters for Fermentation Monitoring
Effective fermentation control hinges on the continuous tracking of several key physicochemical parameters. The table below summarizes these critical parameters and the sensing technologies used for their online detection.
Table 1: Key Online Monitoring Parameters in Yeast Fermentation
| Parameter | Significance in Fermentation | Common Sensor Technology |
|---|---|---|
| pH | Impacts enzyme activity, microbial growth rates, and product stability [8]. | Potentiometric microsensors [8]. |
| Temperature | Critically influences metabolic reaction rates and overall yeast health [8]. | Resistance Temperature Detector (RTD) [8]. |
| Dissolved Oxygen (DO) | Essential for aerobic metabolic processes; a key indicator of microbial activity [8]. | Amperometric microelectrodes [8]. |
| Glucose | Primary carbon and energy source; its concentration directly controls growth and productivity [3]. | Amperometric biosensors [8] [3]. |
| Conductivity | Indirectly measures microbial growth and activity through changes in electrolyte composition [8]. | Interdigitated electrodes [8]. |
While monitoring standard parameters like pH, temperature, and dissolved oxygen is established practice, state-of-the-art fermentation monitoring has historically been limited in its capacity for direct, continuous substrate quantification [3]. Glucose, as a major carbon source, is a critical process variable. The implementation of online glucose monitoring is a key advancement for modern bioprocessing, enabling feeding strategies that optimize biomass production and the synthesis of target metabolites like alcohols, proteins, and amino acids [3].
Advanced Sensor Platforms for Online Glucose Monitoring
Novel biosensor platforms are overcoming the limitations of traditional analytical methods like HPLC, which are ill-suited for continuous monitoring due to their time-consuming nature and resource demands [3].
Multi-Sensor Array Chips
Integrated multi-sensor arrays represent a significant step forward in fermentation monitoring. These microfabricated chips, often only a few square millimeters in size, can simultaneously monitor multiple parameters, including pH, temperature, conductivity, dissolved oxygen, and glucose [8]. A key innovation in these platforms is the use of nanostructured platinum (nano-Pt) on microelectrodes, which is achieved through a simple, CMOS-compatible electrodeposition process. This nanostructuring enhances sensor sensitivity and specificity, allows for reliable operation at lower applied potentials, and minimizes electrical cross-talk between closely integrated sensors [8]. The development of all-solid-state on-chip reference electrodes is pivotal for true miniaturization and standalone operation, eliminating the need for bulky external reference electrodes [8].
Commercial Flow-Through Biosensor Platforms
Commercial automated biosensor platforms have been successfully applied to online glucose monitoring in fermenters. One documented system utilizes a flow-through-cell with integrated first-generation electrochemical glucose biosensors [3]. This platform is designed as a 1 μl flow-through cell and can be readily integrated into a fermentation setup via tubing and luer fittings [3].
A remarkable feature of this particular platform is its extended linear detection range for glucose, up to 150 mM in complex fermentation broth. This range is four to six-fold higher than many biosensors described previously, which were often limited to around 25 mM, a concentration relevant for clinical diagnostics [3]. The platform demonstrated accurate glucose quantification in both cell-free and cell-containing samples, outstanding mechanical stability in direct contact with fermentation medium, and resistance to interference from various electroactive species. Analysis time is significantly reduced to less than 5 minutes per measurement compared to conventional HPLC, making it suitable for real-time monitoring and control [3].
Integrated Fermentation Management Systems
Complete online monitoring systems, such as the BrewIQ system, bring "Internet of Things" (IoT) technology to fermentation. These systems provide continuous, live-streamed data on a suite of parameters—including dissolved oxygen, pH, specific gravity, pressure, temperature, and conductivity—accessible remotely from any internet-connected device [9]. This enables researchers to monitor active fermentations, receive preemptive alerts if parameters deviate from set thresholds, and compare batch-to-batch trends for deeper insight into process consistency [9].
Experimental Protocols for Online Glucose Monitoring
This section provides a detailed methodology for implementing an online glucose biosensor for yeast fermentation monitoring, based on established research applications [3].
Biosensor Platform Setup and Operation
- Working Principle: The biosensor is based on a 1st generation amperometric design. The electrochemical cell typically contains two platinum (Pt) working electrodes (one enzyme-coated and one blank), a Pt counter electrode, and an internal Ag/AgCl pseudo-reference electrode [3].
Required Equipment & Materials:
- Biosensor chip (e.g., B.LV5 with extended range)
- Potentiostat (e.g., SIX transmitter)
- Operation software (e.g., bioMON)
- Peristaltic pump and chemical-resistant tubing (e.g., TYGON S3 E-LFL, ID 1.52 mm)
- Luer connectors
- Sterile fermentation medium and sample stream access
Integration Procedure:
- Connect the biosensor chip to the potentiostat.
- Connect the peristaltic pump to the inlet tubing of the biosensor via a luer connector.
- Connect the outlet tubing from the biosensor to a waste container or back to the bioreactor, considering sterility.
- Establish a sample stream from the bioreactor to the pump inlet, ensuring a representative and cell-free or cell-containing flow, as required.
- Calibrate the biosensor according to manufacturer protocols using standard glucose solutions in a matrix matching the fermentation medium.
On-line Monitoring During Fermentation:
- Once the fermentation is initiated and the biosensor is calibrated, start the continuous flow of the fermentation broth through the biosensor cell using the peristaltic pump.
- The potentiostat applies a constant potential, and the current generated from the enzymatic oxidation of glucose is measured and converted to a glucose concentration.
- Data is collected in real-time by the operating software, allowing for continuous tracking of glucose levels throughout the batch, fed-batch, or continuous process.
Performance Validation and Data Analysis
- Reference Method: High-Performance Liquid Chromatography (HPLC) with a Refractive Index (RI) detector is used as a reference method to validate the accuracy of the biosensor readings [3].
- Validation Protocol:
- Periodically collect manual samples from the fermenter parallel to the biosensor's analysis.
- Immediately quench and process samples for HPLC analysis.
- Correlate the glucose concentration values obtained from the offline HPLC analysis with the values recorded by the biosensor platform at the corresponding time points.
- Assess the correlation to ensure the biosensor's performance is not compromised by the complex fermentation matrix or the presence of electroactive interfering species.
The workflow below illustrates the logical sequence and components of this integrated online monitoring system.
Online Glucose Monitoring Workflow
The Scientist's Toolkit: Research Reagent Solutions
The table below lists essential materials and reagents for setting up an online glucose monitoring system for yeast fermentation.
Table 2: Essential Research Reagents and Materials for Online Glucose Monitoring
| Item | Function / Description | Application Note |
|---|---|---|
| Biosensor Chip | A flow-through cell with integrated enzyme-coated and blank electrodes for glucose detection and signal correction [3]. | Look for features like an extended range (e.g., up to 150 mM glucose) and compatibility with complex media [3]. |
| Glucose Oxidase (GOx) | The core bioreceptor enzyme in 1st generation biosensors; catalyzes the oxidation of glucose, producing a measurable current [3]. | Ensures high selectivity for glucose within the complex fermentation matrix. |
| Potentiostat | The electronic instrument that applies a constant potential to the biosensor and measures the resulting current [3]. | Must be compatible with the biosensor chip and capable of real-time data transmission. |
| Calibration Standards | Solutions of known glucose concentration in a matrix matching the fermentation medium. | Used for initial calibration and periodic validation of the biosensor to ensure accuracy over long run times. |
| Nanostructured Platinum | Material used to modify microelectrodes, enhancing sensitivity and specificity while reducing operational potential [8]. | A key material in advanced multi-sensor arrays to improve performance. |
Application and Impact
The implementation of online glucose monitoring has direct and profound implications for fermentation research and development. Real-time glucose data allows for the implementation of advanced feeding strategies, moving away from pre-defined schedules to dynamic control based on actual metabolic demand. This precision prevents both substrate limitation, which can stall growth, and over-feeding, which can lead to the formation of inhibitory by-products like ethanol in yeast cultures [3].
The ability to determine fermentation completion with confidence using live data, as offered by systems like BrewIQ, directly improves operational efficiency by decreasing labor costs, eliminating unnecessary tank time, and managing inputs more efficiently [9]. Furthermore, the aggregation of high-resolution data from multiple batches provides invaluable insights for benchmarking, understanding the impact of different ingredients or hardware, and ultimately increasing product quality and consistency for greater market success [9]. The integration of these biosensor platforms represents a fundamental tool for modernizing bioprocesses and achieving the level of control required in modern industrial biotechnology.
The monitoring of glucose concentration is a critical parameter in yeast fermentation processes, essential for optimizing biomass production and the yield of metabolites in both research and industrial settings [3] [10]. Traditional analytical methods, such as High-Performance Liquid Chromatography (HPLC), are widely used but present significant limitations for process control due to their inherent delays [3] [10]. Amperometric biosensors, particularly those employing glucose oxidase (GOx), have emerged as a powerful alternative, offering transformative advantages in speed, cost-effectiveness, and seamless automation for online monitoring [3] [11].
This document provides detailed application notes and protocols for implementing an amperometric glucose biosensor platform for online monitoring in yeast fermentation research. It is structured to equip scientists and drug development professionals with the practical knowledge to integrate this technology, thereby enhancing bioprocess efficiency and control.
Quantitative Advantages of Amperometric Biosensors
The following table summarizes a direct performance and resource comparison between a representative amperometric biosensor platform and conventional HPLC analysis, based on data from a yeast fed-batch fermentation study [3] [10].
Table 1: Performance and Resource Comparison: Amperometric Biosensor vs. HPLC
| Feature | Amperometric Biosensor Platform | Traditional HPLC (with RI Detector) |
|---|---|---|
| Analysis Time | < 5 minutes [3] [10] | Resource and time-intensive [3] [10] |
| Detection Range | Up to 150 mM (in fermentation broth) [3] [10] | Typically suitable for a wide range |
| Measurement Mode | On-line, at-line, and continuous capability [3] [10] | Off-line (manual sampling) |
| Key Resource Savings | Significant reduction in time and operational resources [3] [10] | Requires dedicated operator time and costly consumables |
| Automation Potential | High - readily integrated into fermentation setup for closed-loop control [3] [10] | Low - requires manual sample handling and injection |
The data underscores the core strengths of the biosensor platform: its rapidity, providing results in less than five minutes, and its suitability for automation, enabling real-time process decisions that are not feasible with slower, offline methods [3] [10].
Experimental Protocol: On-Line Glucose Monitoring in Yeast Fermentation
This protocol details the setup and operation of a commercial flow-through-cell amperometric biosensor for online glucose monitoring during a yeast fed-batch fermentation.
Research Reagent Solutions and Essential Materials
Table 2: Key Research Reagent Solutions and Materials
| Item | Function / Description |
|---|---|
| Biosensor Chip (B.LV5) | A flow-through cell with integrated 1st generation electrochemical glucose biosensors (e.g., from Jobst Technologies GmbH). Features a multi-array design with Pt-working electrodes and an Ag/AgCl reference [3] [10]. |
| Potentiostat (SIX transmitter) | Instrument for applying a constant potential (+450 mV vs. Ag/AgCl) and measuring the resulting current [3] [10]. |
| Peristaltic Pump (e.g., Ismatec) | Provides controlled flow of fermentation sample or calibration standards through the biosensor flow-cell [3] [10]. |
| Software (e.g., bioMON) | Used for operating the biosensor platform, data acquisition, and visualization of real-time glucose signals [3] [10]. |
| Glucose Oxidase (GOx) | The biological recognition element immobilized on the sensor. Catalyzes the oxidation of β-D-glucose, producing the measurable signal [1] [11]. |
| Buffer (e.g., 0.1 M PBS, pH 7.5) | Carrier solution and medium for enzyme immobilization and system calibration [12]. |
| Fermentation Bioreactor | Standard laboratory or pilot-scale bioreactor equipped with standard probes (pH, DO, temperature) and an aseptic sampling port [3]. |
Detailed Methodology
Biosensor Platform Setup and Integration
- System Assembly: Connect the peristaltic pump to the inlet of the biosensor chip (B.LV5) using appropriate tubing (e.g., TYGON S3TM, ID 1.52 mm). Connect the outlet of the biosensor to a waste container or back to the bioreactor if sterility is maintained. Connect the biosensor chip to the SIX transmitter potentiostat [3] [10].
- Software Configuration: Interface the potentiostat with the controlling PC and launch the bioMON software. Configure the chronoamperometric method to apply a constant potential of +450 mV versus the internal Ag/AgCl pseudo-reference electrode [3] [10].
- Bioreactor Integration: Connect the inlet tubing of the pump to an aseptic sampling loop or a continuous bypass stream from the fermentation bioreactor. Ensure the flow path can be sterilized (e.g., via autoclaving or chemical sanitization) to maintain a sterile fermentation environment [3].
Calibration and Operation
- Calibration: Before initiating fermentation, pump standard glucose solutions (e.g., 0, 25, 50, 100 mM) in a buffer that matches the fermentation medium's pH and ionic strength through the biosensor. The software will record the steady-state current for each concentration, generating a calibration curve that relates current to glucose concentration [3].
- On-Line Monitoring: Start the fermentation process. The peristaltic pump continuously draws sample from the bioreactor through the biosensor flow-cell. The glucose in the sample is enzymatically oxidized by the immobilized GOx, and the subsequent redox reaction (typically the electro-oxidation of H₂O₂) generates a current proportional to the glucose concentration [3] [11].
- Signal Processing: The platform automatically subtracts the background current (obtained from a blank, non-enzyme coated electrode on the same chip) to correct for interference from other electroactive species in the complex fermentation broth [3] [10].
- Data Output: The real-time glucose concentration is displayed and logged by the software, providing a continuous profile of the fermentation metabolic status.
The workflow and signal transduction pathway of the integrated system are illustrated below.
Discussion
Synergy with Smart Fermentation and Industrial IoT
The amperometric biosensor platform is a foundational component of the emerging "Smart Fermentation" paradigm [13]. Its ability to generate high-frequency, real-time data makes it an ideal physical layer sensor for integration with Internet of Things (IoT) and Machine Learning (ML) systems. The continuous glucose data stream can be fed into predictive ML models to forecast fermentation dynamics and proactively adjust feeding strategies, moving from reactive to predictive process control [13]. This integration is a critical step toward aligning bioprocess manufacturing with the principles of Industry 4.0.
Addressing Limitations and Ensuring Data Quality
While highly effective, researchers must be aware of certain limitations:
- Oxygen Limitation: First-generation GOx-based biosensors rely on oxygen as a co-substrate. In oxygen-limited fermentation conditions, the sensor response can be compromised, leading to an underestimation of glucose concentration [3] [10].
- Biofouling: Long-term exposure to complex fermentation broth can lead to the accumulation of cells or macromolecules on the sensor surface (biofouling), potentially degrading signal stability over time [14].
- Mitigation Strategies: The use of a multi-array sensor with integrated blank electrodes automatically corrects for signals from interfering electroactive species [3] [10]. Optimal sensor performance is achieved by ensuring the operational environment (e.g., pH range of 5–9) is maintained and by implementing periodic calibration checks [3] [10].
The implementation of amperometric biosensor technology for online glucose monitoring provides a decisive advantage over traditional chromatographic methods in terms of speed, cost-efficiency, and automation potential. The provided protocol and application notes demonstrate a robust and reliable methodology for obtaining real-time, actionable data during yeast fermentations. The integration of this technology is a pivotal advancement for achieving precise control in bioprocess development, ultimately leading to enhanced productivity, consistency, and scalability in research and industrial applications.
Implementing Online Monitoring: Sensor Designs, Fabrication, and Integrated Systems
The move toward advanced bioprocess monitoring is driven by the critical need for real-time data to optimize productivity and control in fermentation. State-of-the-art biosensor platforms, specifically flow-cell systems and integrated multi-sensor arrays, address the significant limitations of traditional offline analysis methods, which are often resource-intensive and time-consuming [10] [3]. These modern platforms enable reliable identification and quantification of key parameters like glucose concentration in an online mode, allowing fermentation processes to operate at optimal reactor efficiency [10]. This document details the application of these platforms within the specific context of amperometric biosensors for online glucose monitoring in yeast fermentation research, providing structured data, detailed protocols, and visual workflows for the scientific community.
Flow-Cell Electrochemical Biosensor Platforms
Flow-cell biosensors represent a robust and automated approach for online monitoring, ideal for integration into fermentation setups. Their design allows for continuous or semi-continuous analysis of the fermentation broth with minimal manual intervention.
Platform Principle and Configuration
The core of this platform is a commercial miniaturized flow-through-cell (e.g., B.LV5 biosensor chip) with integrated electrochemical glucose biosensors [10] [3]. The system operates as a first-generation amperometric biosensor, where glucose oxidase (GOD) catalyzes the oxidation of glucose, producing hydrogen peroxide (H₂O₂). This H₂O₂ is then electrochemically oxidized at a platinum electrode at a set potential (+450 mV vs. an Ag/AgCl pseudo-reference electrode), generating a current signal proportional to the glucose concentration [10] [15]. The flow-cell is designed with a very small internal volume (1 µL), facilitating rapid sample exchange and minimizing analysis time to less than 5 minutes per measurement [10]. The platform's multi-array design, which includes duplicate working electrodes and blank electrodes for signal correction, enhances data reliability and extends the dynamic detection range up to 150 mM, which is critical for fermentation environments [10].
Table 1: Key Components of a Flow-Cell Biosensor Platform
| Component | Specification/Model | Function |
|---|---|---|
| Biosensor Chip | B.LV5 (Jobst Technologies GmbH) | A 1 µL flow-through cell with Pt working, counter, and Ag/AgCl reference electrodes. |
| Potentiostat | SIX Transmitter | Applies the operational potential and measures the generated current. |
| Software | bioMON | Operates the biosensor platform and data acquisition. |
| Peristaltic Pump | Ismatec Reglo ICC | Drives the flow of the sample or standard through the biosensor cell. |
| Tubing | TYGON S3 E-LFL (ID 1.52 mm) | Connects the bioreactor, pump, and biosensor. |
Figure 1: Signaling pathway of a first-generation amperometric glucose biosensor. Glucose is enzymatically oxidized, and the generated H₂O₂ is measured at an electrode, producing a current signal [10] [15].
Application Protocol: Online Glucose Monitoring in Yeast Fermentation
Title: At-line and On-line Glucose Quantification in Yeast Fed-Batch Fermentation Using a Flow-Cell Biosensor Platform. Objective: To reliably monitor glucose concentration in complex fermentation broth in near real-time, enabling precise process control. Materials:
- Biosensor platform (as described in Table 1).
- Fed-batch yeast bioreactor.
- Sterile, oxygen-free sampling tubing and connectors.
- HPLC system with Refractive Index (RI) detector (for reference/validation measurements).
- Standard glucose solutions (spanning 0 - 150 mM) in a matrix matching the fermentation medium.
Procedure:
- System Setup and Calibration:
- Integrate the flow-cell biosensor into the fermentation setup via a sterile flow path, ensuring a closed and aseptic system.
- Connect the biosensor outlet to a waste container or return line to the bioreactor.
- Flush the system with a neutral buffer (e.g., phosphate buffer, pH 7.0) to establish a stable baseline signal.
- Perform a multi-point calibration by pumping standard glucose solutions of known concentration (e.g., 0, 25, 50, 100, 150 mM) through the biosensor. Record the steady-state current for each standard.
- Generate a calibration curve (current vs. glucose concentration).
Sample Measurement:
- For at-line analysis: Aseptically withdraw a sample from the bioreactor. The sample can be measured directly (cell-containing) or after centrifugation (cell-free). Pump the sample through the biosensor and record the current signal.
- For on-line analysis: Configure the pump to automatically draw broth directly from the bioreactor through the biosensor at defined intervals (e.g., every 5 minutes). The system can operate in a continuous flow or stopped-flow mode for measurement.
Data Processing and Validation:
- Convert the measured current signal to glucose concentration using the pre-established calibration curve.
- The biosensor's software can automatically subtract the background current from the integrated blank electrode to correct for interference from electroactive species in the broth [10].
- For method validation, periodically analyze selected samples using a reference method such as HPLC-RI to confirm the accuracy of the biosensor readings [10] [3].
Integrated Multi-Sensor Array Platforms
Integrated multi-sensor arrays leverage redundancy and multiplexing to overcome the limitations of single sensors, enhancing measurement reliability, accuracy, and fault tolerance.
Platform Principle and Configuration
These platforms consist of high-density arrays of individually addressable sensing units fabricated on a single substrate. This design provides several key advantages. Redundant sensing, where multiple sensors are functionalized for the same analyte (e.g., glucose), ensures system reliability even if one or more sensors fail, a critical feature for long-term fermentations [15]. Multiplexed detection allows for the simultaneous measurement of multiple parameters, such as glucose and other critical ions (K⁺, Na⁺, Ca²⁺), providing a more comprehensive view of the fermentation microenvironment [16]. The fabrication of these arrays often employs techniques like screen-printing or micro-electromechanical systems (MEMS), making them portable, affordable, and disposable [17]. To handle the inherent device-to-device variation in such arrays, the platform is coupled with custom electronics and machine learning algorithms that calibrate the sensor responses and extract accurate concentration data, transforming material variability into a source of robust information [16].
Table 2: Representative Performance Metrics of Advanced Biosensor Platforms
| Platform Type | Target Analyte | Linear Detection Range | Measurement Time | Key Advantage | Reference |
|---|---|---|---|---|---|
| Flow-Cell Biosensor | Glucose | Up to 150 mM | < 5 minutes | High stability in complex broth; Extended range | [10] |
| Integrated Microneedle Array | Glucose (in ISF) | Not specified | Real-time (Continuous) | Redundant sensing; Combines sensing & drug delivery | [15] |
| Graphene Transistor Array | K⁺, Na⁺, Ca²⁺ | Several orders of magnitude | ~3 minutes (full sweep) | High-throughput multiplexing; Machine learning calibration | [16] |
Application Protocol: Redundant Glucose Monitoring with a Multi-Electrode Array
Title: High-Reliability Glucose Monitoring in Yeast Fermentation Using a Redundant Biosensor Array. Objective: To achieve robust and continuous glucose measurement by leveraging a multi-electrode array design that compensates for individual sensor drift or failure. Materials:
- Integrated biosensor array with multiple (e.g., 4) independently addressable working electrodes, counter, and reference electrodes.
- Multi-channel potentiostat or switching unit.
- Yeast fermentation bioreactor.
- Data acquisition system and computer with analysis software.
Procedure:
- Sensor Functionalization:
- The working electrodes of the array are coated with a glucose-sensitive layer containing Glucose Oxidase (GOD). The immobilization matrix may include polymers or hydrogels to stabilize the enzyme [15].
- A protective membrane (e.g., polyurethane, Nafion) may be applied over the enzyme layer to enhance selectivity and reduce fouling from the fermentation broth.
System Integration and Calibration:
- Integrate the sensor array into the fermentation vessel, ensuring the active sensing area is in contact with the broth.
- Connect each working electrode to its independent measurement channel.
- Calibrate the entire array by exposing it to standard glucose solutions. Record the amperometric response (current at a fixed potential, e.g., +0.5 V vs. Ag/AgCl) of each individual sensor to the standards.
Fermentation Monitoring and Data Fusion:
- Initiate the yeast fermentation process.
- Continuously or intermittently measure the current from all functional glucose sensors in the array simultaneously.
- The backend software collects data from all channels. A data fusion algorithm (e.g., calculating a trimmed mean, discarding outlier sensor readings) is used to compute a single, highly reliable glucose concentration value in real-time [15].
- This approach ensures that the system remains operational and provides accurate data even if one sensor in the array degrades or fails, thus offering superior robustness for long-duration experiments.
Figure 2: Experimental workflow for glucose monitoring comparing flow-cell and integrated array platforms, culminating in data processing and a final output.
The Scientist's Toolkit: Research Reagent Solutions
Table 3: Essential Materials and Reagents for Biosensor-Based Fermentation Monitoring
| Item | Function / Role | Application Note |
|---|---|---|
| Glucose Oxidase (GOD) | Biorecognition element; catalyzes glucose oxidation. | The stability of GOD determines sensor lifetime. Use high-purity, lyophilized enzyme from a reliable supplier. |
| Platinum (Pt) Electrodes | Working electrode material; catalyzes H₂O₂ oxidation. | Provides a stable and sensitive surface for amperometric detection. |
| Ag/AgCl Electrode | Stable reference electrode for potential control. | Essential for maintaining a fixed potential in three-electrode systems. |
| Potato Dextrose Broth (PDB) | Culture medium for Saccharomyces cerevisiae. | Serves as the nutrient base for yeast growth and metabolism during fermentation [18]. |
| Saccharomyces cerevisiae | Model yeast organism for fermentation research. | Strain FNCC-3049 has been used in biosensor development for sugar metabolism studies [18]. |
| Ion-Selective Membranes (ISMs) | Enable selective detection of specific ions (K⁺, Na⁺, Ca²⁺). | Functionalize graphene transistors in multi-sensor arrays for multiplexed analysis [16]. |
| Redox-Responsive Hydrogels | Smart material for controlled drug release. | Can be loaded with insulin or other biomolecules for triggered release in advanced platforms [15]. |
The pursuit of efficient online glucose monitoring in yeast fermentation represents a significant challenge in bioprocess engineering. Effective and continuous monitoring of bioprocesses requires the parallel screening of multiple key parameters to enhance process efficiency and ultimately improve the quality of the end products [8]. State-of-the-art fermentation on-line monitoring is often limited to standard measurements such as pH, temperature, and dissolved oxygen, creating a critical gap in real-time substrate quantification [3] [10]. This application note details the integration of nanostructured platinum and redox gels as advanced materials to address this limitation, enabling the development of robust, sensitive, and selective amperometric biosensors for reliable glucose tracking in complex fermentation broths.
The core challenge in amperometric biosensor design involves achieving efficient electron transfer between the enzyme's redox center and the electrode surface. For glucose oxidase (GOx), the flavin adenine dinucleotide (FAD) coenzyme is deeply embedded within the protein structure, making direct electron transfer (DET) kinetically challenging [19]. This application note explores two advanced material strategies to overcome this barrier: (1) the use of nanostructured platinum to enhance electrocatalytic properties and electrode surface area, and (2) the implementation of redox-active gels to mediate electron transfer, thereby improving sensitivity, stability, and operational range for fermentation monitoring applications.
The Scientist's Toolkit: Research Reagent Solutions
The following table details essential materials and their functions for developing and fabricating biosensors based on nanostructured platinum and redox gels.
Table 1: Key Research Reagents and Their Functions in Biosensor Fabrication
| Reagent/Material | Function/Application | Key Characteristics |
|---|---|---|
| Glucose Oxidase (GOx) [3] [19] | Biorecognition element; catalyzes glucose oxidation | From Aspergillus niger; contains FAD coenzyme; high specificity and stability |
| Hexachloroplatinic Acid (H₂PtCl₆) [19] | Precursor for electrochemical deposition of platinum nanostructures | Source of Pt atoms; determines nanostructure morphology |
| 1,10-Phenanthroline-5,6-dione (PD) [19] | Redox mediator for electron shuttling between GOx and electrode | Quinoid compound; fast electron transfer kinetics; stable redox forms |
| Polyvinyl Alcohol with Styryl Pyridinium Groups (PVA-SbQ) [20] | Photo-crosslinkable polymer for enzyme entrapment | Forms a hydrophilic, stable matrix upon UV exposure; reduces enzyme leaching |
| Glutaraldehyde (GA) [20] | Crosslinking agent for covalent enzyme immobilization | Binds to amine groups on enzymes and carrier proteins like BSA |
| Polypyrrole (Ppy) [19] | Conducting polymer for creating protective, permselective membranes | Enzyme-mediated polymerization; protects electrode from interferents |
| meta-Phenylenediamine (m-PD) [20] | Monomer for electro-polymerization of interference-rejection membranes | Forms a semi-permeable poly(m-PD) layer; blocks ascorbic acid and other electroactive species |
Performance Characteristics of Advanced Material-Based Biosensors
The integration of nanostructured platinum and redox gels significantly enhances biosensor performance. The table below summarizes key quantitative metrics reported for various sensor configurations.
Table 2: Performance Comparison of Biosensors Utilizing Nanostructured Platinum and Redox Gels
| Sensor Configuration | Linear Range | Sensitivity | Detection Limit | Key Application Context |
|---|---|---|---|---|
| Graphite Rod/PtNS/PD/GOx [19] | Up to 16.5 mM | 10.1 µA/(mM·cm²) | 0.198 mM | Blood serum analysis |
| Graphite Rod/PtNS/PD/GOx/Ppy [19] | Up to 39.0 mM | 5.31 µA/(mM·cm²) | 0.561 mM | Blood serum analysis (improved interferent resistance) |
| Commercial Flow-Through (B.LV5) [3] [10] | Up to 150 mM | Not specified | Not specified | Yeast fermentation broth (cell-free and cell-containing samples) |
| Lactate Biosensor (Os-Polymer Mediated) [21] | 0.1–9 mM | 1.02 µA/mM | 0.05 mM | L-lactate determination |
| Pt@UiO66–NH₂ AChE Biosensor [22] | 1x10⁻¹⁴ – 1x10⁻⁹ M | Not specified | 4.9x10⁻¹⁵ M | Organophosphorus pesticide detection |
Experimental Protocols
Protocol 1: Fabrication of Nanostructured Platinum Microelectrodes via Electrodeposition
Objective: To create a microsensor array with nanostructured platinum (nano-Pt) electrodes for enhanced sensing performance in a multi-parameter chip for yeast fermentation monitoring [8].
Materials:
- Wafer-level microfabricated platinum microelectrodes
- Hexachloroplatinic acid (H₂PtCl₆) solution [19]
- Supporting electrolyte (e.g., potassium nitrate, sulfuric acid) [19]
- Potentiostat/Galvanostat
- Standard three-electrode setup (Pt working electrode, Pt counter electrode, Ag/AgCl reference electrode)
Procedure:
- Electrode Cleaning: Clean the fabricated platinum microelectrodes thoroughly with solvents and/or oxygen plasma to ensure a pristine surface.
- Electrodeposition Solution: Prepare an aqueous solution containing H₂PtCl₆ (e.g., 1-10 mM) in a suitable supporting electrolyte such as 0.5 M H₂SO₄ or 0.1 M KNO₃ [19].
- Electrodeposition: Immerse the electrode in the deposition solution within a standard three-electrode electrochemical cell.
- Apply a constant potential (e.g., -0.2 V vs. Ag/AgCl) or use a cyclic voltammetry technique (e.g., scanning between +0.60 V and -0.25 V) [19].
- The duration of deposition (from seconds to minutes) and the concentration of H₂PtCl₆ will control the morphology and density of the resulting nanostructures (e.g., clump-like aggregations, leaf-like flakes) [19].
- Rinsing and Drying: After deposition, rinse the electrode meticulously with deionized water and allow it to dry under a gentle stream of inert gas (e.g., N₂).
- Characterization: Characterize the nanostructured surface using techniques such as Scanning Electron Microscopy (SEM) to verify the desired nano-Pt morphology.
Protocol 2: Construction of a Redox Gel-Mediated Glucose Biosensor
Objective: To construct a reagentless glucose biosensor by immobilizing glucose oxidase within a redox-active hydrogel containing 1,10-phenanthroline-5,6-dione (PD) on a nanostructured platinum electrode [19].
Materials:
- Nanostructured Platinum electrode (from Protocol 1)
- Glucose Oxidase (GOx)
- 1,10-Phenanthroline-5,6-dione (PD)
- Polyvinyl Alcohol with Styryl Pyridinium Groups (PVA-SbQ) [20]
- Glutaraldehyde (GA) solution [20]
- Bovine Serum Albumin (BSA)
- Glycerol
- UV Light Source (365 nm)
Procedure:
- Redox Gel/Enzyme Mixture Preparation:
- Immobilization via Photocrosslinking:
- Mix the enzyme gel thoroughly with a photo-crosslinkable polymer solution, PVA-SbQ, to achieve final concentrations (e.g., 1.62 U/µL POx, 13.2% PVA-SbQ) [20].
- Apply a small volume (e.g., 0.15 µL) of the final mixture onto the surface of the nano-Pt electrode.
- Expose the coated electrode to UV light (365 nm) for a defined period (e.g., ~8 min, 2.4 J) to initiate crosslinking and form a rugged, stable hydrogel layer entrapping the enzyme and mediator [20].
- Post-Immobilization Treatment: Rinse the biosensor 2-3 times with working buffer (e.g., phosphate buffer, pH 7.4) for several minutes to remove any unbound molecules.
- Optional Polypyrrole Coating: To enhance resistance to interfering species, a polypyrrole (Ppy) layer can be synthesized on the sensor surface. This can be achieved enzymatically, using the H₂O₂ produced by the GOx reaction to initiate polymerization, forming a uniform protective membrane [19].
- Storage: Store the fabricated biosensors in a dry state at 4-8 °C when not in use [20].
Application in Yeast Fermentation Monitoring
Integrated Multi-Sensor Array for Fermentation
Advanced biosensor platforms have been developed specifically for yeast fermentation monitoring. One notable example is a multi-sensor array chip that integrates nano-Pt modified electrodes for parallel monitoring of pH, temperature, conductivity, dissolved oxygen (DO), and glucose [8]. This platform features all-solid-state on-chip reference electrodes, achieving true miniaturization and standalone operation without external reference electrodes. The nano-Pt modification is pivotal, providing highly sensitive and selective microsensors with minimal cross-talk, enabling reliable sensing at lower applied potentials [8].
On-Line Monitoring with Commercial Biosensor Platforms
Commercial platforms leveraging these material advances demonstrate direct application in fermentation. A flow-through-cell with integrated first-generation electrochemical glucose biosensors has been successfully applied for at-line and on-line monitoring during yeast fed-batch fermentation [3] [10]. This system reliably quantified glucose concentrations up to 150 mM in complex fermentation broth, both in cell-free and cell-containing samples, a significant improvement over previous biosensor limitations. The measurement is fast (<5 minutes), requires significantly fewer resources than HPLC analysis, and shows outstanding mechanical stability in direct contact with fermentation medium [3] [10].
The diagram below illustrates the typical workflow for integrating such a biosensor into a fermentation process for on-line monitoring.
Visualizing Biosensor Architecture and Working Principle
The functionality of these advanced biosensors relies on a coordinated process from molecular recognition to signal transduction. The following diagram illustrates the layered architecture of a combined nanostructured platinum and redox gel biosensor and its operational mechanism.
The integration of amperometric biosensors for online glucose monitoring in yeast fermentation represents a significant advancement in bioprocess control. These sensors provide real-time data critical for optimizing yield and understanding metabolic pathways. However, traditional biosensor fabrication methods often lack the scalability, customization, and integration capabilities required for modern, compact bioreactor systems. This application note details novel fabrication techniques, including 3D printing and microfabrication, which address these challenges by enabling the production of scalable, high-performance, and modular sensing platforms. These methods facilitate the direct integration of biosensors into microfluidic fermentation systems, allowing for precise online monitoring of glucose levels.
Advanced Fabrication Techniques for Biosensors
Multi-Material 3D Printing
Fused filament fabrication (FFF) 3D printing allows for the rapid prototyping of compact, multi-material electrochemical sensors. This technique can produce integrated devices capable of operating with sample volumes as low as 100 µL [23]. A key advantage is the ability to print both conductive and non-conductive components into a single, portable platform, accepting biological modifications like enzyme immobilization for glucose oxidase (GOx)-based sensing [23]. The process parameters, such as layer print orientation and layer thickness, significantly influence electrochemical performance and must be optimized for sensitivity [23].
Movable Type Bioelectronics Printing
Inspired by historical printing methods, this technique uses pre-fabricated master molds to directly transfer bioelectronic materials (e.g., conductive carbon paste, Ag/AgCl paste) onto various rigid or flexible substrates [24]. This approach is simple, low-cost, and suitable for small-scale production. It supports the direct transfer printing of bioactive materials, such as enzymes, alongside electronic components, simplifying the functionalization process and enabling the fabrication of multi-analyte sensors (e.g., for simultaneous glucose and lactate monitoring) on a single platform [24].
Microfabrication and Microfluidic Platform Integration
Microfabrication techniques, including photolithography and soft lithography, are used to create precise microfluidic platforms for bioprocessing [25]. These platforms can be integrated with sensors for real-time monitoring of key parameters like pH, temperature, and dissolved oxygen [25]. For biosensor integration within these systems, materials such as polydimethylsiloxane (PDMS) and polymethylmethacrylate (PMMA) are commonly used due to their optical transparency, biocompatibility, and ease of fabrication [25]. This integration is crucial for creating scaled-down, representative models of fermentation processes that provide a high density of data with minimal sample consumption.
Performance Comparison of Fabricated Glucose Biosensors
The table below summarizes the performance characteristics of glucose biosensors fabricated using different innovative techniques and material systems, highlighting their suitability for quantitative analysis.
Table 1: Performance Metrics of Novel Glucose Biosensors
| Fabrication Technique / Sensor Type | Linear Detection Range | Sensitivity | Detection Limit | Key Advantages |
|---|---|---|---|---|
| Multi-material 3D-printed platform [23] | 1 mM to 12 mM | Data not specified | Data not specified | Compact, operates with 100 µL sample volumes, integrated design |
| Enzyme-Nanozyme (PtCo/GOx) [4] | 0.04 mM to 2.18 mM | 19.38 µA mM⁻¹ cm⁻² | 0.021 mM | High sensitivity, excellent operational and storage stability |
| Chitosan/TiO₂ Nanotube Arrays [26] | 0.3 mM to 1.5 mM | 5.46 µA·mM⁻¹ | Data not specified | Good reproducibility (RSD = 2.5%), 85% activity after 30 days |
| Chitosan Membrane on Pt Electrode [27] | Up to 14.235 mM (Km) | Data not specified | 0.05 mM | Good stability (retained ~36% activity after 2 months), suitable for food analysis |
| MPTMS Sol-Gel Modified [28] | Up to 12.5 mM | Data not specified | Data not specified | Superior stability (~5 months), strong anti-interference ability |
Experimental Protocols
Protocol: Fabrication of a Multi-Material 3D-Printed Glucose Biosensor
This protocol describes the creation of an integrated, portable sensor for small-volume samples [23].
4.1.1 Materials and Equipment
- Multi-material FFF 3D printer
- Conductive (e.g., carbon-filled) and non-conductive (e.g., PLA) filaments
- Glucose oxidase (GOx) from Aspergillus niger
- Phosphate Buffered Saline (PBS), pH 7.4
- Glutaraldehyde ( Grade II, 25% aqueous solution)
- Potentiostat
4.1.2 Procedure
- Design and Printing: Design an integrated three-electrode system (working, reference, counter) within a sample well. Use CAD software to optimize the geometry for a 100 µL working volume. Print the device using conductive filament for the electrodes and non-conductive filament for the body and well.
- Surface Activation (Optional): Clean and activate the surface of the 3D-printed working electrode to improve enzyme adhesion. This may involve electrochemical pretreatment or mild plasma etching.
- Enzyme Immobilization: Prepare a solution of GOx (e.g., 15 mg in 500 µL PBS). Mix the enzyme solution with a crosslinking agent such as glutaraldehyde. Deposit the mixture onto the working electrode and allow it to crosslink and dry.
- Sensor Calibration: Connect the 3D-printed sensor to a potentiostat. Perform chronoamperometry at an applied potential of +0.6 V (vs. the integrated reference) in PBS. Add standard glucose solutions to achieve concentrations from 1 mM to 12 mM and record the steady-state current.
4.1.3 Data Analysis Plot the steady-state current against glucose concentration to generate a calibration curve. The sensor's performance can be assessed using cyclic voltammetry and differential pulse voltammetry in standard redox mediators like hexaammineruthenium(III) chloride to confirm electrochemical functionality before enzyme immobilization [23].
Figure 1: Workflow for fabricating a multi-material 3D-printed biosensor, from design to a ready-to-use integrated device.
Protocol: Movable Type Printing for a Flexible Dual-Channel Biosensor
This protocol outlines the use of movable type printing to create a flexible biosensor for simultaneous monitoring of glucose and lactate, which can be adapted for complex fermentation analyses [24].
4.2.1 Materials and Equipment
- Pre-fabricated molds (e.g., 3D-printed resin)
- Conductive carbon paste, Ag/AgCl paste
- Flexible substrate (e.g., Polyethylene Terephthalate (PET) or Polydimethylsiloxane (PDMS))
- Glucose oxidase (GOx), Lactate oxidase (LOx)
- Spin coater
- Ventilated oven
4.2.2 Procedure
- Master Mold Preparation: Design and fabricate molds for the electrode patterns (working, reference, counter) and, if needed, for enzyme deposition. Molds can be 3D-printed using a light-curing printer.
- Paste Preparation and Spin-Coating: Prepare electronic pastes. For bioactive inks, mix the enzyme (GOx or LOx) directly with a compatible paste or hydrogel. Spin-coat the pastes into thin layers on a flat surface.
- Transfer Printing: Press the pre-fabricated molds onto the thin layers of paste to pick up the material. Then, stamp the molds onto the target flexible substrate (e.g., PET film).
- Curing and Final Assembly: Dry the transferred patterns in a ventilated oven at 40°C. For a dual-channel sensor, ensure spatial separation of the glucose and lactate sensing electrodes.
4.2.3 Data Analysis The performance of the flexible electrodes should be characterized by measuring resistance changes under bending stress. For glucose sensing, amperometric measurements should be conducted at +0.4 V to +0.7 V vs. Ag/AgCl to detect the enzymatically generated H₂O₂ [24] [27].
The Scientist's Toolkit: Essential Research Reagents and Materials
Table 2: Key Reagent Solutions for Biosensor Fabrication and Use
| Item Name | Function / Application | Brief Explanation |
|---|---|---|
| Glucose Oxidase (GOx) | Biological Recognition Element | Enzyme that catalyzes the oxidation of glucose to gluconolactone and hydrogen peroxide, providing the basis for signal generation [26] [27]. |
| Chitosan | Enzyme Immobilization Matrix | A biocompatible hydrogel used to entrap and stabilize enzymes on the electrode surface, preserving their activity [26] [27]. |
| Nafion | Permselective Membrane | A perfluorinated polymer coating used to repel negatively charged interferents (e.g., ascorbic acid, uric acid), enhancing selectivity [4]. |
| Polydimethylsiloxane (PDMS) | Flexible Substrate / Microfluidics | An elastomeric polymer used for flexible sensor substrates and to fabricate microfluidic channels for sample delivery [24] [25]. |
| Glutaraldehyde | Crosslinking Agent | Used to create covalent bonds between enzyme molecules and the immobilization matrix, securing the biorecognition layer [27]. |
| Conductive Carbon Paste | Electrode Material | Forms the conductive trace and working electrode in printed sensors, providing the electrochemical interface [24]. |
| (3-Mercaptopropyl)trimethoxysilane (MPTMS) | Sol-Gel Membrane | Forms a porous, protective layer on the sensor surface, blocking interferents and improving long-term stability [28]. |
The adoption of 3D printing, movable type printing, and microfluidic integration marks a pivotal shift in the development of amperometric biosensors. These novel fabrication techniques directly address the critical need for scalability, customization, and seamless integration within modern yeast fermentation research systems. By providing detailed protocols and performance data, this application note equips researchers and drug development professionals with the tools to implement these advanced sensors, thereby enabling more precise and efficient online glucose monitoring to accelerate bioprocess optimization.
The transition from manual, offline sampling to automated, online monitoring is a cornerstone of modern bioprocess development, particularly for critical parameters like glucose in yeast fermentation. This application note delineates structured integration strategies—at-line, on-line, and in-line—for amperometric biosensors within bioreactor systems. We provide detailed protocols and performance data to guide researchers and scientists in drug development for implementing robust, real-time glucose monitoring to enhance process control, product yield, and overall efficiency.
In bioprocess manufacturing, state-of-the-art fermentation monitoring is often limited to standard measurements such as pH, temperature, and dissolved oxygen, with a notable gap in real-time substrate quantification [10]. Glucose, a major carbon and energy source, exerts a critical influence on the growth of cells and the yield of products; however, its monitoring often relies on time-consuming, offline methods like HPLC that preclude real-time control [29] [10]. Amperometric biosensors, which leverage the specificity of glucose oxidase (GOD), present a powerful solution. The effective integration of these sensors from at-line to true on-line configurations is pivotal for unlocking their potential in advanced process control and optimization, ultimately contributing to more consistent and economical production of biologics and recombinant proteins [30] [31].
Integration Configurations: Definitions and Comparisons
The strategy for connecting a biosensor to a bioreactor fundamentally defines its capabilities and limitations. The following configurations represent a progression toward seamless, real-time monitoring.
Table 1: Comparison of Biosensor Integration Configurations for Glucose Monitoring
| Configuration | Description | Key Advantages | Key Challenges |
|---|---|---|---|
| At-Line | Automated sampling with manual or automated sample transfer to a nearby analyzer. Sample may undergo pre-processing (e.g., dilution, filtration). | Reduced manual intervention; faster than offline analysis; lower contamination risk [29]. | Not truly real-time; time delay between sampling and result. |
| On-Line | Biosensor integrated into a bypass loop or flow-through cell. An automatic sampler continuously draws and may condition the sample. | Near real-time monitoring; allows for sample conditioning (e.g., dilution, filtration) [29] [10]. | System complexity; potential for tube clogging; time lag from bypass loop. |
| In-Line | Sensor placed directly within the bioreactor vessel, exposed to the full fermentation broth. | True real-time data; no time lag; no need for separate sampling unit [8]. | Risk of sensor fouling or drift; must withstand sterilization; difficult to calibrate or replace mid-run. |
The following workflow illustrates the decision-making path and components for implementing these configurations, from sample acquisition to data output for process control.
The Scientist's Toolkit: Essential Research Reagent Solutions
Successful implementation of amperometric glucose monitoring requires specific reagents and materials to ensure sensor sensitivity, selectivity, and stability.
Table 2: Key Research Reagents and Materials for Amperometric Glucose Biosensors
| Item | Function/Description | Application Example |
|---|---|---|
| Glucose Oxidase (GOD) | The core biorecognition element that catalyzes the oxidation of β-D-glucose, producing H₂O₂ [29]. | Enzyme immobilization on electrode surface for specific glucose detection. |
| Nanostructured Platinum (nano-Pt) | Electrocatalyst that enhances electron transfer, improves sensitivity, and enables reliable sensing at lower applied potentials [8]. | Modification of microelectrode surfaces in amperometric dissolved oxygen and glucose sensors. |
| Screen-Printed Electrodes (SPEs) | Disposable, cost-effective, and mass-producible electrode platforms. | Serve as a base for sensor modification with GOD, CNTs, and nanoparticles for at-line/on-line use [29]. |
| Carbon Nanotubes (CNTs) & Gold Nanoparticles (AuNPs) | Increase electrode surface area and conductivity, enhancing the current response and overall sensor performance [29]. | Used in a composite mixture with GOD to modify SPEs for improved signal output. |
| Chitosan | A cationic natural polymer that forms a hydrogel to entrap and stabilize enzymes and nanoparticles on the electrode surface [29]. | Used as a permselective membrane to immobilize the enzyme layer and potentially reject interferents. |
Performance Data from Current Technologies
Recent advancements in sensor design and integration have led to significant improvements in the analytical performance of biosensor platforms for fermentation monitoring.
Table 3: Performance Metrics of Representative Glucose Biosensor Systems
| Sensor Platform / Configuration | Linear Detection Range | Detection Limit | Key Features & Integration Method | Ref |
|---|---|---|---|---|
| Nanostructured Pt Microsensor Array | Not explicitly stated for glucose | Not explicitly stated for glucose | Integrated multi-sensor array (pH, DO, glucose, etc.); on-chip reference electrode; suitable for in-line monitoring in yeast fermentation. [8] | [8] |
| GOD–SWCNTs–AuNPs-modified SPE / Sequential Injection (SI) | Up to 3.8 mM | 0.3 mM | Sequential Injection (SI) system with in-line dilution; amperometric detection at +0.8 V; designed for in-line monitoring in cell culture. [29] | [29] |
| Commercial Flow-Through-Cell (1st Gen. GOD Biosensor) | Up to 150 mM (in fermentation broth) | Not specified | Multi-array design in a 1 μl flow-through-cell; used for at-line and on-line monitoring during yeast fed-batch fermentation; robust in complex broth. [10] | [10] |
Experimental Protocols
Protocol: Modification of Screen-Printed Electrodes (SPEs) for At-Line/On-Line Use
This protocol details the preparation of a sensitive glucose biosensor based on a modified SPE, suitable for integration into at-line or sequential injection systems [29].
Materials:
- Commercial carbon Screen-Printed Electrodes (SPEs)
- Glucose oxidase (GOD), 2.5 U per sensor
- Single-walled carbon nanotubes (SWCNTs)
- Gold nanoparticle (AuNP) suspension
- Chitosan solution (e.g., 0.5-1% w/v in dilute acetic acid)
- Phosphate Buffered Saline (PBS), pH 7.4
Procedure:
- Step 1: Preparation of Modification Mixture. Prepare a homogeneous mixture containing the SWCNTs, AuNPs, and 2.5 Units of GOD in a suitable buffer. The negatively charged nanomaterials and the enzyme will form a composite.
- Step 2: Electrode Modification. Deposit a small, precise volume (e.g., 2-5 μL) of the modification mixture onto the working electrode surface of the SPE.
- Step 3: Chitosan Immobilization. To entrap the composite and form a stable hydrogel matrix, cover the modified working electrode with a layer of chitosan solution. Allow it to dry at room temperature or under mild desiccation.
- Step 4: Curing and Storage. Let the sensor cure fully. Store the modified SPEs dry and refrigerated when not in use.
Calibration:
- Connect the modified SPE to a potentiostat.
- Using a flow cell or in a beaker, expose the sensor to a series of standard glucose solutions prepared in PBS or a simulated fermentation medium.
- Apply a constant potential of +0.8 V (vs. Ag/AgCl pseudo-reference) and record the chronoamperometric current.
- Plot the steady-state current against glucose concentration to generate a calibration curve.
Protocol: Integration of a Commercial Flow-Cell Biosensor for On-Line Monitoring
This protocol outlines the setup for a commercial biosensor platform in an on-line configuration for near real-time glucose monitoring during a fermentation process [10].
System Setup:
- Biosensor Unit: Install the commercial biosensor chip (e.g., a flow-through cell with integrated Pt working and reference electrodes pre-modified with GOD).
- Fluidic Path: Connect the bioreactor's harvest line or a sampling loop to the inlet of the biosensor flow cell using appropriate tubing. A peristaltic or syringe pump should be installed to provide a continuous or sequential flow of the sample.
- Data Acquisition: Connect the biosensor's potentiostat transmitter to a computer running the manufacturer's software (e.g., bioMON) for data acquisition and control.
- Sample Conditioning (Optional): For high glucose concentrations or problematic broths, incorporate an in-line dilution module or a filter between the bioreactor and the biosensor to prevent sensor saturation or fouling [29].
On-Line Operation:
- Sterilization: Ensure the biosensor flow cell and all connections upstream are sterilized (e.g., via autoclaving or chemical sterilants compatible with the sensor) prior to integration.
- Initiation: Start the fermentation process. Once stable conditions are reached, initiate the sample flow pump to direct the broth through the biosensor.
- Measurement: The potentiostat applies a constant potential (e.g., +450 mV vs. Ag/AgCl). The resulting H₂O₂ oxidation current is measured chronoamperometrically, correlated to glucose concentration, and displayed in real-time on the software dashboard.
- System Control: The real-time glucose data can be fed back into the bioreactor control system to automatically adjust nutrient feed pumps, enabling advanced fed-batch strategies.
The following diagram illustrates the typical components and workflow of such an integrated on-line system.
The strategic integration of amperometric biosensors, from at-line to true on-line configurations, is a transformative step toward achieving precise control over yeast fermentation and other critical bioprocesses. The protocols and data presented herein provide a practical framework for researchers to implement these advanced monitoring strategies. By enabling real-time, data-driven decisions, these systems significantly contribute to enhancing process consistency, maximizing product yields, and accelerating development timelines in pharmaceutical and biotechnology industries.
Overcoming Practical Challenges: Stability, Interference, and Sensor Longevity
Addressing the Oxygen Limitation Challenge in Dense Fermentation Broths
In the realm of industrial biotechnology, achieving and maintaining high cell densities in fermentation processes is a primary objective for maximizing the yield of target products, ranging from therapeutic proteins to biofuels. However, as cell density increases, so does the cellular demand for oxygen, a critical substrate for aerobic metabolism. This creates a significant process bottleneck: oxygen limitation. In dense fermentation broths, the oxygen transfer rate (OTR) from the gas phase to the liquid medium often cannot keep pace with the specific oxygen uptake rate (sOUR) of the microbial population, leading to suboptimal growth and productivity [32].
The saturation concentration of dissolved oxygen (DO) in aqueous media is inherently low, and oxygen's poor solubility in water (~7 mg/L at 35°C) makes it the first substrate to become limiting in an actively respiring culture. When cells are deprived of oxygen, metabolic pathways shift, leading to the production of undesirable by-products like ethanol in yeast fermentations or organic acids in bacterial systems. This not only reduces the yield of the target metabolite but also complicates downstream purification. Therefore, online monitoring of key parameters like glucose and dissolved oxygen is not merely beneficial but essential for implementing effective feeding strategies that prevent overflow metabolism and avoid the detrimental effects of oxygen limitation [10] [8].
This application note details protocols for integrating amperometric biosensor technology to monitor glucose levels online, providing a data-driven approach to manage feeding strategies and mitigate oxygen limitation in yeast fermentation research.
Online Monitoring Solution: Amperometric Glucose Biosensors
Biosensor Platform and Working Principle
A robust solution for online glucose monitoring is provided by commercial amperometric biosensor platforms. These systems typically consist of a flow-through-cell with integrated electrochemical biosensors, a potentiostat, and system control software [10]. The core of the biosensor is a multi-array design featuring platinum working electrodes, a counter electrode, and an Ag/AgCl pseudo-reference electrode. A key feature is the inclusion of blank (non-enzyme coated) electrodes, which allow for automatic correction of background current and signal drift, significantly enhancing measurement accuracy in complex media [10].
The fundamental working principle relies on the enzyme glucose oxidase (GOD). GOD catalyzes the oxidation of β-D-glucose to D-glucono-1,5-lactone and hydrogen peroxide (H₂O₂). The subsequent electrochemical detection is most commonly based on the oxidation of the liberated H₂O₂ at a defined anodic potential (typically +450 mV vs. Ag/AgCl) [10] [27]. The resulting current is directly proportional to the glucose concentration in the sample. This reaction sequence is summarized as follows:
- β-D-glucose + O₂ → D-glucono-1,5-lactone + H₂O₂ (catalyzed by GOD)
- H₂O₂ → O₂ + 2H⁺ + 2e⁻ (at the platinum anode)
The integration of this biosensor platform into a fermentation setup involves a flow-through cell connected via tubing to an at-line or in-line sampling port, a pump to drive the sample flow, and the associated electronic control unit [10].
Visualizing the Biosensor System and Process Workflow
The following diagram illustrates the integrated system for online fermentation monitoring and control, highlighting the biosensor's role in the feedback loop.
Figure 1: Online Glucose Monitoring and Control Workflow. The diagram shows the flow of information and materials from the bioreactor, through the amperometric biosensor, to the control system that adjusts process parameters and nutrient feeding.
Performance Specifications of the Biosensor Platform
The presented biosensor platform demonstrates performance characteristics that make it suitable for demanding fermentation environments. The following table summarizes key metrics as reported in the literature.
Table 1: Key Performance Metrics of an Amperometric Glucose Biosensor Platform for Fermentation Monitoring [10].
| Parameter | Specification | Significance for Fermentation |
|---|---|---|
| Linear Detection Range | Up to 150 mM (≈27 g/L) | Covers typical glucose concentrations in fermentation feeds and broths, eliminating the need for manual sample dilution. |
| Response Time | < 5 minutes | Enables near real-time process monitoring and rapid control interventions. |
| Sample Requirement | Cell-free and cell-containing samples | Offers flexibility in sampling methodology; can be used with an external cell-separation unit. |
| Operational pH Range | 5 - 9 | Compatible with the pH range of most yeast fermentation processes. |
| Comparable Method | HPLC with RI detector | Provides equivalent accuracy but with a significantly faster analysis time and less resource consumption. |
Experimental Protocols
Protocol 1: Determination of the Oxygen Mass Transfer Coefficient (kLa)
Principle: The oxygen mass transfer coefficient (kLa) is a critical parameter that defines the maximum oxygen transfer capacity of a bioreactor under specific operating conditions. It is measured using the static gassing-out method [32] [33]. This method involves first deoxygenating the medium by sparging with nitrogen and then monitoring the increase in dissolved oxygen as the liquid is re-aerated.
Procedure:
- Calibration: Calibrate the dissolved oxygen (DO) probe according to the manufacturer's instructions. Set the reading in oxygen-stripped liquid to 0% DO and in air-saturated liquid to 100% DO [32].
- Deoxygenation: Fill the bioreactor with the process medium or water at the desired working volume. Begin agitation and headspace aeration at the test conditions. Sparge nitrogen gas into the vessel until the DO concentration stabilizes near 0% [32].
- Re-aeration: Immediately switch the gas supply from nitrogen to compressed air. Ensure the gas flow rate and agitation speed are maintained constant [32].
- Data Logging: Record the DO concentration at frequent intervals (e.g., every 1-5 seconds) as it increases until it stabilizes at approximately 100% [32].
- Data Analysis: Plot the natural logarithm of the oxygen deficit, ln(C* - C), versus time, where C* is the saturation DO concentration (100%) and C is the measured DO concentration at time t. The kLa is the negative slope of the linear region of this plot, typically between 20% and 80% DO [32] [33]. The governing equation is:
-kLa * t = ln( (C* - C) / (C* - C₀) )where C₀ is the initial DO concentration at the start of re-aeration (time zero).
Protocol 2: Measurement of Specific Oxygen Uptake Rate (sOUR)
Principle: The specific oxygen uptake rate (sOUR) quantifies the oxygen consumption rate per cell, providing insight into the metabolic activity and oxygen demand of the culture. It is measured using the dynamic method [32].
Procedure:
- Culture Preparation: Inoculate and grow the yeast culture in the bioreactor under standard conditions.
- DO Probe Configuration: Ensure the DO probe is properly calibrated and functioning.
- Interruption of Aeration: During the mid-exponential growth phase, when cell density and DO concentration are stable, abruptly stop the air supply and agitation to halt oxygen transfer into the medium.
- Data Logging: Continuously record the declining DO concentration over time. The period of measurement should be brief (a few minutes) to avoid impacting cell viability due to anoxia.
- Data Analysis: Plot the DO concentration against time. The slope of the initial, linear decrease in DO is the oxygen uptake rate (OUR) in mg/L/h.
- Calculation of sOUR: Determine the viable cell density (X, in cells/L or g/L) from a sample taken just before the test. Calculate the sOUR using the formula:
sOUR (mg/cell/h) = OUR (mg/L/h) / X (cells/L)
Protocol 3: Integrating the Glucose Biosensor for Fed-Batch Control
Principle: This protocol outlines the steps for integrating a commercial glucose biosensor platform for at-line monitoring and control of glucose concentration in a yeast fed-batch fermentation, thereby preventing overflow metabolism and reducing oxygen demand [10].
Procedure:
- System Setup:
- Connect the biosensor's flow-through cell to the bioreactor's sampling port via sterilizable tubing.
- Install a peristaltic pump to draw samples from the bioreactor through the biosensor.
- If analyzing cell-free samples, integrate a continuous-flow centrifuge or a filtration probe between the bioreactor and the biosensor.
- Connect the biosensor's potentiostat output to the bioreactor's process control system [10].
- Biosensor Calibration: Prior to sterilization or connection, calibrate the biosensor using standard glucose solutions in a buffer that mimics the fermentation medium. A two-point calibration (e.g., zero and a concentration near the desired set-point) is often sufficient.
- Fermentation Operation:
- Start the fermentation in batch mode. The biosensor will track the initial glucose consumption.
- As the glucose concentration approaches a pre-defined low set-point (e.g., 0.5 g/L), initiate the fed-batch phase.
- The process control software uses the real-time glucose concentration from the biosensor to dynamically control a nutrient feed pump, maintaining the glucose level within a tight range (e.g., 0.1 - 0.5 g/L) [10].
- Validation: Periodically, take manual samples for offline analysis (e.g., HPLC) to validate the accuracy of the biosensor readings throughout the fermentation run.
The Scientist's Toolkit: Research Reagent Solutions
Successful implementation of the aforementioned protocols requires specific reagents and materials. The following table lists the key components and their functions.
Table 2: Essential Research Reagents and Materials for Oxygen Transfer and Biosensor Experiments.
| Item | Function / Application | Experimental Context |
|---|---|---|
| Dissolved Oxygen Probe (e.g., Broadley-James) | Measures the concentration of dissolved oxygen in the broth in real-time. | Essential for kLa and sOUR measurements (Protocols 1 & 2) and for general fermentation monitoring [32]. |
| Amperometric Glucose Biosensor Chip (e.g., B.LV5 from Jobst Technologies) | The core sensing element for selective, enzymatic detection of glucose. | Integrated into the flow-through system for online glucose monitoring (Protocol 3) [10]. |
| Glucose Oxidase (GOD) | The biological recognition element that provides high specificity for β-D-glucose. | The critical, active component of the glucose biosensor [27] [34]. |
| p-Benzoquinone (BQ) | A redox mediator that shuttles electrons from the enzyme's redox center to the electrode surface. | Can be used in biosensor designs to lower the operating potential and reduce interference from other electroactive species [1]. |
| Chitosan Membrane | A biocompatible polymer used for enzyme immobilization via cross-linking. | Serves as a support matrix for stabilizing and retaining Glucose Oxidase on the electrode surface [27]. |
| Glutaraldehyde | A cross-linking agent used to create covalent bonds between enzymes and polymer matrices. | Used to securely immobilize Glucose Oxidase onto the chitosan membrane, ensuring biosensor stability [27]. |
| Nafion Polymer | A charged perfluorosulfonic acid polymer coating applied to the electrode. | Used as a permselective membrane to repel anionic interferents (e.g., ascorbate, urate) found in fermentation broths, improving selectivity [34]. |
| Nutrogen Gas Supply | Used to strip oxygen from the medium to establish a 0% DO baseline. | Required for the kLa determination experiment (Protocol 1) [32]. |
Concluding Remarks
The integration of advanced online monitoring tools, particularly amperometric biosensors, provides a powerful strategy to address the persistent challenge of oxygen limitation in high-cell-density fermentations. By enabling real-time, data-driven control of the glucose feed, these systems allow researchers to maintain metabolic conditions that avoid excessive oxygen demand. Coupling this with a fundamental understanding of the bioreactor's oxygen transfer capacity (kLa) and the microorganism's metabolic demand (sOUR) creates a robust framework for process optimization and scale-up. The protocols and data presented herein offer a practical pathway for researchers in drug development and beyond to enhance the yield and efficiency of their yeast fermentation processes.
Strategies for Mitigating Electroactive Interferences and Biofouling
The deployment of amperometric biosensors for online glucose monitoring in yeast fermentation represents a significant advancement in bioprocess control. However, the complex fermentation broth presents two major challenges that can compromise sensor accuracy and longevity: electroactive interferences and biofouling [3] [35]. Electroactive interferences, such as ascorbate, urate, and various metabolites, are readily oxidized at the working electrode potential, generating non-specific currents that obscure the target glucose signal [35]. Biofouling, the accumulation of proteins, cells, and other biological material on the sensor surface, impedes analyte diffusion, reduces sensitivity, increases response time, and ultimately shortens the functional lifespan of the biosensor [36]. This application note details proven strategies and provides detailed protocols to mitigate these issues, enabling robust and reliable glucose monitoring in yeast fermentation processes.
Strategic Approaches and Comparative Analysis
Mitigation of Electroactive Interferences
Several strategic approaches have been developed to eliminate or correct for signals from electroactive interferents. The choice of strategy depends on the required detection range, sensor design, and application constraints.
Table 1: Strategies for Mitigating Electroactive Interferences in Glucose Biosensors
| Strategy | Mechanism | Key Advantages | Limitations |
|---|---|---|---|
| Permselective Membranes [37] [35] | Electrosynthesis of non-conducting polymers (e.g., poly(o-phenylenediamine), polyphenol) onto the transducer surface to form a size-exclusion or charge-selective barrier. | High selectivity; effective rejection of common anionic interferents (e.g., ascorbate, urate); thin films can be created [37]. | Can introduce additional diffusion resistance; may require optimization for different sensor geometries [35]. |
| Electrochemical Mediators [35] | Incorporation of a redox mediator (e.g., ferrocene, Prussian blue) to shuttle electrons between the enzyme and electrode, allowing operation at a lower detection potential. | Significantly reduces oxidation potential of interferents; improved signal-to-noise ratio [35]. | Potential for mediator leakage; requires long-term stability of the mediator [35]. |
| Differential Sensing [38] | Use of a dual-electrode system where one functionalized electrode measures glucose + interferents, and a blank electrode measures only interferents. The differential signal corresponds to glucose. | Actively corrects for background interference in real-time; highly effective in complex matrices like fermentation broth [38]. | Increases sensor fabrication complexity; requires precise electrode matching [38]. |
| Nanostructured Electrodes [8] | Modification of electrode surfaces with nanomaterials (e.g., nano-Pt, Au nanodendrites) to enhance electron transfer kinetics, enabling reliable sensing at lower applied potentials. | Reduces interferent oxidation by lowering working potential; increases sensitivity and surface area [8]. | Fabrication process can be complex; long-term stability of nanostructures must be evaluated [36]. |
Prevention and Management of Biofouling
Biofouling is a critical concern for long-term in-situ monitoring. Advanced materials and surface modifications are the primary defense mechanisms.
Table 2: Strategies for Mitigating Biofouling in Glucose Biosensors
| Strategy | Mechanism | Key Advantages | Limitations |
|---|---|---|---|
| Antifouling Nanocoatings [36] | Application of nanomaterials (e.g., graphene oxide, zwitterionic polymers, gold nanoparticles) that create a hydrophilic, low-adhesion surface barrier. | Inherently resists protein and cellular adhesion; can be combined with sensing functions [36]. | Requires uniform and stable coating; long-term performance in fermentation broth needs validation [36]. |
| Conductive Polymer Hydrogels [39] | Use of hydrogels (e.g., PEDOT) that facilitate charge transfer while creating a hydrated, bio-inert physical barrier. | Excellent biocompatibility; reduces non-specific adsorption; can be used for enzyme entrapment [39]. | May slow analyte diffusion; mechanical stability on flexible sensors can be a challenge [39]. |
| Non-Enzymatic Sensors (NEGS) [36] | Elimination of the enzyme layer altogether; relies on direct electrocatalytic oxidation of glucose on nanostructured electrode surfaces (e.g., Pt, CuO). | Avoids biofilm formation on the enzyme layer; inherently more stable due to lack of a biological component [36]. | Can suffer from poor specificity and interference from other sugars and electroactive species [36]. |
The following diagram illustrates the logical decision-making process for selecting the appropriate mitigation strategy based on the primary challenge and sensor design constraints.
Experimental Protocols
Protocol: Fabrication of an Interferent-Rejecting Glucose Biosensor with Electrosynthesized Permselective Membrane
This protocol details the creation of a glucose biosensor where enzyme immobilization and the application of a permselective membrane are achieved through all-electrochemically assisted procedures, ensuring high selectivity and spatial control [37].
3.1.1 Materials
- Transducer: Platinum (Pt) wire or disk electrode.
- Cleaning Solutions: Alumina slurry (various grades) and deionized water.
- Enzyme Immobilization Solution: Contains Glucose Oxidase (GOD, from Aspergillus niger), Bovine Serum Albumin (BSA, Fraction V), and Glutaraldehyde (GLU, 25% aqueous solution) [37].
- Polymerization Solution: 5 mM monomer solution (e.g., o-phenylenediamine) in a suitable buffer (e.g., pH 7.0 phosphate buffer) [37].
3.1.2 Procedure
- Electrode Pretreatment: Polish the Pt working electrode sequentially with 1.0, 0.3, and 0.05 μm alumina slurry on a microcloth. Rinse thoroughly with deionized water between each polish. Perform electrochemical cleaning via cyclic voltammetry (e.g., from -0.4 V to +0.6 V vs. Ag/AgCl) in 0.5 M H₂SO₄ until a stable voltammogram is obtained.
- Enzyme Immobilization via Electrophoretic Deposition (EPD):
- Prepare the EPD solution containing GOD (e.g., 1 mg/mL), BSA (e.g., 10 mg/mL), and a low concentration of GLU (e.g., 0.1% v/v) in a suitable buffer [37].
- Immerse the cleaned Pt electrode (as the anode) and a Pt counter electrode in the EPD solution.
- Apply a pulsed current or constant voltage (e.g., +2.5 V for 60 s) to initiate electrophoretic migration of the proteins towards the electrode, triggering in-situ co-crosslinking exclusively on the Pt surface [37].
- Rinse the modified electrode gently with buffer to remove loosely bound molecules.
- Electrosynthesis of the Permselective Membrane:
- Transfer the enzyme-modified electrode to a fresh electrochemical cell containing the polymerization solution (e.g., 5 mM o-phenylenediamine).
- Perform cyclic voltammetry (e.g., 10 cycles between 0.0 V and +0.8 V at a scan rate of 50 mV/s) to electrosynthesize a thin, non-conducting polymer film (e.g., poly(o-phenylenediamine)) over the immobilized enzyme layer [37].
- Rinse the finalized biosensor and store in a refrigerated phosphate buffer (e.g., 4°C, pH 7.4) when not in use.
3.1.3 Validation
- Characterize the biosensor using amperometry at +0.45 V vs. Ag/AgCl.
- Determine the linear range and sensitivity by successive additions of glucose standard solutions.
- Assess selectivity by challenging the sensor with common interferents (e.g., 0.1 mM ascorbic acid) and comparing the response to an equivalent glucose concentration.
Protocol: Implementing a Differential Microneedle Array for Anti-Interference Sensing
This protocol outlines the use of a differential sensing architecture to achieve high selectivity, a method that can be adapted for fermentation online monitoring systems [38].
3.2.1 Principle A dual-working-electrode system is employed. WE1 is functionalized with glucose oxidase (GOx), while WE2 is a non-enzymatic "blank" electrode. In a sample containing glucose and interferents:
- WE1 (GOx-modified): Current (I₁) = Signal from H₂O₂ (from glucose) + Signal from Interferents.
- WE2 (Blank): Current (I₂) = Signal from Interferents. The differential current (ΔI = I₁ - I₂) is directly proportional to the glucose concentration, effectively canceling out the common-mode interference [38].
3.2.2 Procedure
- Sensor Fabrication: Fabricate a sensor array with two identical working electrodes (WE1, WE2), a counter electrode, and a reference electrode.
- Nanostructuring: Electrodeposit a catalytic layer (e.g., Au nanodendrites) on both WE1 and WE2 to enhance sensitivity [38].
- Enzyme Immobilization: Functionalize only WE1 with GOx using a cross-linking method (e.g., with BSA and glutaraldehyde). WE2 undergoes the same procedure but without the addition of GOx.
- Coating: Apply a uniform outer membrane (e.g., a thin polyurethane layer) to both electrodes to provide biofouling resistance and regulate glucose diffusion [38].
The workflow for this differential sensor fabrication is detailed below.
The Scientist's Toolkit: Essential Research Reagents and Materials
Table 3: Key Reagent Solutions for Biosensor Fabrication and Mitigation
| Item | Function/Application | Examples & Notes |
|---|---|---|
| Glucose Oxidase (GOD) | Biological recognition element; catalyzes the oxidation of β-D-glucose, producing H₂O₂ [35]. | From Aspergillus niger; crucial for specificity. Check activity (units/mg) and stability at process pH [37]. |
| Bovine Serum Albumin (BSA) | Inert protein used as a scaffold for enzyme immobilization via co-crosslinking [37]. | Fraction V; used with glutaraldehyde to form a robust, immobilized enzyme layer on the electrode surface [37]. |
| Glutaraldehyde (GLU) | Crosslinking agent; forms covalent bonds between enzyme and BSA, stabilizing the biocomposite layer [37]. | 25% aqueous solution; handle with care. Concentration must be optimized to avoid excessive enzyme denaturation [37]. |
| Permselective Monomers | Precursors for electrosynthesized interference-rejecting membranes [37] [35]. | o-Phenylenediamine, Phenol; form non-conducting polymers (e.g., poly(o-phenylenediamine)) that block interferents [37]. |
| Electrochemical Mediators | Shuttle electrons from enzyme to electrode, enabling low-potential operation [35]. | Prussian Blue, Ferrocene derivatives; reduces interference by lowering the working potential [35]. |
| Antifouling Nanomaterials | Provide a bio-inert surface to resist non-specific adsorption of proteins and cells [36]. | Zwitterionic polymers, Graphene oxide, Gold nanoparticles; integrate into sensor coatings to enhance longevity [36]. |
| Nafion | A cation-exchange polymer; used as an inner membrane to repel anionic interferents like ascorbate and urate [35]. | Can be applied by drop-casting or dip-coating. Also helps to stabilize the enzyme layer [38]. |
Enzyme Immobilization Techniques for Improved Operational and Shelf-Life Stability
In the context of amperometric biosensors for online glucose monitoring during yeast fermentation, the stability of the biological recognition element is paramount. Enzyme-based biosensors, while offering high sensitivity and specificity, often face limitations in operational and shelf-life stability, which can hinder their application in prolonged bioprocess monitoring [40]. Enzyme immobilization techniques address these challenges by stabilizing the enzyme structure on the transducer surface, thereby enhancing biosensor performance, reusability, and longevity [40] [41]. This document details advanced immobilization strategies and their quantitative impact on biosensor stability, providing application notes and protocols tailored for research in yeast fermentation systems.
Core Immobilization Techniques: Mechanisms and Trade-offs
The method chosen for enzyme immobilization fundamentally affects the analytical performance of the resulting biosensor, including its sensitivity, stability, and susceptibility to interference. The following table summarizes the primary techniques, their mechanisms, advantages, and limitations.
Table 1: Comparison of Core Enzyme Immobilization Techniques.
| Immobilization Technique | Mechanism of Attachment | Key Advantages | Key Limitations |
|---|---|---|---|
| Adsorption [40] | Weak bonds (Van der Waals, electrostatic, hydrophobic) | Simple, inexpensive, less destructive to enzyme activity | Weak bonding; susceptible to changes in pH, temperature, and ionic strength |
| Covalent Bonding [40] | Formation of stable covalent bonds with enzyme side chains (e.g., lysine, cysteine) | Strong, stable complexes; superior uniformity and control | Can affect enzyme activity; requires large amounts of bioreagent |
| Entrapment [40] [42] | Enzyme physically confined within a polymeric matrix or membrane | High stability; minimization of enzyme leaching | Gel matrix can hinder substrate diffusion; low enzyme loading capacity |
| Cross-linking [40] | Intermolecular covalent bonds between enzymes using a linker (e.g., glutaraldehyde) | Highly strong and stable bonding; improved efficiency | Use of cross-linking reagents can lead to significant loss of enzyme activity |
The workflow for selecting and implementing an immobilization strategy involves several key decision points, as outlined below.
Quantitative Impact on Biosensor Performance
The choice of immobilization strategy and support material directly translates into measurable biosensor performance characteristics. Recent studies with glucose oxidase (GOx)-based amperometric biosensors highlight the stability achievements possible with advanced techniques.
Table 2: Operational and Shelf-Life Stability Performance of Selected Amperometric Glucose Biosensors.
| Immobilization Technique & Materials | Key Performance Metrics | Operational Stability | Shelf-Life Stability |
|---|---|---|---|
| Bimetallic Nanozyme (PtCo) on Graphite Rod [43] | Sensitivity: 19.38 μA mM⁻¹ cm⁻²LOD: 0.021 mM | Retained 98.93% of initial response after 26 cycles of detection | Retained 95.33% of initial response after 14 days |
| Enzyme Tethered to Electrospun Nanofibers [42] | - | Maintained 100% sensitivity for at least 8 weeks | - |
| GOx Cross-linked on Chitosan/Pt Electrode [27] | LOD: 0.05 mM | Good repeatability (RSD 2.30%) | Retained ~36% of initial activity after 2 months |
| GOx Polygel on MXene Nanosheets [44] | Sensitivity: 48.98 μA mM⁻¹ cm⁻²LOD: 3.1 μM | Current remained at 85.83% of initial value after 200 cycles | - |
Detailed Experimental Protocols
Protocol 1: Covalent Immobilization of Glucose Oxidase on Nanostructured Electrodes using Bifunctional Cross-linkers
This protocol is adapted from a study on a wearable biosensor with exceptional long-term sensitivity [42]. It details the tethering of GOx to nanofiber surfaces, a method that maximizes enzyme stability and activity retention.
Research Reagent Solutions:
- Electrode: Screen-printed electrode with a Prussian Blue layer.
- Support Material: Electrospun nanofibrous polycaprolactone (PCL) mat.
- Enzyme: Glucose Oxidase (GOx) from Aspergillus niger.
- Cross-linker: Bifunctional Poly(ethylene glycol)-hydrazide (PEG-hydrazide).
- Buffer: 0.1 M Phosphate Buffer Saline (PBS), pH 7.4.
Step-by-Step Procedure:
- Electrode Pre-conditioning: Clean the surface of the screen-printed Prussian Blue electrode by rinsing thoroughly with deionized water and dry under a gentle stream of nitrogen gas.
- Support Material Activation: If necessary, activate the surface of the electrospun PCL nanofibers with a plasma treatment to introduce reactive functional groups.
- Cross-linker Application: Apply a uniform layer of the bifunctional PEG-hydrazide solution onto the nanofiber mat. Incubate for 2 hours at room temperature to allow the cross-linker to anchor to the surface.
- Enzyme Coupling: Prepare a 10 mg/mL solution of GOx in 0.1 M PBS, pH 7.4. Deposit this solution onto the cross-linker-activated nanofiber mat. Allow the covalent coupling reaction to proceed for 12 hours at 4°C in a humidified chamber to prevent evaporation.
- Washing: Rinse the modified electrode thoroughly with PBS buffer to remove any physically adsorbed, non-covalently bound enzyme.
- Storage: The finished biosensor should be stored in PBS at 4°C when not in use.
Protocol 2: Entrapment of Viable Yeast Cells in a Polydopamine Biocompatible Coating
For applications beyond single enzymes, such as monitoring metabolic outputs in co-culture systems, whole-cell biosensors are valuable. This protocol describes the immobilization of Saccharomyces cerevisiae for bioelectrochemical sensing [45].
Research Reagent Solutions:
- Electrode: Carbon Screen-Printed Electrode (SPE).
- Biological Element: Viable Saccharomyces cerevisiae cells.
- Immobilization Matrix: Dopamine hydrochloride.
- Electrolyte: MOPS buffer (20 mM, pH 7.0).
- Carbon Source: Glucose, Glycerol, or Ethanol (50 mM).
- Redox Mediator: Potassium ferricyanide (K₃[Fe(CN)₆], 50 µM).
Step-by-Step Procedure:
- Cell Preparation: Grow S. cerevisiae overnight in YPD medium. Centrifuge the culture and resuspend the cell pellet in 20 mM MOPS buffer (pH 7.0) to remove extracellular debris.
- Polydopamine (PDA) Prepolymerization: Prepare a 10 mM dopamine hydrochloride solution in MOPS buffer. Mix this solution with the yeast cell suspension in a 1:1 ratio. Stir the mixture magnetically under aerobic conditions for 1 hour to initiate self-polymerization and form a homogeneous PDA-cell solution.
- Drop-Casting: Deposit 5 µL of the PDA-yeast suspension onto the surface of the carbon working electrode. Allow it to dry at a controlled temperature of 26 ± 1°C for 30 minutes.
- Electrochemical Polymerization: To enhance coating stability, perform electrochemical polymerization by running 20 cycles of Cyclic Voltammetry (CV) between -0.3 V and +0.5 V (vs. Ag pseudo-reference) at a scan rate of 20 mV/s.
- Biosensor Validation: Characterize the biosensor using Chronoamperometry in an electrolyte solution containing MOPS buffer, MgCl₂, a carbon source, and the K₃[Fe(CN)₆] redox mediator.
The entrapment process and the electron transfer pathway within the polydopamine coating are visualized below.
The Scientist's Toolkit: Essential Research Reagent Solutions
The following table catalogs key materials used in the featured protocols and the broader field, with explanations of their specific functions in enzyme immobilization and biosensor operation.
Table 3: Essential Research Reagents for Enzyme Immobilization in Biosensors.
| Reagent Category & Examples | Function in Biosensor Development |
|---|---|
| Enzymes & Biological Elements- Glucose Oxidase (GOx)- Saccharomyces cerevisiae (Viable Cells) | Serves as the primary biorecognition element. GOx specifically catalyzes glucose oxidation. Yeast cells act as robust, genetically tractable whole-cell sensors for metabolic monitoring [27] [45]. |
| Support Matrices & Nanomaterials- Chitosan Membrane- Electrospun Nanofibers (PCL)- Polydopamine (PDA) Coating | Provides a high-surface-area, biocompatible substrate for enzyme attachment. Enhances enzyme loading, stability, and can facilitate electron transfer. PDA forms strong covalent bonds with cell walls and facilitates extracellular electron transfer (EET) [27] [42] [45]. |
| Cross-linking Agents- Glutaraldehyde (GTA)- Bifunctional PEG-hydrazide | Creates stable covalent intermolecular bonds between enzymes (GTA) or between the enzyme and a support matrix (PEG-hydrazide), preventing leaching and improving mechanical stability [40] [42]. |
| Redox Mediators- Potassium Ferricyanide (K₃[Fe(CN)₆])- Ferrocene Derivatives | Acts as an electron shuttle in second-generation biosensors, transferring electrons from the enzyme's redox center to the electrode surface, thereby reducing the operating potential and minimizing interference [44] [45]. |
| Nanomaterials for Electrodes- Bimetallic PtCo Nanoparticles- Prussian Blue | PtCo nanozymes exhibit peroxidase-like activity, electrocatalyzing the reduction of enzymatically produced H₂O₂, which enhances sensitivity. Prussian Blue is an efficient electrocatalyst for H₂O₂ reduction, often used as an internal mediator [43] [42]. |
Optimizing Sensor Calibration and Maintenance for Long-Term Bioprocess Runs
The transition from discrete, offline analytical measurements to continuous, real-time monitoring is a central goal in modern bioprocess engineering, particularly for sensitive processes like yeast fermentation. Amperometric biosensors, which measure the current generated by the electrochemical reaction of a target analyte, have emerged as a cornerstone technology for this application, especially for tracking critical metabolites like glucose [46] [11]. These sensors combine high sensitivity and specificity with the potential for miniaturization and integration into bioreactor systems [8].
However, the deployment of these biosensors in long-term bioprocess runs presents significant challenges. Sensor drift, fouling, and the need for frequent recalibration can compromise data integrity and process control. This application note, framed within a thesis on online glucose monitoring for yeast fermentation research, details proven strategies and protocols to optimize sensor calibration and maintenance, thereby extending operational lifespan and ensuring reliable data throughout extended cultivation processes.
Key Challenges in Long-Term Biosensor Deployment
Maintaining sensor accuracy and stability over timescales relevant to industrial fermentation (often days to weeks) requires addressing several inherent issues:
- Biofouling: The accumulation of cells, proteins, and other biomolecules on the sensor surface physically blocks diffusion and degrades signal response [47].
- Foreign Body Response (FBR): In implantable or in-situ configurations, the host's immune response can encapsulate the sensor, isolating it from the analyte stream and leading to signal attenuation [47].
- Enzyme Degradation: The activity of the biological recognition element (e.g., Glucose Oxidase) inevitably decreases over time and under operational stress [48].
- Calibration Drift: Changes in temperature, pH, and the baseline sensor signal necessitate regular recalibration, which is disruptive to continuous processes [49].
Strategies for Calibration and Maintenance Optimization
Advanced Calibration Methodologies
Moving beyond traditional multi-point calibrations can significantly reduce operational overhead and improve data continuity.
Table 1: Comparison of Calibration Strategies for Long-Term Monitoring
| Calibration Strategy | Key Principle | Experimental Protocol | Advantages for Long-Term Runs |
|---|---|---|---|
| Calibration-Free (dOCP/dt) Potentiometric Sensing | Measures the time-derivative of the open-circuit potential, which is proportional to substrate concentration [50]. | 1. Apply a fixed potential step. 2. Record the subsequent open-circuit potential (OCP) transient. 3. Calculate the derivative (dOCP/dt). 4. Correlate the peak dOCP/dt value to analyte concentration via a one-time factory calibration. | Eliminates need for in-process recalibration; inherently resistant to drift from temperature and pH variations [50]. |
| Multivariate Calibration | Uses chemometric models (e.g., PLS, RBF-ANN) to correlate complex sensor data to analyte concentration [51]. | 1. Build a calibration model using a set of pre-analyzed samples with varying, known concentrations of target and interferents. 2. Deploy the model to deconvolute the sensor signal in real-time. | Compensates for signal interference and cross-talk in complex media; enhances selectivity [51]. |
The dOCP/dt method is particularly promising for fermentation monitoring. Its fundamental principle is that the rate of change in the electrode potential following a perturbation is directly related to the enzyme-catalyzed reaction rate, which in turn is governed by substrate concentration. This relationship remains stable even when environmental factors change, making it inherently robust [50].
Sensor Regeneration and Maintenance Techniques
To combat biofouling and signal decay, proactive regeneration of the sensor interface is essential. Recent advances have yielded several practical methods.
Table 2: Sensor Regeneration and Maintenance Techniques
| Technique | Mechanism | Detailed Protocol | Considerations |
|---|---|---|---|
| Chemical Re-functionalization | Complete removal of the bioreceptor layer and subsequent re-immobilization of fresh elements [49]. | 1. Cleaning: Flow acidic solution (e.g., H₂SO₄) over electrode under CV scanning. 2. Refunctionalization: Flow fresh EDC/NHS coupling agents, followed by bioreceptor (e.g., aptamer, enzyme) solution. 3. Stabilization: Rinse and condition in buffer. | Time-consuming (several hours); requires integrated microfluidics; not suitable for in-vivo use [49]. |
| Stimuli-Responsive Release | Uses external triggers (e.g., light, heat) to break non-covalent bonds between the analyte and bioreceptor [49]. | 1. Functionalize sensor with a stimuli-responsive bioreceptor (e.g., a specific aptamer). 2. After measurement, apply a controlled light pulse or thermal cycle. 3. Confirm signal return to baseline before next measurement. | Allows for rapid, in-situ regeneration; requires careful design of bioreceptors and integrated trigger systems. |
| Smart Biocompatible Coatings | Utilizes coatings that resist fouling or can be sacrificially removed to refresh the sensor surface [49] [47]. | 1. Coat sensor with a hydrogel (e.g., Chitosan) or Nafion film. 2. Post-measurement, a brief ethanol wash can remove the Nafion film and attached fouling agents. 3. Recoat the sensor for the next measurement cycle. | Can extend functional life beyond 3 weeks; sacrificial layer consumption must be managed [47]. |
Experimental Protocols for Validation
Protocol: Assessing Sensor Stability and Lifetime
This protocol is designed to characterize the long-term performance of an amperometric glucose biosensor under simulated fermentation conditions.
- Sensor Preparation: Fabricate glucose biosensors according to your standard method (e.g., immobilize Glucose Oxidase in a Chitosan hydrogel on a nanostructured Pt or TiO₂ nanotube array electrode) [8] [48].
- Initial Calibration: Perform a full calibration in standard buffer (e.g., 0.1 M PBS, pH 7.0) across the expected glucose concentration range (e.g., 0-5 g/L). Record sensitivity and linearity (R²).
- Accelerated Aging: Place sensors in a stirred bioreactor containing a defined yeast growth medium (e.g., YPD) or a synthetic lignocellulosic hydrolysate mimic [52]. Maintain at a controlled temperature (e.g., 30°C).
- Periodic Performance Check: At defined intervals (e.g., every 24 hours), remove sensors, rinse gently with buffer, and perform a single-point calibration check in a standard glucose solution.
- Data Analysis: Plot signal response versus time. The operational lifetime can be defined as the point where the signal degrades by more than 15% from its initial value [48].
Protocol: Implementing a Regeneration Cycle via Chemical Re-functionalization
This protocol details the steps for regenerating a biosensor using a microfluidic system, as might be used in an automated, in-line monitoring setup [49].
- System Setup: Integrate the biosensor with a microfluidic manifold controlled by automated valves and a potentiostat.
- Cleaning Phase:
- Flow 0.5 M H₂SO₄ over the electrode surface for 5-10 minutes while performing cyclic voltammetry (CV) scans (e.g., from -0.2 V to 0.6 V) to strip off immobilized molecules.
- Switch to a 1 mM K₃Fe(CN)₆ solution and continue CV scanning for another 5-10 minutes to further clean the surface.
- Re-functionalization Phase:
- Flow a solution containing EDC (400 mM) and NHS (100 mM) to activate the electrode surface for 30-60 minutes.
- Flush with buffer.
- Immobilize the fresh bioreceptor by flowing a solution of the enzyme (e.g., GOx) or aptamer for 60 minutes, allowing for covalent binding.
- Flush again to remove unbound molecules.
- Re-calibration & Validation: Perform a single-point calibration check to confirm the recovery of sensor sensitivity before reintroducing it to the bioprocess stream.
The Scientist's Toolkit: Essential Research Reagents and Materials
Table 3: Key Reagent Solutions for Biosensor Fabrication and Regeneration
| Item | Function / Application | Specific Example |
|---|---|---|
| Glucose Oxidase (GOx) | Primary biorecognition element for glucose sensing; catalyzes oxidation of glucose [48]. | Type VII from Aspergillus niger [48]. |
| Chitosan Hydrogel | Biocompatible polymer for enzyme immobilization; stabilizes enzyme and allows substrate diffusion [48]. | Low molecular weight Chitosan dissolved in 1% acetic acid [48]. |
| Nanostructured Electrodes | Increases electroactive surface area, enhancing sensitivity and lowering detection limits [8]. | Nanostructured Platinum (nano-Pt) [8] or TiO₂ Nanotube Arrays (TiO₂NTAs) [48]. |
| EDC & NHS | Cross-linking agents for covalent immobilization of biomolecules onto sensor surfaces [49]. | 1-Ethyl-3-(3-dimethylaminopropyl)carbodiimide (EDC) and N-Hydroxysuccinimide (NHS). |
| Simulated Fermentation Media | For validating sensor performance and robustness under realistic, complex conditions with potential interferents. | Synthetic lignocellulosic hydrolysate [52]. |
The successful implementation of amperometric biosensors for long-term bioprocess monitoring hinges on moving from a paradigm of passive use to one of active maintenance and intelligent calibration. The strategies outlined herein—including the adoption of calibration-free methods where possible, the scheduled application of regeneration protocols, and the use of robust, nanostructured materials—provide a roadmap for achieving this goal. By integrating these approaches, researchers can obtain high-fidelity, continuous data on critical process variables like glucose, ultimately leading to better control, higher yields, and more efficient yeast fermentation processes.
Assessing Analytical Performance: Validation, Benchmarking, and Real-World Efficacy
Within the broader scope of thesis research focused on developing an amperometric biosensor for online glucose monitoring in yeast fermentation, establishing a robust correlation with a recognized reference method is paramount. For quantitative analysis of small molecules like glucose in complex biological matrices, High-Performance Liquid Chromatography (HPLC) is often the gold standard [53] [3]. This application note details a standardized protocol for validating the performance of an amperometric glucose biosensor against HPLC, ensuring the generated data is accurate, reliable, and suitable for scientific and regulatory scrutiny.
The core principle involves a head-to-head comparison where multiple fermentation samples are analyzed simultaneously using the biosensor platform and the HPLC method. The resulting data set is then subjected to statistical analysis to quantify the agreement between the two methods, thereby validating the biosensor for its intended use as a rapid, online monitoring tool.
Experimental Design and Workflow
A successful correlation study requires careful planning, from sample preparation to data analysis. The following workflow outlines the key stages of the experimental process.
Correlation Study Workflow
The diagram below illustrates the integrated workflow for correlating biosensor measurements with HPLC analysis during a yeast fermentation process.
Sample Collection and Preparation
- Source: Samples should be collected from a yeast fed-batch fermentation process across its entire duration—lag, exponential, and stationary phases—to ensure a wide range of glucose concentrations are represented [3].
- Handling: Use an automated sampling system, if available, to ensure consistency. For the HPLC arm, immediately centrifuge samples (e.g., at 13,000 rpm for 5 minutes) and filter the supernatant through a 0.2 µm membrane to remove cells and particulates that could damage the HPLC column [3] [54]. The biosensor can often handle cell-containing samples, but consistency demands that the same preparation (i.e., cell-free filtrate) be used for both methods during correlation [3].
Detailed Experimental Protocols
Protocol 1: Glucose Quantification via Amperometric Biosensor
This protocol is adapted from studies employing commercial amperometric biosensors for fermentation monitoring [3].
Principle: The biosensor utilizes glucose oxidase (GOx) immobilized on a platinum working electrode. GOx catalyzes the oxidation of glucose, consuming oxygen and producing hydrogen peroxide. The subsequent electrochemical reduction of H₂O₂ at a specific applied potential (typically +0.4V to +0.7V vs. Ag/AgCl) generates a current proportional to the glucose concentration [3].
Procedure:
- Biosensor Setup: Connect the flow-through biosensor chip (e.g., B.LV5, Jobst Technologies) to a potentiostat and a peristaltic pump via appropriate tubing.
- Calibration: Generate a five-point calibration curve daily using standard glucose solutions in the fermentation base medium (e.g., 0, 2, 5, 10, 20 mM). This controls for matrix effects.
- System Suitability Test: Inject a mid-level calibration standard (e.g., 5 mM) in triplicate. The relative standard deviation (RSD) of the peak current must be ≤5% before proceeding.
- Sample Analysis: Pump the filtered fermentation sample through the biosensor cell. Record the steady-state current or the peak current if using a flow-injection mode.
- Data Recording: The measurement is typically complete within 5 minutes per sample [3]. Record the calculated glucose concentration based on the calibration curve.
Protocol 2: Glucose Quantification via HPLC (Reference Method)
This protocol follows standard HPLC practices for impurity and assay analysis, tailored for glucose quantification [53] [54].
Principle: Analyte separation is achieved based on hydrophilic interaction with the stationary phase. Glucose is detected using a Refractive Index (RI) detector, which is well-suited for carbohydrates lacking strong chromophores.
Procedure:
- HPLC Setup:
- Column: Rezex ROA-Organic Acid H+ (8%) or equivalent (e.g., Aminex HPX-87H)
- Mobile Phase: 5 mM H₂SO₄ in HPLC-grade water.
- Flow Rate: 0.6 mL/min.
- Column Temperature: 60 °C.
- Detector: Refractive Index (RI) detector, temperature stabilized.
- Injection Volume: 10 µL [3].
- Calibration: Prepare a six-point calibration curve from a glucose reference standard (e.g., 0.5, 1, 5, 10, 15, 20 mM).
- System Suitability Test (SST): Before sample analysis, inject a standard solution (e.g., 10 mM) six times. The RSD for the peak area must be ≤2.0% [54]. The asymmetry factor and plate number for the glucose peak should meet predefined criteria.
- Sample Analysis: Inject each filtered fermentation sample in duplicate.
- Data Recording: Integrate the glucose peak and calculate the concentration using the calibration curve. The total run time is approximately 15-30 minutes.
Data Analysis and Validation Parameters
The correlation between the biosensor (test method) and HPLC (reference method) is established through linear regression and statistical analysis of the paired results.
Key Validation Parameters and Acceptance Criteria
Table 1: Key Validation Parameters for Biosensor Correlation with HPLC
| Parameter | Description | Experimental Approach | Acceptance Criterion |
|---|---|---|---|
| Linearity & Range | The ability to obtain results directly proportional to analyte concentration. | Analyze at least 6 different concentrations in triplicate across the expected range (e.g., 1-150 mM [3]). | Correlation coefficient (r) > 0.999 [54]. |
| Accuracy | Closeness of agreement between the biosensor value and the HPLC value. | Use a minimum of 9 determinations over 3 concentration levels (e.g., 80%, 100%, 120% of a target level) [53]. | Mean recovery of 98–102% [54]. |
| Precision | The closeness of agreement among a series of measurements. | Repeatability: Analyze 6 replicates of the same sample. Intermediate Precision: Repeat on a different day with a different analyst. | RSD ≤ 2.0% for both repeatability and intermediate precision [54]. |
| Specificity | The ability to assess the analyte unequivocally in the presence of other components. | Analyze fermentation medium, placebo, and samples spiked with potential interferents (e.g., maltose, fructose). | No significant interference at the glucose retention time; recovery within 98-102%. |
Statistical Correlation of Biosensor vs. HPLC
A statistical summary of a typical correlation study is presented below, demonstrating the level of agreement required for validation.
Table 2: Exemplary Data from a Biosensor-HPLC Correlation Study
| Sample ID | Glucose Concentration by HPLC (mM) | Glucose Concentration by Biosensor (mM) | Relative Difference (%) |
|---|---|---|---|
| Ferm-S1 | 1.5 | 1.45 | -3.3 |
| Ferm-S2 | 5.1 | 5.2 | +2.0 |
| Ferm-S3 | 25.3 | 25.8 | +2.0 |
| Ferm-S4 | 50.5 | 51.5 | +2.0 |
| Ferm-S5 | 75.2 | 74.0 | -1.6 |
| Ferm-S6 | 102.0 | 105.0 | +2.9 |
| Ferm-S7 | 148.0 | 145.0 | -2.0 |
| Statistical Summary | |||
| Slope | 1.02 | ||
| Intercept (mM) | -0.15 | ||
| Correlation Coefficient (r) | 0.9995 | ||
| Mean Relative Difference | +0.3% |
The Scientist's Toolkit
Research Reagent Solutions
Table 3: Essential Materials and Reagents for Biosensor Validation
| Item | Function / Role | Specifications / Examples |
|---|---|---|
| Amperometric Biosensor Platform | Core sensing device for rapid glucose measurement. | Flow-through-cell with integrated Pt working electrode & immobilized Glucose Oxidase (GOx) [3]. |
| HPLC System with RI Detector | Reference method for accurate glucose quantification. | System equipped with a suitable column (e.g., Rezex ROA, Aminex HPX-87H) [3]. |
| Glucose Reference Standard | For calibration of both biosensor and HPLC. | High-purity (>99%) D-(+)-Glucose for preparation of primary standards. |
| Fermentation Base Medium | Matrix for calibration standards to simulate sample conditions. | e.g., Potato Dextrose Broth (PDB) or other defined yeast growth medium [18]. |
| S. cerevisiae Yeast Strain | Bioreactor organism for generating validation samples. | e.g., S. cerevisiae FNCC – 3049 [18]. |
| Membrane Filters | For clarification of samples prior to HPLC analysis. | 0.2 µm pore size, low protein binding [54]. |
This application note provides a detailed framework for validating an amperometric glucose biosensor against HPLC within a yeast fermentation research context. By adhering to the outlined experimental protocols, data analysis methods, and acceptance criteria, researchers can rigorously demonstrate that their biosensor is a precise, accurate, and reliable tool for real-time glucose monitoring. Successful validation paves the way for using the biosensor as an efficient, resource-conscious alternative to HPLC for online fermentation monitoring and control.
Effective monitoring of fermentation processes relies on the accurate and real-time quantification of key analytes, such as glucose. Amperometric biosensors, which combine the specificity of biological recognition with the sensitivity of electrochemical transducers, have emerged as powerful tools for this purpose, particularly in the context of online monitoring during yeast fermentation [3]. For these biosensors to be effectively integrated into bioprocess control systems, a rigorous evaluation of their key performance metrics is essential. This application note details the critical analytical parameters—sensitivity, linear range, limit of detection (LOD), and limit of quantification (LOQ)—within the complex matrix of fermentation media. The protocols and data presented herein are designed to support researchers in the development and validation of robust biosensing platforms for advanced fermentation research.
The core principle of amperometric glucose biosensors is often based on the glucose oxidase (GOx) enzyme, which catalyzes the oxidation of glucose to gluconic acid and hydrogen peroxide. The subsequent electrochemical detection of the consumed oxygen or the generated peroxide provides a quantifiable signal proportional to the glucose concentration [26] [3]. However, the transition from simple buffer solutions to the complex, protein-rich, and dynamically changing environment of a fermentation broth presents significant challenges, including potential fouling, interference from electroactive species, and variations in pH and ionic strength [3] [55]. Therefore, demonstrating performance in a relevant matrix is a prerequisite for reliable online monitoring.
Performance Metrics for Fermentation Monitoring
Key analytical parameters must be thoroughly assessed to validate biosensor performance. The table below summarizes typical target values and the considerations for each metric in the context of fermentation media.
Table 1: Key Performance Metrics for Glucose Biosensors in Fermentation Applications
| Metric | Definition | Target Value / Typical Range for Fermentation | Importance in Fermentation Context |
|---|---|---|---|
| Sensitivity | The change in current output per unit change in glucose concentration (e.g., μA/mM). | Values from 5.46 μA·mM⁻¹ and higher have been reported [26]. | High sensitivity ensures that small changes in glucose concentration, critical for feed control, can be reliably detected. |
| Linear Range | The concentration range over which the sensor's response is linearly proportional to the analyte concentration. | Should cover expected process concentrations; demonstrated up to 150 mM in fermentation broth [3]. | Covers the typical glucose levels from initial batch to controlled feeding phases, avoiding sample dilution. |
| LOD (Limit of Detection) | The lowest analyte concentration that can be reliably detected. | Typically three times the signal-to-noise ratio (S/N). | Ensures the biosensor can signal the depletion of glucose, a crucial metabolic event. |
| LOQ (Limit of Quantification) | The lowest analyte concentration that can be quantitatively determined with stated accuracy and precision. | Typically ten times the signal-to-noise ratio (S/N). | Provides the lower limit for reliable quantitative measurements during the final stages of feeding. |
Beyond these core metrics, a biosensor intended for fermentation monitoring must demonstrate strong repeatability (RSD < 2%), reproducibility (RSD < 2.5%), and long-term stability (e.g., retaining >85% sensitivity after 30 days) to be considered a viable analytical tool [26] [55]. Furthermore, robustness against common interferents in fermentation media (e.g., fructose, ascorbic acid, citric acid) is essential for accurate quantification [26].
Experimental Protocols
This section provides detailed methodologies for characterizing biosensor performance and applying the sensor in a fermentation environment.
Protocol 1: Characterization of Key Performance Metrics
Objective: To determine the sensitivity, linear range, LOD, and LOQ of an amperometric glucose biosensor in a cell-free fermentation-mimicking medium.
Materials:
- Biosensor: Amperometric glucose biosensor (e.g., integrated into a flow-cell or as a rotating disk electrode) [3].
- Potentiostat: To apply potential and measure current.
- Reference Electrode: Ag/AgCl/3 M KCl.
- Counter Electrode: Platinum foil.
- Electrolyte: 0.1 M Phosphate Buffered Saline (PBS), pH 7.0.
- Glucose Standard Solutions: Prepared in 0.1 M PBS at concentrations from 0.1 mM to 200 mM.
- Fermentation Media Mimic: A cell-free, spent, or synthetic fermentation broth to evaluate matrix effects.
Procedure:
- Biosensor Preparation: If newly fabricated, hydrate the biosensor in 0.1 M PBS (pH 7.0) for at least 30 minutes. For a flow-cell system, ensure a stable flow rate (e.g., using a peristaltic pump) [3].
- Electrochemical Setup: Set up a standard three-electrode system. Apply a constant detection potential of -0.4 V vs. Ag/AgCl/3 M KCl to measure the reduction of oxygen or other electroactive products [26] [55].
- Calibration in Buffer:
- With continuous stirring or flow, record a stable baseline current in 100 mL of 0.1 M PBS.
- Sequentially add known volumes of concentrated glucose standard to achieve a range of concentrations (e.g., 0.3, 0.5, 0.8, 1.0, 1.5 mM for a lower range, or up to 150 mM for an extended range).
- After each addition, allow the current to stabilize and record the steady-state value.
- Calibration in Fermentation Mimic:
- Repeat step 3, but using the cell-free fermentation media mimic as the electrolyte. This is critical for assessing matrix effects.
- Data Analysis:
- Plot the steady-state current (or the change in current) against the corresponding glucose concentration.
- Perform a linear regression on the data points within the linear portion of the curve.
- Sensitivity is the slope of the linear calibration curve (e.g., in μA·mM⁻¹).
- Linear Range is the concentration interval over which the R² value of the linear fit is >0.99.
- LOD is calculated as 3.3 × (Standard Deviation of the blank response) / Sensitivity.
- LOQ is calculated as 10 × (Standard Deviation of the blank response) / Sensitivity.
Protocol 2: Online Monitoring in Yeast Fed-Batch Fermentation
Objective: To integrate a flow-through biosensor platform for the quasi-online monitoring of glucose during a yeast fermentation process.
Materials:
- Bioreactor: Equipped with standard probes (pH, dissolved oxygen, temperature).
- Automated Biosensor Platform: Consisting of a biosensor chip (e.g., B.LV5 with integrated Pt-working and reference electrodes), a potentiostat (e.g., SIX transmitter), and control software [3].
- Peristaltic Pump & Tubing: For automated sample delivery.
- Cell Separation Unit (Optional): An inline filter or microfluidic separator to provide cell-free sample streams for the biosensor.
Procedure:
- System Integration: Connect the outlet of the bioreactor, via the peristaltic pump (and optional cell-separation unit), to the inlet of the biosensor flow-cell. The outlet of the flow-cell can be routed to a waste container or back to the bioreactor.
- Biosensor Calibration: Before fermentation, perform an initial calibration of the biosensor platform using standard glucose solutions in a simulated matrix as described in Protocol 1.
- Fermentation Operation: Inoculate and run the yeast fed-batch fermentation according to the established experimental protocol.
- Online Sampling & Measurement:
- Initiate the peristaltic pump to draw broth from the bioreactor through the biosensor cell at defined intervals (e.g., every 5 minutes).
- The potentiostat and software record the amperometric signal in real-time, converting it to a glucose concentration based on the pre-determined calibration.
- Data Validation:
- Periodically, collect manual samples from the bioreactor for offline analysis using a reference method such as HPLC.
- Compare the glucose concentrations obtained from the biosensor with those from the HPLC to validate the sensor's accuracy and reliability throughout the fermentation run [3].
The workflow for this online monitoring setup is summarized in the following diagram:
The Scientist's Toolkit: Essential Research Reagents & Materials
The following table lists key reagents and materials crucial for developing and applying amperometric glucose biosensors in a fermentation context.
Table 2: Essential Research Reagents and Materials
| Item | Function / Role | Specific Example / Note |
|---|---|---|
| Glucose Oxidase (GOx) | Biological recognition element; catalyzes glucose oxidation. | Type VII from Aspergillus niger; ensure high specific activity (>100 units/mg) [26]. |
| Titanium Dioxide Nanotube Arrays (TiO₂NTAs) | High-surface-area electrochemical interface; enhances sensitivity. | Fabricated by anodic oxidation of Ti substrate [26] [55]. |
| Chitosan | Biocompatible hydrogel; used for enzyme immobilization and as a protective membrane. | Stabilizes the enzyme and can reduce fouling [26]. |
| Pentafluorophenyl Methacrylate (PFM) | Anchor for covalent enzyme immobilization; forms stable amide bonds. | Grafted via plasma polymerization to prevent enzyme leakage [55]. |
| Nanostructured Platinum (nano-Pt) | Electrode modification to enhance sensitivity and reduce working potential. | Electrodeposited on microelectrodes to minimize electrical cross-talk in sensor arrays [8]. |
| Flow-Cell Biosensor Platform | Integrated system for online, continuous monitoring. | Commercial platforms (e.g., B.LV5 chip) exist, featuring microfluidic channels and integrated electrodes [3]. |
The rigorous characterization of sensitivity, linear range, LOD, and LOQ directly within fermentation media is a critical step in transitioning amperometric biosensors from laboratory curiosities to reliable tools for bioprocess monitoring. The protocols outlined here provide a framework for this validation. By leveraging robust sensor designs, such as those incorporating nanostructured interfaces and stable immobilization strategies, researchers can achieve the performance necessary for real-time, online glucose monitoring. This capability is a cornerstone for advancing toward more efficient, data-driven, and controlled fermentation processes in both academic and industrial settings.
Comparative Analysis of Commercial Biosensor Platforms for Bioprocess Use
The global large and small-scale bioprocessing market, pivotal to manufacturing advanced biotherapies and products, is projected to grow from USD 80.75 billion in 2024 to USD 248.12 billion by 2034, demonstrating a compound annual growth rate (CAGR) of 11.88% from 2025 to 2034 [56]. This expansion is driven by the critical role of bioprocessing in producing vaccines, cell therapies, biofuels, and food ingredients. Despite this growth, a significant monitoring challenge persists in fermentation processes, which are core to bio-manufacturing. State-of-the-art fermentation on-line monitoring remains largely limited to basic parameters such as pH, temperature, and dissolved oxygen, while reliable, continuous quantification of key substrates like glucose is not yet widely implemented [3] [10]. This monitoring gap often forces operators to rely on manual manipulations based on experience rather than real-time process data, leading to suboptimal reactor efficiency and productivity.
Automated biosensor platforms present a transformative solution for real-time glucose quantification, offering a less resource and time-consuming alternative to conventional analytical methods like High-Performance Liquid Chromatography (HPLC) [3]. The integration of such automation in bioprocessing provides enhanced process control, improved product quality, reduced variability, increased throughput, and significant cost savings [56]. Although sophisticated biosensor solutions are commercially available and have been validated in complex biological matrices like blood, their application for monitoring and controlling fermentation processes has been relatively overlooked outside of clinical diagnostics [10]. This application note provides a comparative analysis of available commercial biosensor platforms, focusing on their implementation for online glucose monitoring within yeast fermentation research.
The biosensor market is experiencing dynamic growth across multiple segments, influenced by technological advancements and shifting end-user demands. The table below summarizes the key drivers and restraints impacting the market's CAGR, along with a segmentation analysis.
Table 1: Biosensor Market Dynamics and Segment Analysis
| Aspect | Detail | Impact / Market Share |
|---|---|---|
| Key Market Drivers | Growing demand for point-of-care diagnostics | +2.1% CAGR [57] |
| Integration of AI and IoT for real-time analytics | +1.8% CAGR [57] | |
| Advancements in nanomaterials for sensitivity | +1.2% CAGR [57] | |
| Key Market Restraints | High development and validation costs | -1.4% CAGR [57] |
| Stringent regulatory approval processes | -1.1% CAGR [57] | |
| Technology Segmentation (2024) | Electrochemical Biosensors | 70.21% share [57] |
| Optical Biosensors | Advancing at 10.67% CAGR [57] | |
| End-User Segmentation (2024) | Point-of-Care Testing | 57.43% of demand [57] |
| Home Healthcare | Projected 11.45% CAGR [57] |
North America dominated the global bioprocessing market in 2024, but the Asia-Pacific region is expected to show the fastest growth over the forecast period [56]. A significant trend in the industry is the growing outsourcing of biomanufacturing processes to Contract Development and Manufacturing Organizations (CDMOs), which allows biopharmaceutical companies to leverage specialized expertise, modernize production, reduce expenses, and enhance quality [56].
Comparative Analysis of Commercial Biosensor Platforms
Several commercial platforms have been developed to address the need for rapid, multi-component analysis in bioprocesses. The following table provides a structured comparison of biosensor technologies and platforms relevant to bioprocess monitoring, particularly for glucose in fermentation.
Table 2: Commercial Biosensor Platforms for Bioprocess Monitoring
| Platform/Technology | Technology Type | Key Features | Reported Performance in Fermentation | Primary Application Context |
|---|---|---|---|---|
| B.LV5 with SIX Transmitter [3] [10] | Electrochemical (Amperometric) | Flow-through-cell (1 μl), multi-array Pt electrodes, operational pH 5–9, +450 mV vs. Ag/AgCl. | Glucose detection up to 150 mM in fermentation broth; measurement in <5 min; outstanding mechanical stability. | Direct at-line and on-line fermentation monitoring (yeast fed-batch demonstrated). |
| ProcessTRACE [10] | Not Specified (Biosensor-based) | Integrated auto-sampling unit with cell-separation. | Enabled glucose monitoring & feed control in long-term fermentations (~600 h) without notable biosensor activity decrease. | Quasi-on-line monitoring on cell-free samples. |
| Cedex Bio (Roche) [10] | Not Specified (Biosensor-based) | Fully automated system for multicomponent analysis. | Rapid quantitative analysis, significantly reducing measurement time and operational errors. | Automated multi-component analysis. |
| YSI Biochemical Analyzer [10] | Not Specified (Biosensor-based) | Fully automated system for multicomponent analysis. | Rapid quantitative analysis, significantly reducing measurement time and operational errors. | Automated multi-component analysis. |
| OmicSense [58] | Computational (Software) | Algorithm using mixture of Gaussian distributions; processes whole omics data (e.g., transcriptome). | High prediction performance (r > 0.8) in benchmark tests; robust against noise and overfitting. | Uses omics data as a "biosensor" to predict environmental/physiological status. |
A critical innovation demonstrated by the B.LV5 biosensor platform is its extended detection range of up to 150 mM glucose, which is four to six-fold higher than the 25-33 mM range typical of many biosensors designed for clinical blood glucose monitoring [3] [10]. This expanded range is essential for addressing the higher glucose concentrations often present in fermentation broths. The platform's multi-array design, which includes two enzyme-coated working electrodes and two blank electrodes, allows for automatic duplicate measurements and subtraction of background current from electroactive interfering species present in the complex fermentation medium [10]. This design is a key factor in its accurate performance in both cell-free and cell-containing samples.
Detailed Experimental Protocol for Online Glucose Monitoring in Yeast Fermentation
This protocol outlines the methodology for integrating an amperometric glucose biosensor platform for at-line and on-line monitoring of a yeast fed-batch fermentation process, based on a validated experimental design [3] [10].
Materials and Equipment
Table 3: Research Reagent Solutions and Essential Materials
| Item | Function/Application | Specifications/Notes |
|---|---|---|
| Biosensor Chip B.LV5 [3] [10] | Electrochemical detection of glucose. | Extended range sensor; flow-through-cell (1 μl); pH range 5–9. |
| SIX Transmitter [3] [10] | Potentiostat. | Applies +450 mV vs. Ag/AgCl; customized connection for biosensor. |
| bioMON Software [10] | System operation and data acquisition. | For operating the biosensor platform. |
| Peristaltic Pump [10] | Facilitates sample flow. | e.g., Ismatec Reglo ICC. |
| Tubing [10] | Sample conveyance. | e.g., TYGON S3TM, ID 1.52 mm. |
| Fermentation Broth | Process medium. | Complex matrix containing yeast cells, nutrients, and metabolites. |
| Glucose Standards | Calibration. | For constructing a standard curve in relevant medium. |
| HPLC System with RI Detector [3] | Reference measurement. | Validates biosensor accuracy; resource and time-intensive. |
Procedure
Step 1: System Setup and Integration
- Connect the biosensor chip B.LV5 to the SIX transmitter.
- Integrate the biosensor into the fermentation setup using luer fittings. Connect the inlet of the biosensor's flow-through-cell to a peristaltic pump via appropriate tubing.
- For on-line monitoring, connect the pump to a sample stream from the bioreactor. For at-line monitoring, the pump can draw from a sample collection vial.
- Ensure the bioMON software is installed and communicating with the SIX transmitter for instrument control and data recording.
Step 2: Calibration
- Prepare a series of glucose standards in a matrix that closely resembles the fermentation medium (e.g., cell-free media or buffer at a similar pH).
- Pump each standard through the biosensor cell sequentially.
- The chronoamperometric current is measured at +450 mV vs. the internal Ag/AgCl pseudo-reference electrode. The bioMON software will record the steady-state current for each standard.
- Generate a calibration curve by plotting the recorded current (after blank subtraction) against the known glucose concentrations.
Step 3: Fermentation Monitoring
- At-line: Manually collect samples from the bioreactor at predetermined time points and immediately pump them through the biosensor system. The result is obtained in less than 5 minutes [3].
- On-line: Continuously pump a small side stream from the bioreactor through the biosensor flow-cell. The system provides near-real-time glucose concentration data, enabling immediate process control decisions.
- The biosensor's multi-array design automatically measures each sample in duplicate using two working electrodes. The current from blank (non-enzyme coated) electrodes is subtracted to correct for signals from electroactive interferents in the broth [10].
Step 4: Data Validation and Analysis
- Periodically validate the biosensor's readings against a reference method, such as HPLC analysis with a refractive index (RI) detector [3].
- Use the software to track glucose concentration trends over time, which can be used to inform feeding strategies for optimizing biomass yield and metabolite production.
Critical Considerations
- Oxygen Limitation: The performance of the 1st generation glucose biosensor (which uses glucose oxidase) can be compromised if oxygen becomes limiting in the sample stream, as oxygen is a co-substrate in the enzymatic reaction [3].
- Sterility: When configured for on-line monitoring, ensure the sampling system is sterilized and maintained to prevent microbial contamination of the bioreactor.
- Matrix Effects: While the biosensor demonstrates good performance in complex broth, calibration in a matrix similar to the sample is crucial for accuracy.
Visualization of Workflows and Principles
Biosensor-Integrated Fermentation Workflow
Diagram Title: Integrated Biosensor Feedback Control for Fermentation
Amperometric Biosensor Working Principle
Diagram Title: Amperometric Glucose Biosensor Detection Principle
The integration of commercial amperometric biosensor platforms, such as the flow-through-cell system described, directly addresses a critical bottleneck in yeast fermentation research and industrial bioprocessing: the lack of real-time, robust glucose monitoring. These platforms offer a compelling combination of performance—with an extended detection range suitable for fermentation broth, rapid analysis sub-5 minutes, and robust operation in complex media—and practical advantages like compact size and simplified operation compared to traditional analytical methods. As the bioprocessing market continues its significant growth and embraces further automation, the adoption of such advanced monitoring tools will be instrumental in maximizing productivity, ensuring product quality, and achieving the integrated process efficiency required for the next generation of biotherapies and bio-based products.
Within biomanufacturing, the cultivation of microorganisms in bioreactors is a core unit operation. Fed-batch processes, which start as a batch and are subsequently supplemented with feed nutrients, are a benchmark for efficient production, prolonging the growth phase and improving productivity [59]. However, a significant challenge in these processes is the lack of real-time monitoring for critical parameters like substrate concentration.
For yeast fermentations, glucose is a major carbon and energy source. Its concentration directly impacts biomass production and the formation of desired metabolites. Traditional methods for glucose quantification, such as High-Performance Liquid Chromatography (HPLC), are resource-intensive and time-consuming, making them unsuitable for prompt process control [3] [10]. This case study details the successful at-line and on-line application of a commercial amperometric biosensor platform for glucose monitoring during a fed-batch yeast fermentation. The integration of this technology provides a path toward intelligent, data-driven biomanufacturing by enabling real-time access to a critical process variable [60].
Experimental Protocol
Biosensor Platform and Working Principle
The core of the monitoring system is a commercial, miniaturized biosensor platform based on a 1st generation amperometric glucose biosensor [3].
- Biosensor Chip: B.LV5 flow-through-cell (Jobst Technologies GmbH – an IST AG company), featuring an electrochemical cell with two Pt-working electrodes (coated with glucose oxidase, GOx), two blank Pt-working electrodes, one Pt-counter electrode, and an internal Ag/AgCl pseudo-reference electrode [3] [10].
- Potentiostat: SIX transmitter for applying potential and measuring current.
- Software: bioMON for system operation.
- Peristaltic Pump: To drive the sample stream through the biosensor flow cell.
Working Principle: The biosensor operates on the principle of amperometry. Glucose oxidase catalyzes the oxidation of glucose, consuming oxygen and producing hydrogen peroxide (H₂O₂). A potential of +450 mV vs. Ag/AgCl is applied, which oxidizes the generated H₂O₂. The resulting current is proportional to the glucose concentration in the sample. The multi-array design allows for duplicate measurements and the use of blank electrodes corrects for signals from interfering electroactive species present in the complex fermentation broth [3] [10].
Integration with Fed-Batch Fermentation
The biosensor platform was adapted for both at-line and on-line monitoring of a yeast fed-batch fermentation process.
- At-Line Monitoring: Manual samples were taken from the bioreactor and injected into the biosensor flow path for immediate analysis.
- On-Line Monitoring: An automated sampling system was connected to the bioreactor, allowing for continuous or frequent intermittent sampling and analysis. The flow-through-cell design facilitated this direct integration [3].
Procedure:
- System Calibration: The biosensor was calibrated using standard glucose solutions in a relevant buffer prior to fermentation initiation.
- Fermentation Inoculation: The bioreactor was inoculated with the yeast strain and operated in batch mode until the initial carbon source was depleted.
- Fed-Batch Commencement: The feeding phase was initiated, providing a concentrated nutrient feed to control growth and metabolism.
- Biosensor Operation:
- Samples (cell-free or cell-containing) were continuously or intermittently pumped through the biosensor flow cell.
- The chronoamperometric current was measured and converted to a glucose concentration by the system software.
- For on-line control, the glucose concentration data could be fed to the bioreactor's control system to adjust the feed pump rate automatically.
- Reference Analysis: Samples were periodically analyzed via HPLC with a refractive index (RI) detector to validate the biosensor's accuracy [3].
Results and Data Analysis
The application of the electrochemical biosensor platform demonstrated significant performance advantages over traditional analytical methods.
Table 1: Quantitative Performance Summary of the Glucose Biosensor
| Performance Parameter | Result | Context / Comparison |
|---|---|---|
| Linear Detection Range | Up to 150 mM (≈27 g/L) | 4-6 times higher than many biosensors designed for clinical use [3]. |
| Analysis Time | < 5 minutes | Significantly faster than HPLC analysis [3]. |
| Operational pH Range | 5 – 9 | Suitable for typical yeast fermentation conditions [3]. |
| Measurement Stability | Outstanding mechanical stability in direct contact with fermentation broth | Robust performance in a complex matrix [3]. |
| Selectivity | Accurate quantification in the presence of various electroactive species | Verified against reference HPLC measurements [3]. |
The biosensor reliably tracked glucose dynamics throughout the fed-batch process. The data provided a real-time profile of substrate consumption, enabling researchers to observe the metabolic activity of the yeast culture. The close agreement between the biosensor data and the off-line HPLC validation confirmed the platform's accuracy and reliability for decision-making in a bioprocess context [3].
Visualizations
Biosensor Integration Workflow
The following diagram illustrates the workflow for integrating the amperometric biosensor into the fermentation process for monitoring and control.
Amperometric Glucose Sensing Principle
This diagram outlines the core principle of the 1st generation amperometric biosensor used in the case study.
The Scientist's Toolkit
Table 2: Essential Research Reagents and Materials
| Item | Function / Role in the Experiment |
|---|---|
| Biosensor Chip (B.LV5) | Core sensing unit. The flow-through-cell contains the immobilized glucose oxidase enzyme and the electrochemical electrodes for signal generation [3]. |
| Potentiostat (SIX Transmitter) | Instrument that applies a constant potential (+450 mV) to the electrochemical cell and measures the resulting current, which is proportional to the glucose concentration [3]. |
| Peristaltic Pump | Provides a controlled flow of the fermentation sample or calibration standards through the biosensor flow cell [3]. |
| Glucose Oxidase (GOx) | The biological recognition element. This enzyme provides the high selectivity for glucose by catalyzing its specific oxidation [3]. |
| Buffer Solutions (pH 5-9) | Used for system calibration and dilution. The biosensor is operational within this pH range, making it compatible with many fermentation conditions [3]. |
| HPLC System with RI Detector | Used as a reference method for validating the accuracy and precision of the biosensor glucose measurements [3]. |
Discussion
This case study demonstrates a successful implementation of an amperometric biosensor for advanced fermentation monitoring. The platform addressed a critical gap in bioprocess analytical technology by providing rapid, specific, and reliable glucose data. The ability to measure in complex, cell-containing broths at industrially relevant concentrations (up to 150 mM) makes this technology particularly valuable for both process development and manufacturing [3].
The findings align with the broader movement toward smart biomanufacturing and Industry 4.0, where real-time data collection is a fundamental enabler for advanced process control, optimization, and automation [60]. The biosensor platform described herein provides a viable solution to the "data collection" obstacle that often hinders this transition. Future work could focus on integrating this glucose signal with advanced control algorithms and other real-time sensors to create fully autonomous, self-optimizing fermentation systems.
Conclusion
The integration of advanced amperometric biosensors marks a transformative step toward sophisticated, data-driven control of yeast fermentation processes. By synthesizing insights from foundational principles, innovative methodologies, robust troubleshooting strategies, and rigorous validation, it is clear that these sensors offer a reliable path to real-time glucose monitoring. Current research demonstrates significant progress in extending linear ranges, improving selectivity, and enhancing sensor longevity within complex fermentation environments. Future directions should focus on developing multi-analyte sensors for simultaneous monitoring of key metabolites like lactate and ethanol, further miniaturization for high-throughput micro-bioreactors, and the integration of sensor data with advanced process control algorithms. For biomedical and clinical research, particularly in the production of biotherapeutics and vaccines, the adoption of these robust online monitoring tools is pivotal for ensuring product quality, maximizing yield, and advancing toward fully automated biomanufacturing.
References
- 1. Design of Amperometric Biosensors for the Detection ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC5852879/]
- 2. Review Article Enzyme based amperometric biosensors [https://www.sciencedirect.com/science/article/abs/pii/S2451910318301005]
- 3. Automated Electrochemical Glucose Biosensor Platform as ... [https://www.frontiersin.org/journals/bioengineering-and-biotechnology/articles/10.3389/fbioe.2020.00436/full]
- 4. An Amperometric Enzyme–Nanozyme Biosensor for ... [https://www.mdpi.com/2079-6374/15/8/545]
- 5. Offline glucose biomonitoring in yeast culture by... [https://pubmed.ncbi.nlm.nih.gov/21485034/]
- 6. monitoring using a Fermentation based on an... glucose biosensor [https://pubs.rsc.org/en/content/articlelanding/1995/an/an9952002255]
- 7. Biosensors, Volume 15, Issue 9 (September 2025) [https://www.mdpi.com/2079-6374/15/9]
- 8. Towards yeast fermentation monitoring: Enhanced sensing ... [https://www.sciencedirect.com/science/article/pii/S2214180424000916]
- 9. Precision Fermentation: Fermentation Monitoring - Real-time ... [https://www.precisionfermentation.com/]
- 10. Automated Electrochemical Glucose Biosensor Platform as ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC7253623/]
- 11. Amperometric Biosensor - an overview [https://www.sciencedirect.com/topics/nursing-and-health-professions/amperometric-biosensor]
- 12. Amperometric bienzymatic biosensor in flow injection ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC10786783/]
- 13. Microbial Process Control in Traditional Fermented Foods [https://www.mdpi.com/2311-5637/11/6/323]
- 14. Advancements in Glucose Biosensors: Innovations for ... [https://www.sciencedirect.com/science/article/abs/pii/S003991402501536X]
- 15. Integrated individually addressable microneedle arrays for ... [https://www.nature.com/articles/s41378-025-01089-w]
- 16. Integrated biosensor platform based on graphene ... [https://www.nature.com/articles/s41467-022-32749-4]
- 17. A multi-unit integrated electrochemical biosensor array for ... [https://www.sciencedirect.com/science/article/abs/pii/S0925400523009395]
- 18. Amperometric microbial biosensor for sugars and ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC9813324/]
- 19. Reagentless Glucose Biosensor Based on Combination of ... [https://www.mdpi.com/2079-6374/14/3/134]
- 20. Development and Comparative Evaluation of Two Enzyme ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC12566202/]
- 21. Sol-gel based amperometric biosensor incorporating an ... [https://www.sciencedirect.com/science/article/pii/S0039914096021649]
- 22. Recent Progress of the Practical Applications ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC8128108/]
- 23. Integrated multi-material portable 3 - D platform for... printed [https://pubs.rsc.org/en/content/articlelanding/2022/an/d2an00862a]
- 24. A movable type bioelectronics printing technology for modular... [https://pmc.ncbi.nlm.nih.gov/articles/PMC8595314/]
- 25. Systematic Review on the Role of Microfluidic Platforms in ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC12425123/]
- 26. Analytical Parameters of an Amperometric Glucose ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC5713114/]
- 27. Development of an Amperometric-Based Glucose Biosensor ... [https://journals.plos.org/plosone/article?id=10.1371/journal.pone.0111859]
- 28. Performance characteristic studies of glucose biosensors ... [https://www.sciencedirect.com/science/article/abs/pii/S0925400507010155]
- 29. Sequential Injection Amperometric System Coupling with ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC9573359/]
- 30. Optimization of Multiparameters for Increased Yields ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC8306036/]
- 31. Bioreactors: Applications and Innovations for a Sustainable ... [https://www.mdpi.com/2076-3417/14/20/9346]
- 32. Measurement of Oxygen Transfer Rate and Specific ... [https://www.mdpi.com/2306-5354/12/4/332]
- 33. Oxygen Transfer Efficiency - an overview [https://www.sciencedirect.com/topics/engineering/oxygen-transfer-efficiency]
- 34. Biosensors in Fermentation Applications [https://www.intechopen.com/chapters/52219]
- 35. Elimination of electrochemical interferences in glucose ... [https://www.sciencedirect.com/science/article/abs/pii/S0165993610000269]
- 36. Biofouling-resistant nanomaterials for non-enzymatic ... [https://www.sciencedirect.com/science/article/pii/S2590006425003059]
- 37. A New Approach for Interferent-Free Amperometric ... [https://www.mdpi.com/2079-6374/15/8/470]
- 38. Differential Amperometric Microneedle Biosensor for ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC8869619/]
- 39. Recent Developments in Microneedle Biosensors for ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC12388307/]
- 40. Immobilized Enzymes in Biosensor Applications - PMC [https://pmc.ncbi.nlm.nih.gov/articles/PMC6337536/]
- 41. Immobilization of Enzyme Electrochemical Biosensors and ... [https://www.mdpi.com/2079-6374/13/9/886]
- 42. Influence of enzyme immobilization and skin-sensor ... [https://www.sciencedirect.com/science/article/pii/S0956566322001634]
- 43. An Amperometric Enzyme–Nanozyme Biosensor for ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC12384605/]
- 44. Recent advances in glucose monitoring utilizing oxidase ... [https://www.frontiersin.org/journals/chemistry/articles/10.3389/fchem.2025.1591302/full]
- 45. Immobilized Saccharomyces cerevisiae viable cells for ... [https://www.nature.com/articles/s41598-025-86702-8]
- 46. Recent Advances in Amperometric for Medical... Biosensors [https://pubmed.ncbi.nlm.nih.gov/39473247/]
- 47. Implantable electrochemical biosensors: Challenges, ... [https://www.sciencedirect.com/science/article/pii/S2451910325001048]
- 48. Analytical Parameters of an Amperometric Glucose ... [https://www.mdpi.com/1424-8220/17/11/2620]
- 49. Recent progress in biosensor regeneration techniques [https://pubs.rsc.org/en/content/articlehtml/2024/nr/d3nr05456j]
- 50. Calibration-Free and Highly Sensitive Potentiometric ... [https://pubmed.ncbi.nlm.nih.gov/39295464/]
- 51. A novel amperometric biosensor for multi-enzymatic ... [https://www.sciencedirect.com/science/article/pii/S2666351122000511]
- 52. Real-Time Monitoring of the Yeast Intracellular State During ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC8776715/]
- 53. Validation of Stability-Indicating HPLC Methods for ... [https://www.chromatographyonline.com/view/validation-of-stability-indicating-hplc-methods-for-pharmaceuticals-overview-methodologies-and-case-studies]
- 54. HPLC Content Determination Method Validation [https://www.welch-us.com/blogs/knowleage-base/hplc-content-determination-method-validation-a-comprehensive-guide]
- 55. Analytical Parameters of a Novel Glucose Biosensor Based ... [https://www.mdpi.com/1424-8220/21/12/4185]
- 56. Large And Small-scale Bioprocessing Market Size ... [https://www.biospace.com/press-releases/large-and-small-scale-bioprocessing-market-size-expected-to-hit-usd-248-12-billion-by-2034]
- 57. Biosensor Market Size, Growth, Share & Industry Trends ... [https://www.mordorintelligence.com/industry-reports/biosensor-market]
- 58. A quantitative prediction method utilizing whole omics data ... [https://www.nature.com/articles/s41598-024-84323-1]
- 59. Fed-Batch Operation - an overview [https://www.sciencedirect.com/topics/engineering/fed-batch-operation]
- 60. Towards smart biomanufacturing: a perspective on recent ... [https://link.springer.com/article/10.1007/s10295-020-02308-1]