Advanced Self-Assembled Monolayers for Eliminating Non-Specific Adsorption on Gold Surfaces in Biomedical Sensing
This article provides a comprehensive resource for researchers and drug development professionals on utilizing self-assembled monolayers (SAMs) to combat non-specific adsorption (NSA) on gold surfaces.
Advanced Self-Assembled Monolayers for Eliminating Non-Specific Adsorption on Gold Surfaces in Biomedical Sensing
Abstract
This article provides a comprehensive resource for researchers and drug development professionals on utilizing self-assembled monolayers (SAMs) to combat non-specific adsorption (NSA) on gold surfaces. It covers fundamental principles of SAM formation and antifouling mechanisms, explores diverse molecular designs and functionalization methodologies, details optimization strategies for enhanced stability and specificity, and evaluates analytical techniques for performance validation. The content synthesizes recent scientific advances to guide the development of reliable biosensors and biomedical devices with improved signal-to-noise ratios and diagnostic accuracy.
Understanding SAMs: The Foundation for Low-Noise Gold Surfaces
Core Challenge: Non-Specific Adsorption in Biosensing
Non-specific adsorption (NSA), also referred to as non-specific binding or biofouling, occurs when molecules irreversibly adsorb to a sensor's surface through physisorption, leading to high background signals that are often indistinguishable from specific binding events [1]. This phenomenon severely compromises biosensor performance by decreasing sensitivity, specificity, and reproducibility, ultimately increasing false-positive rates and limiting detection capabilities [1].
The underlying mechanisms of NSA involve intermolecular forces such as hydrophobic interactions, ionic interactions, van der Waals forces, and hydrogen bonding [1]. For biosensors utilizing self-assembled monolayers (SAMs) on gold surfaces, instability in the monolayer can create false signals, as demonstrated in developing an erythromycin aptasensor where initial SAM desorption mimicked target binding [2].
Quantitative Data: SAM Performance and NSA Impact
Table 1: Analytical Performance of SAM-Based Biosensors for Different Targets
| Target Analyte | SAM Composition | Electrode Platform | Linear Detection Range | Limit of Detection | Reference |
|---|---|---|---|---|---|
| Erythromycin | MCH/Thiolated Aptamer | Pure [111] Gold Electrode | 1 × 10⁻⁶ M to 2 × 10⁻⁴ M | 3.2 × 10⁻⁷ M | [2] |
| α-Synuclein | Cysteamine (CYS) | FTO Electrode | 10 to 1000 ng/mL | 1.13 ng/mL | [3] |
Table 2: Common Blocking Agents and Their Efficacy in Reducing NSA
| Blocking Agent / Method | Mechanism of Action | Advantages | Limitations |
|---|---|---|---|
| 6-Mercapto-1-hexanol (MCH) | Displaces non-specifically adsorbed aptamers; dilutes and reorients surface probes [2]. | Well-established; creates a hydrophilic barrier [2]. | Can desorb over time, causing signal drift [2]. |
| Serum Albumin (e.g., BSA) | Proteins adsorb to vacant surface sites, preventing further NSA [1]. | Easy to use; effective for many immunoassays [1]. | Can be unstable and add its own non-specific background [1]. |
| Polyethylene Glycol (PEG) | Forms a dense, hydrated layer that sterically hinders protein approach [4]. | High reduction of NSA and non-specific cellular uptake [4]. | Requires precise surface density (>0.96 PEG/nm² for optimal effect) [4]. |
| Zwitterionic Moieties | Creates a strong hydration layer via electrostatic interactions [2]. | Excellent antifouling properties; highly resistant to protein adsorption [2]. | More complex synthesis and attachment chemistry [2]. |
Detailed Experimental Protocols
Protocol: Optimized SAM Formation and Stabilization on Gold Electrodes
This protocol outlines a method for constructing a stable, low-NSA mixed SAM of a thiolated aptamer and MCH on a gold electrode, derived from research on an erythromycin aptasensor [2].
I. Materials Required
- Pure [111] gold electrode or gold nanoparticle-coated electrode
- Thiol-modified DNA aptamer (e.g., 5'-SH-(CH₂)₆-AGT ATT GCG GAG GAA GGG GTC GAC CCC ATC ATC AAT GAC CAG ACA CG-3' for erythromycin) [2]
- 6-Mercapto-1-hexanol (MCH) solution (e.g., 1-10 mM in ultrapure water)
- Tris-EDTA or phosphate buffer (for aptamer dilution)
- Electrolyte solution (e.g., 0.1 M PBS, pH 7.4)
- Ultrapure water
II. Step-by-Step Procedure
Critical Surface Pre-treatment:
- Clean the bare gold electrode electrochemically via cyclic voltammetry (e.g., in 0.5 M H₂SO₄ or 0.1 M HCl) until a stable voltammogram characteristic of a clean Au surface is obtained [5].
- Alternatively, mechanically polish the electrode with alumina or diamond slurry, followed by thorough rinsing and sonication in ultrapure water and ethanol [5].
- Note: The polishing material (alumina vs. diamond) can influence the final SAM structure and performance [5].
Aptamer Immobilization:
- Prepare a 1-5 µM solution of the thiolated aptamer in an appropriate buffer (e.g., Tris-EDTA with added MgCl₂).
- Incubate the pre-treated gold electrode with the aptamer solution for a defined period (e.g., 1-24 hours) at room temperature. Extended immobilization times (e.g., overnight) have been shown to enhance mixed SAM stability [2].
Surface Blocking with MCH:
- Rinse the electrode gently with ultrapure water to remove physisorbed aptamers.
- Incubate the aptamer-functionalized electrode in a 1-10 mM aqueous solution of MCH for 30-60 minutes. This step displaces any remaining non-specifically adsorbed aptamers and creates a well-diluted, oriented monolayer, reducing subsequent NSA [2].
Sensor Stabilization:
- Rinse the modified electrode with buffer.
- For enhanced signal stability, condition the electrode by soaking it in the measurement electrolyte (buffered solution) for up to 12 hours before use. This allows the MCH/aptamer SAM to reorganize into a more stable configuration, minimizing signal drift [2].
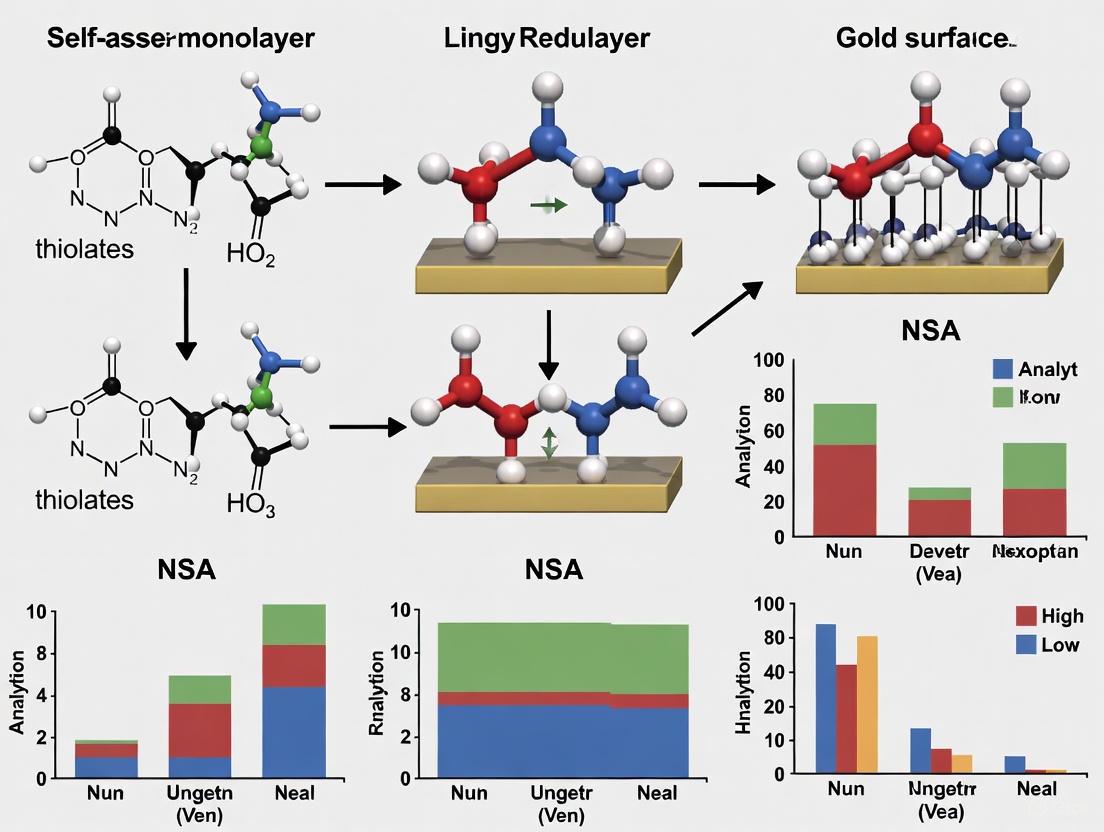
Workflow Visualization: SAM Fabrication and NSA Mitigation
The following diagram illustrates the complete process of creating a low-NSA SAM-based biosensor.
Protocol: Evaluation of SAM Stability and NSA
This protocol describes how to electrochemically characterize the quality of the formed SAM and test its resistance to NSA.
I. Materials Required
- SAM-modified gold electrode from Protocol 3.1
- Redox probe solution (e.g., 5 mM K₄[Fe(CN)₆]/K₃[Fe(CN)₆] in PBS)
- Buffer solution (for blank incubation)
- Complex solution (e.g., 1-10% serum or a solution of non-target proteins)
II. Step-by-Step Procedure
Electrochemical Characterization:
- Using cyclic voltammetry (CV) or electrochemical impedance spectroscopy (EIS), record the response of the SAM-modified electrode in the redox probe solution.
- A well-formed, dense SAM will significantly suppress the Faradaic current (in CV) or increase the charge transfer resistance (Rct in EIS) compared to a bare gold electrode.
Blank Signal Stability Test:
- Incubate the sensor in a pure buffer solution (blank) for a duration mimicking the real assay.
- Periodically measure the electrochemical signal (e.g., via differential pulsed voltammetry - DPV). A stable signal over time indicates a robust SAM that is resistant to desorption and reorganization, which is critical for avoiding false positives [2].
NSA Challenge Test:
- Expose the sensor to a complex solution containing non-target proteins or other potential interferents.
- After washing, measure the signal again in the redox probe. A minimal change in signal compared to the measurement before the challenge indicates effective NSA reduction.
The Scientist's Toolkit: Essential Research Reagents
Table 3: Key Reagent Solutions for SAM Research on Gold Surfaces
| Reagent / Material | Function / Application | Key Considerations |
|---|---|---|
| Thiolated Alkanes (e.g., MCH) | Backfill agent to create mixed SAMs; displaces physisorbed DNA and reduces NSA [2]. | Intermediate (C6) chain length offers a good compromise between SAM stability and target accessibility [2]. |
| Thiol-Modified DNA Aptamers | Biorecognition element; confers specificity to the sensor. | Requires a spacer (e.g., C6 alkyl) between thiol group and sequence; incubation time affects surface coverage and stability [2]. |
| PEGylated Thiols | Creates a highly effective antifouling layer to minimize protein NSA [4]. | A high surface density (>0.96 PEG molecules per nm²) is critical for maximum NSA reduction [4]. |
| Redox Probes (e.g., [Fe(CN)₆]³⁻/⁴⁻) | Electrochemical label for characterizing SAM integrity and monitoring binding events [2]. | Signal suppression indicates successful SAM formation; signal changes can indicate desorption or target binding [2]. |
| Zwitterionic Thiols | Forms ultra-low fouling SAMs via a strong bound water layer [2]. | Emerging alternative to PEG; excellent for use in complex biological fluids [2]. |
NSA Reduction Strategy Visualization
The diagram below categorizes the main strategies for combating Non-Specific Adsorption in biosensing.
Self-assembled monolayers (SAMs) of alkanethiolates on gold represent a cornerstone of surface science, enabling precise control over interfacial properties for applications ranging from biosensing to fundamental studies of non-specific adsorption (NSA). These highly ordered organic assemblies form spontaneously when alkanethiols chemisorb onto gold surfaces, creating robust molecular architectures with tailored terminal functionalities. The molecular-level control afforded by SAMs makes them indispensable tools for investigating and mitigating NSA on gold surfaces, a critical requirement for the development of reliable biosensors and diagnostic platforms. This application note provides detailed protocols and key data for the formation, characterization, and application of alkanethiolate SAMs, with particular emphasis on their role in creating bio-inert surfaces that resist non-specific protein adsorption and cell attachment.
Molecular Structure and System Composition
Structural Components of Alkanethiolate SAMs
The molecular architecture of alkanethiolate SAMs consists of three distinct regions that collectively determine their physical and chemical properties. The headgroup (thiol moiety) forms a coordinative bond with gold surfaces, creating a stable thiolate-gold interface with well-defined geometry. The alkyl chain (typically C6-C18) provides structural integrity through van der Waals interactions between adjacent chains, driving the self-assembly process and determining monolayer packing density. The terminal functional group (e.g., -CH3, -OH, -COOH, -NH2, or ethylene glycol) defines surface chemistry and interfacial behavior, with specific functionalities engineered to control wettability, biocompatibility, and resistance to non-specific adsorption [6].
Key Research Reagent Solutions
Table 1: Essential Materials for Alkanethiolate SAM Formation and Characterization
| Reagent/Material | Function/Application | Specifications/Notes |
|---|---|---|
| Gold Substrates | Foundation for SAM formation | Typically 10-100 nm Au films on Si wafers with 1-5 nm Ti or Cr adhesion layer [7] |
| Alkanethiols | SAM precursor molecules | Examples: Hexadecanethiol [HS(CH₂)₁₅CH₃], 6-amino-1-hexanethiol [HS(CH₂)₆NH₂], EG3-thiol [HS(CH₂)₁₁(OCH₂CH₂)₃OH] [6] [8] |
| Ethanol (Absolute) | Primary solvent for thiol solutions | High-purity, anhydrous for optimal SAM formation (1-10 mM thiol concentration) |
| Piranha Solution | Substrate cleaning | 3:1 H₂SO₄:30% H₂O₂; Highly corrosive [7] |
| Redox Probes | Electrochemical characterization | Ru(NH₃)₆Cl₃, K₃Fe(CN)₆ for CV and EIS measurements [8] |
| Plasma Cleaning Systems | SAM removal/substrate regeneration | Hydrogen or oxygen plasma for complete monolayer removal [7] |
Experimental Protocols
Substrate Preparation and Cleaning
Objective: To obtain atomically flat, contamination-free gold surfaces essential for reproducible SAM formation.
Materials:
- Gold substrates (as in Table 1)
- Piranha solution (3:1 v/v concentrated H₂SO₄:30% H₂O₂)
- CAUTION: Piranha solution is extremely corrosive and must be handled with appropriate PPE in a fume hood
- Ultrapure water (18.2 MΩ·cm)
- Ethanol (HPLC grade)
- Nitrogen gas (high purity)
Procedure:
- Initial Cleaning: Sonicate gold substrates in ultrapure water for 1 minute to remove particulate contamination [7].
- Oxidative Cleaning: Immerse substrates in freshly prepared piranha solution for 15 minutes to remove organic residues.
- Rinsing: Thoroughly rinse substrates with copious amounts of ultrapure water (minimum 500 mL per substrate) to completely remove acid residues.
- Solvent Rinsing: Rinse substrates sequentially with ethanol to facilitate drying and remove any organic impurities.
- Drying: Dry substrates under a stream of dry nitrogen gas.
- Immediate Use: Use prepared substrates immediately for SAM formation to prevent surface contamination.
SAM Formation via Solution Deposition
Objective: To form densely packed, well-ordered alkanethiolate monolayers on gold surfaces.
Materials:
- Prepared gold substrates
- Alkanethiol of choice (e.g., 6-amino-1-hexanethiol for amino-terminated surfaces)
- Ethanol (absolute, anhydrous)
- Inert atmosphere glove box or sealed deposition vessels (optional)
Procedure:
- Solution Preparation: Prepare 1-10 mM alkanethiol solution in absolute ethanol under inert atmosphere if possible to prevent thiol oxidation.
- SAM Deposition: Immerse clean gold substrates in the thiol solution for 12-24 hours at room temperature to ensure complete monolayer formation.
- Rinsing: Remove substrates from thiol solution and rinse thoroughly with pure ethanol to remove physisorbed molecules.
- Drying: Dry SAM-functionalized substrates under a stream of nitrogen gas.
- Storage: Store prepared SAMs under nitrogen or in vacuum until use to minimize contamination.
Microcontact Printing for Patterned SAMs
Objective: To create spatially defined regions with different surface functionalities for controlling cell attachment and protein adsorption [6].
Materials:
- Poly(dimethylsiloxane) (PDMS) stamps with desired relief patterns
- Two different alkanethiols with contrasting terminal groups (e.g., hexadecanethiol and EG3-thiol)
- Ethanol for rinsing
- Fibronectin or other extracellular matrix proteins
Procedure:
- Stamp Inking: Apply hexadecanethiol (1-2 mM in ethanol) to a flat PDMS stamp and dry gently with nitrogen.
- Contact Printing: Bring the inked stamp into conformal contact with a contoured gold surface for 10-20 seconds.
- Backfilling: Immerse the printed substrate in a solution of EG3-thiol [HS(CH₂)₁₁(OCH₂CH₂)₃OH] for 1-2 hours to form a protein-resistant monolayer in the non-printed regions [6].
- Protein Adsorption: Incubate the patterned substrate with fibronectin solution (10-50 μg/mL in PBS) for 1 hour.
- Cell Seeding: Apply bovine capillary endothelial cells (or other cell types) to the functionalized surface to achieve patterned cell attachment exclusively on the methyl-terminated regions.
Data Presentation and Analysis
Quantitative Characterization Data
Table 2: Electrochemical Characterization of 6-Amino-1-Hexanethiol (AHT) SAMs Using Ru(NH₃)₆³⁺/²⁺ Redox Probe [8]
| Parameter | Acidic Conditions (pH < 5) | Basic Conditions (pH > 9) | Measurement Technique |
|---|---|---|---|
| Current Density | Decreased | Increased | Cyclic Voltammetry |
| Peak Potential (Eₚ) | Shifted positively | Shifted negatively | Cyclic Voltammetry |
| Heterogeneous Rate Constant (k⁰) | Lower values | Higher values | EIS and CV |
| Reductive Desorption Charge | Decreased due to H₂ evolution side reaction | Increased | Linear Sweep Voltammetry |
| Contact Angle | More hydrophilic (~40-50°) | Less hydrophilic (~60-70°) | Static Water Contact Angle |
| SAM Organization | Less ordered, protonated NH₃⁺ groups | More ordered, neutral NH₂ groups | XPS, Electrochemical Analysis |
Table 3: SAM Removal Efficiency Comparison [7]
| Parameter | Hydrogen Plasma | Oxygen Plasma | Piranha Etching |
|---|---|---|---|
| Treatment Time | < 60 seconds | < 60 seconds | 15 minutes |
| Sulfur Removal | Complete (XPS detection limit) | Incomplete (oxidized S species remain) | Complete |
| Surface Chemistry After | Pure gold | Gold oxide with sulfonates/sulfate | Pure gold |
| Surface Roughness Change | Minimal alteration | Minimal alteration | Induces recrystallization |
| Additional Steps | None required | None required | Extensive rinsing required |
| Practical Considerations | No chemical waste | No chemical waste | Corrosive waste generation |
Biological Application Data
Table 4: Cell Attachment Control on Patterned SAMs [6]
| Surface Region | SAM Composition | Protein Adsorption | Cell Attachment |
|---|---|---|---|
| Raised Plateaus | Hexadecanethiol [HS(CH₂)₁₅CH₃] | Fibronectin adsorption observed | Bovine capillary endothelial cells attach |
| Grooves | EG3-thiol [HS(CH₂)₁₁(OCH₂CH₂)₃OH] | Protein adsorption resisted | No cell attachment |
| Reverse Pattern | Complementary patterning approach | Confined to groove regions | Cells attach only in grooves |
Visualization of Experimental Workflows
SAM Formation and Characterization Workflow
SAM Formation Workflow: This diagram illustrates the sequential process for preparing and characterizing alkanethiolate SAMs on gold surfaces, from substrate cleaning through biological application.
Microcontact Printing Process
Microcontact Printing Process: This visualization shows the step-by-step procedure for creating patterned SAMs using microcontact printing to control cellular attachment.
Technical Notes and Troubleshooting
Optimization of SAM Quality
- Solvent Quality: Always use high-purity, anhydrous ethanol for thiol solutions to prevent oxidation and ensure reproducible SAM formation.
- Dissolved Oxygen: Degas solutions with nitrogen or argon when working with thiols prone to oxidation, particularly those with reactive terminal groups.
- Contamination Control: Handle SAMs in clean environment and store under nitrogen to prevent hydrocarbon contamination that can affect surface properties.
- Characterization Validation: Combine multiple characterization techniques (electrochemical, spectroscopic, and microscopic) to verify SAM quality and organization.
Common Issues and Solutions
- Incomplete SAM Coverage: Extend deposition time to 24 hours or increase thiol concentration to 10 mM for complete monolayer formation.
- High Non-specific Adsorption: Ensure proper backfilling with protein-resistant thiols like EG3-terminated alkanethiols and verify monolayer quality using reductive desorption.
- Inconsistent Patterning: Optimize stamp pressure and contact time during microcontact printing, and verify stamp integrity before use.
- SAM Removal Challenges: For complete monolayer removal, hydrogen plasma treatment for 60 seconds effectively regenerates gold surfaces without leaving sulfur residues [7].
The protocols and data presented herein provide a comprehensive framework for the fabrication, characterization, and application of alkanethiolate SAMs on gold surfaces, with specific utility for controlling non-specific adsorption in biological contexts.
The prevention of nonspecific adsorption (NSA) on sensor surfaces is a critical challenge in biomedical research and diagnostics. For gold-surface-based biosensors, self-assembled monolayers (SAMs) provide a powerful platform for engineering surfaces that resist fouling from proteins, cells, and other biomolecules. This application note examines three fundamental antifouling mechanisms—steric repulsion, hydration layers, and electrostatic effects—within the context of SAM-functionalized gold surfaces. We present quantitative data, detailed protocols, and experimental tools to guide researchers in developing effective antifouling coatings for biosensors, implants, and other devices that interface with biological systems.
Antifouling Mechanisms of SAMs
Steric Repulsion
Steric repulsion operates through a physical barrier mechanism where surface-grafted polymer chains resist compression when biomolecules approach. Poly(ethylene glycol) (PEG) and its derivative oligo(ethylene glycol) (OEG) are the most extensively studied SAMs employing this mechanism [9]. The resistance is primarily entropy-driven: when biomaterials compress the polymer chains, the loss of conformational freedom generates a repulsive elastic force that prevents adsorption [9]. While this mechanism is highly effective for longer, flexible polymer brushes, it is less dominant in densely packed SAMs where chain mobility is constrained [9].
Hydration Layer Mechanism
Hydration layers form when water molecules strongly bind to hydrophilic surface groups via hydrogen bonding, creating an energetic barrier to adsorption [9] [10]. Biomolecular adsorption requires displacing these bound water molecules, a process that is thermodynamically unfavorable due to high activation energy [9]. This mechanism is particularly effective with zwitterionic SAMs and short-chain hydrophilic materials where tightly bound water forms a protective barrier [9] [10]. Unlike steric repulsion, the hydration mechanism does not require extensive polymer chain mobility and is therefore highly effective in densely packed SAMs [9].
Electrostatic Effects
Electrostatic interactions provide antifouling through long-range repulsive forces between charged surfaces and biomolecules. Recent research using total internal reflection microscopy (TIRM) has revealed that even supposedly "electrically neutral" polymer brushes, including zwitterionic and PEG-based surfaces, exhibit significant electrostatic interactions that influence contaminant distribution [10]. These long-range forces (detectable beyond 300 nm) operate before short-range steric or hydration effects become relevant and are highly responsive to ionic strength variations [10].
Table 1: Comparative Analysis of Antifouling Mechanisms
| Mechanism | Key Materials | Range of Effect | Dominant Driving Force | Dependence on Ionic Strength |
|---|---|---|---|---|
| Steric Repulsion | PEG, OEG, POEGMA [9] [10] | Short-range (< polymer brush thickness) [10] | Entropic penalty from chain compression [9] | Low to moderate |
| Hydration Layer | Zwitterions, OEG, PEG [9] [10] | Short-range (molecular water layer) [10] | Energetic cost of dehydration [9] | Moderate |
| Electrostatic Effects | Zwitterionic PCBMA, charged SAMs [10] | Long-range (up to 300+ nm) [10] | Electrostatic repulsion/attraction [10] | High (screened at high ionic strength) |
Table 2: Quantitative Performance of Antifouling SAMs on Gold Surfaces
| SAM Composition | Substrate | Fouling Reduction | Test Conditions | Key Mechanism |
|---|---|---|---|---|
| Si-MEG-OH (direct) | Gold | 88% [11] | Undiluted goat serum | Hydration layer [11] |
| βME/Si-MEG-OH (tandem) | Gold | ~75% [11] | Undiluted goat serum | Combined hydration/steric [11] |
| PCBMA brushes | Glass slides | Significant reduction in bacterial colonization [10] | Variable ionic strength | Electrostatic (long-range) [10] |
| POEGMA brushes | Glass slides | Effective antifouling performance [10] | Variable ionic strength | Combined steric/electrostatic [10] |
Experimental Protocols
Protocol: Tandem Antifouling Coating on Gold Surfaces
This protocol details the application of a tandem β-mercaptoethanol (βME)/monoethylene glycol silane (Si-MEG-OH) antifouling coating on gold surfaces, achieving approximately 75% fouling reduction against undiluted goat serum [11].
Materials and Equipment
- Gold substrates (e.g., gold-quartz crystals, gold-coated glass slides)
- β-mercaptoethanol (βME) (≥95%)
- Si-MEG-TFA precursor (synthesized as in [11])
- Anhydrous toluene
- Ethanol (95% and absolute)
- Methanol
- Acetone
- Sodium dodecyl sulfate (SDS) solution (1%)
- Nitrogen gas supply
- Plasma cleaner (e.g., Harrick PDC-3XG)
- Humidity chamber (70% relative humidity, saturated MgNO₃·6H₂O)
- Nitrogen glovebox
- Orbital shaker
Step-by-Step Procedure
Step 1: Gold Substrate Cleaning
- Place gold substrates in clean test tubes
- Rinse three times with distilled water
- Rinse twice with 1% SDS solution
- Shake vigorously in SDS solution for 15 minutes
- Rinse three times with acetone
- Rinse twice with methanol
- Shake in methanol for 5 minutes
- Dry under nitrogen stream
- Plasma clean under vacuum in ambient air for 5 minutes [11]
Step 2: βME SAM Formation
- Submerge clean gold substrates in 95% ethanol solution
- Add 0.5% v/v βME to the solution
- Place on orbital shaker for minimum 2 hours
- Rinse with methanol and dry with nitrogen gas [11]
Step 3: Si-MEG-OH Coating
- Place βME-coated substrates in test tubes dried at 180°C for 90 minutes
- Transfer to humidity chamber (70% relative humidity) for 30 minutes
- Move to nitrogen glovebox
- Submerge in anhydrous toluene with 1% v/v Si-MEG-TFA precursor
- Seal tubes with rubber stoppers
- Shake on orbital shaker for at least 2 hours
- Unseal and rinse thoroughly three times with toluene
- Rinse three times with 95% ethanol
- Submerge in 50% ethanol for 12 hours to convert terminal groups to Si-MEG-OH
- Rinse with methanol, dry with nitrogen, and store in glass vials [11]
Quality Control and Characterization
- Contact Angle Goniometry: Measures hydrophilicity/hydrophobicity changes
- Atomic Force Microscopy (AFM): Visualizes layer formation and detects defects
- X-ray Photoelectron Spectroscopy (XPS): Confirms chemical composition and monolayer formation
- Antifouling Tests: Use thickness shear mode (TSM) sensors with undiluted goat serum to quantify fouling reduction [11]
Protocol: Direct Measurement of Long-Range Interactions Using TIRM
This protocol describes the use of total internal reflection microscopy (TIRM) to directly measure long-range interactions near polymer-grafted surfaces, revealing significant electrostatic effects even on supposedly "neutral" antifouling surfaces [10].
Materials and Equipment
- Glass slides grafted with zwitterionic PCBMA or nonionic POEGMA brushes
- Sulfated polystyrene microspheres (common tracer particles, ~1μm diameter)
- NaCl solutions of varying concentrations (0.1mM to 10mM)
- Total internal reflection microscope with appropriate optics
- Temperature-controlled stage
- Data acquisition system
Surface Preparation
- Graft PCBMA or POEGMA brushes onto glass slides via atom transfer radical polymerization (ATRP)
- Target molecular weight: ~52,000 g/mol for PCBMA
- Target graft density: ~0.2 chains/nm² [10]
- Characterize brush thickness and quality using AFM and ellipsometry
TIRM Measurement Procedure
- Place polymer-grafted slide in flow chamber
- Introduce suspension of sulfated PS microspheres in desired NaCl concentration
- Focus laser beam for total internal reflection at sample interface
- Record scattering signals from freely diffusing microspheres near surface
- Track particle position and motion continuously for minimum 30 minutes per condition
- Repeat measurements across various ionic strengths (0.1-10mM NaCl) [10]
Data Analysis
- Calculate interaction potential from probability distribution of particle positions
- Identify equilibrium position (hm) where repulsion and attraction balance
- Fit repulsive portions of interaction curves to determine Debye length (κ⁻¹)
- Compare measured Debye lengths with theoretical values for electrostatic interactions
- Analyze salt-dependent changes in hm to confirm electrostatic origin of interactions [10]
The Scientist's Toolkit: Essential Research Reagents
Table 3: Key Research Reagent Solutions for Antifouling SAM Research
| Reagent/Material | Function/Application | Key Characteristics | Research Context |
|---|---|---|---|
| 6-amino-1-hexanethiol (AHT) | Amino-terminated alkanethiol for SAM functionalization [8] | Appropriate chain length for organized SAMs; facilitates derivatization [8] | Building block for functional platforms; enables further chemical modification |
| Si-MEG-TFA precursor | Forms monoethylene glycol silane antifouling coating [11] | Trichlorosilane-based; forms covalent siloxane network [11] | Creates ultrathin coatings with SAM and polymer brush-like properties |
| β-mercaptoethanol (βME) | Hydroxylating agent for gold surfaces [11] | Thiol-terminated with hydroxyl group; forms SAM on gold [11] | Provides hydroxyl groups on gold for subsequent silane chemistry |
| Zwitterionic silanes | Creates surfaces with stable hydration layers [12] | Contains both positive and negative charges; highly hydrophilic [12] | Modulates Schiff base and Michael addition reactions in coating formation |
| PLL-g-PEG | Electrostatic adsorption coating for oxidized surfaces [13] | Poly(l-lysine)-graft-PEG; adsorbs on negatively charged surfaces [13] | Easy application without covalent bonding; suitable for PDMS and oxidized surfaces |
| Pluronic surfactants | Physical coating for hydrophobic surfaces [13] | PEO-PPO-PEO triblock copolymer; adsorbs via hydrophobic interactions [13] | Dynamic coating for PDMS microchannels; reduces electroosmotic flow |
Mechanism Interrelationships and Experimental Design
The three antifouling mechanisms do not operate in isolation but often function cooperatively. The relative contribution of each mechanism depends on surface chemistry, environmental conditions, and the nature of potential foulants. Steric repulsion dominates with longer, flexible polymer chains, while hydration effects are primary with short-chain, highly hydrophilic SAMs. Electrostatic interactions provide long-range protection that operates before shorter-range mechanisms become relevant [10].
Diagram 1: Interrelationship of Antifouling Mechanisms on Functionalized Gold Surfaces. Surface chemistry determines dominant mechanism, though multiple mechanisms often operate concurrently.
Environmental conditions significantly influence mechanism dominance. Electrostatic effects are pronounced at low ionic strength but become screened as salt concentration increases [10]. Hydration layers remain stable across various ionic conditions but may be compromised by extreme temperatures or dehydrating agents. Steric repulsion effectiveness depends on polymer chain mobility, which can be affected by surface grafting density and molecular weight.
Understanding the interplay between steric repulsion, hydration layers, and electrostatic effects enables rational design of antifouling SAMs for gold surfaces in biomedical applications. While traditional approaches emphasized short-range interactions, recent research reveals that long-range electrostatic forces play a crucial role in initial fouling prevention. The protocols and data presented here provide researchers with practical tools for developing and characterizing advanced antifouling coatings, ultimately enhancing the performance and reliability of biosensors, implants, and diagnostic devices that interface with complex biological environments.
Self-assembled monolayers (SAMs) engineered to minimize nonspecific adsorption (NSA) are critical for advancing technologies in biosensing, medical implants, and drug development. Within this field, hydrophilic SAMs—particularly those based on oligo(ethylene glycol) (OEG), zwitterions, and natural peptides—have emerged as leading strategies to create ultra-low fouling surfaces. This Application Note provides a consolidated overview of these designs, focusing on their performance data, underlying mechanisms, and detailed protocols for their implementation on gold surfaces, a common substrate in biomedical devices.
Quantitative Performance Comparison
The following table summarizes key performance metrics for different hydrophilic SAM designs as reported in the literature, providing a basis for material selection.
Table 1: Performance Summary of Hydrophilic SAM Designs for Reducing Nonspecific Adsorption
| SAM Design | Specific Composition | Protein Adsorption (ng/cm²) | Cell Adhesion & Fouling Resistance | Key Findings | Reference |
|---|---|---|---|---|---|
| Zwitterionic Peptides | EK (Glu-Lys) repeating sequence | < 0.3 (Fibrinogen, QCM-D) | Excellent resistance to platelet adhesion | Strong interfacial water layer induces ~8 nm repulsion; superior anti-biofouling. | [14] [15] |
| Zwitterionic Peptides | DK (Asp-Lys) repeating sequence | < 0.3 (Fibrinogen, QCM-D) | Excellent resistance to platelet adhesion | Behavior similar to EK peptides; effective ultra-low fouling. | [14] [15] |
| Mixed OEG/Zwitterion | Multidentate polymer with ZW & OEG groups | Not quantitatively specified | Substantial improvement for fixed and living cells | Synergistic effect; combination outperforms ZW or OEG alone in complex biological environments. | [16] |
| Zwitterionic Peptides | ER (Glu-Arg) repeating sequence | High protein adsorption | Significant platelet adhesion | No significant hydration layer or repulsive force; poor anti-fouling performance. | [14] [15] |
| Zwitterionic Peptides | DR (Asp-Arg) repeating sequence | High protein adsorption | Significant platelet adhesion | Behavior similar to ER peptides; lacks the necessary hydration. | [14] [15] |
Experimental Protocols
Protocol: Fabrication of Peptide-Based SAMs on Gold
This protocol details the formation of peptide self-assembled monolayers on gold substrates, adapted from foundational research [14].
Research Reagent Solutions
Table 2: Essential Reagents for Peptide-SAM Formation
| Item | Function / Description |
|---|---|
| Gold-coated substrates | (e.g., silicon wafers with 100 nm Au layer over a 5 nm Ge adhesion layer) |
| Synthetic peptides | Custom sequences (e.g., C-terminus amide, EKEKEKE-PPPPC-Am) with a terminal thiol (-C) for gold anchoring. |
| Phosphate Buffered Saline (PBS) | (pH 7.4, ionic strength 167 mM) Used as the solvent for peptide solution. |
| Pure water and solvents | (e.g., acetone, ethanol) For cleaning substrates. |
Step-by-Step Procedure
- Substrate Preparation: Clean gold-coated substrates (e.g., silicon wafers) sequentially by ultrasonic cleaning in acetone, ethanol, and pure water for approximately 10 minutes each. Dry the substrates using a stream of nitrogen gas. For optimal monolayer formation, further clean the substrates using UV-Ozone treatment for 15 minutes.
- Peptide Solution Preparation: Dissolve the thiol-terminal peptide in degassed, pure PBS (pH 7.4) to a final concentration of 0.14 mM. Ensure complete dissolution.
- SAM Formation: Immerse the clean, dry gold substrates into the peptide solution. Allow the self-assembly process to proceed for 24 hours at room temperature, protected from light.
- Rinsing and Drying: After incubation, remove the substrates from the peptide solution and rinse them thoroughly with copious amounts of pure water to remove any physisorbed molecules. Dry the substrates under a gentle stream of nitrogen gas.
- Storage: Store the prepared SAMs in a clean, dry environment. For best results, use them within 24 hours of preparation.
Protocol: Quantitative Analysis of Protein Adsorption via QCM-D
Quartz Crystal Microbalance with Dissipation monitoring (QCM-D) is a highly sensitive method for measuring adsorbed mass and viscoelastic properties of the adlayer in real-time [14].
Procedure
- Sensor Preparation: Fabricate peptide-SAMs directly on gold-coated QCM-D sensors following the protocol in Section 3.1.
- Baseline Establishment: Mount the sensor in the QCM-D chamber and flow PBS buffer until a stable baseline in both resonant frequency (Δf) and energy dissipation (ΔD) is achieved (overtone n=3 is typically used).
- Protein Adsorption: Introduce a solution of the target protein (e.g., 1 mg/mL human fibrinogen in PBS) into the chamber at a constant flow rate.
- Rinsing: Once the frequency stabilizes, flush the chamber with PBS buffer again to remove any loosely bound proteins.
- Data Analysis: The change in frequency (Δf) before injection and after rinsing is used to calculate the adsorbed mass per unit area (Δm) using the Sauerbrey equation: Δm = -C * (Δf / n), where C is the sensor constant (17.7 ng cm⁻² Hz⁻¹ for a 5 MHz crystal). The ΔD/Δf ratio provides insights into the viscoelasticity (rigidity) of the adsorbed layer.
Mechanisms and Workflow Visualization
The anti-fouling performance of SAMs is closely linked to the formation of a tightly bound hydration layer. Surface force measurements have revealed that effective zwitterionic peptides like EK and DK generate a long-range (~8 nm) water-induced repulsive force, which acts as a physical barrier against approaching proteins and cells [14].
The following diagram illustrates the experimental workflow for creating and evaluating anti-fouling SAMs, integrating the protocols above.
The Scientist's Toolkit
Table 3: Key Research Reagent Solutions
| Item | Function / Description | Key Consideration |
|---|---|---|
| Thiol-Terminal Peptides | Provides covalent anchor to gold surface via Au-S bond. The peptide sequence defines surface properties. | Requires custom synthesis. Purity and correct sequence verification (e.g., via mass spectrometry) are critical. |
| Multidentate Polymers (ZW/OEG) | Coating for nanoparticles; combines stability with ultra-low fouling from synergistic ZW and OEG groups. | Polymer length and ratio of functional groups can be tuned to optimize size and performance [16]. |
| QCM-D Instrument | Real-time, label-free measurement of adsorbed mass (via frequency shift, Δf) and layer viscoelasticity (via energy dissipation, ΔD). | Highly sensitive to mass changes; the Sauerbrey equation applies best for rigid, thin adlayers. |
| Colloidal Probe AFM | Direct measurement of interfacial forces (e.g., hydration repulsion) between the SAM and a probe particle. | Quantifies the physical barrier responsible for anti-fouling, correlating force with performance [14]. |
Non-specific adsorption (NSA) of biomolecules such as proteins to solid surfaces is a fundamental challenge in biomedical research, diagnostics, and therapeutic development. It interferes with the accuracy of biosensors, reduces the efficiency of drug delivery vehicles, and can compromise implantable medical devices. Within the context of self-assembled monolayers (SAMs) on gold surfaces, research has focused on developing surface chemistries that can effectively resist this fouling. While surfaces presenting poly(ethylene glycol) (PEG) have been the gold standard for preventing NSA, hydrophobic and amphiphilic films present compelling alternative strategies. These materials leverage molecular-level control over surface energy, topography, and chemical functionality to create barriers against unwanted protein adhesion. This application note details the protocols and quantitative data underlying the use of hydrophobic and amphiphilic SAMs as advanced fouling-resistant coatings, providing researchers with methodologies to implement and characterize these surfaces in their own work.
Theoretical Background and Key Principles
Molecular Basis of Fouling Resistance
The interaction of proteins with a surface is primarily governed by the surface's chemical functionality and energy.
- Hydrophilic Surfaces: These surfaces, such as those terminated with PEG or oligo(ethylene glycol) (OEG), resist protein adsorption through the formation of a tightly bound layer of water molecules. This hydration layer creates a thermodynamic barrier that must be disrupted for proteins to adsorb, an energetically unfavorable process [17] [18]. The PEG chains are highly solvated and exhibit molecular mobility, which further repels approaching proteins.
- Hydrophobic Surfaces: The fouling resistance of hydrophobic surfaces operates on a different principle. These surfaces, featuring non-polar groups like methyl (-CH₃) or fluorinated chains (-CF₃), exhibit very low surface energy. While proteins can adsorb to hydrophobic surfaces via hydrophobic interactions, highly uniform and densely packed hydrophobic SAMs can present a surface with minimal defects and high chemical inertia, reducing the driving force for protein adhesion and biofilm formation [18].
- Amphiphilic Surfaces: Amphiphilic materials combine hydrophilic and hydrophobic elements within the same molecular structure. This combination can lead to superior fouling resistance compared to homogeneous surfaces. The proposed mechanism suggests that amphiphilic surfaces disrupt the orderly adsorption of proteins, as the heterogeneous surface chemistry does not provide a contiguous domain for proteins to bind to strongly or denature upon [18].
Self-Assembled Monolayers as Model Systems
SAMs of alkanethiolates on gold are an ideal platform for studying these interactions due to the exceptional control they offer over surface properties at the molecular level [17]. They form spontaneously upon immersion of a gold-coated substrate into a solution of alkanethiols, resulting in a densely packed, well-ordered film. The terminal functional group of the alkanethiol dictates the surface properties, allowing for the precise engineering of hydrophobicity, hydrophilicity, or amphiphilicity. Furthermore, mixed monolayers can be created from solutions containing two or more different alkanethiols, enabling fine control over the ratio and presentation of different chemical motifs [17].
Quantitative Data and Performance Comparison
The efficacy of fouling-resistant strategies is often quantified by the amount of non-specifically bound protein measured via techniques like Surface Plasmon Resonance (SPR) or Quartz Crystal Microbalance (QCM). The following tables summarize key performance data and structural properties for different SAM types.
Table 1: Fouling Resistance of Different SAM Termini Against Model Proteins
| SAM Terminal Group | Surface Type | Protein/Medium Tested | Fouling Resistance / Adsorption | Reference |
|---|---|---|---|---|
| Tri(ethylene glycol) | Hydrophilic | Fibronectin, Fibrinogen | Very High (~99% reduction vs bare gold) | [17] |
| Oligo(ethylene glycol) | Hydrophilic | Blood Serum, Cell Lysate | High (Adequate for SPR in lysate) | [19] |
| Methyl (-CH₃) | Hydrophobic | Rat Brain Lysate | Low (Significant protein binding) | [19] |
| Mixed Amphiphilic | Amphiphilic | Complex Biofluids | Moderate to High (Disrupts adhesion) | [18] |
Table 2: Structural and Experimental Parameters for Stable SAMs
| Parameter | Impact on SAM Stability & Fouling Resistance | Optimal Range / Example |
|---|---|---|
| Alkanethiol Chain Length (n) | Determines SAM stability and density. Longer chains enhance stability. | n ≥ 11 (for stable SAMs in aqueous solutions) [19] |
| Ligand Density | Controls availability of binding sites and steric hindrance. | 0.01% - 1% (of functional ligand in inert background) [17] |
| Substrate | Provides foundation for SAM formation. | Evaporated or sputtered gold film (≥ 100 Å thick) [17] |
Experimental Protocols
Protocol: Preparation of Hydrophobic and Amphiphilic SAMs on Gold
Objective: To form a stable, fouling-resistant self-assembled monolayer on a gold substrate using hydrophobic or mixed amphiphilic alkanethiols.
Materials:
- Gold Substrates: Glass slides or SPR chips coated with a thin gold film (≥ 100 nm).
- Alkanethiols:
- For Hydrophobic SAMs: 1-Dodecanethiol (CH₃(CH₂)₁₁SH) or 1-Hexadecanethiol (CH₃(CH₂)₁₅SH).
- For Amphiphilic SAMs: A mixture of a hydrophobic thiol (e.g., 1-Dodecanethiol) and a hydrophilic thiol (e.g., 11-Mercapto-1-undecanol, HO(CH₂)₁₁SH).
- Solvents: Absolute ethanol (high purity), Anhydrous toluene.
- Equipment: UV-Ozone cleaner or plasma cleaner, Chemical fume hood, Clean glassware, Nitrogen gas (N₂) stream.
Procedure:
- Substrate Cleaning:
- Clean the gold substrates in a UV-ozone cleaner for 20 minutes or under an oxygen plasma for 5-10 minutes.
- This step removes organic contaminants and creates a pristine, hydrophilic gold surface.
Solution Preparation:
- Hydrophobic SAM: Prepare a 1 mM solution of the chosen hydrophobic alkanethiol (e.g., 1-Dodecanethiol) in absolute ethanol.
- Amphiphilic SAM: Prepare a 1 mM total thiol solution in ethanol with the desired molar ratio of hydrophobic to hydrophilic thiol (e.g., 1:1 mol/mol). The total concentration of alkanethiols should be 1 mM.
SAM Formation:
- Immediately after cleaning, immerse the gold substrates in the prepared thiol solution.
- Seal the container to prevent solvent evaporation and contamination.
- Allow the self-assembly to proceed for a minimum of 12-24 hours at room temperature.
Post-Assembly Rinsing and Drying:
- Carefully remove the substrates from the thiol solution using tweezers.
- Rinse thoroughly by immersing and agitating in fresh, pure ethanol for 60 seconds to remove physically adsorbed thiols.
- Dry the substrates under a gentle stream of clean, dry nitrogen gas.
- Store the prepared SAMs in a clean, dry environment if not used immediately.
Protocol: Quantifying Non-Specific Adsorption via Surface Plasmon Resonance (SPR)
Objective: To quantitatively evaluate the fouling resistance of the prepared SAMs by measuring the adsorption of proteins from a complex medium.
Materials:
- Prepared SAM substrates from Protocol 4.1.
- SPR Instrument.
- Running Buffer: Phosphate Buffered Saline (PBS), pH 7.4.
- Challenge Solutions:
- 10% (v/v) Fetal Bovine Serum (FBS) in PBS.
- Or a clarified cell lysate (e.g., E. coli lysate expressing a protein of interest) in an appropriate buffer.
- Regeneration Solution (if needed): 10 mM Glycine-HCl, pH 2.0.
Procedure:
- Instrument Priming:
- Install the prepared SAM substrate into the SPR instrument according to the manufacturer's instructions.
- Prime the fluidic system with running buffer (PBS) until a stable baseline is achieved.
Baseline Acquisition:
- Flow the running buffer over the SAM surface at a constant rate (e.g., 10-30 μL/min) for at least 10 minutes to establish a stable refractive index baseline.
Sample Injection and Adsorption Phase:
- Switch the flow to the challenge solution (e.g., 10% FBS).
- Inject the solution for a sufficient time (e.g., 10-15 minutes) to allow for protein adsorption to reach equilibrium. Observe the sensorgram for an increase in response units (RU), which indicates mass binding.
Dissociation Phase:
- Switch back to the running buffer and flow for another 10-15 minutes. This allows any loosely bound proteins to dissociate from the surface.
Data Analysis:
- Measure the total change in RU from the start of the injection to the end of the dissociation phase. This value corresponds to the amount of protein irreversibly bound to the surface.
- Compare the final RU values for different SAMs. A lower RU indicates superior fouling resistance.
The Scientist's Toolkit: Essential Research Reagents
Table 3: Key Reagent Solutions for SAM-based Fouling Resistance Research
| Reagent / Material | Function and Role in Research | Key Considerations |
|---|---|---|
| Alkanethiols (e.g., 1-Dodecanethiol, 11-Mercapto-1-undecanol) | The molecular building blocks that form the SAM. Their terminal group defines surface properties. | Purity is critical. Store under inert atmosphere (N₂/Ar) to prevent oxidation. |
| PEG-Terminated Alkanethiols (e.g., HS-(CH₂)₁₁-EG₆) | Creates a highly hydrophilic, protein-resistant background. Often used as a benchmark. | EG₃ (tri-ethylene glycol) is often sufficient for strong resistance [17]. |
| Absolute Ethanol | The preferred solvent for alkanethiol solutions during SAM formation. | Must be of high purity and anhydrous to prevent contamination and ensure uniform SAM formation. |
| Gold-coated Substrates (SPR chips, slides) | The solid support that facilitates the covalent attachment of thiols to form the SAM. | A gold thickness of ≥ 100 Å ensures optical transparency for microscopy while providing a continuous film [17]. |
| Complex Biofluids (Serum, Lysate) | Real-world challenge media used to test the efficacy of fouling-resistant surfaces under realistic conditions. | Clarify by centrifugation and filtration (0.22 μm) before use in fluidic systems to prevent clogging. |
Visualization of Experimental Workflows and Concepts
Workflow for SAM Fabrication and Testing
Diagram 1: SAM fabrication and testing workflow.
Structural Concepts of SAM-based Fouling Resistance
Diagram 2: Fouling resistance mechanisms of SAMs.
Self-assembled monolayers (SAMs) represent a cornerstone of surface science, enabling precise control over interfacial properties for applications ranging from biomedical devices to biosensing. For research focused on reducing non-specific adsorption (NSA) on gold surfaces, a deep understanding of three core surface properties is paramount: wettability, packing density, and terminal group functionality. These properties collectively determine the effectiveness of a SAM in creating a non-fouling surface. Wettability, often quantified by the water contact angle, dictates the surface's hydrophobic or hydrophilic character, which influences protein and cell adhesion. Packing density, governed by the molecular structure and assembly conditions, determines the structural integrity and barrier properties of the monolayer. Finally, the chemical nature of the terminal group provides the primary interface for interacting with the biological environment. This Application Note provides detailed protocols and data for the fabrication and characterization of SAMs on gold, with an emphasis on correlating these fundamental properties with NSA performance for researchers and drug development professionals.
Experimental Protocols
Synthesis of Alkanethiols and Perfluoroalkanethiols
The molecular structure of the thiol precursor is a critical determinant of the final SAM properties. The following protocol, adapted from studies on perfluorinated alkanethiols, can be modified for various hydrocarbon and fluorocarbon chains [20].
Materials:
- Precursor: 1-iodo-1H,1H,2H,2H-perfluoroalkane (select chain length as needed, e.g., perfluorooctane).
- Reagents: Thiol acetic acid (freshly distilled), Sodium hydride (NaH, 60% dispersion in mineral oil), Lithium aluminum hydride (LAH), Tetrahydrofuran (THF, distilled from CaH₂), Methanol (MeOH), Methylene chloride, Hexanes.
- Equipment: Flame-dried round bottom flasks, Schlenk line for nitrogen atmosphere, magnetic stirrer, heating mantle, chromatography equipment for purification.
Procedure:
- Synthesis of Thioacetate Intermediate: In a flame-dried 250 ml round bottom flask under nitrogen, add dry THF (40 ml) and NaH (18.99 mmol, 0.76 g of 60% dispersion). Cool the slurry to 0°C. Add thiol acetic acid (18.99 mmol, 1.44 g) dropwise over 15 minutes. Stir at 0°C for 45 minutes, then cool further to -78°C. Add the 1-iodo-perfluoroalkane (e.g., 1-iodo-perfluorooctane, 6.32 mmol, 3 g) dropwise to the slurry. After 3 hours, remove the cold bath and allow the mixture to warm to room temperature. Quench the reaction by adding methanol (20 ml) and stirring for 15 minutes.
- Purification: Evaporate volatile organics under vacuum. Add de-ionized H₂O (40 ml) and extract the product with methylene chloride (3 × 20 ml). Purify the crude product via flash chromatography using hexanes as the eluent. The thioacetate intermediate (e.g., 1H,1H,2H,2H-perfluorooctanethiol acetate) is obtained as a yellow oil after solvent evaporation [20].
- Reduction to Thiol: In a second flame-dried 250 ml flask under nitrogen, add LAH (1.18 mmol, 0.095 g) and dry THF (30 ml). Cool the slurry to -78°C. Dissolve the thioacetate intermediate (1.18 mmol, e.g., 0.50 g) in dry THF and add it dropwise to the LAH slurry over 20 minutes. Stir at -78°C for 45 minutes. Carefully quench the reaction with methanol (20 ml) and stir for an hour while warming to room temperature.
- Work-up: Extract the final perfluoroalkanethiol product using standard aqueous workup and purification techniques. Confirm the structure and purity via ( ^1 )H NMR spectroscopy [20].
Fabrication of SAMs on Gold Substrates
This protocol describes the formation of SAMs from synthesized or commercially available alkanethiols on polycrystalline gold surfaces.
Materials:
- Substrate: Template-stripped or evaporated gold on a silicon wafer/mica substrate.
- SAM Solution: 1 mM solution of the alkanethiol in absolute ethanol.
- Cleaning Reagents: Piranha solution (3:1 v/v concentrated H₂SO₄:30% H₂O₂) - CAUTION: Highly corrosive and reactive. Handle with extreme care in a fume hood., absolute ethanol.
- Equipment: UV-Ozone cleaner, Teflon sample holders, glass vials with PTFE-lined caps, nitrogen gun.
Procedure:
- Substrate Cleaning: Clean the gold substrates thoroughly. This can be achieved by treatment with UV-ozone for 20-30 minutes or by immersion in piranha solution for 10-15 seconds, followed by extensive rinsing with ultra-pure water and absolute ethanol. Dry the substrates under a stream of nitrogen.
- SAM Formation: Immerse the clean, dry gold substrates into the 1 mM alkanethiol solution in ethanol. Ensure the substrate is fully submerged.
- Incubation: Allow the self-assembly process to proceed for a minimum of 18-24 hours at room temperature in a sealed vial to prevent solvent evaporation.
- Rinsing and Drying: After incubation, remove the substrates from the solution and rinse them thoroughly with copious amounts of pure ethanol to remove physisorbed molecules. Dry the SAM-coated substrates under a stream of clean, dry nitrogen gas.
- Storage: Store the prepared SAMs in a clean, dark, and dry environment. For best results, use them within 24-48 hours of preparation.
Characterization of Surface Properties
A multi-technique approach is essential for comprehensively characterizing SAM properties.
I. Wettability by Contact Angle Goniometry
- Principle: Measures the angle a liquid droplet makes with the solid surface to quantify surface energy and wettability.
- Protocol: Use a contact angle goniometer. Place a 2-5 µL droplet of ultra-pure water on the SAM surface. Capture an image of the droplet and measure the static contact angle using the sessile drop method. Take measurements at multiple locations on the sample to ensure homogeneity. A higher contact angle indicates greater hydrophobicity [21] [22].
II. Packing Density and Molecular Orientation by NEXAFS
- Principle: Near Edge X-ray Absorption Fine Structure (NEXAFS) probes the orientation of molecular orbitals via the dependence of absorption on the angle of incident X-rays.
- Protocol: Perform NEXAFS experiments at a synchrotron beamline. Record partial electron yield (PEY) spectra at the carbon K-edge. Vary the angle of incidence (e.g., 20°, 55°, and 90° relative to the surface). The intensity of π* resonances associated with C=C or C=O bonds is strongly angle-dependent. The difference in spectral intensity at different angles is used to calculate the molecular tilt angle and degree of ordering within the SAM [20].
III. Terminal Group Composition by X-ray Photoelectron Spectroscopy (XPS)
- Principle: XPS determines the elemental composition and chemical bonding environment in the top ~10 nm of a material.
- Protocol: Use a monochromatic Al Kα X-ray source. Acquire high-resolution spectra for relevant core levels (e.g., C 1s, F 1s, S 2p). For perfluorinated SAMs, the C 1s spectrum will show distinct peaks for CF₃, CF₂, and C-S/C-C components. Angle-dependent XPS (taking measurements at take-off angles of 90° and, for example, 30°) can provide depth distribution information, confirming the surface enrichment of the terminal CF₃ group [20].
Data Presentation and Analysis
Quantitative Data on SAM Properties
Table 1: Impact of Perfluoroalkanethiol Chain Length on SAM Structure and Wettability [20]
| Perfluoroalkanethiol Chain Length (Rf Carbons) | Average Molecular Tilt Angle (from NEXAFS) | Surface CF₃ Group Enrichment (from XPS) | Water Contact Angle (°) |
|---|---|---|---|
| F4 (CF₃(CF₂)₃CH₂CH₂SH) | High disorder, poorly organized | Low (<50%, significant hydrocarbon contamination) | Not Reported |
| F6 (CF₃(CF₂)₅CH₂CH₂SH) | Moderate order | Significant | Not Reported |
| F8 (CF₃(CF₂)₇CH₂CH₂SH) | High order | Significant | Not Reported |
| F10 (CF₃(CF₂)₉CH₂CH₂SH) | Highest order, nearly perpendicular to surface | Substantial | Not Reported |
Table 2: Critical Contact Angles for Effective Flotation of Solid Particles [22]. This data illustrates the concept of a wettability threshold for macroscopic surface behavior, which is analogous to NSA prevention.
| Electrostatic Interaction Condition | Critical Contact Angle (°) for Flotation | Implication for NSA |
|---|---|---|
| Attractive or Weakly Repulsive | ~25° | A minimum hydrophobicity is required to prevent wetting and initial adsorption. |
| Strongly Repulsive | ~62° | In challenging (e.g., highly charged) environments, a much higher surface hydrophobicity is needed to resist interactions. |
Visualizing the SAM Fabrication and Structure-Property Relationship
The following diagram illustrates the workflow from molecule synthesis to functional SAM, highlighting the key controlled variables and resulting surface properties.
Diagram 1: From Molecule to Function: The SAM Development Workflow. This chart outlines the logical progression from molecular design choices, through fabrication, to the final surface properties that determine efficacy in reducing Non-Specific Adsorption (NSA).
The Scientist's Toolkit: Research Reagent Solutions
Table 3: Essential Materials for SAM Research on Gold Surfaces
| Item/Category | Specific Examples | Function & Rationale |
|---|---|---|
| Thiol Precursors | 1-iodo-1H,1H,2H,2H-perfluoroalkanes; n-alkanethiols (e.g., C6, C10, C16); ω-functionalized thiols (e.g., OH-terminated) | The molecular building blocks. Determines terminal group functionality, chain length, and internal packing structure of the SAM [20]. |
| Solvents | Absolute Ethanol (anhydrous), Tetrahydrofuran (THF, distilled from CaH₂), Methylene Chloride | High-purity solvents are essential for thiol synthesis (THF) and for the self-assembly process (ethanol) to prevent contamination and ensure high-quality monolayer formation [20]. |
| Reducing Agents | Lithium Aluminum Hydride (LAH), Sodium Borohydride (NaBH₄) | Used in the synthetic pathway to reduce thioacetate intermediates to the final free thiol, enabling SAM formation [20]. |
| Gold Substrates | Template-stripped gold, Evaporated gold on mica/silicon | Provides an atomically flat, clean, and chemically defined (111) surface for highly ordered, epitaxial thiolate binding and SAM formation. |
| Cleaning Agents | Piranha Solution (H₂SO₄/H₂O₂), UV-Ozone Cleaner | Removes organic contaminants from the gold substrate prior to SAM assembly, which is critical for achieving uniform and densely packed monolayers. |
| Characterization Tools | Contact Angle Goniometer, XPS, NEXAFS, ToF-SIMS | A suite of techniques to quantitatively measure the resulting surface properties: wettability, elemental composition, molecular orientation, and surface chemical structure [20]. |
Application in Reducing Non-Specific Adsorption
The data and protocols presented herein provide a roadmap for designing SAMs to minimize NSA. The key conclusions for application are:
- Achieve High Packing Density: Use longer chain molecules (e.g., ≥ C10 for hydrocarbons, ≥ F8 for fluorocarbons) to promote van der Waals interactions and form well-ordered, dense monolayers that act as a physical barrier to adsorbates [20].
- Control Wettability Strategically: Understand the critical wettability threshold required for your specific environment. Highly hydrophobic surfaces (e.g., from CF₃ termination) are effective against many biological adsorbates, but the exact requirement depends on the interplay with electrostatic forces [22].
- Select Terminal Groups Judiciously: Inert terminal groups like oligo(ethylene glycol) or perfluorinated groups (CF₃) are known to resist protein adsorption. The terminal group defines the outermost chemical interface and is the first point of contact with potential adsorbates [21] [20].
By systematically varying the thiol structure and using the characterized SAM properties—especially a high packing density, optimized contact angle, and a bio-inert terminal group—researchers can rationally design gold surfaces with significantly reduced NSA, thereby enhancing the performance of sensors, biomedical implants, and diagnostic platforms.
Building Effective Barriers: SAM Compositions and Biosensor Applications
Self-assembled monolayers (SAMs) on gold surfaces provide a powerful platform for controlling the interface between a sensor or biomedical device and its biological environment. A primary challenge in developing such interfaces is minimizing nonspecific adsorption (NSA) of proteins and other biomolecules, which can lead to signal interference, biofouling, and reduced performance. The selection of appropriate molecules for SAM construction is therefore critical. This guide details the properties, applications, and practical protocols for three key classes of molecules used to create low-fouling surfaces: 6-mercapto-1-hexanol (MCH), ethylene glycol-based thiols (EG), and zwitterionic thiols. The content is framed within the context of a broader thesis on reducing NSA on gold surfaces, providing researchers with the necessary tools to design effective and stable SAMs.
Key Molecule Classes and Their Properties
The effectiveness of a SAM in resisting NSA is largely determined by the chemical properties of its constituent thiols. Below is a detailed comparison of the most commonly used molecules.
Table 1: Key Molecule Classes for Constructing Low-Fouling SAMs on Gold
| Molecule Class | Specific Examples | Key Properties & Mechanism | Typical Application Context | Advantages | Limitations |
|---|---|---|---|---|---|
| Short-Chain Alkanethiols | 6-Mercapto-1-hexanol (MCH) | Hydrophilic terminal group; displaces non-specifically adsorbed aptamers and dilutes the sensing layer to reduce steric hindrance and NSA [2]. | Primarily used as a diluent or blocking agent in electrochemical aptasensors [2]. | Good compromise between stability and sensitivity; widely available and easily implemented [2]. | Can exhibit signal drift due to SAM desorption over time; offers moderate antifouling resistance [2]. |
| Zwitterionic Thiols | Sulfobetaine thiol (e.g., DPS: 3-((3-mercaptopropyl)dimethylammonio)propane-1-sulfonate) [23] | Possess both positive and negative charges; form a hydration layer via electrostatic interactions that resists protein adsorption [23]. | Excellent for sensors operating in complex, protein-rich media like blood plasma [23]. | Superior resistance to nonspecific interactions and biofouling in human plasma; high stability and biocompatibility [23]. | Requires synthesis; may need optimization of mixing ratios with functional thiols (e.g., MUA). |
| Mixed Short-Chain Thiols | 2-Thiophenethiol (TT) / 2-Mercaptoethanol (ME) mixtures [24] | Aromatic TT and hydrophilic ME allow fine-tuning of surface properties. The 1:1 ratio promoted specific biological responses (neurite outgrowth) while minimizing adverse reactions [24]. | Neural interfaces and applications where specific cell-surface interactions are desired beyond just antifouling [24]. | Enables precise control over surface chemistry and cellular responses; intermediate electrochemical properties. | Requires characterization of mixed monolayer structure and composition. |
Quantitative Performance Data
To guide the selection process, it is essential to consider quantitative data on the performance of different SAM formulations. The following table summarizes key findings from recent studies.
Table 2: Quantitative Performance Comparison of SAM Formulations
| SAM Composition | Assembly Method | Key Performance Metrics | Results |
|---|---|---|---|
| MCH/Aptamer (Mixed SAM) [2] | Passive incubation (conventional) | SAM Stability | Extended thiol immobilization time enhanced mixed SAM stability [2]. |
| MUA + MCH (Binary Mixed SAM) [23] | Potential-assisted assembly | Fabrication Time | >200 times shorter (5 minutes) than passive incubation [23]. |
| MUA + DPS (Zwitterionic Binary SAM) [23] | Potential-assisted assembly | Antifouling Performance | Superior resistance to nonspecific interactions compared to MCH; negligible matrix effect in LPS-spiked human plasma [23]. |
| TLR4/MUA-DPS/Au Sensor [23] | Potential-assisted assembly | Biosensor Performance (Detection of E. coli endotoxin) | Detection Limit: 4 ng mL⁻¹Dynamic Range: Up to 1000 ng mL⁻¹ [23] |
| TT:ME (1:1 Ratio Mixed SAM) [24] | Passive incubation | Biological Response | Promoted enhanced neurite outgrowth while minimizing astrocytic activation, indicating improved tissue integration for neural interfaces [24]. |
Detailed Experimental Protocols
Protocol 1: Conventional Passive Incubation for Mixed MCH/Aptamer SAMs
This protocol is adapted from the development of an erythromycin aptasensor and is typical for constructing electrochemical biosensors [2].
Research Reagent Solutions:
- Thiol-modified DNA Aptamer Solution: Dilute the aptamer in a suitable buffer (e.g., Tris-EDTA or phosphate buffer) to a concentration typically ranging from 0.1 to 1 µM.
- MCH Blocking Solution: Prepare a 1-10 mM solution of 6-mercapto-1-hexanol in absolute ethanol.
- Immobilization Buffer: Typically 10-50 mM Tris or phosphate buffer, often containing 50-100 mM NaCl and 5-10 mM MgCl₂ to shield the negative charge of the DNA backbone and promote adsorption [2].
Procedure:
- Substrate Preparation: Clean the polycrystalline gold electrode or gold nanoparticle-modified substrate. A standard method involves electrochemical cycling in 0.5 M H₂SO₄ or piranha solution treatment, followed by thorough rinsing with deionized water and drying under a stream of nitrogen.
- Aptamer Immobilization: Incubate the clean gold electrode in the thiol-modified aptamer solution for a defined period, typically 1 to 18 hours, at room temperature.
- Rinsing: Rinse the electrode gently with nuclease-free water and then with immobilization buffer to remove physisorbed aptamer strands.
- MCH Backfilling: Incubate the aptamer-functionalized electrode in the MCH solution for 15 minutes to 2 hours. This step displaces any non-specifically adsorbed aptamers and creates a well-diluted, oriented monolayer.
- Final Rinsing and Drying: Rinse the electrode sequentially with ethanol and immobilization buffer to remove excess MCH. Dry gently with nitrogen before use or further characterization.
Protocol 2: Rapid, Potential-Assisted Assembly of Mixed Zwitterionic SAMs
This advanced protocol enables the formation of highly reproducible, compact SAMs in minutes rather than hours, using a potentiostat [23].
Research Reagent Solutions:
- Mixed Thiol Solution: Prepare a 1 mM total thiol solution in absolute ethanol. The optimal ratio for MUA and the zwitterionic thiol (DPS) was found to be 1:9 (MUA:DPS) [23].
- Electrolyte Solution: 0.1 M KNO₃ in deionized water.
- PBS Buffer: Standard phosphate-buffered saline, pH 7.4.
Procedure:
- Electrochemical Cell Setup: Place the gold working electrode, a platinum counter electrode, and a reference electrode (e.g., Ag/AgCl) in the electrolyte solution.
- Application of Potential: Apply a constant DC potential of -0.4 V vs. Ag/AgCl to the gold working electrode.
- SAM Formation: While the potential is applied, add the mixed thiol solution directly to the electrochemical cell to achieve a final concentration of ~1 µM. Allow the self-assembly to proceed for 5 minutes [23].
- Rinsing and Drying: After 5 minutes, remove the electrode, rinse it copiously with absolute ethanol to remove loosely adsorbed thiols, and dry under a stream of nitrogen.
- Functionalization: The resulting SAM presents a surface rich in carboxylic acid groups from MUA, which can be activated with EDC/NHS for the covalent immobilization of biorecognition elements like proteins or antibodies [23].
The Scientist's Toolkit: Essential Research Reagents
Table 3: Key Reagents for SAM Construction on Gold
| Reagent / Material | Function / Role in SAM Development |
|---|---|
| 11-Mercaptoundecanoic acid (MUA) | A long-chain thiol that provides a terminal carboxylic acid group for the covalent attachment of biorecognition elements (proteins, antibodies) via EDC/NHS chemistry [23]. |
| 6-Mercapto-1-hexanol (MCH) | A short-chain alkanethiol used as a diluent and blocking agent to displace non-specifically adsorbed biomolecules, orient probe strands, and reduce nonspecific binding [2]. |
| Zwitterionic Sulfobetaine Thiol (DPS) | A thiol that forms an ultra-low fouling surface by creating a strong hydration layer, highly effective for use in complex biological samples like blood plasma [23]. |
| 2-Thiophenethiol (TT) & 2-Mercaptoethanol (ME) | Short-chain thiols used in combination to fine-tune surface properties for specific biological applications, such as neural interfaces [24]. |
| EDC & NHS Crosslinkers | Activating agents used to convert the terminal carboxylic acids of thiols like MUA into reactive esters for coupling to primary amines on proteins [23]. |
| Potassium Ferri/Ferrocyanide Redox Probe | Used in electrochemical characterization (Cyclic Voltammetry, EIS) to assess the quality, packing density, and barrier properties of the formed SAM [2]. |
Visualization of SAM Design and Signaling Pathways
Self-assembled monolayers (SAMs) of alkanethiolates on gold represent a class of model organic surfaces that provide exceptional control over interfacial structure and properties, making them indispensable for fundamental research on reducing nonspecific adsorption (NSA) [17]. These monolayers form spontaneously when a gold-coated substrate is immersed in a solution of alkanethiols, resulting in a densely packed, ordered surface [17]. A primary application of well-constructed SAMs is the creation of bio-inert surfaces that minimize the non-specific binding of proteins and other biomolecules, a critical requirement for the reliability of biosensors, clinical diagnostics, and studies of specific cell-matrix interactions [19] [17]. These protocols detail the materials and methods for constructing SAMs on gold surfaces, with a focus on techniques to mitigate NSA.
Research Reagent Solutions
The following table lists key reagents essential for the formation and characterization of SAMs on gold.
| Reagent/Material | Function/Explanation |
|---|---|
| Alkanethiols (e.g., 1-Octadecanethiol) | The molecular building blocks of the SAM. Their long alkane chains (e.g., C18) form a stable, crystalline-like layer on the gold surface via the thiol group, creating a hydrophobic surface [19] [17]. |
| EG(_n)-terminated Alkanethiols (e.g., tri(ethylene glycol)) | Serves as an inert, non-fouling component of mixed SAMs. The ethylene glycol (EG) groups are highly effective at preventing the non-specific adsorption of proteins due to their highly hydrated nature [17]. |
| PEG-type Hydrophilic Spacer (e.g., Compound 1 from [19]) | A specialized poly(ethylene glycol) spacer used as a hydrophilic layer between the gold surface and a ligand. It significantly reduces nonspecific interactions from complex mixtures like cell lysates [19]. |
| Absolute Ethanol | A high-purity solvent used for preparing alkanethiol solutions. It ensures clean formation of SAMs without water-induced defects or contamination. |
| Gold-coated Substrates (e.g., on glass or silicon wafer) | Provides the foundational surface for SAM formation. The gold (111) crystal face is typically used for forming highly ordered, well-defined monolayers [17]. |
| Bovine Serum Albumin (BSA) | Often used as a "blocking" protein in an attempt to passivate any remaining "sticky" sites on a surface after SAM formation, though its effectiveness on well-defined SAMs can be limited [17]. |
Quantitative Data on SAM Stability and Protein Binding
The stability of the SAM and its effectiveness in reducing NSA are critically dependent on the molecular structure of the alkanethiols. The data below summarize key experimental findings.
Table 1: Impact of Alkanethiol Chain Length and Surface Chemistry on SAM Properties
| Parameter Tested | Experimental Finding | Implication for SAM Performance |
|---|---|---|
| Critical Methylene Chain Length (n) for stable SAMs [19] | SAMs with a methylene chain length (n) of 11 or greater demonstrated stability in aqueous solutions. Shorter chains resulted in less stable monolayers. | Using alkanethiols with sufficiently long chains (e.g., n ≥ 11) is essential for creating a stable, defect-free SAM that can withstand subsequent experimental conditions. |
| Surface Hydrophilicity vs. NSA [19] [17] | Introduction of a PEG-based hydrophilic spacer significantly reduced nonspecific protein binding from rat brain lysate compared to standard hydrophobic alkanethiol SAMs. | Hydrophilic surfaces, particularly those presenting oligo(ethylene glycol) groups, are highly effective at creating non-fouling surfaces for biomedical applications. |
| Ligand Density for Specific Binding [17] | Cell adhesion mediated by RGD peptides was efficient on SAMs presenting the ligand at densities of 0.5% mixed with tri(ethylene glycol) groups. Higher crowding (e.g., with hexa(ethylene glycol)) reduced affinity. | Both the density and local microenvironment of a bioactive ligand on a SAM are critical for controlling its specific interactions with target receptors. |
Experimental Protocol: Fabrication of Mixed SAMs for Reduced NSA
This protocol describes the creation of a mixed SAM consisting of an inert, protein-resistant background and a small fraction of a functionalized alkanethiol for ligand immobilization.
Substrate Preparation and SAM Formation
Materials:
- Gold-coated glass chips or slides (≥ 100 Å gold thickness)
- 1 mM solution of tri(ethylene glycol)-terminated alkanethiol (EG(_3)-thiol) in absolute ethanol
- 1 mM solution of a functional alkanethiol (e.g., carboxylate- or amine-terminated) in absolute ethanol
- Absolute ethanol
- Nitrogen gas stream
Procedure:
- Gold Substrate Cleaning: Clean the gold substrates immediately before use. A standard method involves immersion in freshly prepared piranha solution (Note: Piranha solution is extremely dangerous and must be handled with extreme care, using appropriate personal protective equipment and protocols) for 10-15 minutes, followed by thorough rinsing with copious amounts of pure water and absolute ethanol. Alternatively, plasma cleaning can be used.
- Preparation of Mixed Alkanethiol Solution: Prepare a mixed alkanethiol solution in absolute ethanol with a molar ratio of 99.5% EG(_3)-thiol to 0.5% functionalized thiol. This ratio creates a surface that is predominantly non-fouling while presenting a low density of sites for specific ligand attachment [17].
- SAM Assembly: Immerse the clean, dry gold substrate into the mixed alkanethiol solution. Allow the self-assembly process to proceed for a minimum of 12-24 hours at room temperature in a sealed container, protected from light.
- Rinsing and Drying: After assembly, remove the substrate from the solution and rinse it thoroughly with pure ethanol to remove any physisorbed thiols. Dry the substrate under a gentle stream of nitrogen gas.
Ligand Immobilization via Covalent Coupling
This section outlines a common carbodiimide chemistry approach for coupling amine-containing ligands to a carboxylate-terminated SAM.
Materials:
- SAM from Section 4.1 with terminal carboxylate groups
- Amine-containing ligand (e.g., a peptide, aptamer)
- 0.4 M EDC·HCl (N-Ethyl-N'-(3-dimethylaminopropyl)carbodiimide hydrochloride) in water
- 0.1 M NHS (N-Hydroxysuccinimide) in water
- Reaction buffer (e.g., 10 mM MES, pH 5.5)
Procedure:
- Activation of Carboxylates: Place the carboxyl-terminated SAM in a reaction vessel. Incubate the surface with a freshly prepared mixture of EDC and NHS in reaction buffer for 30-60 minutes at room temperature. This step converts the stable carboxyl groups into an active NHS ester, which is more reactive with amine groups.
- Rinsing: Rinse the substrate with reaction buffer to remove excess EDC/NHS.
- Ligand Coupling: Immediately incubate the activated SAM with a solution of your amine-containing ligand (typically in a phosphate buffer at pH 7.0-7.4) for 2-4 hours.
- Quenching and Storage: After coupling, rinse the substrate with buffer and then incubate it in a quenching solution (e.g., 1M ethanolamine, pH 8.5, or 100 mM glycine) for 30 minutes to deactivate any remaining active esters. The functionalized SAM can be stored in an appropriate buffer at 4°C.
The following workflow diagram illustrates the complete experimental process from substrate preparation to final application.
Characterization and Troubleshooting
- Verification of Non-fouling Properties: The effectiveness of the SAM in reducing NSA should be validated using a relevant analytical technique. Surface Plasmon Resonance (SPR) is ideal for real-time, in-situ measurement of protein adsorption [19] [17]. A successful non-fouling SAM will show minimal signal change when exposed to complex protein mixtures like blood serum or cell lysates.
- Troubleshooting Common Issues:
- High NSA: This can result from an unstable SAM. Ensure the alkanethiols have a sufficient chain length (n ≥ 11) and that the gold substrate was thoroughly cleaned before assembly [19].
- Low Specific Signal: This may be due to over-crowding or incorrect orientation of the immobilized ligand. Optimize the ratio of functional to inert alkanethiol in the initial solution and ensure the coupling chemistry is appropriate for the ligand [25] [17].
The performance of biosensors and diagnostic assays is critically dependent on the interface chemistry between the biological recognition element and the transducer surface. Self-assembled monolayers (SAMs) on gold provide a versatile platform for immobilizing ligands such as antibodies, aptamers, or carbohydrates while minimizing non-specific adsorption (NSA) through incorporation of backfill molecules. Achieving optimal biosensor sensitivity and specificity requires precise control over the surface density of functional ligands, which is primarily governed by the ratio of ligand-to-backfill molecules during SAM formation. An improper ratio can lead to excessive ligand density causing steric hindrance, or insufficient density reducing capture capacity, while inadequate backfill coverage results in increased NSA and false-positive signals. This Application Note details protocols for determining and optimizing these critical ratios across various application scenarios, providing researchers with methodologies to enhance assay performance through controlled surface engineering.
Theoretical Background and Significance
Mixed SAMs typically consist of two primary components: a thiolated ligand with specific biorecognition capability (e.g., antibodies, aptamers, carbohydrates) and a diluting or backfill molecule (e.g., oligo(ethylene glycol) [OEG]-terminated thiols) that resists non-specific protein adsorption. The central challenge lies in balancing sufficient ligand density for effective target capture against sufficient backfill density to minimize NSA. This balance is governed by the cluster glycoside effect in carbohydrate systems and optimal spacing requirements for protein ligands, which facilitate multivalent interactions while preventing steric interference between adjacent recognition elements.
Research indicates that the optimal ratio varies significantly depending on the application:
For carbohydrate-lectin systems, mixed SAMs containing 8-mercaptooctyl α-D-mannopyranoside with n-octanethiol showed maximal Concanavalin A (Con A) binding at 1:9 solution molar ratio on flat gold surfaces, while nanoporous gold (NPG) surfaces performed optimally at 1:19 solution molar ratio [26]. This demonstrates how substrate morphology influences optimal composition.
For his-tagged protein immobilization, mixed SAMs containing nitrilotriacetic acid (NTA) headgroups and OEG moieties achieved effective site-specific immobilization with controlled density, though specific optimal ratios weren't provided [27].
For antibody-based detection, several functionalization strategies based on mercaptoundecanoic acid SAMs demonstrated effectiveness for protein detection but proved inadequate for whole bacteria capture, highlighting how target size influences optimal surface chemistry [28].
Table 1: Optimal Ligand:Backfill Ratios for Different Applications
| Ligand Type | Backfill Molecule | Substrate | Optimal Ratio (Ligand:Backfill) | Target | Performance Metric |
|---|---|---|---|---|---|
| 8-mercaptooctyl α-D-mannopyranoside [26] | n-octanethiol | Flat Au | 1:9 (solution molar ratio) | Con A | Highest binding response |
| 8-mercaptooctyl α-D-mannopyranoside [26] | n-octanethiol | Nanoporous Au (NPG) | 1:19 (solution molar ratio) | Con A | Highest binding response |
| NTA thiol [27] | OEG thiol | Flat Au | Varied (sequential adsorption) | his-tagged proteins | Controlled immobilization |
Table 2: Additional Parameters Affecting SAM Performance
| Parameter | Effect | Optimal Conditions |
|---|---|---|
| SAM incubation time [29] | Reduced NSA with increased time; short-chain SAMs respond more favorably | Varies by chain length |
| Surface roughness [29] | Long-chain SAMs reduced NSA by 75% on rougher surfaces (4.4 nm vs 0.8 nm RMS) | Smo surfaces (0.8 nm RMS) preferred |
| Gold crystal orientation [29] | Profound NSA reduction on short-chain SAMs with (1 1 1) orientation | Strong directional alignment along (1 1 1) |
Experimental Protocols
Protocol 1: Determining Optimal Ratio for Carbohydrate-Lectin Systems
4.1.1 Materials and Reagents
- 8-mercaptooctyl α-D-mannopyranoside (ligand)
- n-octanethiol (backfill molecule) or alternative OEG-terminated thiol
- Absolute ethanol (200-proof)
- Gold substrates (flat or nanoporous)
- Phosphate buffered saline (PBS), pH 7.4
- Target lectin (e.g., Concanavalin A)
4.1.2 Procedure
- Prepare mixed SAM solutions with varying ligand:backfill molar ratios (e.g., 1:1, 1:4, 1:9, 1:19, 1:99) in absolute ethanol.
- Clean gold substrates with oxygen plasma or piranha solution followed by thorough rinsing with ethanol and drying under nitrogen.
- Immerse substrates in prepared solutions for 24 hours at room temperature in sealed containers.
- Remove substrates and rinse sequentially with pure ethanol, ethanol with 2% (v/v) acetic acid, and deionized water.
- Dry substrates under nitrogen stream and store under nitrogen until use.
- Characterize SAM formation using electrochemical impedance spectroscopy (EIS) or surface plasmon resonance (SPR).
- Expose to target lectin (e.g., 0.1-100 µg/mL Con A in PBS) for 30-60 minutes.
- Quantify binding using EIS, SPR, or other appropriate detection methods.
- Identify optimal ratio that provides maximal specific binding with minimal non-specific adsorption.
Protocol 2: Sequential Adsorption for NTA/OEG Mixed SAMs
4.2.1 Materials and Reagents
- NTA-thiol (e.g., nitrilotriacetic acid-terminated tetra(ethylene glycol) undecythiol)
- OEG-thiol (e.g., hydroxyl-terminated tetra(ethylene glycol) undecythiol)
- Absolute ethanol
- Gold substrates
- Nickel sulfate solution (for NTA activation)
4.2.2 Procedure
- Clean gold substrates as described in Protocol 1.
- Prepare pure NTA thiol solution (0.1 mM in ethanol).
- Immerse substrates in NTA thiol solution for 5 minutes to form initial monolayer.
- Rinse thoroughly with ethanol to remove loosely bound NTA thiol.
- Prepare OEG thiol solution (0.1 mM in ethanol).
- Backfill by immersing NTA-covered samples in OEG thiol solution for varying times (0.5-48 hours).
- Remove and rinse sequentially with ethanol, ethanol with 2% acetic acid, and water.
- Dry under nitrogen and characterize using XPS or ToF-SIMS.
- Activate with Ni(II) by treating with NiSO₄ solution (10-50 mM for 5-10 minutes).
Protocol 3: Optimization for Antibody-Based Detection Systems
4.3.1 Materials and Reagents
- Mercaptoundecanoic acid (MUA) or similar carboxylic acid-terminated thiol
- OEG-terminated backfill thiol (e.g., mercaptohexanol or longer-chain equivalents)
- Antibodies (specific to target)
- EDC/NHS coupling reagents
- Blocking solution (e.g., BSA, casein)
4.3.2 Procedure
- Prepare mixed SAM solutions with varying MUA:OEG ratios.
- Form SAMs on gold substrates by immersion for 18-24 hours.
- Activate carboxyl groups using EDC/NHS chemistry (typically 0.2M EDC/0.05M NHS for 30 minutes).
- Immobilize antibodies by exposing to specific concentration (typically 10-100 µg/mL) for 1-2 hours.
- Block remaining active sites with appropriate blocking solution.
- Evaluate performance by exposing to target analyte and measuring specific vs. non-specific binding.
- For bacterial detection, consider physisorption as an alternative to SAM-based incorporation [28].
The Scientist's Toolkit: Essential Research Reagents
Table 3: Key Reagents for Mixed SAM Formation and Characterization
| Reagent/Material | Function/Application | Examples/Specifications |
|---|---|---|
| NTA-thiol | Immobilization of his-tagged proteins | Nitrilotriacetic acid-terminated tetra(ethylene glycol) undecythiol [27] |
| Carbohydrate-thiol conjugates | Lectin binding studies | 8-mercaptooctyl α-D-mannopyranoside [26] |
| OEG-terminated thiols | Resistance to non-specific adsorption | Hydroxyl-terminated tetra(ethylene glycol) undecythiol [27] |
| Carboxylic acid-terminated thiols | Antibody immobilization via EDC/NHS chemistry | Mercaptoundecanoic acid [28] |
| Short-chain alkane thiols | Diluting agents in mixed SAMs | n-octanethiol, 6-mercapto-1-hexanol [2] [26] |
| Gold substrates | SAM support | Flat gold films, nanoporous gold, gold nanoparticles [29] [26] |
Workflow and Signaling Pathways
Figure 1: Workflow for optimizing mixed monolayer ratios, showing the iterative process of SAM formation, characterization, and performance testing to determine optimal ligand:backfill ratios.
Figure 2: Effects of ligand:backfill ratio variations on biosensor performance, demonstrating how improper ratios lead to suboptimal performance while balanced ratios maximize specific binding and minimize interference.
Troubleshooting and Technical Notes
Surface roughness effects: Long-chain SAMs reduce NSA by 75% on rougher surfaces (4.4 nm RMS vs. 0.8 nm RMS) [29]. Characterize surface topography before SAM formation.
Gold crystal orientation: Strong directional alignment along (1 1 1) profoundly reduces NSA on short-chain SAMs [29]. Consider using template-stripped gold for optimal results.
Incubation time: Extended SAM incubation time reduces NSA, with short-chain SAMs responding more favorably than long-chain SAMs [29]. Optimize incubation time for each system.
Stability concerns: For aptamer-based sensors, ensure mixed SAM stability by optimizing immobilization time and considering pure gold surfaces rather than gold-coated carbon electrodes to prevent false signals [2].
Verification methods: Include control experiments without ligands to quantify non-specific adsorption, and use multiple characterization techniques (EIS, SPR, XPS) to confirm surface composition and density.
Optimizing the ratio of ligand to backfill molecules in mixed SAMs represents a critical step in developing high-performance biosensing platforms. The protocols outlined herein provide researchers with systematic approaches to determine optimal compositions for various applications, from carbohydrate-lectin interactions to antibody-based detection. The quantitative data presented demonstrates that optimal ratios vary significantly based on substrate morphology, ligand type, and target size, emphasizing the need for empirical optimization in each specific system. Through careful implementation of these protocols and consideration of the troubleshooting guidelines, researchers can significantly enhance biosensor performance by maximizing specific binding interactions while minimizing deleterious non-specific adsorption effects.
Covalent immobilization serves as a foundational technique in biotechnology for permanently affixing biomolecular probes to solid supports. This method generates stable complexes between functional groups on the substrate and complementary groups on the biomolecule, ensuring minimal leakage into the surrounding environment and significantly enhancing the stability of the immobilized molecules [30]. Unlike physical adsorption methods that rely on weak van der Waals forces, hydrophobic interactions, or electrostatic attraction, covalent bonding provides durable attachment that withstands changes in temperature, pH, and surface charge [30].
The performance of immobilized biomolecules critically depends on both the substrate properties and the immobilization method employed [30]. Within the context of self-assembled monolayers (SAMs) on gold surfaces—a system renowned for its well-defined chemical functionality and structural control—covalent immobilization strategies enable precise positioning of biomolecular probes for applications ranging from biosensing to drug development. Gold substrates provide an excellent platform for SAM formation due to gold's resistance to oxidation and corrosion, as well as the relative ease of generating SAMs via adsorption of various surfactants [31].
Fundamental Chemistries for Covalent Immobilization
Covalent immobilization techniques typically utilize the inherent functional groups of biomolecules, including amino, carboxylic, thiol, imidazole, indole, and hydroxyl groups [30]. The selection of appropriate immobilization chemistry depends on which functional groups are available on both the biomolecule and the support material.
Amine-Reactive Immobilization Strategies
Primary amines are the most common targets for protein immobilization, as they exist at the N-terminus of each polypeptide chain and in the side chain of lysine residues [32]. Due to their positive charge under physiological conditions, primary amines are typically displayed on the outer surface of proteins, making them readily accessible for conjugation without denaturing the protein structure [32].
NHS Ester-Activated Supports: NHS esters react with primary amines under slightly alkaline conditions (pH 7.2-8.5) to form stable amide bonds [32]. This reaction is typically performed in phosphate buffer for 0.5 to 4 hours at room temperature or 4°C. Primary amine buffers such as Tris must be avoided as they compete in the reaction, though they can be useful for quenching the reaction upon completion.
Aldehyde-Activated Supports: This reliable method employs reductive amination, where an initial Schiff base forms between aldehyde and amine groups, which is then reduced to a secondary amine by sodium cyanoborohydride (NaCNBH₃) [32]. This reducing agent is mild enough to avoid cleaving disulfide bonds in most proteins. Coupling yields can exceed 85%, with some protocols suggesting a two-step process at different pH levels for optimal efficiency.
Azlactone-Activated Supports: Azlactone groups react with primary amines through a ring-opening process that generates an amide bond without requiring additives or catalysts [32]. The reaction occurs spontaneously within approximately one hour, with coupling efficiency often enhanced by lyotropic salts like sodium citrate that drive protein molecules toward the bead surface.
Carbodiimide and Schiff Base Techniques
Carbodiimide chemistry and Schiff base reactions represent the two most prevalent covalent bond techniques for immobilization, particularly advantageous due to their utilization of amino and carboxylic acid groups commonly found on enzyme surfaces [25]. Carbodiimide-mediated coupling typically employs EDC (1-Ethyl-3-(3-dimethylaminopropyl)carbodiimide) to activate carboxyl groups for subsequent reaction with primary amines, forming stable amide bonds. Schiff base formation occurs through reactions between aldehydes and amines, often stabilized by reduction with cyanoborohydride to create stable secondary amine linkages [25].
Table 1: Comparison of Common Covalent Immobilization Chemistries
| Immobilization Chemistry | Reactive Groups | Bond Formed | Key Advantages | Key Limitations |
|---|---|---|---|---|
| NHS Ester [32] | NHS ester reacts with primary amine | Amide bond | Rapid reaction; stable amide linkage; well-established protocols | Susceptible to hydrolysis; requires controlled pH conditions |
| Aldehyde/Reductive Amination [32] | Aldehyde reacts with primary amine | Secondary amine (after reduction) | High coupling yields (>85%); stable linkage; suitable for antibodies | Requires reducing agent; potential for disulfide reduction with strong agents |
| Azlactone [32] | Azlactone reacts with primary amine | Amide bond | Spontaneous reaction; no additives needed; durable support | May require lyotropic salts for optimal efficiency |
| Carbodiimide (e.g., EDC) [25] [30] | Carboxylate with primary amine | Amide bond | Versatile; utilizes common functional groups; zero-length crosslinker | May require NHS for efficiency; can cause cross-linking |
| Epoxy [30] | Epoxide reacts with amine, thiol, or hydroxyl | Secondary amine, thioether, or ether | Stable bonds; reacts with multiple functional groups; long spacer arms | Slower reaction rate; may require elevated temperatures |
Considerations for SAMs on Gold Surfaces
When working with self-assembled monolayers on gold surfaces, stability emerges as a critical concern. SAMs can be vulnerable to various environmental factors, including elevated temperatures, oxidants, and solvents [31]. Several strategies have been developed to enhance SAM stability:
- Multidentate Adsorbates: Utilizing adsorbates with multiple anchoring groups (e.g., dithiols, trithiols) significantly improves monolayer stability compared to monodentate thiols through enhanced coordination to the gold surface [31].
- Cross-linking Between Adsorbates: Creating physical or chemical bonds between adjacent molecules in the SAM enhances film integrity and resistance to displacement [31].
- Post-assembly Chemical Modification: Transforming the terminal functionality of SAMs after assembly can create more robust interfaces for subsequent biomolecule immobilization [31].
Quantitative Comparison of Immobilization Techniques
The selection of an appropriate immobilization method requires careful consideration of multiple performance parameters. The following table summarizes key characteristics across different covalent approaches, particularly as they relate to creating stable functional interfaces on gold surfaces.
Table 2: Performance Characteristics of Covalent Immobilization Methods
| Method | Stability | Residual Activity | Resistance to Leaching | Orientation Control | Recommended Biomolecules |
|---|---|---|---|---|---|
| NHS Ester [32] | High (amide bond) | Moderate to High | Excellent | Low to Moderate | Antibodies, proteins with available lysines |
| Aldehyde [32] | High (secondary amine) | Moderate to High | Excellent | Moderate | Antibodies, proteins, amines at specific sites |
| EDC/NHS [30] | High (amide bond) | Variable (can be low) | Excellent | Low | Enzymes, proteins with carboxyl/amine pairs |
| Glutaraldehyde [30] | High | Often Low (rigidification) | Excellent | Low | Enzymes, proteins (when amine-rich) |
| Click Chemistry [30] | Very High | High | Excellent | High | Site-specifically modified biomolecules |
The immobilization process can significantly impact biomolecule functionality. For enzymes, covalent immobilization typically enhances stability across various pH and temperature ranges but often at the cost of reduced enzymatic activity [25] [30]. Activity losses up to 98% have been reported, primarily attributed to alterations in active site conformation or immobilization in orientations that sterically hinder substrate access [30]. Quantifying this impact involves comparing specific activity (enzyme activity per milligram of total protein) before and after immobilization, often expressed as residual activity [30].
Experimental Protocols
Protocol 1: Covalent Immobilization via Amine Coupling on NHS-Activated Surfaces
This protocol describes a standard procedure for immobilizing biomolecules containing primary amines onto NHS-activated surfaces, suitable for functionalized gold substrates.
Research Reagent Solutions:
| Reagent/Material | Function in Experiment |
|---|---|
| NHS-activated surface or support [32] | Provides reactive ester groups for covalent amine coupling |
| Biomolecule solution (0.1-1 mg/mL in coupling buffer) [32] | The probe to be immobilized (antibody, enzyme, protein) |
| Coupling Buffer (e.g., 0.1 M phosphate, pH 7.2-8.0) [32] | Provides optimal alkaline pH for efficient amine reaction |
| Quenching Solution (1 M Tris-HCl, pH 7.4) [32] | Blocks unreacted NHS esters by reacting with excess Tris |
| Washing Buffer (e.g., PBS with 0.05% Tween 20) | Removes non-covalently bound biomolecules and excess reagents |
Step-by-Step Procedure:
- Preparation: Equilibrate the NHS-activated surface with 10-15 volumes of coupling buffer. Avoid buffers containing primary amines (e.g., Tris, glycine).
- Immobilization: Incubate the biomolecule solution with the activated surface for 0.5-4 hours at room temperature or 4°C with gentle agitation. Use a biomolecule concentration optimized for your application to avoid over-crowding.
- Quenching: Remove the biomolecule solution and incubate the surface with 1 M Tris-HCl, pH 7.4, for 30-60 minutes to block any remaining reactive groups.
- Washing: Wash sequentially with coupling buffer, washing buffer, and storage buffer to remove non-covalently attached molecules.
- Storage: Store the functionalized surface in appropriate buffer at 4°C until use.
Protocol 2: Covalent Immobilization via Reductive Amination on Aldehyde-Activated Surfaces
This protocol utilizes reductive amination for immobilizing biomolecules through primary amines, particularly effective for antibody immobilization on functionalized gold surfaces.
Research Reagent Solutions:
| Reagent/Material | Function in Experiment |
|---|---|
| Aldehyde-activated surface or support [32] | Provides aldehyde groups for Schiff base formation with amines |
| Sodium Cyanoborohydride (NaCNBH₃) [32] | Selective reducing agent that converts Schiff bases to stable amines |
| Coupling Buffer (e.g., 0.1 M phosphate, pH 7.2, or carbonate, pH 10) [32] | Provides appropriate pH for efficient Schiff base formation |
| Washing Buffer (e.g., PBS) | Removes unbound reagents and biomolecules |
Step-by-Step Procedure:
- Preparation: Equilibrate the aldehyde-activated surface with coupling buffer. For proteins stable at high pH, use carbonate buffer (pH 10) for more efficient Schiff base formation.
- Immobilization: Incubate with the biomolecule solution for 1-2 hours at room temperature.
- Reduction: Add NaCNBH₃ to a final concentration of 10-20 mM and continue incubation for 30 minutes to 2 hours to reduce the Schiff bases to stable secondary amine linkages.
- Washing: Wash thoroughly with washing buffer to remove excess reducing agent and unbound biomolecules.
- Storage: Store in appropriate buffer at 4°C.
The experimental workflow for covalent immobilization, from surface preparation to application, can be visualized as follows:
Experimental Workflow for Covalent Immobilization
Application in Biomolecular Probe Development
Covalent immobilization strategies have enabled the development of sophisticated molecular probes for investigating complex biological systems. Functionalized covalent probes—bioactive ligands that form covalent bonds with their target biomacromolecules—represent particularly powerful tools for target validation, off-target identification, and as starting points for drug discovery programs [33]. These probes typically incorporate three functional elements: a high-affinity ligand for target recognition, a reactive group (warhead) for covalent attachment, and a reporter group for detection [34].
In GPCR research, for example, affinity-based probes (AfBPs) have been developed for a multitude of receptors, including cannabinoid, dopamine, and serotonin receptors [34]. These tools allow robust investigation of GPCRs across expanded experimental methods, including SDS-PAGE and pull-down proteomics, by maintaining the covalent bond between probe and receptor through rigorous washing steps that include surfactants, reductants, and oxidants [34].
The strategic selection of covalent chemistry profoundly impacts probe performance. Different warhead classes offer distinct advantages:
Covalent Probe Warhead Strategies
Covalent immobilization provides a versatile and robust methodology for creating stable, functional interfaces on gold surfaces and other substrates. The selection of appropriate chemistry—whether amine-reactive, carbodiimide-mediated, or based on other principles—must be guided by the specific application requirements, considering trade-offs between stability, activity retention, orientation control, and minimization of non-specific binding. Within the framework of self-assembled monolayers on gold, these immobilization strategies enable the precise presentation of biomolecular probes for advanced applications in biosensing, diagnostics, and drug development. As the field progresses, continued refinement of covalent immobilization techniques will further enhance our ability to engineer functional biointerfaces with precisely controlled properties.
Application in Electrochemical Aptamer-Based (E-AB) Sensors
Electrochemical aptamer-based (E-AB) sensors represent a modular platform for real-time, reagentless molecular monitoring directly in complex biological fluids like blood and even in situ in the living body [35] [36]. Their functionality is intrinsically linked to the precise engineering of the electrode interface, where self-assembled monolayers (SAMs) play a critical role in reducing nonspecific adsorption (NSA), ensuring optimal sensor performance, and maintaining signal stability [36]. A typical E-AB sensor is constructed by tethering a redox-tagged DNA or RNA aptamer to a gold electrode surface. Upon binding to a specific target molecule, the aptamer undergoes a conformational change, altering the electron transfer kinetics of the redox tag and producing a measurable electrochemical signal [36]. The SAM, co-immobilized with the aptamer, serves to passivate the gold surface, minimizing fouling from proteins and other biomolecules present in the sample matrix. This is paramount for achieving reliable measurements in undiluted biological fluids, as NSA can severely degrade sensor sensitivity and lifespan [35] [36]. Therefore, the development of robust SAMs on gold surfaces is a foundational element in the advancement of E-AB sensing technology for applications in therapeutic drug monitoring and personalized medicine.
Working Principle and Signaling Mechanism
The core signaling mechanism of an E-AB sensor relies on a binding-induced conformational change in a surface-immobilized aptamer, which modulates electron transfer from a redox reporter to the electrode.
Figure 1: E-AB Sensor Signaling Pathway. The binding of the target molecule causes the aptamer to fold, bringing the redox reporter closer to the electrode surface and increasing the electron transfer rate, which is detected via voltammetry. The most common redox reporter used is methylene blue [36]. This change in electron transfer kinetics is quantified using electrochemical techniques, most notably square-wave voltammetry (SWV), which is well-suited for monitoring the fast electron transfer kinetics and provides a rapid, sensitive readout ideal for real-time sensing [35] [36]. The entire process is reagentless and reversible, allowing for continuous, real-time monitoring of target analyte concentration.
Quantitative Performance Data for E-AB Sensors
The analytical performance of E-AB sensors has been demonstrated for various targets, particularly for drugs with narrow therapeutic windows. The following table summarizes key performance metrics from recent studies.
Table 1: Analytical Performance of Selected E-AB Sensors
| Target Analyte | Sensor Platform | Detection Range | Limit of Detection (LOD) | Sample Matrix | Key Performance Features |
|---|---|---|---|---|---|
| Vancomycin [35] | Wire Electrode | Entire 6–35 μM clinical range | Not Specified | Undiluted Whole Blood | ~120% signal change; ~9s equilibration; ±20% accuracy in 100 μL blood |
| Vancomycin [37] | Microneedle (MN-EAB) | Clinically Relevant | Not Specified | Undiluted Blood, Porcine Skin | Quantitative measurements in body-temperature blood; signal maintained in skin |
| Small Molecules (e.g., Cocaine) [36] | Gold Electrode | Analyte-Dependent | Not Specified | Buffer Solutions | Greatest signal gain at low aptamer packing density |
| Proteins (e.g., Thrombin) [36] | Gold Electrode | Analyte-Dependent | Not Specified | Buffer Solutions | Greatest signal gain at intermediate aptamer packing density |
These data underscore the capability of E-AB sensors to perform rapid, calibration-free measurements directly in complex, clinically relevant samples like whole blood, which is a significant advantage over traditional laboratory-based methods [35].
Detailed Experimental Protocols
This section provides a standardized protocol for fabricating and characterizing a gold-surface E-AB sensor, with integrated steps for SAM formation aimed at minimizing nonspecific adsorption.
Protocol: Fabrication of an E-AB Sensor on a Gold Electrode
Principle: This protocol outlines the steps to construct a functional E-AB sensor by covalently immobilizing a thiol-modified, redox-tagged DNA aptamer onto a gold disk electrode and backfilling with a passivating SAM to create a low-NSA interface.
Materials:
- Gold Working Electrode (e.g., 2 mm diameter gold disk electrode)
- Thiol-Modified DNA Aptamer with a Methylene Blue redox tag on the 3' or 5' end.
- SAM Passivation Molecule (e.g., 6-mercapto-1-hexanol (6-MCH))
- Ultrapure Water (18.2 MΩ·cm resistivity)
- Phosphate Buffered Saline (PBS) (1X, pH 7.4)
- Potentiostat with a three-electrode setup
- UV-Ozone Cleaner or Plasma Cleaner
Procedure:
- Gold Electrode Pretreatment:
- Mechanically polish the gold electrode surface with successive alumina slurries (e.g., 1.0, 0.3, and 0.05 μm) on a microcloth pad. Rinse thoroughly with ultrapure water after each polishing step.
- Electrochemically clean the polished electrode in 0.5 M H₂SO₄ solution using cyclic voltammetry (CV) between -0.2 V and +1.5 V (vs. Ag/AgCl) until a stable voltammogram characteristic of a clean gold surface is obtained.
- Critical SAM Pre-step: Treat the clean electrode with UV-ozone or oxygen plasma for 10-20 minutes. This step is crucial for removing trace organic contaminants from the gold surface, which is essential for forming a dense, well-ordered SAM and effectively reducing NSA [38]. Rinse the electrode with ethanol and ultrapure water, then dry under a stream of nitrogen or argon gas.
Aptamer Immobilization:
- Prepare a 1 μM solution of the thiol-modified, MB-tagged aptamer in deaerated 1X PBS.
- Spot 10-20 μL of the aptamer solution onto the cleaned gold electrode surface.
- Incubate the electrode in a humidified chamber at room temperature for a minimum of 12 hours (or overnight) to allow for the formation of gold-thiol bonds.
SAM Backfilling for NSA Reduction:
- After immobilization, rinse the electrode gently with PBS to remove any physisorbed aptamer strands.
- Incubate the electrode in a 1 mM solution of 6-MCH in PBS for 30-60 minutes. This step passivates the uncovered areas of the gold surface, creating a hydrophilic and neutrally charged monolayer that dramatically reduces nonspecific adsorption of interferents [36].
- Rinse the electrode thoroughly with PBS to remove excess 6-MCH.
Sensor Conditioning and Storage:
- Before the first measurement, condition the sensor by performing square-wave voltammetry (SWV) in a blank PBS buffer (e.g., 50-100 cycles) until a stable voltammogram is achieved.
- The fabricated sensor can be stored dry at 4°C for several days or used immediately for measurement.
Protocol: Real-Time Measurement of Vancomycin in Whole Blood
Principle: This protocol describes the use of a fabricated E-AB sensor for the quantitative, calibration-free measurement of vancomycin concentration in small-volume, whole blood samples, simulating a point-of-care testing scenario.
Materials:
- Fabricated Vancomycin E-AB Sensor (from Protocol 4.1)
- Fresh, undiluted whole blood (bovine or rat)
- Vancomycin stock solution (e.g., 1 mM in PBS)
- Potentiostat
- Eppendorf tubes (0.5 mL)
Procedure:
- Sensor Baseline Acquisition:
- Place 100 μL of blank, undiluted whole blood into a 0.5 mL Eppendorf tube.
- Immerse the E-AB sensor (as the working electrode) along with the reference and counter electrodes into the blood sample.
- Using the potentiostat, acquire a baseline SWV measurement. The parameters may vary, but a typical setup includes a frequency of 10-100 Hz, a step potential of 1 mV, and an amplitude of 25-50 mV, scanning around the formal potential of the methylene blue tag [35].
Sample Measurement:
- Spike the 100 μL blood sample with a known concentration of vancomycin (e.g., 2 μL of a 1 mM stock for a final concentration of ~20 μM). Mix gently.
- Immediately after spiking, initiate continuous SWV measurement. The sensor signal (peak current) will equilibrate within seconds (e.g., ~9 s as reported) [35].
Calibration-Free Quantification:
- Utilize the "signal-on" and "signal-off" response of the sensor at different square-wave frequencies. The ratio of currents at these frequencies is concentration-dependent but largely independent of sensor-to-sensor variation, enabling calibration-free operation [35].
- Determine the target concentration by comparing the measured signal ratio to a pre-established calibration curve or model.
Figure 2: Workflow for Real-Time Blood Measurement. The process from sensor immersion to quantitative readout, highlighting the rapid, single-step nature of E-AB sensing.
The Researcher's Toolkit: Essential Materials and Reagents
The following table lists key reagents and materials critical for the fabrication and application of E-AB sensors, with an emphasis on components essential for creating low-NSA surfaces.
Table 2: Essential Research Reagents for E-AB Sensor Development
| Item Name | Function/Application | Critical Specifications |
|---|---|---|
| Gold Electrode | Provides the conductive substrate for aptamer and SAM immobilization via gold-thiol chemistry. | High surface purity and roughness factor; available as disk, wire, or screen-printed chips. |
| Thiol-Modified Aptamer | The biological recognition element; its conformational change upon target binding generates the signal. | Must be designed with a thiol group (e.g., C6-SH) for immobilization and a redox tag (e.g., Methylene Blue). |
| 6-Mercapto-1-hexanol (6-MCH) | A short-chain alkanethiol used to form a passivating SAM. Reduces NSA by blocking free gold sites. | Critical for NSA Reduction. Creates a hydrophilic, non-fouling surface. SAM thickness impacts electron transfer [36]. |
| Methylene Blue | A redox reporter tag. Electron transfer between this tag and the electrode is modulated by aptamer folding. | Covalently attached to the distal end of the DNA aptamer. |
| UV-Ozone Cleaner | Used for pre-cleaning gold substrates to remove organic contaminants prior to SAM formation. | Critical for SAM Quality. Ensures robust thiol-gold bonding and formation of a dense, uniform monolayer [38]. |
| Potentiostat | The electronic instrument that applies potential and measures current in electrochemical experiments. | Must be capable of performing Square-Wave Voltammetry (SWV) with high sensitivity and temporal resolution. |
The detection of low molecular weight contaminants, such as antibiotics, in aquatic environments presents a significant analytical challenge. Erythromycin, a commonly used macrolide antibiotic, has been detected in environmental samples, leading to increased risks of bacterial resistance and potential ecosystem damage [39] [40]. Conventional detection methods like chromatography, while sensitive, often require sophisticated instrumentation, skilled personnel, and lack portability for field applications [41]. Biosensors, particularly those utilizing aptamers as recognition elements, have emerged as promising alternatives due to their specificity, sensitivity, and potential for miniaturization [39] [41].
A critical challenge in developing robust biosensors for long-term monitoring applications is signal drift, which compromises measurement accuracy over time. This case study details the development of an erythromycin aptasensor that integrates self-assembled monolayers (SAMs) on gold surfaces with a dual-aptamer drift cancellation scheme to achieve stable, reliable detection. This work is framed within broader thesis research focused on optimizing SAM architectures to minimize non-specific adsorption (NSA) and enhance sensor performance.
Background and Principles
Aptamers as Recognition Elements
Aptamers are short, single-stranded DNA or RNA oligonucleotides selected in vitro through the Systematic Evolution of Ligands by EXponential enrichment (SELEX) process [41]. For erythromycin detection, a specific DNA aptamer (Ery_06) was identified via Capture-SELEX, exhibiting high affinity with a dissociation constant (Kd) of 20 ± 9 nM and excellent specificity in distinguishing erythromycin from other antibiotics [39]. Aptamers offer advantages over antibodies, including superior stability, easier modification, and lower production costs, making them ideal for biosensing applications [41].
The Challenge of Signal Drift
Signal drift in electrochemical aptasensors arises from various factors, including:
- Non-specific adsorption of interferents on the sensor surface.
- Structural rearrangement of SAMs and immobilized biomolecules.
- Environmental fluctuations in temperature and pH during long-term operation.
Conventional drift correction methods, such as Kinetic Differential Measurements (KDM), are limited as their effectiveness depends on the specific structure-switching characteristics and electron transfer kinetics of the aptamer-redox reporter pair, which may not be universally applicable [42].
Experimental Design and Protocols
Gold Surface Preparation and SAM Formation
The foundation of a reliable aptasensor is a well-ordered SAM that minimizes NSA.
Protocol: Gold Electrode Pretreatment and SAM Formation
- Electrode Cleaning: Immerse polycrystalline gold disk electrodes (e.g., 2 mm diameter) in fresh piranha solution (3:1 v/v concentrated H₂SO₄:30% H₂O₂) for 10 minutes. Caution: Piranha solution is highly corrosive and must be handled with extreme care.
- Rinsing: Thoroughly rinse the electrodes with copious amounts of absolute ethanol and deionized water.
- Electrochemical Activation: Perform cyclic voltammetry (CV) in 0.5 M H₂SO₄ from -0.2 V to 1.5 V (vs. Ag/AgCl) at a scan rate of 100 mV/s until stable voltammograms characteristic of clean gold are obtained.
- SAM Formation: Incubate the clean, dry gold electrodes in a 1 µM solution of thiol-modified aptamers (Ery_06 and its paired aptamer) and 1 mM 6-mercapto-1-hexanol (MCH) in Tris-EDTA (TE) buffer (pH 7.4) for 1 hour at room temperature. This co-deposition method, where the thiolated aptamer and MCH diluent are deposited simultaneously, has been shown to improve signal gain and sensor stability [43].
- Rinsing and Storage: Gently rinse the modified electrodes with TE buffer to remove physically adsorbed molecules. Store in TE buffer at 4°C if not used immediately.
Table 1: Key Reagents for SAM Formation and Aptasensor Construction
| Research Reagent | Function / Explanation |
|---|---|
| Thiol-modified DNA Aptamers | The biorecognition element. The thiol group (-SH) forms a covalent gold-sulfur (Au-S) bond for stable anchoring to the electrode surface. |
| 6-Mercapto-1-hexanol (MCH) | A diluent molecule that fills empty spaces on the gold surface, creating a well-ordered SAM. This passivates the surface, reducing non-specific adsorption and facilitating proper aptamer folding. |
| Tris-EDTA (TE) Buffer | A standard buffer for storing and handling nucleic acids, maintaining pH and stability. |
| Gold Electrodes / Chips | The substrate for SAM formation. Gold is inert, biocompatible, and forms strong bonds with thiols. |
| Erythromycin Standard | The target analyte, used for sensor calibration and validation. |
Dual-Aptamer Drift Cancellation Strategy
To address the critical issue of long-term signal drift, a dual-aptamer scheme was implemented [42].
Principle: This technique utilizes two different aptamers immobilized on the same sensor platform. Both aptamers are exposed to the same physical and chemical environment and thus experience a common drift. However, they are engineered to exhibit differential signaling toward the target molecule, erythromycin.
- Aptamer A (Sensor): The primary, high-affinity erythromycin aptamer (Ery_06) whose signal changes upon target binding.
- Aptamer B (Reference): A second aptamer that responds minimally to erythromycin but is sensitive to environmental drift.
The signal from Aptamer B is used as a real-time reference to computationally cancel out the common-mode drift from the signal of Aptamer A, yielding a drift-corrected measurement.
Protocol: Implementing the Dual-Aptamer Sensor
- Aptamer Pair Selection: Screen existing aptamer pools or engineer redox reporters to identify a pair of aptamers that show differential response to erythromycin but common drift in complex media like serum or wastewater [42].
- Co-immobilization: Co-deposit the thiolated versions of both Aptamer A and Aptamer B with MCH on the same gold electrode, as described in Section 3.1.
- Electrochemical Measurement: Perform square-wave voltammetry (SWV) to monitor the individual voltammetric peaks of the redox reporters tagged on each aptamer.
- Data Processing: Record the signals from both aptamer channels. Subtract the reference signal (Aptamer B) from the sensor signal (Aptamer A) to generate a drift-corrected differential signal.
The following diagram illustrates the operational principle and workflow of the dual-aptamer drift cancellation technique.
Sensor Characterization and Validation
Protocol: Analytical Performance Evaluation
- Dose-Response Calibration: Incubate the aptasensor with a series of erythromycin standards in Tris-buffered saline (TBS, pH 7.4) at 37°C, covering a concentration range from 1 pM to 100 nM.
- Electrochemical Measurement: After a fixed incubation time (e.g., 15 minutes), record SWV scans.
- Data Analysis: Plot the drift-corrected signal (e.g., peak current change) against the logarithm of erythromycin concentration. Fit the data to a sigmoidal function (e.g., 4-parameter logistic equation) to determine the dynamic range and half-maximal effective concentration (EC50).
- Limit of Detection (LOD) Calculation: The LOD is determined as the concentration corresponding to the signal from the blank (buffer) plus three times the standard deviation of the blank.
- Specificity Testing: Challenge the sensor with other macrolide antibiotics (e.g., clarithromycin, azithromycin) and potential interferents found in environmental water samples to confirm the specificity of the Ery_06 aptamer.
- Stability Assessment: Continuously monitor the sensor signal in a complex matrix (e.g., diluted goat serum or spiked tap water) over several hours or days to quantify the reduction in drift achieved by the dual-aptamer scheme.
Table 2: Summary of Key Performance Metrics for the Erythromycin Aptasensor
| Performance Parameter | Result / Value | Experimental Conditions / Notes |
|---|---|---|
| Detection Principle | Electrochemical, Structure-Switching | Square-Wave Voltammetry (SWV) transduction |
| Assay Time | ~15 minutes | Includes incubation and measurement |
| Linear Range | Not explicitly stated; covers pM to nM | Calibration performed with erythromycin standards in buffer |
| Limit of Detection (LOD) | 3 pM (from aptamer-based LFS [39]) | Demonstrates high sensitivity of the selected aptamer |
| Drift Reduction | 370-fold improvement [42] | Achieved via dual-aptamer cancellation in undiluted goat serum |
| Specificity | High (distinguished erythromycin from other tested antibiotics) [39] | Validated against other macrolides and interferents |
| Sample Matrix | Validated in spiked tap water [39] | Proof-of-concept for environmental application |
Results and Discussion
The integration of a well-constructed SAM with the dual-aptamer strategy successfully addresses the core challenge of signal drift. The co-deposition method for SAM formation ensures a dense, well-oriented monolayer that effectively minimizes non-specific adsorption, a common source of false signals and baseline drift [43]. The 370-fold reduction in sensor drift demonstrated by the dual-aptamer technique is a significant advancement for the long-term stability of real-time aptasensors [42]. This approach is more universally applicable than KDM, as it does not rely on specific aptamer kinetics.
The high affinity (Kd = 20 ± 9 nM) and specificity of the Ery_06 aptamer are crucial for the sensor's performance in complex samples like tap water [39]. The achieved LOD of 3 pM meets the requirement for detecting trace levels of erythromycin in environmental samples, where even low concentrations can contribute to antimicrobial resistance (AMR) [39] [40].
The overall experimental workflow, from surface preparation to data analysis, is summarized below.
This protocol provides a detailed framework for constructing an electrochemical erythromycin aptasensor with minimized signal drift. The critical steps for success are:
- Surface Preparation: Impeccable cleanliness of the gold substrate is non-negotiable for forming a high-quality, stable SAM.
- SAM Optimization: The ratio of thiolated aptamer to MCH and the co-deposition time should be optimized to maximize target accessibility and minimize non-specific binding.
- Aptamer Pair Validation: The dual-aptamer drift cancellation requires thorough validation of the selected aptamer pair to ensure effective common-drift capture with sufficient differential signaling.
This case study demonstrates that the strategic combination of optimized self-assembled monolayers and a dual-aptamer readout scheme effectively mitigates the pervasive problem of signal drift. The developed aptasensor presents a promising, robust, and sensitive tool for the on-site monitoring of erythromycin, contributing to the fight against antimicrobial resistance within a One-Health framework. The principles outlined here can be adapted for developing stable biosensors for other small molecule targets in clinical, environmental, and food safety applications.
Troubleshooting SAM Performance: Stability, Specificity, and Signal Drift
This application note provides a detailed examination of common failure modes in self-assembled monolayers (SAMs) on gold surfaces, with particular relevance to research aimed at reducing non-specific adsorption (NSA). SAMs are widely used to control surface and interface properties, and their stability is paramount for applications in biosensors, biointerfaces, and molecular electronic devices [44]. The formation of highly ordered, dense molecular layers is critical for effectively blocking undesirable interactions at gold surfaces. This document outlines standardized protocols for identifying, characterizing, and mitigating failure modes related to desorption, oxidation, and structural defects to enhance SAM performance and reliability.
Failure Modes: Mechanisms and Quantitative Analysis
Desorption
Desorption involves the loss of molecules from the gold surface, leading to a decrease in surface coverage and a loss of function. It can be triggered thermally, chemically, or by irradiation.
- Thermal Desorption: The Se–Au bond in alkaneselenolate SAMs exhibits higher thermal stability than the S–Au bond in alkanethiolates. Thermal desorption spectroscopy shows the main desorption peak for CH₃Se species occurs at approximately 416 K, whereas for CH₃S species, it occurs at around 366 K [44].
- Chemical Desorption (Reductive Desorption): Alkaneselenolate SAMs on gold electrodes undergo reductive desorption at more negative potentials than their thiolate counterparts, confirming the stronger Se–Au bond [44].
- Laser-Induced Desorption: A thermo-kinetics model can predict feature sizes during laser patterning. Higher laser power and larger beam sizes result in broader desorption features. The surface composition of the SAM after laser irradiation correlates with the theoretical model and can be interpreted through wetting properties [45].
Oxidation
A significant problem with thiol-based SAMs is their susceptibility to oxidation.
- Oxidation in Thiols: Organic thiols can oxidize in solution to form disulfides or other oxidized groups during SAM formation, leading to undesirable surface properties [44].
- Alternative Precursors: To address oxidation, precursors like organic thiosulfates, thioacetates, and thiocyanates have been proposed. Notably, molecules containing selenium offer higher chemical stability. Selenolate SAMs demonstrate superior stability in solutions where thiolates would oxidize [44].
Structural Defects
Structural defects compromise SAM density and order, creating pathways for non-specific adsorption.
- Defect Types: Common defects include pinholes (exposed surface sites) and collapsed sites (where alkyl chains are physisorbed onto the gold surface) [46]. Defects can arise from impurities in the molecule or surface, as well as restructuring of the underlying gold [46].
- Formation and Healing: Studies on nanoporous gold show that highly ordered alkanethiol SAMs form rapidly, within seconds of immersion. While larger defects can form initially, they are often filled by thiol molecules within minutes of functionalization [46].
- Impact of Surface Morphology: Nanoporous gold, with its high surface area and curvature, can lead to intermolecular interactions within pores. However, research indicates that variations in pore size do not significantly affect the final molecular layer ordering or defect levels [46].
Table 1: Summary of Common SAM Failure Modes and Characteristics
| Failure Mode | Primary Causes | Key Characteristics | Impact on SAM Performance |
|---|---|---|---|
| Desorption | Thermal energy, reducing potentials, laser irradiation | Loss of surface coverage, change in wet properties | Reduced surface passivation, increased NSA |
| Oxidation | Exposure to oxidants in solution or air | Formation of disulfides or other oxidized species | Altered surface chemistry, unreliable functionalization |
| Structural Defects | Surface impurities, rapid deposition, substrate restructuring | Pinholes, collapsed sites, low-density domains | Localized NSA, reduced structural integrity |
Table 2: Quantitative Data on SAM Stability and Defects
| Parameter | Alkanethiolate on Au(111) | Alkaneselenolate on Au(111) | Measurement Technique |
|---|---|---|---|
| Thermal Desorption Peak | ~366 K (CH₃S) | ~416 K (CH₃Se) | Thermal Desorption Spectroscopy (TDS) [44] |
| Reductive Desorption Potential | Less negative | More negative | Cyclic Voltammetry [44] |
| Ordered Structure | (√3 × √3)R30° or c(4 × 2) | (2 × 2√7)rect or (√3 × √23)rect | Scanning Tunneling Microscopy (STM) [44] |
| Areal Density | Varies with phase | 23.3–28.9 Ų/molecule | STM [44] |
| Rapid Formation Time | Seconds to minutes | Hours (solution) / 6-24h (vapor) | IRRAS, Electrochemistry [46] |
Experimental Protocols
Protocol for Quantifying Defect Density using Electrochemistry
This protocol uses ferrocene-labeled thiols to electrochemically quantify defect density in alkanethiol SAMs [46].
- Principle: A ferrocenylalkylthiolate (e.g., FcC₁₁SH) is used to label defect sites. The ferrocene group undergoes a reversible redox reaction, and the charge measured during this reaction is proportional to the number of defects.
- Materials:
- Gold substrate (planar or nanoporous)
- Primary alkanethiol (e.g., 1-Octadecanethiol, C₁₈SH)
- Ferrocene-labeled thiol (11-ferrocenyl-undecanethiol, FcC₁₁SH)
- Electrolyte solution (e.g., 0.1 M HClO₄ or 0.1 M KCl)
- Potassium ferricyanide, K₃[Fe(CN)₆] (for pinhole assessment)
- Procedure:
- SAM Formation: Immerse the clean gold substrate in a 1 mM solution of the primary alkanethiol in ethanol for a specified time (e.g., 24 hours) to form a dense monolayer.
- Defect Labeling: Rinse the SAM and immerse it in a 1 mM solution of FcC₁₁SH in ethanol for a short period (e.g., 30-60 minutes). The ferrocene molecules will preferentially adsorb at defect sites.
- Electrochemical Measurement:
- Transfer the SAM to an electrochemical cell containing a deaerated electrolyte.
- Using Cyclic Voltammetry (CV), scan the potential around the formal potential of the ferrocene/ferrocenium couple (typically ~0.3 V vs. Ag/AgCl).
- Measure the integrated charge (Q) under the oxidation (or reduction) peak.
- Calculation: The defect density (Γ, in mol/cm²) is calculated using the equation: Γ = Q / (nFA), where n is the number of electrons (1 for ferrocene), F is Faraday's constant, and A is the electrode area.
- Pinhole Assessment (Optional): Perform CV in a solution containing 1 mM K₃[Fe(CN)₆]. A well-packed SAM will block the redox reaction of [Fe(CN)₆]³⁻/⁴⁻. The presence of significant current indicates pinhole defects.
Protocol for Assessing Thermal Stability using STM
This protocol outlines the use of Scanning Tunneling Microscopy (STM) to visualize structural changes and order in SAMs under thermal stress [44].
- Principle: STM provides real-space, molecular-scale images of the SAM structure. By observing the same sample area before and after heating, one can directly quantify domain size, defect density, and structural phase changes.
- Materials:
- SAM sample on a well-defined Au(111) substrate.
- Variable-temperature STM with a controlled atmosphere or ultra-high vacuum (UHV) chamber.
- Procedure:
- Baseline Imaging: Obtain high-resolution STM images of the SAM at room temperature. Identify characteristic features like molecular packing, domain boundaries, and inherent defects.
- Thermal Treatment: In the STM chamber or a connected preparation chamber, heat the sample to a specific temperature (e.g., 363 K, 400 K) for a set duration (e.g., 10-30 minutes).
- Post-Treatment Imaging: After cooling to room temperature, re-image the same sample region.
- Analysis:
- Domain Size: Measure the average size of ordered domains before and after heating.
- Defect Density: Quantify the number of pinholes or disordered areas per unit area.
- Packing Structure: Note any changes in the molecular lattice (e.g., from c(4 × 2) to a less ordered phase).
Protocol for Vapor Deposition to Minimize Oxidation
This protocol describes vapor deposition as a method to fabricate high-quality, oxidation-resistant alkaneselenolate SAMs [44].
- Principle: Vapor deposition eliminates solvent molecules that can hinder the formation of well-ordered selenolate SAMs and reduces the risk of oxidative side reactions that occur in solution.
- Materials:
- Gold substrate (Au(111) preferred)
- Dioctyl diselenide (DODSe) or other dialkyl diselenide precursor.
- Vapor deposition apparatus: A sealed glass vessel connected to a vacuum line, placed in an oven.
- Procedure:
- Preparation: Place the clean gold substrate and a small amount (e.g., 50-100 mg) of solid DODSe in the glass vessel.
- Evacuation: Seal the vessel and evacuate it to a low pressure (e.g., 10⁻² mbar) to remove air and moisture.
- Deposition: Place the entire vessel in an oven at an elevated temperature (e.g., 363 K for 6 hours). The DODSe will vaporize and adsorb onto the gold surface, dissociating into octaneselenolate (C₈Se) and forming a SAM.
- Completion: After the deposition time, remove the sample and rinse it with a clean solvent (e.g., ethanol) to remove any physisorbed molecules.
Visualization of Experimental Workflows
The following diagrams outline the core experimental and failure analysis pathways described in this note.
Defect Analysis Workflow
Diagram 1: Electrochemical Defect Analysis
SAM Failure Mode Pathways
Diagram 2: SAM Failure Pathways
The Scientist's Toolkit: Research Reagent Solutions
Table 3: Essential Materials for SAM Failure Mode Research
| Reagent / Material | Function / Application | Example & Key Details |
|---|---|---|
| Ferrocenylalkylthiolates | Electrochemical quantification of defect density in SAMs. | 11-Ferrocenyl-undecanethiol (FcC₁₁SH); selectively labels defect sites for Cyclic Voltammetry (CV) measurement [46]. |
| Dialkyl Diselenides | Precursors for forming oxidation-resistant SAMs. | Dioctyl Diselenide (DODSe); used in vapor deposition to form octaneselenolate SAMs with strong Se-Au bonds and high thermal stability [44]. |
| Methyl-β-Cyclodextrin (MβCD) | Cholesterol-depleting agent for studying membrane domain effects on cell adhesion. | Used in metastasis studies; removes cholesterol from cell membranes, disrupting lipid microdomains and altering adhesion protein distribution (e.g., CD44) [47]. |
| Alkanethiols | Standard precursors for forming SAMs on gold. | 1-Octadecanethiol (C18SH); forms dense, ordered monolayers. Chain length influences packing density and stability [46]. |
| Nanoporous Gold | High-surface-area substrate for studying SAM formation on complex morphologies. | Prepared by dealloying Ag/Au leaf; provides a model to investigate SAM formation kinetics, defect filling, and stability on curved surfaces [46]. |
Self-assembled monolayers (SAMs) on gold surfaces are crucial for investigating intermolecular interactions in fields ranging from biosensing to drug development. A primary challenge in these applications, particularly in surface plasmon resonance (SPR) studies, is the reduction of nonspecific adsorption (NSA) of proteins to the sensor surface. The structural quality and stability of the SAM are paramount for device performance, and these properties are directly influenced by the formation conditions. This application note details the critical parameters—solvent, incubation time, and temperature—for optimizing SAM formation on gold to create highly ordered, stable monolayers that effectively minimize NSA, thereby improving the accuracy and reliability of biomolecular interaction analyses [19].
The Scientist's Toolkit: Essential Research Reagents
The following table lists key reagents and materials essential for the formation of high-quality SAMs on gold surfaces.
Table 1: Key Research Reagent Solutions for SAM Formation on Gold
| Reagent/Material | Function/Description |
|---|---|
| Gold-coated Substrates | Provides the foundational surface for thiol-based SAM formation. Typically used as gold foils or gold-coated glass/ silicon chips [19]. |
| Alkanethiols | The primary molecules that form the SAM. Consist of a thiol head group (binds to gold), an alkane chain (provides stability), and a terminal functional group [19]. |
| Poly(ethyleneglycol) (PEG)-type Spacers | Hydrophilic spacers (e.g., compound 1 from cited research) inserted between the SAM and ligand to drastically reduce nonspecific protein binding [19]. |
| Absolute Ethanol | A common and effective solvent for the preparation of alkanethiol SAMs on gold [48]. |
| Isopropyl Alcohol (IPA) | An alternative solvent for SAM formation [48]. |
| Tetrahydrofuran (THF) | An alternative solvent for SAM formation [48]. |
Critical Parameters for Optimized SAM Formation
The quality of a SAM, defined by its order, packing density, and homogeneity, is highly dependent on the preparation procedure. Optimization of the following parameters is essential for achieving reproducible and high-performance monolayers.
Solvent Selection
The choice of solvent is a critical, yet often overlooked, parameter that significantly impacts the structural quality of the resulting SAM. Research on analogous systems has demonstrated that the solvent choice can make the difference between a disordered, liquid-like monolayer and a highly ordered, crystalline one [48].
Table 2: Impact of Solvent on SAM Structural Quality
| Solvent | Impact on SAM Quality |
|---|---|
| Ethanol | Leads to the formation of the most densely packed and highly ordered SAMs. Recommended as the primary solvent choice for optimal results [48]. |
| Isopropyl Alcohol (IPA) | Results in SAMs with lower packing density and structural order compared to those prepared from ethanol [48]. |
| Tetrahydrofuran (THF) | Produces the lowest quality SAMs, with poor structural order, and is not recommended for forming high-quality monolayers [48]. |
Incubation Time and Temperature
The kinetics of SAM formation are governed by incubation time and temperature. Longer incubation times and elevated temperatures can facilitate the reorganization of molecules on the surface, leading to a more thermodynamically stable and ordered monolayer.
- Incubation Time: The formation of a complete monolayer is a two-stage process: a rapid initial chemical adsorption of molecules to the surface, followed by a slower process of organization and crystallization. While a basic monolayer forms quickly, allowing for an incubation time of at least 12-24 hours is recommended to achieve superior structural quality and packing density [48].
- Incubation Temperature: Preparation at elevated temperatures (e.g., ~60°C) significantly enhances the final quality of the SAM. Higher thermal energy enables molecules to overcome kinetic barriers, facilitating better surface mobility and reorganization into a more perfectly ordered crystalline state [48].
The following workflow integrates these critical parameters into a cohesive experimental protocol.
Detailed Experimental Protocol: Forming a PEG-SAM on Gold
This protocol provides a step-by-step methodology for creating a SAM on a gold surface using a Poly(ethyleneglycol) (PEG)-type compound, optimized to reduce nonspecific binding [19].
Objective: To form a high-quality, hydrophilic SAM on a gold-coated glass chip that minimizes nonspecific protein adsorption for SPR studies.
Materials:
- Gold-coated glass chips (e.g., ~100 nm gold layer) [19] [48].
- PEG-type alkanethiol compound (e.g., compound 1 from Furuya et al. [19]).
- Absolute ethanol (≥ 99.9%).
- Nitrogen gas (high purity).
- Cleaning solutions: Piranha solution (CAUTION: Extremely corrosive and reactive. Handle with extreme care and consult all relevant safety procedures before preparation or use.), or as an alternative, oxygen plasma treatment.
- Ultrasonic bath.
Procedure:
- Substrate Cleaning:
- Clean the gold substrate prior to use to remove organic contaminants.
- Option A (Chemical): Immerse the gold chip in piranha solution (a 3:1 mixture of concentrated sulfuric acid [H₂SO₄] and hydrogen peroxide [H₂O₂]) for 10-15 minutes. *
- Option B (Physical): Treat the gold chip with oxygen plasma for 5-10 minutes.
- Thoroughly rinse the cleaned chip with copious amounts of pure water (e.g., Milli-Q grade) followed by a rinse with absolute ethanol.
- Dry the chip under a stream of clean, dry nitrogen gas.
SAM Solution Preparation:
- Prepare a fresh solution of the PEG-alkanethiol in absolute ethanol. A typical concentration range is 0.1 - 1.0 mM.
- Ensure the solution is well-mixed.
SAM Formation (Incubation):
- Place the cleaned, dry gold substrate into a clean vial or container.
- Add the alkanethiol solution, ensuring the substrate is fully immersed.
- Seal the container to prevent solvent evaporation and contamination.
- Incubate the substrate in the solution for 24 hours at 60°C to form a highly ordered and stable monolayer [48].
Post-Assembly Processing:
- After incubation, carefully remove the substrate from the solution using tweezers.
- Rinse the substrate thoroughly with absolute ethanol to remove physisorbed molecules.
- Dry the substrate under a stream of clean, dry nitrogen gas.
- The functionalized chip is now ready for characterization or immediate use.
Verification and Application in Reducing Nonspecific Adsorption
The success of the SAM formation and its effectiveness in reducing NSA can be verified through several characterization techniques and functional assays.
Table 3: Techniques for Characterizing SAM Quality and NSA
| Technique | Information Obtained |
|---|---|
| Contact Angle Goniometry | Measures surface wettability. A successful PEG-SAM will yield a highly hydrophilic surface (low water contact angle), indicating reduced nonspecific binding [48]. |
| Infrared Reflection-Absorption Spectroscopy (IRRAS) | Probes the molecular order and orientation of the alkane chains within the SAM. Sharp vibrational peaks indicate a well-ordered, crystalline-like film [19] [48]. |
| X-ray Photoelectron Spectroscopy (XPS) | Determines the elemental composition and chemical states on the surface, confirming the presence and coverage of the SAM [48]. |
| Functional SPR Assay | The ultimate test involves challenging the surface with a complex protein mixture like E. coli lysate. A well-formed PEG-SAM will show minimal nonspecific binding while allowing accurate quantification of specific interactions (e.g., Kd estimation) [19]. |
The application of an optimized PEG-SAM enables precise measurement of molecular interactions, even in crude biological mixtures. For instance, research has demonstrated that a gold chip with a PEG spacer allowed for adequate estimation of the dissociation constant (Kd) between FK506 and FKBP12, yielding values of 22 nM with purified FKBP12 and 15 nM with Escherichia coli lysate expressing FKBP12. These results confirm the spacer's effectiveness in mitigating nonspecific interactions, which would otherwise obscure the specific signal [19].
The formation of high-quality SAMs on gold is a foundational step in creating reliable biosensing platforms. Meticulous optimization of the formation parameters—specifically the use of ethanol as a solvent, extended incubation times (24 hours), and elevated temperatures (~60°C)—is essential for achieving well-ordered, stable monolayers. Incorporating a hydrophilic PEG spacer into the SAM design is a proven strategy to drastically reduce nonspecific adsorption. Adherence to the detailed protocols and verification methods outlined in this application note will provide researchers and drug development professionals with robust, reproducible surfaces for highly accurate biomolecular interaction analysis.
The Role of the Alkanethiol Chain Length in SAM Stability and Performance
Self-assembled monolayers (SAMs) of alkanethiols on gold surfaces represent a cornerstone of surface functionalization in numerous scientific and industrial applications, ranging from electrochemical biosensing to molecular electronics. Within the broader context of research on reducing non-specific adsorption (NSA) on gold surfaces, the alkyl chain length of the alkanethiol emerges as a critical parameter dictating the final SAM's physicochemical properties, stability, and performance. This application note provides a detailed examination of the chain length effect, consolidating key quantitative data and providing standardized protocols for researchers and drug development professionals. The objective is to furnish a practical guide for the rational design of SAMs with tailored characteristics, enabling enhanced performance in specific applications, particularly where signal stability and low background interference are paramount.
Theoretical Foundations: Chain Length and SAM Properties
The stability and performance of an alkanethiol SAM are governed by a complex interplay of molecular interactions, with chain length serving as a primary modulator. Two key interactions are significantly influenced: the van der Waals (vdW) forces between adjacent alkyl chains and the electron transfer efficiency through the monolayer.
Longer alkyl chains enhance inter-chain vdW interactions, leading to a more densely packed and crystalline-like monolayer structure. This improved packing creates a formidable physical and energetic barrier, which enhances the SAM's stability against desorption and its effectiveness as a passivation layer to block non-specific adsorption [49] [50]. This principle is directly demonstrated in the context of germanium surface passivation, where longer-chain alkanethiols (e.g., C12) provide superior oxidation resistance compared to shorter chains (e.g., C4) [49]. Furthermore, in adhesion studies, longer chains (C14, C16) exhibit higher activation barriers for adhesive bond formation, correlating with lower adhesion and improved mechanical stability [51].
However, this enhancement in stability comes with a trade-off in electrochemical applications. A densely packed, long-chain SAM forms a thick insulating layer that can impede electron transfer between the electrode surface and a solution-based redox probe, thereby diminishing the sensitivity of electrochemical biosensors [52] [2]. Consequently, a critical compromise must be struck: shorter chains favor electron transfer but offer limited stability, while longer chains provide superior stability at the cost of reduced signal strength. This balance is a central theme in SAM design.
The following tables consolidate experimental data from the literature, providing a clear comparison of how alkanethiol chain length impacts key SAM properties.
Table 1: Impact of Alkanethiol Chain Length on SAM Physicochemical Properties
| Chain Length (Carbon Atoms) | Adsorption Energy on Ge(100) (eV) [49] | Water Contact Angle (°) [49] | Surface pKa of ω-carboxy Termini [53] | Adhesion Activation Barrier (kBT) [51] |
|---|---|---|---|---|
| C2 | - | 65 ± 2 | 6.1 (C2) | - |
| C3 | - | - | 5.3 (C3) | - |
| C4 | -1.75 | 72 ± 2 | - | - |
| C6 | -1.93 | 78 ± 1 | - | ~40 (C10)† |
| C8 | -2.06 | 85 ± 1 | - | - |
| C10 | - | - | - | ~44 |
| C11 | - | - | 7.3 (C11) | - |
| C12 | -2.31 | 93 ± 1 | - | - |
| C14 | - | - | - | ~47 |
| C16 | - | - | 7.9 (C16) | ~49 |
†Data for C10 (1-decanethiol) is included from [51] for comparison with longer chains.
Table 2: Performance of SAMs in Electrochemical Biosensing Applications
| SAM Description / Chain Length | Apparent Electron Transfer Rate (s⁻¹) [52] | Limit of Detection (LOD) [54] | Stability (Signal Retention) [52] |
|---|---|---|---|
| C6 Monothiol | 40 - 70 | - | ~40% after 50 days in buffer |
| Flexible C6 Trithiol | 40 - 70 | - | ~75% after 50 days in buffer |
| Rigid C6 Trithiol | 40 - 70 | - | ~40% after 50 days in buffer |
| C11 Monothiol | ~7* | - | - |
| Sulfuric Acid-treated Au | - | 13.4 nM (Dopamine) | - |
†Value estimated from context stating C11 SAMs lead to "sluggish electron transfer" [52].
Detailed Experimental Protocols
Protocol 1: Vapor-Phase Passivation of a Solid Substrate
This protocol, adapted from the study on germanium passivation, details the formation of alkanethiol SAMs via a vapor-phase method, which is particularly useful for nanostructured surfaces to avoid destructive capillary forces [49].
1. Reagents and Materials:
- Substrate: Ge(100) wafer (or other target substrate like Au).
- Alkanethiols: Ethanethiol (C2), 1-Butanethiol (C4), 1-Hexanethiol (C6), 1-Octanethiol (C8), 1-Dodecanethiol (C12). Purity: ≥97%.
- Etchant: 20% (v/v) Hydrochloric Acid (HCl) solution.
- Inert Gas: Nitrogen (N₂), high purity.
2. Equipment:
- Vapor-phase deposition chamber or a sealed desiccator.
- Chemical fume hood.
- AFM for surface characterization.
- X-ray Photoelectron Spectrometer (XPS).
- Contact Angle Goniometer.
3. Procedure:
- Step 1: Surface Preparation. Place the Ge wafer in a 20% HCl solution for 10 minutes to remove the native oxide and achieve a Cl-terminated surface.
- Step 2: Rinsing and Drying. Remove the substrate from the HCl solution and rinse thoroughly with deionized water. Dry the substrate under a stream of N₂ gas.
- Step 3: Vapor-Phase Passivation. In a fume hood, place a small open container of the liquid alkanethiol (∼1 mL) inside the deposition chamber alongside the dried substrate. Seal the chamber to allow the thiol vapor to saturate the environment. Incubate for a minimum of 168 hours (7 days) at room temperature.
- Step 4: Post-Assembly Cleaning. After incubation, remove the substrate from the chamber and rinse it gently with ethanol to remove any physisorbed molecules. Dry again under a stream of N₂.
- Step 5: Quality Control. Characterize the resulting SAM by:
- Water Contact Angle (WCA): Assess hydrophobicity and monolayer quality. Expected values range from ~65° for C2 to ~93° for C12 SAMs [49].
- XPS: Confirm successful thiolate bonding to the surface and quantify surface oxidation after environmental exposure.
- AFM: Evaluate surface morphology and uniformity.
Protocol 2: Fabrication of an Electrochemical Aptasensor
This protocol outlines the formation of a mixed SAM on a gold electrode for electrochemical biosensing, highlighting the critical steps to ensure a stable and functional interface [2].
1. Reagents and Materials:
- Electrode: Pure [111] gold electrode.
- DNA Aptamer: Thiol-modified oligonucleotide specific to the target (e.g., erythromycin aptamer).
- Blocking Agent: 6-Mercapto-1-hexanol (MCH).
- Buffer: Immobilization buffer (e.g., 6X SSC: 90 mM sodium citrate, 0.9 M NaCl, pH 7.0).
- Redox Probe: K₄[Fe(CN)₆]/K₃[Fe(CN)₆] solution in buffer.
2. Equipment:
- Electrochemical Workstation with a standard 3-electrode setup (Gold Working Electrode, Ag/AgCl Reference Electrode, Pt Counter Electrode).
- Micropipettes.
3. Procedure:
- Step 1: Electrode Pretreatment. Clean the gold electrode electrochemically in 0.5 M H₂SO₄ via cyclic voltammetry (e.g., scanning from -0.1 V to +1.5 V) until a stable voltammogram is achieved. Rinse with deionized water.
- Step 2: Aptamer Immobilization. Incubate the clean gold electrode in a 1 µM solution of the thiol-modified aptamer in 6X SSC buffer for a prolonged period (e.g., 12-24 hours) at room temperature in the dark. Extended immobilization times enhance mixed SAM stability [2].
- Step 3: Backfilling with MCH. Rinse the electrode with DI water to remove loosely bound aptamer. Incubate the electrode in a 3 mM solution of MCH in DI water for 1 hour at room temperature in the dark. This step displaces non-specifically adsorbed DNA and creates a well-ordered, diluting monolayer that reduces non-specific adsorption.
- Step 4: Electrochemical Characterization. Perform characterization using Differential Pulse Voltammetry (DPV) or Cyclic Voltammetry (CV) in a solution containing the [Fe(CN)₆]³⁻/⁴⁻ redox probe. A stable and reproducible signal indicates successful SAM formation.
- Step 5: Sensing Measurements. Incubate the functionalized aptasensor with the target analyte and record the DPV response. The change in current or peak potential is correlated with the target concentration.
The Scientist's Toolkit: Essential Research Reagents
Table 3: Key Reagent Solutions for SAM Research on Gold Surfaces
| Reagent Name | Function / Role in SAM Research | Example Application & Rationale |
|---|---|---|
| Short-Chain Alkanethiols(e.g., C2 - C6) | Facilitate efficient electron transfer; higher vapor pressure for vapor-phase deposition. | Used in electrochemical biosensors where signal strength is prioritized [49] [52]. |
| Long-Chain Alkanethiols(e.g., C12 - C18) | Enhance SAM stability and oxidation resistance via strong van der Waals interactions; improve hydrophobicity. | Ideal for surface passivation applications to prevent oxidation or as robust non-fouling layers [49] [51]. |
| 6-Mercapto-1-hexanol (MCH) | A short-chain hydroxyl-terminated thiol used as a blocking agent in mixed SAMs. | Backfilling agent to displace non-specifically adsorbed DNA on aptasensors, orient probes, and reduce NSA [52] [2]. |
| Multi-Thiol Anchors(e.g., Flexible Trithiol) | Provide multiple Au-S bonds, drastically improving SAM stability without severely compromising electron transfer. | Anchoring layer for DNA probes in biosensors requiring long-term storage stability and thermal robustness [52]. |
| ω-Functionalized Thiols(e.g., ω-carboxy) | Introduce terminal functional groups (-COOH, -NH₂) for further covalent immobilization of biomolecules. | Forming a surface for coupling amines via EDC/NHS chemistry, enabling the creation of more complex interfaces [53]. |
| Acid Etchants(HCl, H₂SO₄) | Remove native oxides and contaminants; terminate surfaces for subsequent reaction. | HCl for creating a Cl-terminated Ge surface [49]; H₂SO₄ for electrochemical cleaning of Au electrodes [54] [2]. |
The chain length of alkanethiols is a fundamental design parameter that directly dictates the critical balance between SAM stability and performance. Longer chains (≥ C12) are the clear choice for applications demanding maximum passivation stability and oxidation resistance, as they form a robust, crystalline barrier. In contrast, for electrochemical biosensing, shorter chains (C6) or innovative multi-thiol anchors provide the optimal compromise, offering sufficient stability while maintaining high electron transfer efficiency. The protocols and data summarized in this application note provide a framework for the rational selection of alkanethiols and the fabrication of reliable SAMs, ultimately contributing to the advancement of research aimed at reducing non-specific adsorption and enhancing the performance of functionalized gold surfaces.
Nonspecific adsorption (NSA) of biomolecules and other interfering species onto sensor surfaces remains a significant challenge in the development of reliable diagnostic devices and fundamental biological studies. Self-assembled monolayers (SAMs) on gold surfaces have emerged as a powerful platform for creating well-defined interfaces that minimize NSA while maintaining specific biological recognition. Traditional alkanethiol-based SAMs, while widely used, suffer from limitations in stability and packing density that restrict their performance. This application note details three advanced strategies—crosslinking, N-heterocyclic carbenes (NHCs), and pulse-assisted assembly—that address these limitations by creating more robust, densely packed, and ordered monolayers. These approaches significantly enhance surface engineering capabilities for biosensing, molecular electronics, and drug development applications where interface purity and stability are paramount. By systematically implementing these strategies, researchers can achieve unprecedented control over surface properties, leading to substantial reductions in NSA and improved signal-to-noise ratios in complex biological environments.
N-Heterocyclic Carbenes as Superior Molecular Anchors
Fundamental Advantages Over Traditional Thiol Chemistry
N-heterocyclic carbenes have emerged as exceptional molecular anchors for creating ultra-stable SAMs on gold and other materials. The primary advantage of NHCs stems from their stronger metal-carbon bond formation compared to the gold-sulfur bond of traditional thiols. Research demonstrates that NHCs form SAMs with significantly higher thermal stability—able to withstand temperatures up to ≥573 K compared to thiol SAMs—and greater chemical resilience against harsh conditions including boiling water, organic solvents, pH extremes, and chemical oxidants like hydrogen peroxide [55] [56]. This robust bonding translates directly to reduced NSA by maintaining monolayer integrity in complex biological environments where thiol-based SAMs would degrade and create adsorption sites.
The structural properties of NHC SAMs contribute directly to their NSA reduction capabilities. Studies show that properly designed NHCs can form highly crystalline, densely packed monolayers with defect concentrations reduced by 2-3 orders of magnitude compared to traditional SAMs [57]. This dense packing leaves minimal gaps for nonspecific interactions. Notably, benzimidazole-derived carbenes provide films with the highest stabilities and evidence of short-range molecular ordering, making them particularly effective for biosensing applications [56].
Recent Advances in NHC Design and Performance
Recent research has optimized NHC structural parameters to enhance monolayer performance. Contrary to earlier beliefs that bulky side groups were necessary for stable NHC SAMs, studies now show that NHCs with small methyl side groups form aromatic monolisters with at least double the surface density and upright molecular orientation compared to bulkier NHC analogs [58]. This increased packing density directly correlates with reduced NSA by eliminating interstitial spaces where unwanted adsorption occurs.
The exceptional insulating properties of NHC SAMs further contribute to their effectiveness in electrochemical biosensing applications. Densely packed NHC monolayers exhibit conductivity ~4 orders of magnitude lower than standard alkanethiols of comparable length, significantly reducing background noise and interference in sensing applications [57]. This combination of high stability, dense packing, and superior insulating properties makes NHCs ideal anchors for creating biointerfaces with minimal NSA.
Table 1: Comparative Analysis of NHC vs. Thiol SAM Properties
| Property | NHC SAMs | Traditional Thiol SAMs | Significance for NSA Reduction |
|---|---|---|---|
| Bond Strength | Strong Au-C bond [56] | Weaker Au-S bond [56] | Maintains monolayer integrity under harsh conditions |
| Thermal Stability | Stable up to ≥573 K [55] [57] | Degrades at lower temperatures | Prevents thermal decomposition that creates adsorption sites |
| Chemical Stability | Resists pH extremes, oxidants, sonication [55] [56] | Vulnerable to oxidation and displacement | Maintains consistent surface properties in biological buffers |
| Packing Density | Potentially higher with small substituents [58] | Limited by steric constraints of S-Au bonding | Reduces interstitial spaces for nonspecific binding |
| Defect Concentration | 2-3 orders of magnitude lower [57] | Higher defect density | Fewer defects mean fewer sites for unwanted adsorption |
Crosslinking Strategies for Enhanced SAM Stability
Molecular Design for Lateral Crosslinking
Crosslinking represents a powerful strategy for enhancing SAM stability and reducing NSA by creating interconnected monolayer networks that resist displacement and molecular penetration. The design of crosslinkable NHCs has recently been demonstrated with Di-AlkNHC, which features two alkyne groups aligned parallel to the surface, enabling lateral crosslinking between adjacent molecules [55]. This horizontal networking creates a more rigid two-dimensional matrix that maintains molecular orientation while eliminating dynamic movements that can create temporary adsorption sites.
The crosslinking process typically employs copper-catalyzed azide-alkyne cycloaddition (CuAAC) either through chemical or electrochemical activation. The electrochemical approach offers particular advantages for biosensor fabrication, applying a constant potential of -0.2 V for 15 minutes in the presence of 0.1 mM Cu(II) to generate catalytically active Cu(I) in situ [55]. This controlled activation enables spatial patterning of crosslinking density across the surface, creating domains with varying resistance to NSA tailored to specific application requirements.
Functional Benefits for NSA Reduction
Crosslinked NHC SAMs demonstrate markedly improved resistance to molecular displacement, even when challenged with thiols or thioethers that typically disrupt conventional monolayers [56]. This stability is crucial for long-term biosensing applications where surface fouling progressively degrades performance. Additionally, crosslinked networks minimize molecular reorganization under electrochemical cycling, maintaining consistent interfacial properties through multiple measurement cycles.
The enhanced structural integrity of crosslinked SAMs directly impacts NSA by preventing the formation of defects that typically develop over time in conventional monolayers. Studies show that crosslinked NHC films maintain their barrier properties after extended exposure to biological fluids, with XPS analysis confirming minimal nitrogen signal changes after challenging conditions [55]. This durability makes crosslinked NHC SAMs particularly valuable for implantable sensors and reusable diagnostic platforms where surface regeneration is required.
Pulse-Assisted Assembly Techniques
Potential-Assisted Thiol Exchange for DNA SAMs
Pulse-assisted assembly methods provide dynamic control over monolayer formation, enabling precise regulation of surface density and organization. Potential-assisted thiol exchange has been successfully demonstrated for creating DNA SAMs with controlled surface densities on single crystal gold bead electrodes [59]. This approach applies potential pulses to modulate the adsorption/desorption equilibrium during SAM formation, resulting in nonuniformly distributed DNA SAMs with varying surface densities across different crystallographic facets of the gold substrate.
The significance of this method for NSA reduction lies in its ability to create surfaces with optimized probe density—a critical parameter influencing both specific hybridization efficiency and nonspecific interference. Research shows that lower coverage regions achieve full hybridization 5× faster than higher coverage regions, approaching solution-phase kinetics while maintaining surface confinement advantages [59]. This kinetic advantage translates to reduced NSA by minimizing incubation times required for target recognition, thereby limiting opportunities for nonspecific interactions.
Electrochemical Deposition of NHC Monolayers
Pulse-assisted methods have been adapted for NHC deposition, employing potential pulses to generate reactive oxygen species that deprotonate imidazolium precursors to form reactive carbenes. One optimized protocol uses 25 pulses of 5 seconds at -1.2 V, each followed by a 5-second relaxation period, to deposit NHCs on various substrates including gold, copper, steel, and glassy carbon [55]. This pulsed approach produces more homogeneous films compared to continuous potential application, with thicknesses of approximately ~4 nm that balance barrier properties with electrochemical accessibility.
The reduced film thickness of electrochemically deposited NHC SAMs compared to traditional diazonium salt approaches (typically >10 nm) offers advantages for electrochemical biosensing by minimizing electron transfer barriers while maintaining effective blocking capabilities [55]. This optimal thickness profile enables efficient electron transfer for redox-labeled probes while effectively blocking interferents, creating an ideal platform for sensitive detection in complex biological samples.
Table 2: Pulse-Assisted Assembly Parameters and Outcomes
| Method | Pulse Parameters | Resulting SAM Properties | Impact on NSA |
|---|---|---|---|
| Potential-Assisted Thiol Exchange | Optimized for target coverage [59] | Controlled surface density variation (ΓDNA) | Enables optimization of probe spacing to minimize crowding effects that promote NSA |
| NHC Electrodeposition | 25 pulses of 5 s at -1.2 V, 5 s relaxation [55] | ~4 nm thin films with low passivation | Reduced film thickness decreases diffusion barriers while maintaining blocking function |
| Pulse Laser Annealing | Femtosecond pulses [60] | Reshaping and fragmentation of gold nanoparticles | Creates uniform nanostructures with consistent SAM coverage, eliminating defect sites |
Experimental Protocols
Protocol 1: Electrochemical Deposition of NHC SAMs on Gold
Principle: This protocol describes the electrochemical deposition of alkyne-functionalized NHCs on gold surfaces for subsequent functionalization via click chemistry, creating stable monolayers with superior resistance to NSA.
Materials:
- iPrNHCAlk or Di-AlkNHC imidazolium bicarbonate salt precursors
- Acetonitrile with 0.1% v/v H2O
- Supporting electrolyte (NBu4PF6, KPF6, or NaI depending on substrate)
- Gold substrate (polycrystalline or single crystal bead electrode)
- Reference electrode (Ag/AgCl) and platinum counter electrode
Procedure:
- Prepare gold substrates by standard cleaning protocols, including Aqua Regia (1:3 HNO3:HCl) etching and flame annealing [59].
- Prepare deposition solution containing 1-5 mM NHC precursor in CH3CN with 0.1% v/v H2O and 50 mM supporting electrolyte.
- Assemble electrochemical cell with gold working electrode, reference electrode, and counter electrode.
- Apply 25 cycles of 5-second potentiostatic pulses at -1.2 V vs. Ag/AgCl, each followed by a 5-second relaxation at open circuit potential [55].
- Remove electrode and rinse thoroughly with CH3CN and ethanol to remove physisorbed precursor, electrolyte, and byproducts.
- Characterize film quality by XPS (N 1s signal at 400.2 eV confirms NHC binding) and AFM.
Validation: Successful deposition shows increased N 1s signal by XPS with characteristic binding energy shift from precursor (401.1 eV) to surface-bound NHC (400.2 eV) [55]. Film thickness should be approximately ~4 nm by AFM.
Protocol 2: Crosslinking NHC SAMs via Electro-Click Reaction
Principle: This protocol describes the functionalization and crosslinking of alkyne-bearing NHC SAMs via electrochemical copper-catalyzed azide-alkyne cycloaddition, creating a networked monolayer with enhanced stability against NSA.
Materials:
- NHC SAM-functionalized electrode from Protocol 1
- Azide-functionalized crosslinker (e.g., 1-azidohexane or custom azides)
- CuSO4 (0.1 mM) in CH3CN with supporting electrolyte (50 mM)
Procedure:
- Prepare clicking solution containing 1-10 mM azide crosslinker and 0.1 mM CuSO4 in CH3CN with 50 mM supporting electrolyte.
- Assemble electrochemical cell with NHC-modified working electrode.
- Apply constant potential of -0.2 V vs. Ag/AgCl for 15 minutes to generate catalytically active Cu(I) in situ [55].
- Remove electrode and rinse thoroughly with CH3CN and ethanol to remove copper catalyst and unreacted azide.
- For characterization, employ XPS to detect triazole formation (increased N% signal) and contact angle measurements to confirm surface property modification.
Validation: Successful clicking shows increased nitrogen atomic percentage (N%) by XPS survey scans, typically tripling after the click reaction [55]. For fluorophore-tagged azides, fluorescence microscopy confirms functionalization.
Protocol 3: FRET Imaging for DNA SAM Hybridization Kinetics
Principle: This protocol employs Förster resonance energy transfer (FRET) imaging to quantify DNA hybridization kinetics on different SAM regions, enabling optimization of surface density to minimize NSA while maintaining rapid target recognition.
Materials:
- Thiolated DNA strands (30-mer) labeled with AlexaFluor488 (donor) or AlexaFluor647 (acceptor)
- TCEP reduction solution
- TRIS buffer (10 mM, pH 7.5) with NaCl (100 mM) and MgCl2 (25 mM)
- Single crystal gold bead electrode
- MCH (1 mM in MeOH) for passivation
- Complementary DNA target sequence
Procedure:
- Reduce thiolated DNA with 100× excess TCEP for 3 hours at room temperature, then filter using G-50 columns [59].
- Prepare mixed DNA solution (400 nM total concentration) with desired donor:acceptor ratio in immobilization buffer.
- Assemble electrochemical cell with gold bead working electrode and apply potential-assisted thiol exchange to form mixed DNA SAMs.
- Passivate with 1 mM MCH for 1 hour to displace nonspecifically adsorbed DNA.
- Image FRET signal across different crystallographic regions using appropriate filter sets.
- Introduce complementary DNA target and monitor FRET intensity changes over time.
- Fit FRET increase to second-order Langmuir adsorption rate equation to determine hybridization kinetics [59].
Validation: Successful implementation shows coverage-dependent FRET signals consistent with 2D FRET models, with lower coverage regions hybridizing 5× faster than higher coverage regions [59].
The Scientist's Toolkit: Essential Research Reagents and Materials
Table 3: Key Research Reagent Solutions for Advanced SAM Fabrication
| Reagent/Material | Function/Application | Key Characteristics | Representative Examples |
|---|---|---|---|
| Imidazolium Bicarbonate Salts | NHC precursors for electrochemical deposition | Stable carbene precursors soluble in organic electrolytes | iPrNHCAlk, Di-AlkNHC [55] |
| Alkyne-Functionalized Azides | Crosslinking agents via CuAAC | Variable spacer lengths, terminal functionalities | 1-azidohexane, PEG-azides, fluorophore-tagged azides [55] |
| Thiolated Fluorophore-Labeled DNA | FRET-based SAM characterization | 30-mer sequences with disulfide bonds for gold attachment | AlexaFluor488 or AlexaFluor647 labeled [59] |
| Single Crystal Gold Bead Electrodes | Substrates with multiple crystallographic facets | Enable correlation of SAM properties with surface structure | Facet-dependent coverage studies [59] |
| Mercaptohexanol (MCH) | Passivation agent | Displaces nonspecifically adsorbed DNA | 1 mM in MeOH [59] |
Analytical Methods for SAM Characterization
Spectroscopic Techniques
X-ray photoelectron spectroscopy (XPS) provides essential quantitative data on NHC SAM composition and binding characteristics. For NHCs on steel substrates, the N 1s signal shows characteristic peaks at 401.8 eV (quaternary ammonium impurities), 400.2 eV (surface-bound NHC), and 398.9 eV (neutral byproducts) [55]. The successful electro-click reaction with azides is confirmed by a tripling of nitrogen atomic percentage from approximately 0.9% to 2.7% after functionalization [55]. This quantitative assessment ensures monolayer quality and functionalization efficiency directly correlated with NSA resistance.
FRET imaging serves as a powerful tool for investigating local DNA SAM environments and hybridization kinetics. The FRET signal shows strong dependence on surface DNA density (ΓDNA) and donor-to-acceptor ratio, enabling quantification of local crowding effects that influence NSA [59]. The increase in FRET intensity during hybridization follows a second-order Langmuir adsorption model, providing direct assessment of probe environment and its influence on recognition efficiency [59].
Stability Assessment Protocols
Standardized stability testing is essential for evaluating SAM performance under application-relevant conditions. Recommended testing includes:
- Thermal stability: Heating samples to 573K while monitoring structural changes [57]
- Chemical stability: Exposure to pH extremes (2-12), organic solvents, 1% H2O2, and electrochemical cycling above 0V [56]
- Sonication resistance: Subjecting samples to ultrasonic baths for defined periods [55]
- Biological challenges: Incubation in serum-containing media to assess fouling resistance
NHC SAMs consistently outperform thiol counterparts across all these metrics, with benzimidazole-derived carbenes showing particular resilience [56]. This comprehensive characterization ensures SAMs will maintain their NSA resistance throughout their intended application lifetime.
Comparative Workflow Analysis
Diagram 1: Comparative workflow analysis of traditional thiol versus advanced NHC strategies for creating NSA-resistant SAMs. The NHC approach incorporates crosslinking and pulse-assisted assembly for enhanced performance.
The integration of N-heterocyclic carbenes, crosslinking strategies, and pulse-assisted assembly represents a significant advancement in SAM technology for reducing nonspecific adsorption on gold surfaces. The quantitative data presented demonstrates that NHC-based SAMs offer superior thermal stability (withstanding temperatures ≥573K), enhanced chemical resistance (tolerating pH extremes, oxidants, and sonication), and reduced defect concentrations (2-3 orders of magnitude lower than thiol SAMs) [55] [57] [56]. These properties directly translate to improved NSA resistance by maintaining monolayer integrity in challenging biological environments.
Future developments in this field will likely focus on expanding the substrate scope of NHC chemistry, particularly for carbon-based materials like glassy carbon, which has recently been demonstrated as a viable substrate for NHC anchoring [55]. Additionally, the combination of these advanced SAM strategies with spatially patterned surfaces will enable multiplexed biosensing platforms with minimal cross-talk between detection zones. As characterization techniques like FRET imaging continue to improve, researchers will gain even deeper insights into the relationship between local monolayer structure, hybridization kinetics, and NSA, enabling rational design of next-generation biointerfaces with optimized performance for drug development and clinical diagnostics.
The reliability of biosensing and diagnostic assays is critically dependent on minimizing non-specific adsorption (NSA) and background interference. These factors are major contributors to false-positive signals, particularly when working with complex biological media such as blood and cell lysates. These media contain a multitude of proteins, lipids, nucleic acids, and other cellular components that can adhere non-specifically to sensor surfaces, compromising assay specificity and accuracy.
Self-assembled monolayers (SAMs) on gold surfaces represent a powerful and versatile platform for creating well-defined, reproducible, and functionalizable interfaces that can mitigate these challenges. When properly engineered, these organic assemblies form highly ordered, dense films that present specific chemical terminal groups to the solution, effectively creating a molecular barrier against non-specific interactions. This application note details practical strategies and optimized protocols for utilizing SAMs to enhance assay fidelity in complex environments, providing researchers with a toolkit for developing robust diagnostic and research applications.
Strategic Approaches for Minimizing Non-Specific Adsorption
Effectively preventing false positives requires a multi-faceted strategy that combines surface chemistry, sample pre-treatment, and detection methodology. The following table summarizes the core strategic pillars and their underlying principles.
Table 1: Strategic Pillars for Preventing False Positives in Complex Media
| Strategy | Principle | Key Technique | Targeted Interference |
|---|---|---|---|
| Surface Passivation | Creates a physical and energetic barrier on the sensor surface to reduce non-specific binding. | Self-Assembled Monolayers (SAMs) of ethylene glycol (EG) or zwitterionic molecules [61]. | Serum proteins, cellular debris, lipids. |
| Host Biomass Depletion | Selectively removes background biological material from the sample before analysis. | Filtration membranes that capture human cells based on surface charge [62]. | Host leukocyte DNA (>98% removal) [62]. |
| Target Enrichment | Amplifies the specific signal from the target of interest relative to the background. | Targeted Next-Generation Sequencing (tNGS) panels [62]. | Background nucleic acids, low-abundance targets. |
| Microfluidic Sorting | Physically isolates the target analyte from the complex matrix. | Antibody-coated (e.g., anti-EpCAM) microfluidic chips [63]. | Blood cells, non-target components. |
The logical relationship between these strategies, from sample to analysis, can be visualized as an integrated workflow. The following diagram outlines the pathway for processing a complex sample like blood to achieve a specific readout while minimizing false positives.
Experimental Protocols
Protocol: Fabrication of Protein-Resistant SAMs on Gold Surfaces
This protocol describes the formation of a mixed self-assembled monolayer (SAM) on a gold surface, optimized to resist non-specific adsorption of proteins and other biomolecules from complex media like blood serum and cell lysates.
Principle: A monolayer composed of a high proportion of oligo(ethylene glycol)-terminated alkanethiols creates a hydrophilic, neutrally charged, and highly hydrated surface that is sterically and energetically unfavorable for protein adsorption. Co-adsorption with a functional thiol (e.g., carboxylic acid-terminated) provides sites for subsequent covalent immobilization of specific capture probes.
Research Reagent Solutions:
Table 2: Essential Reagents for SAM Formation and Passivation
| Reagent | Function / Role | Example / Note |
|---|---|---|
| Gold Substrates | Sensor substrate that forms a stable covalent bond with thiols. | Template-stripped gold, gold-coated glass/silicon slides. |
| Alkanethiols (EG-based) | Forms the protein-resistant background of the SAM. | HS-(CH2)11-(EG)6-OH (e.g., OEG6). The EG group is critical for resistance [61]. |
| Carboxylic Acid-Terminated Thiol | Provides functional groups for activating and immobilizing specific capture molecules (e.g., antibodies, DNA). | HS-(CH2)15-COOH (e.g., 16-Mercaptohexadecanoic acid). |
| Absolute Ethanol | High-purity solvent for thiol dissolution and SAM formation. | Prevents contamination. |
| Passivating Protein Solution | Blocks any remaining non-specific binding sites on the SAM. | 1% (w/v) Bovine Serum Albumin (BSA) in PBS. |
Procedure:
- Substrate Cleaning: Clean gold substrates prior to use. A standard protocol involves immersion in freshly prepared "piranha solution" (a 3:1 mixture of concentrated sulfuric acid and 30% hydrogen peroxide) for 10-15 minutes at 80°C. Caution: Piranha solution is extremely corrosive and must be handled with extreme care. Rinse the substrates thoroughly with copious amounts of Milli-Q water and absolute ethanol, then dry under a stream of nitrogen or argon gas.
- SAM Solution Preparation: Prepare a 1 mM total thiol concentration solution in absolute ethanol. For a mixed SAM, use a 90:10 molar ratio of protein-resistant thiol (e.g., OEG6-OH) to functional thiol (e.g., 16-Mercaptohexadecanoic acid). This ratio maximizes passivation while providing sufficient functional groups for target capture.
- SAM Formation: Immerse the clean, dry gold substrates in the prepared thiol solution. Incubate for a minimum of 18 hours (overnight) at room temperature in a sealed, dark container to prevent solvent evaporation and photodegradation.
- Rinsing and Drying: After incubation, remove the substrates and rinse them thoroughly with absolute ethanol to remove physically adsorbed thiols. Dry the substrates under a gentle stream of inert gas (N2 or Ar).
- Post-Assembly Rinsing (Optional but Recommended): Immerse the SAM-functionalized substrates in a large volume of Milli-Q water for 1-2 hours to remove any residual solvent and facilitate the reorientation of the SAM into its lowest energy, most ordered state.
- Final Drying: Dry the substrates again under a stream of inert gas. They can be stored under nitrogen atmosphere for short periods before use.
Protocol: Sample Pre-Treatment for Blood-Based Assays
This protocol details a filtration-based method to deplete host nucleated cells from whole blood, significantly reducing background host DNA and improving the sensitivity of downstream pathogen or biomarker detection.
Principle: A specialized filtration membrane with tailored surface charge properties (electrostatically attractive to leukocytes) selectively captures nucleated human cells while allowing pathogens and other analytes to pass through, thereby enriching the target-to-background ratio [62].
Procedure:
- Sample Collection: Collect whole blood into appropriate anticoagulant tubes (e.g., EDTA or citrate).
- Filtration Setup: Assemble the filtration apparatus according to the manufacturer's instructions, ensuring the human cell-specific filtration membrane is properly seated.
- Sample Filtration: Gently pass the whole blood sample (typically 1-5 mL) through the filtration membrane. Apply gentle pressure or vacuum as required by the specific device to maintain a steady flow rate. Avoid excessive force that could rupture cells.
- Filtrate Collection: Collect the filtrate, which is now significantly depleted of host leukocytes and their associated DNA, into a sterile tube.
- Downstream Application: The processed filtrate can now be used directly in downstream assays such as nucleic acid extraction for targeted sequencing (tNGS) or applied to a SAM-functionalized biosensor. This pre-treatment has been shown to reduce host DNA by over 98% and can boost pathogen-specific reads by 6- to 8-fold in sequencing applications [62].
The workflow for this sample pre-treatment and its integration with a targeted detection system is illustrated below.
The Scientist's Toolkit
Successful implementation of the aforementioned strategies requires a suite of reliable reagents and tools. The following table catalogues essential solutions for researchers developing assays for complex media.
Table 3: Research Reagent Solutions for Complex Media Assays
| Category / Item | Function / Application | Key Characteristic |
|---|---|---|
| Surface Chemistry | ||
| OEG6-Alkanethiol | Forms protein-resistant SAM on gold to minimize NSA. | Presents a hydrated, neutral barrier [61]. |
| Carboxy-Terminated Alkanethiol | Co-adsorbs in SAM to provide sites for probe immobilization. | Enables EDC-NHS chemistry for coupling amines. |
| Bovine Serum Albumin (BSA) | Blocks residual non-specific binding sites on sensor surfaces. | Inert, low-cost, and effective passivating agent. |
| Sample Pre-Treatment | ||
| Human Cell-Specific Filtration Membrane | Selectively depletes leukocytes from whole blood. | Electrostatic capture of host cells; >98% host DNA removal [62]. |
| Lysis Buffers (Gentle) | Releases intracellular targets without fragmenting host DNA. | Critical for minimizing background in nucleic acid tests. |
| Capture & Detection | ||
| Anti-EpCAM Antibody | Immobilized on surfaces or chips to capture epithelial cells (e.g., CTCs) [63]. | Key for positive selection from blood. |
| Targeted NGS (tNGS) Panel | Amplifies specific genomic regions of interest from pathogens or hosts. | Focuses sequencing power; increases sensitivity and reduces cost vs. mNGS [62]. |
| Microfluidic CTC-Chip | Captures rare circulating tumor cells from blood based on surface markers. | High-sensitivity cell capture for liquid biopsy [63]. |
The fight against false positives in complex biological media is winnable through a disciplined, multi-layered approach. The integration of well-designed self-assembled monolayers for surface passivation with robust sample pre-treatment methods, such as selective filtration, creates a powerful defensive front against non-specific adsorption and background interference. When combined with highly specific detection techniques like targeted sequencing or antibody-based capture on functionalized devices, researchers can achieve the levels of sensitivity and specificity required for modern diagnostics and advanced research. The protocols and strategies outlined herein provide a concrete foundation for developing reliable and reproducible assays in even the most challenging biological milieus.
The development of analytical platforms using self-assembled monolayers (SAMs) on gold surfaces requires distinct methodological approaches depending on the biological target. Detecting whole bacterial cells presents fundamentally different challenges compared to detecting proteins, impacting SAM design, detection methodologies, and experimental protocols. This application note details the specific considerations, protocols, and key reagents for these two target classes within the context of biosensor and biomaterial research. The core difference lies in the target complexity: proteins are molecular entities detected through binding affinity, whereas bacteria are living cellular organisms whose detection must account for viability, adhesion mechanisms, and complex surface interactions [64] [65].
Comparative Challenges: Bacteria vs. Protein Detection
The table below summarizes the core challenges and strategic considerations when designing SAM-based detection systems for bacteria versus proteins.
Table 1: Key Challenges and Strategic Considerations for Detecting Bacteria vs. Proteins on SAM-Coated Surfaces
| Parameter | Bacterial Detection | Protein Detection |
|---|---|---|
| Target Complexity | Living cells (3-5 µm) with complex, dynamic surfaces [65]. | Molecules (5-50 nm) with defined binding epitopes [66]. |
| Primary Challenge | Non-specific adhesion and subsequent biofilm formation [65]. | Non-specific adsorption (fouling) on the SAM surface [66] [67]. |
| Critical Consideration | Must account for the multi-stage biofilm lifecycle (reversible/irreversible attachment, maturation, dispersion) [65]. | Must preserve protein conformation and binding activity after immobilization. |
| Detection Signal | Often relies on viability indicators (e.g., ATP, respiration) or adhesion force [64] [67]. | Directly measures binding affinity, mass, or refractive index change [66]. |
| SAM Design Strategy | Incorporate biocidal agents (e.g., silver ions) or anti-adhesion motifs (e.g., tri(ethylene glycol)) to prevent initial attachment [65] [67]. | Control ligand density and orientation; use non-fouling backgrounds (e.g., PEG) to minimize false positives [66] [68]. |
| Representative Analytical Techniques | Atomic Force Microscopy (AFM) for adhesion force, fluorescence microscopy for viability, SEM for morphology [67]. | Surface Plasmon Resonance (SPR), ellipsometry, label-free mass spectrometry [66] [69]. |
Experimental Protocols
Protocol 1: Measuring Bacterial Adhesion Forces on SAMs using AFM
This protocol is adapted from studies investigating Staphylococcus epidermidis interactions with SAMs, crucial for understanding the initial stages of biofilm formation on functionalized surfaces [67].
1. SAM Formation on Gold Substrates:
- Surface Preparation: Clean gold-coated substrates (e.g., on glass slides or AFM discs) via UV-ozone treatment or piranha solution (Caution: Extremely corrosive), followed by thorough rinsing with ethanol and deionized water.
- SAM Preparation: Immerse the clean gold substrates in a 1 mM ethanolic solution of the desired alkanethiol (e.g., IPA [isophthalic acid] or IAG [isophthalic acid with silver]) for 18-24 hours at room temperature [67].
- Post-Assembly Processing: Rinse the SAM-coated substrates rigorously with pure ethanol to remove physisorbed molecules and dry under a stream of inert gas (e.g., N₂).
2. Bacterial Probe Functionalization:
- Culture Preparation: Grow S. epidermidis (or target bacterium) in Tryptic Soy Broth (TSB) to mid-exponential growth phase [67].
- Harvesting and Immobilization: Centrifuge the bacterial culture, wash the pellet with a buffer like phosphate-buffered saline (PBS), and re-suspend to a defined optical density.
- AFM Tip Coating: Immerse tipless AFM cantilevers in a poly-L-lysine solution for 1 hour, rinse, and then incubate in the bacterial suspension for 15 minutes. Gently rinse the functionalized tip to remove loosely attached cells [67].
3. Adhesion Force Measurement:
- AFM Setup: Mount the SAM substrate and the bacterial-functionalized cantilever in the AFM liquid cell. Use an appropriate buffer (e.g., PBS).
- Force Curve Acquisition: Program the AFM to collect force-distance curves at multiple (e.g., 64-256) random locations on the SAM surface.
- Data Analysis: Analyze the retraction portion of the force curves to quantify the adhesion force (maximum pull-off force, in nN) and the work of adhesion (area under the curve). Compare forces across different SAM terminal groups and in the presence of model proteins like fibronectin [67].
Protocol 2: Label-Free Protein Detection via Surface Plasmon Resonance (SPR)
SPR is a powerful technique for real-time, label-free analysis of protein interactions on SAM-functionalized surfaces [66].
1. SPR Sensor Chip Functionalization:
- Surface Preparation: Use a commercial gold SPR sensor chip. Clean as per manufacturer's instructions (often similar to AFM substrate cleaning).
- SAM Formation: Assemble a mixed SAM by exposing the chip to a solution containing ~90% of a non-fouling alkanethiol (e.g., oligo(ethylene glycol) thiol) and ~10% of a functional thiol (e.g., carboxyl-terminated thiol) to create a low-fouling background with available sites for protein immobilization [66] [68].
- Ligand Immobilization: Activate the carboxyl groups on the SAM using a mixture of EDC (1-ethyl-3-(3-dimethylaminopropyl)carbodiimide) and NHS (N-hydroxysuccinimide). Immediately inject the purified protein ligand (e.g., an antibody or antigen) in sodium acetate buffer (pH 5.0) over the activated surface. Deactivate any remaining active esters with ethanolamine hydrochloride [66].
2. Binding Kinetics Analysis:
- Baseline Stabilization: Run the continuous flow of running buffer (e.g., HBS-EP: 10 mM HEPES, 150 mM NaCl, 3 mM EDTA, 0.005% v/v Surfactant P20, pH 7.4) over the functionalized sensor surface until a stable baseline is achieved.
- Sample Injection (Association Phase): Inject a series of concentrations of the analyte protein over the sensor surface at a constant flow rate (e.g., 30 µL/min) for a set time (e.g., 3-5 minutes).
- Dissociation Phase: Switch back to running buffer flow to monitor the dissociation of the bound complex for 5-10 minutes.
- Surface Regeneration: If the interaction is reversible, inject a regeneration solution (e.g., 10 mM glycine-HCl, pH 2.0) for 30 seconds to remove all bound analyte without damaging the immobilized ligand.
3. Data Processing:
- Reference Subtraction: Subtract the signal from a reference flow cell (coated with a non-reactive SAM) from the ligand flow cell data to correct for bulk refractive index changes and non-specific binding.
- Kinetic Analysis: Fit the processed sensorgrams (response vs. time) for all concentrations simultaneously to a suitable interaction model (e.g., 1:1 Langmuir binding) using the SPR instrument's software to determine the association rate constant (kₐ), dissociation rate constant (kd), and equilibrium dissociation constant (KD) [66].
Conceptual Workflows and Signaling Pathways
Bacterial Adhesion and Biofilm Formation on Surfaces
This diagram illustrates the multi-stage process of biofilm formation, which SAM-based strategies aim to disrupt.
Label-Free Protein Detection Principle
This diagram outlines the core working principle of optical label-free detection techniques like SPR, which can be applied to SAM-functionalized surfaces.
The Scientist's Toolkit: Research Reagent Solutions
The table below lists key materials and their functions for conducting experiments on SAM-based detection of bacteria and proteins.
Table 2: Essential Research Reagents for SAM-Based Detection Studies
| Reagent/Material | Function/Description | Example Application |
|---|---|---|
| Gold-coated Substrates | Provides a smooth, chemically uniform surface for covalent thiol-gold bond formation and SAM assembly. | AFM studies, SPR sensor chips [67] [68]. |
| Functional Alkanethiols | Molecules that form the SAM; terminal group (e.g., -EG, -COOH, -CH₃) dictates surface properties and functionality. | Creating non-fouling, protein-binding, or bactericidal surfaces [65] [67] [68]. |
| EDC & NHS | Cross-linking agents for activating carboxyl-terminated SAMs to enable covalent immobilization of protein ligands. | Preparing biosensor surfaces for protein capture [66] [68]. |
| Tryptic Soy Broth (TSB) | A rich nutrient medium for the cultivation and maintenance of a wide variety of fastidious and non-fastidious bacteria. | Growing bacterial cultures for adhesion assays [67]. |
| Fibronectin (FN) | A host extracellular matrix protein that can adsorb to surfaces and promote specific, ligand/receptor-mediated bacterial adhesion. | Modeling competitive protein-bacteria interactions on SAMs [67]. |
| Atomic Force Microscope (AFM) | An instrument used to measure nanoscale forces, such as the adhesion force between a single bacterium and a SAM surface. | Quantifying bacterial adhesion forces [67]. |
| Surface Plasmon Resonance (SPR) | An optical technique that detects changes in refractive index at a metal surface, allowing real-time monitoring of biomolecular binding events. | Label-free protein interaction kinetics [66]. |
Validating SAM Efficacy: Analytical Techniques and Performance Benchmarking
The study of self-assembled monolayers (SAMs) on gold surfaces for reducing non-specific adsorption (NSA) represents a critical area of interface science with profound implications for biomedical applications, including biosensors, medical implants, and drug delivery systems. Prof. George Whitesides' pioneering work established that surfaces presenting certain hydrophilic functional groups can exhibit remarkable resistance to protein adsorption and cell adhesion, forming the basis of "Whitesides' Rules" for bioinert surface design [70]. The advancement of this research field heavily depends on sophisticated analytical tools that can probe molecular interactions, layer properties, and binding events with high sensitivity. This application note provides a detailed examination of four essential analytical techniques—Surface Plasmon Resonance (SPR), Quartz Crystal Microbalance with Dissipation Monitoring (QCM-D), Spectroscopic Ellipsometry (SE), and Electrochemical Methods—within the specific context of characterizing SAMs on gold for NSA reduction. We present standardized protocols, comparative performance data, and practical guidance to enable researchers to select and implement the most appropriate characterization strategies for their specific biointerface projects.
Technique Comparisons and Capabilities
Comparative Analysis of Characterization Techniques
Table 1: Comparison of key analytical techniques for SAM characterization
| Technique | Measurement Principle | Information Output | Mass Sensitivity | Thickness Range | SAM Applications |
|---|---|---|---|---|---|
| SPR | Optical: Measures changes in plasmon resonance angle [71] | Binding/adsorption/desorption events; kinetic rate constants; affinity constants; concentration; optical mass (dry mass) [71] [72] | ~pg/mm² | < 10⁻¹⁰ m to 2-3·10⁻⁷ m [71] | Real-time monitoring of protein resistance; binding affinity studies; kinetics of molecular interactions [71] [72] |
| QCM-D | Acoustic: Measures shifts in resonance frequency and energy dissipation of oscillating crystal [71] | Hydrated mass (includes coupled water); layer softness/viscoelastic properties; conformational changes; thickness when modeled [71] | ~ng/cm² | 10⁻¹⁰ m to 10⁻⁶ m [71] | Hydration state of bioinert SAMs; structural changes in adsorbed layers; water coupling as signal enhancement [71] [70] |
| Ellipsometry | Optical: Measures change in polarization state of reflected light [73] | Film thickness; refractive index; optical constants; adsorption/desorption events [73] | Sub-nm thickness resolution | Monolayers to multilayers [73] | Ultrathin film characterization; thickness of SAMs and protein layers; in situ monitoring of binding [73] |
| Electrochemical Methods | Electrical: Measures electron transfer processes at electrode interface [74] | Electron transfer kinetics; surface coverage; SAM integrity and defects; redox activity [74] | Sub-monolayer coverage | N/A | SAM quality assessment; defect density; barrier properties; functional group accessibility [74] |
Technical Specifications and Experimental Considerations
Table 2: Technical specifications and experimental requirements
| Parameter | SPR | QCM-D | Ellipsometry | Electrochemical Methods |
|---|---|---|---|---|
| Sample Volume | Typically < µl - ~ µl per channel [71] | 15-40 µl (sample volume over surface) [71] | Platform dependent | Small volume cells possible |
| Substrate Requirements | Gold, silver (noble metals) [71] [75] | Any coating that is thin and rigid [71] | Any reflective surface | Conducting surfaces (gold ideal) |
| Liquid Compatibility | Excellent; microfluidic flow cells standard [72] | Excellent; flow cells available [71] | In situ liquid cells possible | Designed for liquid electrolytes |
| Throughput | Moderate to high; multi-channel systems [72] | Moderate; typically 1-4 sensors simultaneously | Low to moderate | High; rapid measurements |
| Key Strengths | Excellent for kinetics and affinity; small molecule detection; gold standard for binding studies [71] [72] | Sensitive to hydrated mass and viscoelastic properties; detects structural changes [71] | Non-invasive; accurate thickness measurement; can detect molecular absorptions [73] | Highly sensitive to SAM defects; provides quantitative surface coverage [74] |
| Limitations | Limited to ~200-300 nm thickness; insensitive to water content [71] | Larger sensing area requires more sample; sensitive to viscosity changes [76] | Data analysis can be complex for complex layers | Requires redox-active species or conductive SAMs |
Technique Selection Guide
Application-Based Technique Selection
The choice of analytical technique depends heavily on the specific research questions and the properties of the SAM system under investigation. For comprehensive characterization of SAMs for NSA reduction, a combination of techniques is often necessary to obtain a complete picture of both structural and functional properties.
For investigators focused primarily on protein resistance performance, SPR provides exceptional sensitivity for quantifying non-specific binding events in real-time and is considered the gold standard for kinetic analysis [71] [72]. When hydration states and water coupling are of interest, particularly for oligo(ethylene glycol) or zwitterionic SAMs where interfacial water structure is crucial for bioinertness, QCM-D offers unique capabilities as it senses the hydrated mass including water molecules associated with the SAM [71] [70]. For structural characterization of the SAM itself, including thickness, packing density, and optical properties, spectroscopic ellipsometry provides precise, non-invasive measurement capabilities [73]. When assessing SAM quality, defect density, and electrochemical barrier properties, electrochemical methods including cyclic voltammetry and electrochemical impedance spectroscopy are unparalleled in their sensitivity to even minor defects in the monolayer [74].
Complementary Techniques for Comprehensive Analysis
The most insightful studies of bioinert SAMs often employ multiple complementary techniques. For example, combining QCM-D and SPR provides both hydrated and dry mass measurements, offering insight into the water content of the SAM and any adsorbed layers [71] [76]. Similarly, ellipsometry can be combined with electrochemical methods to correlate SAM thickness with barrier properties [73] [74]. For investigating the mechanism of bioinertness, each technique provides a different perspective: QCM-D can detect changes in coupled water, SPR precisely measures binding kinetics, electrochemical methods assess SAM integrity, and ellipsometry provides structural information.
Experimental Protocols
Protocol 1: SPR Analysis of Protein Resistance on SAMs
Purpose: To quantitatively evaluate the resistance of SAM-coated gold surfaces to non-specific protein adsorption using SPR.
Materials:
- SPR instrument with gold sensor chip
- Alkanethiols for SAM formation (e.g., HS-C11-EG3-OH, HS-C16-EG3-NTA)
- Absolute ethanol (≥99.8%)
- Phosphate buffered saline (PBS), pH 7.4
- Protein solution (e.g., 1 mg/mL fibrinogen in PBS)
- Regeneration solution (e.g., 10 mM glycine-HCl, pH 2.0)
Procedure:
- Surface Preparation: Clean gold SPR sensor chip using oxygen plasma treatment or piranha solution (Caution: extremely corrosive).
- SAM Formation: Immerse clean gold chip in 1 mM thiol solution in ethanol for 18-24 hours at room temperature.
- SAM Rinsing: Remove chip from thiol solution, rinse thoroughly with ethanol, and dry under stream of nitrogen.
- Instrument Priming: Prime SPR instrument with running buffer (PBS) until stable baseline is achieved.
- Baseline Establishment: Mount SAM-functionalized chip in SPR instrument and establish stable baseline in buffer flow.
- Protein Challenge: Inject protein solution (1 mg/mL in PBS) over SAM surface at flow rate of 30 µL/min for 5-10 minutes.
- Dissociation Phase: Switch to buffer flow to monitor dissociation of loosely bound protein.
- Surface Regeneration: If necessary, inject regeneration solution to remove any bound protein (optional for NSA studies).
- Data Analysis: Calculate response units (RU) corresponding to protein adsorption; compare against bare gold and positive controls.
Data Interpretation: Bioinert SAMs such as oligo(ethylene glycol)-terminated thiols typically show >90% reduction in protein adsorption compared to bare gold surfaces [70]. The real-time binding curves provide information on both the amount of non-specific adsorption and the adsorption kinetics.
Protocol 2: QCM-D Analysis of Hydrated SAM Properties
Purpose: To characterize the hydrated mass, viscoelastic properties, and hydration state of bioinert SAMs on gold.
Materials:
- QCM-D instrument with gold-coated quartz crystals
- Alkanethiol solutions for SAM formation
- Absolute ethanol
- PBS buffer, pH 7.4
Procedure:
- Crystal Preparation: Clean gold-coated QCM-D crystals using UV-ozone treatment or plasma cleaning.
- Baseline Measurement: Mount clean crystal in QCM-D instrument and establish frequency (f) and dissipation (D) baselines in air or liquid.
- SAM Formation: For in situ SAM formation, inject 1-2 mM thiol solution in ethanol over gold surface at flow rate of 50-100 µL/min.
- SAM Stabilization: Monitor f and D shifts until stable (typically 1-2 hours for full formation).
- Buffer Exchange: Rinse with pure ethanol followed by PBS buffer to remove unbound thiols.
- Hydration Assessment: Compare frequency shifts between air and liquid environments to assess water coupling.
- Protein Challenge: Expose SAM to protein solution (as in SPR protocol) while monitoring f and D.
- Data Analysis: Calculate hydrated mass using Sauerbrey relation for rigid layers or apply viscoelastic modeling for soft layers.
Data Interpretation: Frequency decreases (Δf) indicate mass increases, while dissipation changes (ΔD) reveal structural changes: increased dissipation indicates softer, more viscous layers. Bioinert SAMs typically show significant water coupling (large Δf in liquid vs air) and minimal Δf and ΔD changes during protein challenge [71] [70].
Protocol 3: Spectroscopic Ellipsometry of SAM Thickness and Optical Properties
Purpose: To determine the thickness and optical constants of SAMs on gold substrates.
Materials:
- Spectroscopic ellipsometer
- Gold substrates on silicon or glass
- Alkanethiol solutions
- Ethanol for rinsing
Procedure:
- Substrate Preparation: Clean gold substrates thoroughly before SAM formation.
- Reference Measurement: Measure ellipsometry spectra (Ψ and Δ) of bare gold substrate.
- SAM Formation: Form SAM on gold as described in previous protocols.
- SAM Measurement: Measure ellipsometry spectra of SAM-functionalized surface.
- Data Analysis: Calculate difference spectra (δΔ and δΨ) to emphasize SAM contribution [73].
- Optical Modeling: Fit data using appropriate optical model (typically transparent layer on metallic substrate).
- Thickness Determination: Extract SAM thickness from best-fit model.
Data Interpretation: Well-ordered alkanethiol SAMs on gold typically show thicknesses of 1.5-3.0 nm, depending on chain length [73]. The difference spectra method helps distinguish specific molecular features and can detect even submonolayer changes.
Protocol 4: Electrochemical Characterization of SAM Integrity
Purpose: To assess the quality, defect density, and barrier properties of SAMs using electrochemical methods.
Materials:
- Potentiostat with three-electrode setup
- Gold working electrode
- Platinum counter electrode
- Reference electrode (Ag/AgCl)
- Redox probe (e.g., 1 mM K₃Fe(CN)₆ in 0.1 M KCl)
- Electrolyte solution (0.1 M K₂SO₄ or KCl)
Procedure:
- Electrode Preparation: Clean gold working electrode by polishing and electrochemical cycling.
- SAM Formation: Form SAM on gold working electrode as described previously.
- Electrochemical Cell Assembly: Assemble three-electrode cell with SAM-modified working electrode.
- Cyclic Voltammetry: Record CV curves in redox probe solution at scan rates from 10-500 mV/s.
- Impedance Spectroscopy: Perform EIS measurements from 10⁵ to 10⁻² Hz at formal potential of redox couple.
- Data Analysis: Calculate surface coverage from suppression of Faradaic current; analyze impedance data using equivalent circuit modeling.
Data Interpretation: High-quality, defect-free SAMs exhibit strong suppression of Faradaic current (>95% reduction compared to bare gold) and high charge transfer resistance [74]. The surface coverage (θ) can be calculated from CV data using: θ = 1 - (QSAM/Qbare), where Q represents charge under oxidation or reduction peak.
Research Reagent Solutions
Table 3: Essential materials for SAM research and their functions
| Material | Function/Application | Examples/Specifications |
|---|---|---|
| Gold Substrates | Platform for thiol-based SAM formation | Template-stripped gold; evaporated gold films; gold-coated QCM crystals; SPR chips |
| Alkanethiols | SAM-forming molecules | Varying chain lengths (C6-C16); terminal groups (OH, OEG, CH₃, COOH); mixture compositions for gradient studies [70] [77] |
| Protein Solutions | Biofouling challenge | Fibrinogen, serum albumin, full serum; typical concentration 1 mg/mL in PBS [70] |
| Redox Probes | Electrochemical characterization | Ferricyanide ([Fe(CN)₆]³⁻/⁴⁻); ruthenium hexamine ([Ru(NH₃)₆]³⁺/²⁺); concentration typically 1-5 mM [74] |
| Buffer Systems | Physiological relevant conditions | Phosphate buffered saline (PBS), HEPES; typically 10-100 mM, pH 7.4 |
| Solvents | SAM formation and cleaning | Absolute ethanol (>99.8%) for thiol solutions; ultrapure water (18.2 MΩ·cm) |
Experimental Workflow Visualization
Diagram 1: Comprehensive SAM characterization workflow illustrating the sequential process from substrate preparation through technique-specific analysis to data integration for performance assessment.
The strategic combination of SPR, QCM-D, ellipsometry, and electrochemical methods provides researchers with a powerful toolkit for comprehensive characterization of self-assembled monolayers designed to reduce non-specific adsorption on gold surfaces. Each technique offers unique capabilities and insights, with SPR excelling in binding kinetics, QCM-D revealing hydrated mass and viscoelastic properties, ellipsometry providing precise structural information, and electrochemical methods assessing SAM integrity and defect density. The protocols and comparisons presented in this application note enable researchers to select appropriate techniques, implement standardized methodologies, and interpret results within the framework of bioinert surface design. As research in biointerface science continues to advance, these analytical tools will remain essential for developing next-generation surfaces with enhanced resistance to non-specific adsorption for biomedical and biotechnological applications.
Within the field of surface science and biotechnology, non-specific adsorption (NSA) poses a significant challenge to the performance of biosensors, diagnostic assays, and biomedical devices. For surfaces functionalized with self-assembled monolayers (SAMs) on gold, quantifying the extent of NSA is paramount to evaluating and optimizing their antifouling properties. This application note provides detailed protocols and performance metrics for researchers and drug development professionals engaged in the development of SAM-based coatings, focusing on standardized methods to accurately measure and compare protein adsorption on gold surfaces. The protocols outlined herein enable the quantitative assessment of SAM performance, facilitating the development of highly specific sensing platforms and reliable biomedical implants.
Quantifying NSA on SAM-Coated Gold: Core Principles and Metrics
The primary goal of quantifying NSA is to determine the amount of protein that adsorbs to a surface independently of specific biorecognition events. This measurement directly informs on the efficacy of an antifouling SAM coating.
- Key Performance Metrics: The fundamental quantitative metric is the surface mass density of adsorbed protein, often expressed in ng/cm². This value is crucial for comparing the performance of different SAM formulations. A lower surface mass density indicates superior antifouling performance. The data is typically presented as the mean ± standard deviation from replicate measurements (e.g., n ≥ 3) to ensure statistical significance.
- The Critical Role of SAM Terminal Chemistry: Research consistently demonstrates that the chemical terminal group of the SAM is a dominant factor in controlling NSA. A foundational study utilizing surface plasmon resonance (SPR) revealed that the surface concentration of human IgG adsorbed on different SAMs decreased in the order: CH₃ > C₆H₄OH > COO⁻ > NH₂ > OH > oligoethylene oxide (OEO). The OEO-terminated SAM exhibited exceptional performance, with protein adsorption below the detection limit of 0.5 ng/cm², approximately 1000 times lower than on a hydrophobic CH₃-terminated SAM [78].
Table 1: Quantitative Protein Adsorption on SAMs with Different Terminal Groups as Measured by SPR
| SAM Terminal Group | Human IgG Adsorption (Relative) | BSA Adsorption (Relative) | Key Interaction Forces |
|---|---|---|---|
| CH₃ (Methyl) | Very High | High | Hydrophobic interactions |
| C₆H₄OH (Phenol) | High | Very High | Hydrophobic, π-π, H-bonding |
| COO⁻ (Carboxylate) | Medium | Medium | Electrostatic, ionic |
| NH₂ (Amino) | Low | Low | Electrostatic, H-bonding |
| OH (Hydroxyl) | Very Low | Very Low | Hydrophilic, H-bonding |
| OEO (Oligoethylene Oxide) | Undetectable (<0.5 ng/cm²) | Undetectable (<0.5 ng/cm²) | Steric repulsion, hydration |
Detailed Experimental Protocols
The following sections provide step-by-step protocols for quantifying NSA, ranging from a widely accessible bulk method to more advanced surface-sensitive techniques.
Protocol 1: Quantifying NSA via a Micro-BCA Assay (Bulk Measurement)
This protocol is ideal for initial screening of SAM performance and does not require specialized instrumentation beyond a microplate reader [79].
- Principle: Proteins adsorbed on a surface are desorbed via sonication and quantified using a micro-bicinchoninic acid (BCA) assay, which detects reduced copper ions in the presence of peptide bonds.
- Materials:
- SAM-coated gold substrates
- Protein solution (e.g., 2 mg/mL BSA, Fibrinogen, or diluted serum in PBS or deionized water)
- 24-well plate
- Deionized water
- Ultrasonic bath
- Micro-BCA protein assay kit
- Microplate reader
- Procedure:
- Incubation: Place the SAM-coated gold substrate in a 24-well plate and incubate with 1 mL of the protein solution (2 mg/mL) at 37°C for 2 hours.
- Rinsing: Gently remove the sample from the protein solution and wash it five times with copious amounts of deionized water to remove loosely adsorbed proteins.
- Desorption: Transfer the sample to a clean tube containing 1 mL of fresh deionized water. Subject it to sustained ultrasound cleaning for 10 minutes to desorb the firmly bound proteins.
- Quantification: Perform the micro-BCA assay on the resulting solution according to the manufacturer's instructions. Use standard solutions of the same protein to generate a calibration curve for accurate quantification.
- Calculation: The mass of adsorbed protein is calculated from the BCA results. The surface mass density is then determined by dividing the total adsorbed protein mass by the total surface area of the substrate exposed to the protein solution.
Protocol 2: Quantifying NSA via Surface Plasmon Resonance (SPR)
SPR provides real-time, label-free monitoring of adsorption kinetics and is a gold standard for surface-specific quantification [78].
- Principle: SPR measures changes in the refractive index at the surface of a thin gold film. The adsorption of biomolecules (with a higher refractive index than the buffer) causes a measurable shift in the resonance angle, which is directly proportional to the surface mass density.
- Materials:
- SPR instrument equipped with a flow cell
- SAM-coated gold sensor chip
- Running buffer (e.g., PBS)
- Protein solutions in running buffer (e.g., 100 µg/mL IgG or BSA)
- Procedure:
- Baseline Establishment: Dock the SAM-coated sensor chip in the SPR instrument. Flow running buffer at a constant rate (e.g., 20-50 µL/min) until a stable baseline is achieved.
- Sample Injection: Inject the protein solution over the sensor surface for a fixed period (e.g., 10-15 minutes) to allow for adsorption.
- Dissociation Phase: Switch back to running buffer to monitor the dissociation of loosely bound proteins.
- Regeneration (Optional): A regeneration solution (e.g., mild acid or base) may be used to remove all adsorbed protein and prepare the surface for a new experiment.
- Data Analysis: The response difference (in resonance units, RU) between the final baseline and the baseline before injection is used to calculate the adsorbed mass. A conversion factor of 1000 RU ≈ 1 ng/mm² is often used for proteins. Kinetic parameters (association/dissociation rates) can also be extracted from the sensorgram.
Figure 1: SPR Experimental Workflow for Quantifying NSA.
Protocol 3: Quantifying Immobilized Antibody Activity on Gold Nanoparticles
For functionalized surfaces, it is critical to distinguish between total adsorbed protein and the fraction that is biologically active. This enzyme-mediated protocol quantifies both the loading and the activity of antibodies immobilized on gold nanoparticles (AuNPs) [80].
- Principle: Anti-horseradish peroxidase (HRP) antibody is immobilized onto AuNPs. The total antibody loading is determined by quantifying the unbound antibody in the supernatant. The activity is determined by saturating the AuNP-antibody conjugates with HRP antigen and quantifying the bound HRP via its enzymatic activity.
- Materials:
- Gold nanoparticles (e.g., 60-100 nm)
- Anti-HRP antibody
- Horseradish Peroxidase (HRP)
- Protein A (for oriented immobilization, optional)
- Modified Bradford assay reagents
- HRP enzyme activity substrate (e.g., TMB)
- Microplate reader
- Procedure:
- Conjugate Formation: Incubate excess anti-HRP antibody with AuNPs (via direct adsorption or protein A-mediated binding) for 1 hour.
- Quantify Loading: Separate the AuNP conjugates and quantify the unbound antibody in the supernatant using a modified Bradford assay. The loaded antibody is calculated as the difference between added and unbound antibody.
- Determine Activity: Incubate the Ab-AuNP conjugates with excess HRP to saturate all accessible binding sites. Wash to remove unbound HRP.
- Enzymatic Quantification: Add HRP substrate and measure the enzymatic reaction rate. Compare this rate to a standard curve of free HRP to determine the number of active HRP molecules bound, which corresponds to the number of active antibodies.
- Calculation:
- Total Antibody Loading:
(Initial Ab - Unbound Ab) / Number of AuNPs = Abs/AuNP - Active Fraction:
(Number of active Abs / Total loaded Abs) * 100 = % Active Antibodies
- Total Antibody Loading:
Table 2: The Scientist's Toolkit: Essential Reagents for NSA Quantification
| Research Reagent / Material | Function / Explanation |
|---|---|
| SAM-forming Thiols (e.g., OEG-terminated) | Creates a well-defined, antifouling surface on gold; the OEG group provides steric repulsion and hydration to resist protein adsorption [9] [78]. |
| Model Proteins (BSA, IgG, Fibrinogen) | Representative proteins used to challenge surfaces; BSA is a small, abundant serum protein, while IgG and Fibrinogen are larger and more sticky, providing a robust NSA test [78] [79]. |
| Micro-BCA Assay Kit | A colorimetric method for total protein quantification; highly sensitive and compatible with the eluates from desorbed proteins [79]. |
| SPR Sensor Chip (Gold Film) | The core substrate for real-time, label-free monitoring of biomolecular adsorption kinetics and quantification of surface mass density [78]. |
| Anti-HRP Antibody | A specialized antibody used in activity assays; its binding to the HRP antigen produces a quantifiable enzymatic signal, enabling measurement of the active fraction of immobilized antibodies [80]. |
| Protein A | An immobilization ligand that binds the Fc region of antibodies, promoting an oriented attachment on gold surfaces which maximizes antigen-binding site availability and activity [80]. |
The rigorous quantification of non-specific adsorption is a critical step in the development of effective SAM-based coatings for gold surfaces. The protocols detailed in this document—from the accessible micro-BCA assay to the sophisticated real-time analysis provided by SPR and the functional activity assessment—provide researchers with a comprehensive toolkit for evaluating antifouling performance. By applying these standardized methods and focusing on key quantitative metrics like surface mass density and active antibody fraction, scientists can make informed, data-driven decisions to optimize surface chemistry, ultimately leading to more reliable and high-performing biosensors, diagnostic assays, and biomedical devices.
Self-assembled monolayers (SAMs) on gold surfaces are a cornerstone technology for controlling biointerfacial interactions in diagnostics, biosensing, and biomedical research. A primary function of SAMs is to minimize non-specific adsorption (NSA) of biomolecules, which can cause false signals, reduce sensitivity, and compromise the accuracy of analytical results. This application note provides a comparative analysis of prominent SAM chemistries, evaluating their efficacy in reducing NSA. We present quantitative performance data, detailed experimental protocols for replication, and a practical toolkit to guide researchers in selecting and implementing optimal surface functionalization strategies for their specific applications.
Non-specific adsorption (NSA), the undesirable physisorption of biomolecules to a sensor or assay surface, remains a persistent challenge in biotechnology. It leads to elevated background noise, reduced signal-to-noise ratios, decreased sensitivity, and false-positive results, ultimately undermining the reliability of data [1]. For biosensors operating in complex matrices like serum, plasma, or cell lysate—which can contain 40-80 mg/mL of non-target protein—controlling NSA is not merely beneficial but essential for functionality [81].
SAMs, formed by the spontaneous organization of thiolated molecules onto gold surfaces, provide a powerful and versatile method to create chemically well-defined interfaces. By presenting specific terminal functional groups, SAMs can be engineered to resist biomolecular adsorption while providing anchor points for the covalent immobilization of capture molecules like antibodies or aptamers [82] [83]. The performance of a SAM, however, is highly dependent on its molecular composition, assembly protocol, and the nature of the target analyte. This report delivers a head-to-head comparison of these critical parameters.
Comparative Performance of SAM Chemistries
The effectiveness of a SAM in resisting NSA is a function of its ability to form a dense, well-ordered, and hydrophilic barrier. The following table summarizes the performance of various SAM chemistries as reported in the literature.
Table 1: Comparative Performance of Different SAM Chemistries in Reducing NSA
| SAM Chemistry / Coating | Key Characteristics | Reported NSA Performance | Best For | Key Considerations |
|---|---|---|---|---|
| Alkanethiolates (e.g., mercaptoundecanoic acid) | Well-established; terminal COOH for biomolecule conjugation [28] [83]. | Good for protein detection; inadequate for direct bacteria detection [28] [83]. | Protein detection in buffer; fundamental studies. | Performance is highly dependent on chain length and packing. |
| Polyethylene Glycol (PEG) | Hydrophilic polymer; industry standard for "stealth" coatings [82] [1]. | Good NSA reduction, but outperformed by newer chemistries in complex media [81]. | General purpose NSA reduction in moderate conditions. | Can be susceptible to oxidative degradation. |
| Peptide-based (Afficoat) | Zwitterionic, hydrophilic peptide sequence; forms a stable SAM [81]. | Superior NSA reduction in crude serum (76 mg/mL protein) vs. PEG and CM-Dextran [81]. | Biosensing in complex, undiluted biological samples (serum, lysate). | Proprietary peptide; requires specific conjugation knowledge. |
| Co-adsorbed SAMs | Mixed monolayer (e.g., 2PACz/PyCA-3F) to reduce molecular aggregation [84]. | Smoother, more uniform surfaces with improved electronic properties for photovoltaics [84]. | Electronic devices; interfaces where morphology and work function are critical. | Emerging strategy; protocols are less standardized for biosensing. |
| Dextran Hydrogel | 3D hydrogel matrix that resists fouling [85]. | Moderate NSA reduction; performance varies [85]. | SPR applications where a 3D matrix is beneficial for loading. | Not a monolayer; thicker 3D structure can alter sensor physics. |
| Simple Physisorption | Random adsorption of antibodies directly onto bare gold [28] [83]. | Poor NSA resistance, but surprisingly effective for capturing large targets like bacteria [28]. | Cost-effective capture of large cells/organisms where orientation is less critical. | High background likely; not suitable for sensitive protein detection. |
Detailed Experimental Protocols
The following sections provide step-by-step protocols for constructing and evaluating two of the most performant SAM types from the comparison.
Protocol 1: Optimized Alkanethiolate SAM for Protein Detection
This protocol is adapted from studies that systematically optimized SAM parameters for immunoassays [28] [83].
Research Reagent Solutions:
- Gold Substrate: Template-stripped gold or polished gold electrode.
- SAM Solution: 1 mM mercaptoundecanoic acid (MUA) in absolute ethanol.
- Activation Buffer: 0.1 M MES, 0.5 M NaCl, pH 6.0.
- Coupling Agents: 0.4 M EDC (1-Ethyl-3-(3-dimethylaminopropyl)carbodiimide) and 0.1 M NHS (N-hydroxysuccinimide) in Activation Buffer.
- Antibody Solution: 10-50 µg/mL capture antibody in 10 mM phosphate buffer, pH 7.4.
Methodology:
- Gold Substrate Preparation: Clean the gold substrate using oxygen plasma treatment or by immersion in piranha solution (Caution: Piranha solution is extremely corrosive and must be handled with extreme care), followed by thorough rinsing with deionized water and absolute ethanol. Dry under a stream of nitrogen.
- SAM Formation: Immerse the clean gold substrate in the 1 mM MUA/ethanol solution. Allow self-assembly to proceed for a minimum of 12 hours at room temperature in a sealed, dark container.
- Surface Activation: Rinse the SAM-functionalized substrate thoroughly with ethanol and dry with nitrogen. Incubate the surface with the EDC/NHS mixture for 15 minutes to activate the terminal carboxylic acid groups to form NHS esters.
- Antibody Conjugation: Rinse the activated surface with Activation Buffer. Immediately incubate with the Antibody Solution for 30-60 minutes at room temperature.
- Quenching & Blocking: Rinse the surface with PBS. To block any remaining activated esters and non-specific sites, incubate with a 1 M ethanolamine solution (pH 8.5) for 10 minutes, followed by a 1% (w/v) BSA in PBS solution for 30 minutes.
- Storage: The functionalized surface can be stored in PBS at 4°C for short-term use.
Protocol 2: Peptide SAM (Afficoat-like) for Complex Samples
This protocol is based on the development of ultra-low fouling peptide SAMs for use in serum and cell lysate [81].
Research Reagent Solutions:
- Gold Sensor Chip: (e.g., SPR chip).
- Peptide Solution: 1 mM zwitterionic, thiol-terminated peptide (e.g., sequence pattern with alternating hydrophilic/hydrophobic amino acids) in nuclease-free water.
- Complex Sample: Crude bovine serum or cell lysate.
Methodology:
- Gold Cleaning: Clean the gold sensor chip as described in Protocol 1, Step 1.
- Peptide SAM Formation: Incubate the clean, dry gold surface with the 1 mM Peptide Solution for a minimum of 4 hours at room temperature.
- SAM Stabilization: Rinse the surface extensively with PBS and water to remove physisorbed peptides. Stabilize the monolayer by soaking in PBS for 1 hour.
- NSA Challenge Test: To quantify NSA resistance, expose the peptide SAM to a complex biological sample (e.g., 76 mg/mL protein bovine serum) for 20 minutes.
- Rinsing and Quantification: Rinse the surface with PBS for 5 minutes to remove loosely adsorbed material. Quantify the amount of non-specifically adsorbed protein using a sensitive technique like Surface Plasmon Resonance (SPR) or quartz crystal microbalance (QCM).
Workflow Visualization
The following diagram illustrates the logical flow for developing and testing an effective SAM-based interface.
Diagram 1: SAM Selection and Testing Workflow
The Scientist's Toolkit: Essential Reagents and Materials
Successful SAM development relies on a set of key reagents and instruments. The following table outlines the essential components of the toolkit.
Table 2: Research Reagent Solutions for SAM Development
| Item / Reagent | Function / Role | Example & Notes |
|---|---|---|
| Gold Substrates | Provides the foundational surface for thiol-gold covalent bonding. | SPR chips, template-stripped gold, gold-coated electrodes. Purity and surface roughness are critical. |
| Thiolated Molecules | The building blocks of the SAM. | Alkanethiols (e.g., MUA, MCH), thiol-terminated PEG, custom peptides (e.g., Afficoat). |
| Blocking Agents | Passivates unreacted gold and SAM defects to minimize NSA. | Bovine Serum Albumin (BSA), casein, ethanolamine, 6-Mercapto-1-hexanol (MCH). |
| Coupling Chemistry | Activates SAM terminal groups for biomolecule conjugation. | EDC/NHS carbodiimide chemistry for carboxyl groups; Sulfo-SMCC for amine-maleimide coupling. |
| Characterization Instruments | Validates SAM formation, quality, and NSA performance. | Surface Plasmon Resonance (SPR), Electrochemical Impedance Spectroscopy (EIS), Ellipsometry, AFM. |
The choice of SAM chemistry is a decisive factor in the success of any surface-based bioanalytical application. While traditional alkanethiols and PEG coatings provide a solid foundation, advanced strategies like zwitterionic peptide SAMs and co-adsorbed monolayers demonstrate superior performance in demanding environments. The data and protocols provided herein offer a roadmap for researchers to make informed decisions, enabling the development of more sensitive, specific, and reliable biosensors and assays. Future directions in SAM research will continue to focus on enhancing stability, dynamic control, and performance in ever-more complex biological milieus.
Assessing Specific Binding Affinity (Kd) in the Presence of Complex Samples
The accurate determination of the equilibrium dissociation constant (Kd) is fundamental to biophysical characterization in drug discovery, providing critical information about the strength of interactions between therapeutic candidates and their targets. However, measuring true binding affinity becomes significantly more challenging when performed in complex, biologically relevant samples such as serum, cell lysates, or crude extracellular mixtures. These matrices introduce numerous complicating factors including non-specific binding, spectral interference, and molecular crowding that can obscure specific interaction signals.
For research focused on self-assembled monolayers (SAMs) on gold surfaces for reducing nonspecific adsorption (NSA), this challenge presents both a hurdle and a validation opportunity. Well-designed SAM coatings intrinsically resist fouling from complex samples, thereby enhancing the signal-to-noise ratio for measuring specific binding events. This application note provides detailed methodologies for assessing specific binding affinity within complex samples, leveraging the non-fouling properties of SAM-coated gold surfaces to obtain reliable Kd measurements that more closely mimic in vivo conditions.
Technical Background
The Critical Role of Kd Measurements in Complex Environments
Characterizing molecular interactions under idealized buffer conditions provides valuable baseline data, but often fails to predict performance in physiological environments. Complex biological samples like serum and cell lysates contain numerous confounding components:
- Serum proteins (e.g., albumin, immunoglobulins) that can bind analytes or surfaces
- Lipids and extracellular vesicles that may sequester hydrophobic compounds
- Proteases and nucleases that can degrade protein or nucleic acid-based therapeutics
- Soluble receptors and binding partners that compete with the intended interaction
These factors collectively alter the effective concentration of free analyte, potentially leading to significant overestimation of Kd values if not properly controlled. Performing affinity measurements in these matrices provides crucial data on whether a binding interaction remains functionally specific under biologically competitive conditions—a key predictor of therapeutic efficacy.
SAMs as a Platform for Reducing Nonspecific Background
Self-assembled monolayers (SAMs) of organothiols on gold surfaces provide a highly controlled interface that can be engineered to minimize nonspecific interactions. The fundamental structure consists of three components:
- Headgroup (typically thiol, disulfide, or phosphonic acid) that chemisorbs to the gold surface
- Spacer chain (often alkane or oligoethylene glycol) that provides structural integrity
- Terminal functional group (e.g., hydroxyl, carboxyl, or oligoethylene glycol) that presents specific chemical properties to the solution interface
When properly designed, SAMs create a tightly packed, homogeneous surface that effectively resists protein adsorption from complex samples. This property is crucial for maintaining assay specificity when working with biological fluids, as it ensures that detected binding signals originate primarily from the specific interaction of interest rather than adventitious adsorption.
Experimental Protocols
Protocol 1: Surface Plasmon Resonance (SPR) with SAM-Modified Sensor Chips
Surface Plasmon Resonance (SPR) is a powerful label-free technique for measuring binding affinities and kinetics in real-time. The following protocol details its application with SAM-functionalized surfaces for complex sample analysis [86] [87] [88].
Sensor Chip Preparation
Gold Surface Cleaning
- Clean bare gold sensor chips (e.g., CM5 from GE Healthcare) with fresh piranha solution (3:1 H₂SO₄:H₂O₂) for 2 minutes
- Caution: Piranha solution is extremely corrosive and must be handled with appropriate personal protective equipment
- Rinse thoroughly with absolute ethanol and dry under a stream of nitrogen
SAM Formation
- Prepare a 1 mM solution of hydroxyl-terminated thiol (e.g., 11-mercapto-1-undecanol) in absolute ethanol
- Immerse the clean gold chip in the thiol solution for 24 hours at room temperature under nitrogen atmosphere
- Rinse sequentially with fresh ethanol, ethanol:toluene (1:1), and toluene to remove physisorbed thiols
- Dry under nitrogen and characterize by contact angle goniometry (expected water contact angle: 27.6° ± 4.3°) [89] [90]
Ligand Immobilization
- For covalent attachment, activate the SAM surface with NHS/EDC chemistry for 7 minutes
- Dilute the ligand to 10-50 µg/mL in 10 mM sodium acetate buffer (pH 4.0-5.5) and inject until desired immobilization level is reached (typically 50-100 RU for small molecules, 500-5000 RU for proteins)
- Deactivate excess activated esters with 1 M ethanolamine-HCl (pH 8.5)
- For capture methods, use pre-functionalized surfaces (e.g., streptavidin, anti-His antibody)
SPR Analysis with Complex Samples
Running Buffer Preparation
- Prepare HBS-EP+ buffer (10 mM HEPES, 150 mM NaCl, 3 mM EDTA, 0.05% v/v surfactant P20, pH 7.4)
- For serum-containing experiments, supplement with 1-10% (v/v) heat-inactivated fetal bovine serum
- For cell lysate experiments, clarify lysate by centrifugation (16,000 × g, 10 minutes) and filtering (0.22 µm) before adding to buffer (final concentration 0.1-1 mg/mL total protein)
Analyte Preparation in Complex Matrices
- Prepare serial dilutions of the analyte in running buffer containing the complex matrix
- For small molecules dissolved in DMSO, ensure consistent DMSO concentration (typically ≤1%) across all samples and running buffer
- Centrifuge all samples (16,000 × g, 10 minutes) immediately before injection to remove aggregates
Binding Measurement
- Prime the SPR instrument with running buffer containing the appropriate complex matrix
- Inject analyte solutions over both active and reference surfaces at a flow rate of 30 µL/min for 2-3 minutes
- Monitor dissociation for 5-10 minutes
- Regenerate the surface with appropriate conditions (e.g., 10 mM glycine pH 2.0 for 30 seconds)
- Include blank injections (matrix without analyte) for double-referencing
Data Analysis
- Subtract reference surface signals and blank injections
- Fit the resulting sensorgrams to appropriate binding models (1:1 Langmuir, two-state, or conformational change)
- Calculate kinetic parameters (ka, kd) and derive equilibrium Kd from the ratio kd/ka
Table 1: Key SPR Experimental Parameters for Complex Sample Analysis
| Parameter | Standard Condition | Complex Sample Adaptation |
|---|---|---|
| Flow Rate | 30 µL/min | 30-50 µL/min (higher to minimize mass transport) |
| Association Time | 2-3 minutes | 3-5 minutes (for slower equilibration) |
| Dissociation Time | 5-10 minutes | 5-15 minutes (for slower dissociation) |
| Surface Regeneration | Glycine pH 2.0, 30 sec | Varied based on complex matrix (may require optimization) |
| Running Buffer | HBS-EP+ | HBS-EP+ + 1-10% serum or 0.1-1 mg/mL lysate |
| DMSO Tolerance | ≤1% | ≤1% (must be matched in all solutions) |
Protocol 2: Competitive Fluorescence Binding Assay with SAM-Functionalized Surfaces
This protocol describes a fluorescence-based method for determining binding affinity in complex samples using SAM-coated nanoparticles to minimize nonspecific background [68].
SAM-Coated Nanoparticle Preparation
Gold Nanoparticle Synthesis
- Prepare gold nanoparticles (AuNPs) by citrate reduction method: Heat 100 mL of 0.25 mM HAuCl₄ to boiling, then rapidly add 2.5 mL of 1% sodium citrate solution while stirring
- Continue heating and stirring for 15 minutes until the solution develops a deep red color
- Cool to room temperature and characterize by UV-Vis spectroscopy (λmax ≈ 520 nm) and dynamic light scattering (size ≈ 15-20 nm)
SAM Formation on AuNPs
- Add hydroxyl-terminated thiol (e.g., 11-mercapto-1-undecanol) to the AuNP solution at 1 mM final concentration
- Stir gently for 24 hours at room temperature
- Purify by repeated centrifugation (14,000 × g, 15 minutes) and resuspension in ethanol
- Transfer to aqueous buffer via sequential solvent exchange (ethanol:PBS mixtures)
Fluorophore-Ligand Conjugation
- Activate the SAM surface with NHS/EDC chemistry for 30 minutes
- Incubate with fluorescently-labeled ligand (e.g., FITC-conjugated) at 10 µg/mL for 2 hours
- Block excess activated esters with 1 M ethanolamine-HCl (pH 8.5)
- Purify by centrifugation and resuspend in assay buffer
Competitive Binding Assay
Sample Preparation
- Prepare serial dilutions of unlabeled analyte in assay buffer (PBS with 0.1% BSA) containing 10% serum or 0.5 mg/mL cell lysate
- Mix equal volumes of analyte solutions with constant concentration of SAM-coated, ligand-functionalized nanoparticles
- Include controls: no competitor (max fluorescence), and excess unlabeled ligand (min fluorescence)
Measurement and Analysis
- Incubate samples for 1 hour at room temperature with gentle shaking
- Centrifuge briefly (5,000 × g, 5 minutes) to pellet nanoparticles
- Measure fluorescence intensity of supernatant (or resuspended pellets based on experimental design)
- Fit competition data to a sigmoidal dose-response curve to determine IC50
- Convert IC50 to Ki using the Cheng-Prusoff equation: Ki = IC50 / (1 + [L]/Kd), where Kd is measured separately for the fluorescent ligand
Data Analysis and Interpretation
Accounting for Matrix Effects in Kd Determination
When working with complex samples, several analytical corrections must be applied to obtain accurate Kd values:
Free Analyte Concentration Correction
- In complex matrices, a portion of the analyte may be sequestered by matrix components
- Measure the free fraction by equilibrium dialysis or ultrafiltration
- Adjust the nominal analyte concentration by multiplying by the free fraction
Non-specific Binding Correction
- Include reference surfaces with scrambled or non-functional SAMs
- Subtract reference signals from specific binding signals
- For fluorescence assays, include controls with non-specific competitors
Signal Normalization
- Normalize signals to the maximum specific binding in buffer
- This facilitates comparison between buffer and complex sample conditions
Table 2: Troubleshooting Guide for Kd Measurements in Complex Samples
| Problem | Potential Cause | Solution |
|---|---|---|
| High background signal | Nonspecific adsorption to surface | Optimize SAM packing density; incorporate oligoethylene glycol termini |
| Irreversible binding | Strong nonspecific interactions or aggregation | Increase salt concentration (up to 500 mM NaCl); add mild detergent (0.01% Tween-20) |
| Poor data fitting | Heterogeneous binding or mass transport limitation | Reduce ligand density; increase flow rate; try different binding models |
| Signal attenuation | Matrix interference or quenching | Use longer association times; try label-free methods (SPR); include internal standards |
| Inconsistent replicates | Matrix variability or surface fouling | Pre-condition surface with multiple injections; use fresh matrix samples |
Validation of Specific Binding
To confirm that measured signals represent specific binding interactions:
Concentration-Dependent Saturation
- Demonstrate that binding signals saturate at high analyte concentrations
- Show that the saturation level is consistent with the immobilized ligand density
Competition with Unlabeled Ligands
- Show that excess unlabeled ligand competes with the signal
- Demonstrate specificity with structurally unrelated competitors
Surface Density Dependence
- Measure binding at different ligand surface densities
- Confirm that Kd is independent of density (suggesting minimal avidity effects)
The Scientist's Toolkit: Research Reagent Solutions
Table 3: Essential Materials for Kd Measurements with SAM Surfaces
| Reagent/Category | Specific Examples | Function/Application |
|---|---|---|
| SAM Forming Thiols | 11-Mercapto-1-undecanol, EG6-thiol | Create non-fouling surfaces resistant to nonspecific adsorption |
| Activation Reagents | NHS (N-hydroxysuccinimide), EDC (1-ethyl-3-(3-dimethylaminopropyl)carbodiimide) | Activate terminal groups for ligand immobilization |
| Sensor Chips | CM5 (carboxymethylated dextran), HPA (hydrophobic association), SA (streptavidin) | Provide specialized surfaces for different immobilization strategies |
| Capture Tags | Biotin, His₆-tag, Fc region | Enable oriented immobilization through specific interactions |
| Running Buffers | HBS-EP+, PBS-P+, TBS with 0.05% Tween-20 | Maintain pH and ionic strength while minimizing nonspecific binding |
| Regeneration Solutions | Glycine-HCl (pH 2.0-3.0), NaOH (10-50 mM), SDS (0.01-0.05%) | Remove bound analyte without damaging immobilized ligand |
| Matrix Components | Fetal bovine serum, human plasma, cell lysates | Provide complex biological environment for relevant binding measurements |
| Reference Molecules | Scrambled peptides, isotype controls, structurally related inactive compounds | Distinguish specific from nonspecific binding interactions |
Experimental Workflow and Data Analysis
The following diagram illustrates the complete workflow for assessing binding affinity in complex samples using SAM-functionalized surfaces:
Accurate determination of specific binding affinity in complex samples requires careful experimental design, appropriate surface engineering, and rigorous data analysis. Self-assembled monolayers on gold surfaces provide an excellent platform for these measurements, offering controlled surface chemistry that minimizes nonspecific adsorption. The protocols described herein enable researchers to obtain reliable Kd values that more accurately reflect binding interactions under physiologically relevant conditions, thereby bridging the gap between idealized in vitro assays and complex biological environments. As drug discovery increasingly focuses on targeting difficult interactions in native contexts, these methodologies will continue to grow in importance for predicting therapeutic efficacy.
Long-Term Stability and Durability Testing for Commercial Viability
The integration of self-assembled monolayers (SAMs) on gold surfaces represents a foundational technology in sensing and diagnostic applications, where interfacial stability directly dictates device performance and commercial readiness. This application note establishes a standardized framework for evaluating the long-term stability and durability of SAM-modified gold surfaces, with a specific focus on meeting the rigorous requirements for drug development and diagnostic platforms. The protocols outlined herein are designed to provide researchers with a systematic methodology to assess SAM integrity under accelerated aging conditions, enabling the prediction of functional lifespan and the identification of failure mechanisms critical for commercial deployment.
Quantitative Stability Benchmarks for SAMs on Gold
Stability testing for SAMs must simulate both storage and operational environments. The table below summarizes key stability metrics and performance thresholds derived from accelerated aging studies, providing critical benchmarks for commercial viability assessment.
Table 1: Key Stability Metrics and Performance Thresholds for SAM-Modified Gold Surfaces
| Stress Factor | Test Condition | Performance Threshold | Validated SAM System | Key Measurement |
|---|---|---|---|---|
| Thermal Cycling | -40°C to 85°C, 1200 cycles | <3% efficiency loss post-testing | Friedel–Crafts cross-linked SABs on ITO [91] | Power Conversion Efficiency |
| Damp Heat | 85°C / 85% Relative Humidity, 2000 hours | <4% efficiency loss post-testing | Friedel–Crafts cross-linked SABs on ITO [91] | Power Conversion Efficiency |
| Continuous Heating | 100°C, 200 hours | Resistance to thermal degradation | Polymerized SAB Network [91] | XPS, FTIR, Adhesion Energy |
| Desorption Resistance | >65°C | Prevention of SAM disordering and desorption | Covalently Interconnected Bilayers [91] | Surface Analysis (XPS, TOF-SIMS) |
Experimental Protocols for Stability Assessment
This section provides detailed methodologies for conducting standardized stability tests, which are essential for generating comparable and reliable data across different SAM formulations.
Protocol: Thermal Cycling Test
Objective: To evaluate the mechanical and chemical resilience of SAMs on gold against repeated temperature fluctuations, which induce stress through mismatched thermal expansion coefficients.
Materials & Equipment:
- Thermal cycling chamber (capable of -40°C to 85°C)
- Nitrogen or argon glovebox (for inert atmosphere storage, optional)
- Characterization equipment (e.g., Contact Angle Goniometer, XPS, Electrochemical Impedance Spectroscopy (EIS) setup)
Procedure:
- Baseline Characterization: Perform initial surface characterization on at least five (n≥5) SAM-gold samples. Key measurements include:
- Contact Angle: Quantifies surface energy and molecular order.
- Electrochemical Response: Measures charge transfer resistance or redox probe accessibility via EIS/Cyclic Voltammetry.
- XPS: Determines elemental composition and chemical states at the surface.
- Cycle Programming: Program the thermal chamber to execute cycles defined by the International Electrotechnical Commission (IEC) 61215:2021 standards [91]:
- Ramp from -40°C to +85°C.
- Dwell at extremes for a minimum of 10 minutes.
- Ensure a cycle time that allows for complete thermal equilibration.
- Interim Monitoring: Periodically remove samples (e.g., every 250 cycles) for non-destructive analysis to track degradation progression.
- Endpoint Analysis: After completing the target number of cycles (e.g., 1200), perform a full suite of characterization identical to the baseline. Compare results to quantify degradation.
Protocol: Damp Heat Testing
Objective: To assess SAM stability under combined thermal and humidity stress, which accelerates hydrolytic desorption and oxidation.
Materials & Equipment:
- Environmental chamber (capable of 85°C and 85% RH)
- Analytical balance (for weighing samples to monitor desorption)
Procedure:
- Preparation: Place SAM-gold samples in the chamber maintained at 85°C and 85% Relative Humidity.
- Sample Monitoring: Remove samples at predetermined intervals (e.g., 500h, 1000h, 2000h).
- Characterization: Analyze the samples for:
- Mass Loss: Use a microbalance to detect the mass loss of molecular components.
- Surface Wettability: Track changes in water contact angle.
- Chemical State: Employ XPS to monitor oxidation of sulfur-gold bonds or the appearance of new oxygen species.
- Functional Integrity: For biosensors, measure the binding affinity towards a target analyte.
The Scientist's Toolkit: Essential Research Reagents & Materials
The following table catalogues critical materials and their functions for preparing and evaluating stable SAMs on gold surfaces.
Table 2: Essential Reagents and Materials for SAM Formation and Stability Testing
| Item | Function & Importance | Application Notes |
|---|---|---|
| Gold Substrates (with Cr/Ti adhesion layer) | Provides the foundational surface for thiolate bond formation. An adhesion layer prevents gold delamination. | Must have an adhesion layer (Cr or Ti); otherwise, gold will delaminate during sonication steps [92]. |
| Thiol Compounds (e.g., Alkanethiols, Bipyridine-SH) | The active molecules that form the SAM. Terminal group defines surface functionality (e.g., bio-recognition). | For amine-terminated thiols, adjust solution pH to ~12; for carboxy-terminated, adjust pH to ~2 [92]. |
| High-Purity Ethanol (200 proof) | Primary solvent for thiol solution preparation. Purity is critical to prevent contamination. | Elevated levels of copper impurities can disrupt the assembly process and impair SAM quality [92]. |
| Sealed Sample Containers (Glass or Polypropylene) | Vessels for SAM formation. Must be sealable to minimize oxygen exposure and solvent evaporation. | Containers can be reused but must be dedicated to the same thiol to avoid cross-contamination [92]. |
| Dry Nitrogen Gas | Used for drying samples after rinsing and for creating an inert atmosphere during storage. | Minimizes oxidation of the SAM during storage and handling. |
| Molecular Sieves (5 Å) | Scavengers that sequester CO2 and water during N-Heterocyclic Carbene (NHC) SAM formation. | Improves monolayer formation and long-range ordering by shifting reaction equilibrium [93]. |
Advanced Stabilization Strategies
Beyond standard alkanethiol SAMs, recent advancements introduce novel chemical approaches to significantly enhance durability.
Covalent Stabilization via Bilayer Formation
A leading strategy to overcome the thermal instability of monolayers is the formation of a self-assembled bilayer (SAB). This involves covalently interconnecting a bottom SAM with an upper layer, creating a polymerized network.
Experimental Workflow: The following diagram illustrates the key steps in creating a covalently stabilized self-assembled bilayer on a substrate surface.
Key Characterization for SABs:
- Time-of-Flight SIMS (TOF-SIMS): Used to detect high mass-to-charge ratio fragments (e.g., m/z 862.4, 804.3) confirming the formation of cross-linked products via methylene bridges [91].
- X-ray Photoelectron Spectroscopy (XPS): Monitors the disappearance of the carbonyl carbon peak (at 289.2 eV) from the alkylating agent, confirming its decomposition and reaction [91].
- Adhesion Testing: A 1.7-fold improvement in adhesion energy has been demonstrated with SABs compared to standard SAMs, indicating superior mechanical stability [91].
N-Heterocyclic Carbene (NHC) SAMs
NHCs are an emerging class of ligands for gold surfaces that offer superior chemical stability compared to thiolates [93].
Improved Deposition Protocol:
- Solution Preparation: Use low-cost benzimidazolium hydrogen carbonate salts as the NHC precursor.
- CO2 Sequestration: Add molecular sieves (5 Å) to the deposition solution to sequester CO2, a key side product. This shifts the equilibrium towards the free carbene, improving monolayer formation and long-range order [93].
- Broad Functional Group Tolerance: The methodology using a bicarbonate source and scavengers expands the variety of functional groups that can be incorporated into the NHC-SAM.
The commercial viability of SAM-based gold platforms in drug development and diagnostics is inextricably linked to their demonstrated long-term stability. The application notes and protocols detailed herein provide a standardized roadmap for researchers to rigorously validate SAM performance against industry-relevant stress factors. By adopting these testing methodologies and leveraging emerging stabilization strategies such as covalently cross-linked bilayers and NHC SAMs, scientists can de-risk the development pathway, bridge the gap between laboratory innovation and market-ready products, and deliver highly reliable sensing and diagnostic solutions.
Non-specific adsorption (NSA) remains a significant challenge in biosensing and biomedical applications, particularly on ubiquitous gold surfaces, as it leads to false-positive signals, reduced sensitivity, and poor reproducibility [1]. Within the broader research on self-assembled monolayers (SAMs) for reducing NSA on gold surfaces, BSA blocking and direct physisorption have emerged as critical techniques. This application note provides a detailed benchmark of these methods, offering structured quantitative data and validated protocols to guide researchers and drug development professionals in selecting and implementing the optimal surface passivation strategy for their specific applications.
Background and Principle
The Problem of Non-Specific Adsorption
NSA occurs when biomolecules such as proteins, vesicles, or other bio-foulants adhere to sensing surfaces through physisorption, driven by hydrophobic forces, ionic interactions, van der Waals forces, and hydrogen bonding [1]. On gold surfaces, which are vulnerable to nonspecific molecular adsorption, this phenomenon is particularly problematic for assays relying on surface plasmon resonance (SPR), quartz crystal microbalance (QCM), and other label-free detection techniques [94] [9]. The resulting biofouling diminishes sensor performance by increasing background noise and obscuring specific binding signals.
Self-Assembled Monolayers as a Foundational Strategy
SAMs of alkanethiolates on gold provide a powerful platform for creating well-defined, chemically tunable surfaces to combat NSA [17] [9]. These monolayers form spontaneously upon immersion of a gold substrate into a solution of alkanethiols, creating a densely packed, ordered interface [17]. The properties of the surface are determined by the terminal functional group of the alkanethiol, allowing for precise control over surface chemistry and interactions with biological materials [17]. SAMs presenting oligo(ethylene glycol) (OEG) termini have been shown to be particularly effective at preventing the non-specific adsorption of proteins [17].
Quantitative Benchmarking of BSA Blocking and Physisorption
The following section provides a comparative analysis of the performance of traditional chemisorption with thiol linkers, direct physisorption, and BSA blocking, based on experimental data.
Table 1: Performance Comparison of Surface Modification Methods for EV Detection on Gold
| Method | Specific EV Capture (Anti-CD63) | Non-specific EV Binding (IgG Control) | Specific Capture Ratio | Reproducibility (CV) |
|---|---|---|---|---|
| Physisorption (Citrate-capped Au) | Not significantly different from chemisorption [94] | 134.9 [94] | >200 [94] | 6.4% [94] |
| Chemisorption (MUA Linker) | Not significantly different from physisorption [94] | 2,430 [94] | 11.7 (inferred) [94] | 18.1% [94] |
| Chemisorption (SH-PEG Linker) | Not significantly different from physisorption [94] | 2,088 [94] | 14.6 (inferred) [94] | 8.6% [94] |
Table 2: BSA Blocking Efficiency on Different Surfaces
| Surface Type | BSA Blocking Efficiency | Optimal BSA Incubation Conditions | Notes |
|---|---|---|---|
| Hydrophobic Surface | 90-100% (against Con A, IgG, SpA) [95] | 1 mg/mL, 30 min incubation [95] | Efficient blocking with low surface coverage ( ~35% of a monolayer) [95] |
| Hydrophilic Surface | 68-100% (against Con A, IgG, SpA) [95] | 1 mg/mL, 30 min incubation [95] | Blocking efficacy is more variable and depends on the challenging protein [95] |
Detailed Experimental Protocols
Protocol 1: Direct Physisorption for Antibody Immobilization
This protocol describes the functionalization of a plain or nanostructured gold surface via direct physisorption of antibodies, a method demonstrated to achieve ultra-low nonspecific binding of extracellular vesicles (EVs) [94].
4.1.1 Research Reagent Solutions
- Citrate Solution: 1% (w/v) trisodium citrate in deionized water. Used to create a hydrophilic, negatively charged gold surface.
- Antibody Solution: 20 µg/mL of the capture antibody (e.g., anti-CD63 for EV studies) in phosphate-buffered saline (PBS). This concentration was found to yield the highest net specific binding [94].
- Blocking Solution: 1% (w/v) Bovine Serum Albumin (BSA) in PBS. Used to passivate any remaining uncovered surface sites.
- Wash Buffer: PBS, pH 7.4.
4.1.2 Step-by-Step Procedure
- Surface Pretreatment: Clean the gold substrate (e.g., plain film or nanowell structures) using an oxygen plasma cleaner or piranha solution (Caution: Piranha is highly corrosive and must be handled with extreme care). Rinse thoroughly with deionized water and ethanol, then dry under a stream of nitrogen.
- Citrate Capping: Immerse the clean gold substrate in the 1% citrate solution for 30 minutes at room temperature. This step creates a citrate-capped surface that facilitates subsequent physisorption.
- Antibody Immobilization: Incubate the citrate-capped gold surface with the 20 µg/mL antibody solution for 60 minutes at room temperature.
- Blocking: Rinse the surface gently with wash buffer to remove unbound antibodies. Incubate with the 1% BSA blocking solution for 30 minutes to passivate any remaining non-specific binding sites.
- Rinsing and Storage: Rinse the functionalized surface thoroughly with wash buffer. The sensor is now ready for immediate use or can be stored in PBS at 4°C for short-term use.
Protocol 2: BSA Blocking for SAM-Modified Gold Surfaces
This protocol outlines the use of BSA to block non-specific binding on a gold surface already functionalized with a SAM, such as one created with mercaptoundecanoic acid (MUA).
4.2.1 Research Reagent Solutions
- SAM Solution: 1 mM alkanethiol (e.g., MUA or a mixed SAM with OEG-terminated thiols) in ethanol.
- BSA Blocking Solution: 1 mg/mL BSA in PBS for optimized blocking [95]. A higher concentration (e.g., 10 mg/mL) with longer incubation may be used but can lead to competitive adsorption-desorption dynamics.
- Activation Buffer: PBS, pH 7.4.
4.2.2 Step-by-Step Procedure
- SAM Formation: Immerse the clean, dry gold substrate in the 1 mM SAM solution for a minimum of 12 hours (overnight) at room temperature to form a dense, well-ordered monolayer.
- Rinsing: Remove the substrate from the thiol solution and rinse it copiously with absolute ethanol to remove physically adsorbed thiols. Dry under a stream of nitrogen.
- BSA Blocking: Incubate the SAM-functionalized gold surface with the 1 mg/mL BSA solution in PBS for 30 minutes at room temperature.
- Final Rinsing: Rinse the surface gently with activation buffer to remove any excess, unbound BSA. The surface is now blocked and ready for subsequent specific biofunctionalization or use in an assay.
Conceptual Workflow and Signaling Pathways
The following diagram illustrates the key mechanistic differences between the physisorption and chemisorption methods, highlighting why physisorption can lead to superior blocking against NSA.
Diagram: Mechanistic Comparison of Physisorption and Chemisorption
The diagram delineates the two functionalization pathways. The physisorption process achieves a uniform, stable protein layer through direct adsorption onto a citrate-capped gold surface, facilitated by hydrophobic, electrostatic, and cysteine-gold affinity interactions [94]. This results in effective blocking and low NSA. In contrast, the chemisorption process relies on multi-step thiol-based SAM formation and EDC/NHS chemistry, which is prone to defective SAM formation at grain boundaries and the generation of non-reactive byproducts (e.g., N-acylurea) during activation [94]. These defects create sites for weak immobilization and subsequent NSA.
The Scientist's Toolkit: Essential Research Reagents
Table 3: Key Reagents for SAM and Blocking Experiments on Gold
| Reagent / Material | Function / Role | Example & Notes |
|---|---|---|
| Alkanethiols | Form the Self-Assembled Monolayer (SAM) base on gold. | Mercaptoundecanoic acid (MUA) provides a carboxyl terminus for EDC/NHS chemistry [94]. Oligo(ethylene glycol) (OEG)-terminated thiols create non-fouling surfaces [17]. |
| BSA (Bovine Serum Albumin) | A blocking agent that physisorbs to surfaces to passivate non-specific binding sites. | Effective at ~1 mg/mL for 30 min incubation [95]. Blocking efficiency varies with surface hydrophobicity [95]. |
| EDC / NHS | Cross-linking chemistry for covalent immobilization of ligands to carboxylated SAMs. | 1-Ethyl-3-(3-dimethylaminopropyl)carbodiimide / N-Hydroxysuccinimide. The reaction is fast and can form non-reactive byproducts that hinder coupling [94]. |
| Citrate | A physisorbed capping agent that creates a hydrophilic and negatively charged gold surface. | Enables direct, uniform physisorption of antibodies and blocking proteins [94]. |
| Antibodies | The affinity capture ligands for specific detection of analytes. | Can be immobilized via physisorption or chemisorption. Physisorption avoids chemical modification and preserves activity [94] [96]. |
Conclusion
Self-assembled monolayers represent a powerful and versatile strategy for engineering gold surfaces that effectively resist non-specific adsorption, which is a critical hurdle in biosensing and diagnostic applications. Success hinges on a holistic approach that integrates a foundational understanding of antifouling mechanisms, careful selection of molecular building blocks, meticulous optimization of formation protocols, and rigorous validation using appropriate analytical techniques. Future directions will likely involve the development of even more robust and smart SAMs with enhanced stability in physiological conditions, dynamic responsivity, and tailored functionality for next-generation point-of-care diagnostics, implantable medical devices, and high-throughput drug discovery platforms. The continued refinement of SAM technology promises to significantly improve the reliability and sensitivity of biomedical analyses.
References
- 1. Non-Specific Adsorption Reduction Methods in Biosensing [https://pmc.ncbi.nlm.nih.gov/articles/PMC6603772/]
- 2. Optimizing self-assembled monolayer construction for ... [https://www.sciencedirect.com/science/article/abs/pii/S0013468625004591]
- 3. Applications of Self-Assembled Monolayers [https://www.ossila.com/pages/applications-of-self-assembled-monolayers]
- 4. Gold Nanoparticles Surface Design and Modification [https://www.cytodiagnostics.com/pages/gold-nanoparticles-surface-design-and-modification?srsltid=AfmBOorCTofNGVRUieBZNUwhk8-DaLFP0kVbzhOPc8ymrbpYVzryQCKD]
- 5. Analytical application of self assembled monolayers on ... [https://www.sciencedirect.com/science/article/pii/0956566395992179]
- 6. Controlling cell attachment on contoured surfaces with self ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC38231/]
- 7. Removal of self-assembled monolayers of alkanethiolates ... [https://www.sciencedirect.com/science/article/abs/pii/S0039602805008927]
- 8. Self-assembled monolayers of 6-amino-1-hexanethiol on ... [https://www.sciencedirect.com/science/article/abs/pii/S1572665725004412]
- 9. Self-Assembled Monolayer Coatings on Gold and Silica ... [https://www.mdpi.com/2079-6412/12/10/1462]
- 10. Direct Measurements of Overlooked Long-Range ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC12004931/]
- 11. Antifouling Modification of Gold Surfaces for Acoustic Wave ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC12191305/]
- 12. Co-hydrolyzed bio-inspired antifouling coatings for ... [https://www.sciencedirect.com/science/article/pii/S0021979725015887]
- 13. Anti-fouling Coatings of Poly(dimethylsiloxane) Devices for ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC4414934/]
- 14. Protein- and Cell-Resistance of Zwitterionic Peptide-Based ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC8527039/]
- 15. Ultra-low fouling peptide surfaces derived from natural ... [https://www.sciencedirect.com/science/article/abs/pii/S0142961209006887]
- 16. Zwitterion and Oligo(ethylene glycol) Synergy Minimizes ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC7321848/]
- 17. Using self-assembled monolayers to model the ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC2771169/]
- 18. Hydrophilicity and Hydrophobicity in Advanced Material Applications... [https://ncstate.pressbooks.pub/advancesinpolymerscience/chapter/hydrophilicity-and-hydrophobicity-in-advanced-material-applications/]
- 19. Reduction of nonspecific binding proteins to self ... [https://www.sciencedirect.com/science/article/abs/pii/S096808960500876X]
- 20. Quantitative evaluation of perfluorinated alkanethiol ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC10264085/]
- 21. A simulation study on the thermal and wetting behavior of ... [https://www.sciencedirect.com/science/article/pii/S1002007114000872]
- 22. Insight into the influence of surface wettability on flotation ... [https://www.sciencedirect.com/science/article/pii/S0032591023008392]
- 23. Rapid assembly of mixed thiols for toll-like receptor-based ... [https://pubs.rsc.org/en/content/articlehtml/2024/ay/d4ay00983e]
- 24. Engineering Gold Biointerfaces with Mixed Short-Chain ... [https://www.sciencedirect.com/science/article/abs/pii/S0927776525007374]
- 25. Covalent immobilization: A review from an enzyme ... [https://www.sciencedirect.com/science/article/pii/S1385894724095457]
- 26. Comparative Study of the Binding of Concanavalin A ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC3601678/]
- 27. XPS, ToF-SIMS, NEXAFS and SPR Characterization of ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC3781950/]
- 28. Gold immuno-functionalisation via self-assembled ... [https://pubmed.ncbi.nlm.nih.gov/18534611/]
- 29. Methods of reducing non-specific adsorption in microfluidic ... [https://asu.elsevierpure.com/en/publications/methods-of-reducing-non-specific-adsorption-in-microfluidic-biose]
- 30. A Comprehensive Review of the Covalent Immobilization ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC7692479/]
- 31. A key issue for self-assembled monolayers on gold as thin ... [https://www.sciencedirect.com/science/article/abs/pii/S0927775711005838]
- 32. Covalent Immobilization of Affinity Ligands [https://www.thermofisher.com/us/en/home/life-science/protein-biology/protein-biology-learning-center/protein-biology-resource-library/pierce-protein-methods/covalent-immobilization-affinity-ligands.html]
- 33. Covalent chemical probes | Communications Chemistry [https://www.nature.com/articles/s42004-025-01652-6]
- 34. Covalent functionalization of G protein-coupled receptors ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC11827490/]
- 35. Electrochemical Aptamer-Based Sensors for Improved ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC6886665/]
- 36. Electrochemical aptamer-based biosensors [https://en.wikipedia.org/wiki/Electrochemical_aptamer-based_biosensors]
- 37. Microneedle electrochemical aptamer-based sensing [https://www.sciencedirect.com/science/article/pii/S0956566323003500]
- 38. Gold cleaning methods for preparation of cell culture ... [https://www.sciencedirect.com/science/article/abs/pii/S1389172317308897]
- 39. Preparation of DNA aptamer and development of lateral ... [https://pubmed.ncbi.nlm.nih.gov/33756378/]
- 40. Updates on developing and applying biosensors for the ... [https://www.frontiersin.org/journals/public-health/articles/10.3389/fpubh.2023.1240584/full]
- 41. Optical Biosensors for Label-Free Detection of Small ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC6308632/]
- 42. Dual-aptamer Drift Cancelling Techniques to Improve Long ... [https://arxiv.org/abs/2212.14491]
- 43. Optimizing self-assembled monolayer construction for ... [https://www.semanticscholar.org/paper/Optimizing-self-assembled-monolayer-construction-of-Lupoi-Baccara/26c20c07a9c94e9b113d6dc7ec24dfede3124771]
- 44. Formation and Structure of Highly Ordered Self-Assembled ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC12471213/]
- 45. Thermo-kinetics study of laser-induced desorption of self ... [https://pubmed.ncbi.nlm.nih.gov/16852480/]
- 46. Ordering and defects in self-assembled monolayers on ... [https://www.sciencedirect.com/science/article/abs/pii/S0169433216311795]
- 47. Cholesterol depletion decreases adhesion of non-small cell ... [https://pubmed.ncbi.nlm.nih.gov/37399498/]
- 48. Anchoring groups for ordered and highly stable ... [https://www.sciencedirect.com/science/article/abs/pii/S0169433224009127]
- 49. Effect of alkanethiol chain length on the oxidation ... [https://www.sciencedirect.com/science/article/abs/pii/S0040609023002055]
- 50. Understanding the Phase Diagram of Self-Assembled ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC4904245/]
- 51. Effect of the Chain Length and Temperature on ... [https://pubmed.ncbi.nlm.nih.gov/28965416/]
- 52. Improving the Stability and Sensing of Electrochemical ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC3956047/]
- 53. Effect of chain length on the surface properties of ω- ... [https://pubs.rsc.org/en/content/articlelanding/2001/cp/b104570a]
- 54. Optimizing gold electrode surface treatments for enhanced ... [https://www.sciencedirect.com/science/article/pii/S0927775725007769]
- 55. N-heterocyclic carbenes as clickable molecular anchors for ... [https://pubs.rsc.org/en/content/articlehtml/2025/sc/d5sc03908h]
- 56. Ultra stable self-assembled monolayers of N-heterocyclic ... [https://pubmed.ncbi.nlm.nih.gov/24755592/]
- 57. N-Heterocyclic Carbenes for the Self-Assembly of Thin and ... [https://pubmed.ncbi.nlm.nih.gov/32343123/]
- 58. N-Heterocyclic Carbenes - the Design Concept for Densely ... [https://chemrxiv.org/engage/chemrxiv/article-details/6440131d7be842788de9706d]
- 59. FRET Imaging of Nonuniformly Distributed DNA SAMs on ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC10302881/]
- 60. Reshaping, Fragmentation, and Assembly of Gold ... [https://pubmed.ncbi.nlm.nih.gov/27035211/]
- 61. Self-assembling biomolecules for biosensor applications [https://pmc.ncbi.nlm.nih.gov/articles/PMC10696764/]
- 62. a novel filtration and targeted next-generation sequencing ... [https://www.frontiersin.org/journals/microbiology/articles/10.3389/fmicb.2025.1538265/full]
- 63. Target sequence of single cells captured by a polymeric ... [https://www.nature.com/articles/s41598-025-14826-y]
- 64. Microbial Whole-Cell Biosensors - PubMed Central - NIH [https://pmc.ncbi.nlm.nih.gov/articles/PMC8376793/]
- 65. Versatile Strategies to Combat Bacterial Biofilm Formation [https://pmc.ncbi.nlm.nih.gov/articles/PMC9415113/]
- 66. Label‐free detection techniques for protein microarrays [https://pmc.ncbi.nlm.nih.gov/articles/PMC7167936/]
- 67. Adhesion forces between Staphylococcus epidermidis and ... [https://www.sciencedirect.com/science/article/abs/pii/S0142961208005346]
- 68. Advanced drug delivery via self-assembled monolayer- ... [https://www.aimspress.com/article/doi/10.3934/bioeng.2017.2.275?viewType=HTML]
- 69. Label-free quantification [https://en.wikipedia.org/wiki/Label-free_quantification]
- 70. Prof. George Whitesides' Contributions to Self-Assembled ... [https://www.mdpi.com/2624-8549/7/1/9]
- 71. QCM-D vs. SPR - What are the differences, and which ... [https://www.nanoscience.com/blogs/qcm-d-vs-spr-what-are-the-differences-and-which-technique-fits-your-application/]
- 72. Applications of Surface Plasmon Resonance for ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC3989105/]
- 73. Investigating organic multilayers by spectroscopic ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC4901546/]
- 74. Electrochemistry of redox-active self-assembled monolayers [https://pmc.ncbi.nlm.nih.gov/articles/PMC2885823/]
- 75. Study of self-assembled monolayers of n-alkanethiol on a ... [https://www.sciencedirect.com/science/article/abs/pii/S0040609095082956]
- 76. Technical Guide: Comparing SPR and QCM [https://nicoyalife.com/blog/quartz-crystal-microbalance-vs-surface-plasmon-resonance/]
- 77. Impact of self-assembled monolayer templates on ... [https://www.sciencedirect.com/science/article/abs/pii/S1572665723000541]
- 78. SPR Studies of the Nonspecific Adsorption Kinetics ... [https://www.sciencedirect.com/science/article/pii/S0021979796945865]
- 79. 2.6. Measurement of nonspecific protein adsorption [https://bio-protocol.org/exchange/minidetail?id=18212208&type=30]
- 80. Quantifying Bound and Active Antibodies Conjugated to ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC6072236/]
- 81. Afficoat™ for Reducing Non-Specific Adsorption in SPR ... [https://www.affiniteinstruments.com/post/afficoat-for-reducing-non-specific-adsorption]
- 82. Chemically well-defined self-assembled monolayers for ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC4159093/]
- 83. Gold immuno-functionalisation via self-assembled ... [https://www.sciencedirect.com/science/article/abs/pii/S0022175908001476]
- 84. Co-adsorbed self-assembled monolayer enables high ... [https://www.nature.com/articles/s41467-024-51760-5]
- 85. Non-specific adsorption of serum and cell lysate on 3D ... [https://www.sciencedirect.com/science/article/pii/S1878535215002099]
- 86. Use of Surface Plasmon Resonance (SPR) to Determine ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC8489108/]
- 87. Surface plasmon resonance spectroscopy for ... [https://www.sciencedirect.com/science/article/pii/S0005273613001466]
- 88. Surface Plasmon Resonance (SPR) Analysis Services [https://kactusbio.com/pages/surface-plasmon-resonance-spr-services?srsltid=AfmBOorFrZXXGyEXx6OtL94D4sHLS0txbZgjNStMU_-M00yGlLgcfCTL]
- 89. Drug delivery from gold and titanium surfaces using self- ... [https://pubmed.ncbi.nlm.nih.gov/18790530/]
- 90. Drug delivery from gold and titanium surfaces using self- ... [https://www.sciencedirect.com/science/article/abs/pii/S0142961208006145]
- 91. Self-assembled bilayer for perovskite solar cells with ... [https://www.nature.com/articles/s41560-024-01689-2]
- 92. Preparing Self-Assembled Monolayers [https://www.sigmaaldrich.com/US/en/technical-documents/protocol/materials-science-and-engineering/organic-electronics/preparing-self-assembled?srsltid=AfmBOop4OZRYwUEFb0W7ZtE1NQvWS9FjhUz2wzMYB6uwsDjILEm3KsvD]
- 93. Sieving for Gold─An Efficient Method for Generating N ... [https://pubmed.ncbi.nlm.nih.gov/41251396/]
- 94. Physisorption of Affinity Ligands Facilitates Extracellular ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC9715849/]
- 95. Efficiency of blocking of non-specific interaction of different ... [https://www.sciencedirect.com/science/article/abs/pii/S0021979709011540]
- 96. A Physisorbed Interface Design of Biomolecules for ... [https://www.sciencedirect.com/science/article/pii/S1535553509002135]