Afficoat vs PEG vs CM-Dextran: A Comparative Analysis of Non-Specific Adsorption Performance for Advanced Biosensing
Non-specific adsorption (NSA) remains a critical challenge that compromises the sensitivity and accuracy of biosensors, particularly when analyzing complex biological samples like serum and cell lysate.
Afficoat vs PEG vs CM-Dextran: A Comparative Analysis of Non-Specific Adsorption Performance for Advanced Biosensing
Abstract
Non-specific adsorption (NSA) remains a critical challenge that compromises the sensitivity and accuracy of biosensors, particularly when analyzing complex biological samples like serum and cell lysate. This article provides a comprehensive, evidence-based comparison of three prominent antifouling surface chemistries: the novel zwitterionic Afficoat, established polyethylene glycol (PEG), and carboxymethyl dextran (CM-Dextran). Tailored for researchers and drug development professionals, we dissect the foundational principles, practical methodologies, and optimization strategies for these coatings. By synthesizing data from recent SPR studies and real-world applications, we deliver a validated performance assessment to guide the selection and development of robust, low-fouling biosensor platforms for clinical and pharmaceutical applications.
Understanding the Battle Against Fouling: Core Principles of Non-Specific Adsorption
The Critical Impact of NSA on Biosensor Signal, Sensitivity, and Selectivity
Non-specific adsorption (NSA) represents a fundamental barrier in biosensing, negatively impacting sensitivity, specificity, and reproducibility by generating background signals indistinguishable from specific binding events [1]. When biomolecules such as proteins adsorb indiscriminately onto sensing surfaces through physisorption, they occlude binding sites, degrade signal-to-noise ratios, and produce false positives that compromise analytical accuracy [1] [2]. This challenge intensifies in complex matrices like serum, plasma, and cell lysate, where high concentrations of interferents (30-80 mg/mL total protein) compete with target analytes [3] [4]. The selection of appropriate surface chemistry is therefore critical for biosensor performance, particularly for applications in clinical diagnostics, drug discovery, and biomanufacturing where reliable detection of low-abundance biomarkers is essential.
The following comparison guide objectively evaluates the NSA reduction performance of three surface chemistry approaches: the zwitterionic peptide-based coating Afficoat, traditional polyethylene glycol (PEG), and carboxymethyl dextran (CM-Dextran). Through systematic analysis of experimental data, methodological protocols, and real-world applications, this guide provides researchers with evidence-based insights for selecting optimal surface modifications to mitigate fouling in biosensing platforms.
Surface Chemistry Comparison: Mechanisms and Experimental Performance
Coating Technologies and Their Antifouling Mechanisms
Afficoat: This proprietary coating forms a self-assembled monolayer (SAM) composed of thiol-terminated peptides with hydrophilic and zwitterionic properties. It immobilizes to gold sensor surfaces via thiol-gold chemistry, creating a hydrated boundary layer that minimizes protein adhesion through electrostatic neutrality and hydrophilicity. The carboxyl terminal allows subsequent functionalization with capture biomolecules such as antibodies or enzymes [3].
Polyethylene Glycol (PEG): A well-established polymer coating that reduces NSA through steric hindrance and hydration effects. PEG chains form a dynamic, highly hydrated physical barrier that prevents foulant proteins from reaching the underlying surface. The effectiveness depends on molecular weight, density, and conformation of the polymer chains [1] [4].
Carboxymethyl Dextran (CM-Dextran): A hydrogel-based 3D matrix that provides a porous, hydrophilic environment for biomolecule immobilization. While offering high loading capacity, its polysaccharide structure can still attract certain protein types through residual hydrophobic or ionic interactions, potentially leading to significant NSA in complex samples [3] [4].
Quantitative Performance Comparison in Complex Media
Direct comparative studies reveal significant differences in NSA resistance when these coatings are exposed to biologically relevant samples. The table below summarizes key experimental findings:
Table 1: Comparative NSA Performance of Surface Coatings in Complex Biological Media
| Surface Coating | Sample Matrix | Protein Concentration | NSA Level | Reference Method |
|---|---|---|---|---|
| Afficoat | Crude bovine serum | 76 mg/mL | Lowest (~15% of PEG) | SPR [3] |
| PEG | Crude bovine serum | 76 mg/mL | Moderate (Reference level) | SPR [3] |
| CM-Dextran | Crude bovine serum | 76 mg/mL | Highest (~200% of PEG) | SPR [3] |
| SIP-based surface | Human serum & cell lysate | Complex mixture | Minimal adsorption | SPRi [4] |
| Dextran hydrogel | Human serum & cell lysate | Complex mixture | High adsorption | SPRi [4] |
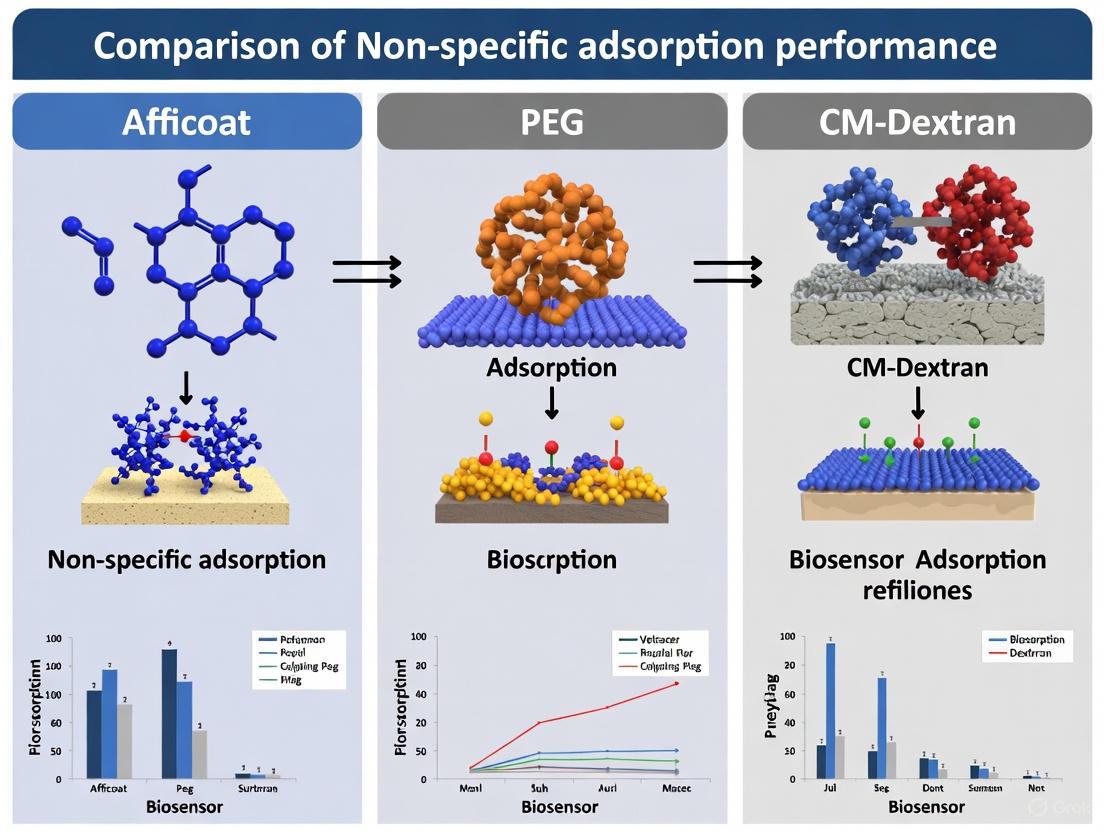
In a critical comparative SPR study, Afficoat demonstrated superior NSA reduction, exhibiting approximately 85% less adsorption than PEG and 90% less than CM-Dextran when exposed to crude bovine serum containing 76 mg/mL of total protein [3]. This performance advantage stems from its optimized peptide sequence that creates a densely packed, neutrally charged interface that effectively repels proteins through combined hydration and electrostatic effects.
Impact on Bioanalytical Assay Performance
Beyond basic NSA metrics, the retention of biorecognition element functionality is essential for functional biosensors. Experimental data confirms that Afficoat maintains biological activity while providing antifouling protection:
Enzyme Activity Retention: Immobilized His-tagged human dihydrofolate reductase (hDHFR) retained significant enzymatic activity ((4.4 ± 0.8) × 10⁻⁵ U) when coupled via Afficoat, equivalent to (5 ± 0.9) ng of fully active enzyme in solution [3].
Affinity Binding Preservation: For a His-tagged maltose binding protein-IgG system immobilized on Afficoat, the equilibrium dissociation constant (K_D) was determined to be (9.6 ± 0.3) nM, consistent with values obtained by other techniques for antigen-antibody interactions [3].
These results demonstrate that Afficoat provides not only exceptional NSA resistance but also maintains the conformational integrity and biological function of immobilized recognition elements—a critical advantage over traditional coatings where surface-induced denaturation can compromise assay performance.
Experimental Protocols for NSA Evaluation
Standardized SPR-Based NSA Assessment
The experimental workflow for quantitatively comparing surface coating performance involves carefully controlled conditions to ensure reproducible and meaningful results:
Diagram 1: SPR NSA Assessment Workflow
Key Protocol Details:
Surface Functionalization: Gold sensor chips are immersed in 1 mM ethanolic solutions of the respective coating molecules (Afficoat peptides, PEG-thiols, or dextran derivatives) for 16 hours to form dense SAMs, followed by thorough rinsing to remove unbound material [3] [4].
Complex Sample Exposure: Surfaces are exposed to crude bovine serum with a standardized protein concentration of 76 mg/mL for 20 minutes under continuous flow conditions, simulating challenging biological environments [3].
Quantification Method: NSA levels are determined by measuring the response unit shift after a 5-minute PBS rinse phase, with normalized comparisons to baseline signals [3].
Complementary NSA Evaluation Techniques
Beyond SPR, researchers employ multiple orthogonal methods to comprehensively characterize NSA:
Surface Plasmon Resonance Imaging (SPRi): Enables high-throughput, simultaneous evaluation of multiple surface chemistries under identical conditions, with applications demonstrated for serum and cell lysate analysis [4].
Matrix-Assisted Laser Desorption Ionization Time-of-Flight Mass Spectrometry (MALDI-TOF/TOF MS): Identifies specific proteins and lipids non-specifically adsorbed to sensor surfaces, providing molecular-level insights into fouling mechanisms [4].
Electrochemical (EC) Methods: Monitor interfacial electron transfer resistance changes resulting from surface fouling, particularly valuable for combined EC-SPR biosensing platforms [2].
The Researcher's Toolkit: Essential Reagents and Materials
Table 2: Key Research Reagents for NSA Reduction Studies
| Reagent/Material | Function | Example Application |
|---|---|---|
| Afficoat Coating Reagent | Zwitterionic peptide SAM formation | Gold surface functionalization for SPR [3] |
| SH-(PEG)ₙ-OH/-OCH₃ | PEG-based SAM formation | Reference antifouling surface preparation [4] |
| CM-Dextran | Hydrogel matrix formation | 3D surface coating comparison [3] [4] |
| ω-Mercaptoundecyl bromoisobutyrate | ATRP initiator for SIP | Surface-initiated polymerization coatings [4] |
| Crude bovine serum | Complex test matrix | NSA challenge studies (76 mg/mL protein) [3] |
| EDC/NHS chemistry | Carboxyl group activation | Biomolecule immobilization on functionalized surfaces [4] |
Implications for Biosensor Applications in Complex Matrices
The differential NSA performance of these surface coatings has profound implications for real-world biosensing applications:
Clinical Diagnostic Applications: Afficoat-modified sensor chips have successfully enabled detection of antibodies against SARS-CoV-2 in clinical samples (serum, plasma, and dried blood spots), demonstrating reliable performance in complex, minimally processed biological fluids [3] [5].
Therapeutic Drug Monitoring: The P4SPR platform with Afficoat surfaces achieved accurate methotrexate monitoring in human serum, with results correlating well with LC-MS/MS and fluorescence polarization immunoassay reference methods [3].
Biomanufacturing Quality Control: Surface coatings with superior NSA resistance facilitate real-time monitoring of bioprocesses and quality attribute assessment without extensive sample cleanup, as demonstrated in AAV production pipeline applications [5].
The experimental evidence clearly demonstrates that zwitterionic peptide-based coatings like Afficoat provide superior NSA reduction compared to traditional PEG and CM-Dextran surfaces in complex biological samples. This performance advantage translates directly to enhanced biosensor sensitivity, reliability, and functionality in real-world applications.
For researchers developing biosensors for clinical diagnostics, bioprocess monitoring, or drug discovery, surface chemistry selection should be guided by rigorous NSA performance data in relevant matrices. While traditional coatings like PEG remain viable for certain applications, advanced materials like Afficoat represent the evolving standard for demanding biosensing environments where maximum signal fidelity and minimum false positives are critical requirements.
Future directions in NSA reduction will likely focus on high-throughput screening of novel materials, machine learning-assisted coating design, and multifunctional surfaces that combine exceptional antifouling properties with enhanced bioreceptor stability and activity [2]. As biosensing technologies continue to advance toward point-of-care and resource-limited settings, the critical importance of effective NSA mitigation will only intensify, underscoring the essential role of optimized surface chemistries in the next generation of bioanalytical devices.
In the design of biosensors, implants, and drug delivery systems, controlling nonspecific adsorption (NSA), commonly known as fouling, is a paramount challenge [6]. When a biosensor interface is exposed to complex biological fluids like blood, serum, or cell lysate, a layer of non-target molecules (e.g., proteins, lipids) can adsorb onto the surface, compromising the sensor's function [4] [6]. This fouling is primarily driven by three fundamental physical interactions: electrostatic, hydrophobic, and van der Waals forces [2]. Electrostatic interactions occur between charged groups on the protein and the surface; hydrophobic attractions drive non-polar protein regions to associate with non-polar surfaces; and van der Waals forces involve induced dipole interactions that are always present [7] [8] [9]. This non-specifically adsorbed layer can block access to recognition ligands, generate false positive signals, and lead to a significant underestimation or overestimation of the target analyte concentration, thereby reducing the sensitivity, reliability, and operational lifespan of the biosensor [6] [2].
To combat NSA, biosensor surfaces are often modified with hydrophilic, bio-inert coatings. Among the most common are polyethylene glycol (PEG), carboxymethyldextran (CM-Dextran), and newer alternatives like Afficoat, a proprietary zwitterionic peptide monolayer [3] [6]. PEG is widely used due to its biocompatibility and ability to form a hydrated barrier that sterically hinders protein approach [6]. CM-Dextran, a 3D hydrogel, is popular in surface plasmon resonance (SPR) biosensors for its high loading capacity for bioreceptors, but its effectiveness against fouling in complex media can be variable [3] [4]. Afficoat represents an approach based on a self-assembled monolayer (SAM) of hydrophilic, zwitterionic peptides that create a strong hydration layer via ionic solvation, theoretically providing a robust shield against various foulants [3]. This guide provides a direct, data-driven comparison of the non-specific adsorption performance of these three surface chemistries, equipping researchers with the information needed to select the optimal coating for their specific application.
Comparative Experimental Data on Non-Specific Adsorption
The performance of Afficoat, PEG, and CM-Dextran has been evaluated under controlled experimental conditions, providing quantitative data for direct comparison. Key findings from these studies are summarized in the table below.
Table 1: Summary of Non-Specific Adsorption Performance in Complex Media
| Surface Coating | Composition & Structure | Test Sample & Concentration | Non-Specific Adsorption Level | Key Experimental Findings |
|---|---|---|---|---|
| Afficoat | Zwitterionic peptide SAM [3] | Crude Bovine Serum (76 mg/mL protein) [3] | Lowest (Superior performance) [3] | • Showed the least non-specific adsorption among tested peptide sequences and outperformed PEG & CM-Dextran [3]. • Enabled detection of biomarkers in undiluted clinical samples (serum, plasma) [3]. |
| PEG (Polyethylene Glycol) | Polyether chain; often grafted as a brush [3] [6] | Crude Bovine Serum (76 mg/mL protein) [3] | Moderate (Intermediate performance) [3] | • More effective than CM-Dextran but less effective than Afficoat in serum [3]. • Performance is highly dependent on chain length and brush density [6]. |
| CM-Dextran | Polysaccharide hydrogel matrix [3] | Crude Bovine Serum (76 mg/mL protein) [3] | Highest (Inferior performance) [3] | • Exhibited the highest level of non-specific adsorption in the comparative SPR study [3]. • Can entangle proteins like BSA, leading to increased adsorption in some configurations [7]. |
Further context from independent studies reveals nuances in PEG and dextran performance. One study investigating PDMS modified with dextran found that the coating caused more Bovine Serum Albumin (BSA) to adsorb than unmodified PDMS, attributing this to sparse surface coverage and potential entanglement of BSA within the dextran matrix [7] [8]. Another study noted that while dextran-based surfaces are valued for their high loading capacity, they still exhibit a high non-specific adsorption response from complex media like serum and cell lysate [4].
Detailed Experimental Protocols for Performance Evaluation
The comparative data presented hinge on robust and reproducible experimental methodologies. Below are detailed protocols for the key experiments cited in this guide.
SPR-Based NSA Evaluation in Complex Serum
This protocol is adapted from the study that directly compared Afficoat, PEG, and CM-Dextran [3].
- Surface Functionalization: Gold sensor chips are functionalized with the coating of interest (e.g., Afficoat via thiol-gold chemistry, CM-Dextran via carbodiimide coupling, or PEG grafting).
- Instrument Setup: An SPR instrument (e.g., Affinité's P4SPR or a comparable system) is set up in the Kretschmann configuration. A stable baseline signal is established using a running buffer, typically phosphate-buffered saline (PBS) at pH 7.4.
- Sample Exposure: The functionalized sensor chip is exposed to the complex biological sample—crude bovine serum with a protein concentration of 76 mg/mL—for a defined period (e.g., 20 minutes) under continuous flow.
- Rinsing: The chip is rinsed with PBS for 5 minutes to remove loosely bound material.
- Quantification: The change in the SPR signal (response units) between the initial baseline and the signal after rinsing is measured. This change is directly correlated with the mass of non-specifically adsorbed proteins on the surface.
Assessment of Immobilized Bioactivity
A critical test for any biosensor coating is its ability to immobilize capture molecules without compromising their function [3].
- Ligand Immobilization: A His-tagged protein (e.g., human dihydrofolate reductase, hDHFR, or maltose binding protein) is immobilized onto the Afficoat surface using a nitrilotriacetic acid (NTA) analog and copper ions.
- Activity Assay (Enzymatic): For an enzyme like hDHFR, its activity is determined by flowing its substrate and measuring the conversion rate. The activity of the surface-immobilized enzyme is compared to that of the same amount of fully active enzyme in solution.
- Affinity Assay (Binding): For a binding protein, increasing concentrations of its binding partner (e.g., IgG) are flowed over the surface. The interaction is monitored in real-time, and the equilibrium dissociation constant (KD) is calculated from the binding curves to confirm the retained affinity of the immobilized protein.
Mechanisms and Workflow: A Visual Guide
The following diagrams illustrate the core concepts of fouling mechanisms and the experimental process for evaluating surface coatings.
Fundamental Forces Driving Non-Specific Adsorption
The fouling of biosensor surfaces is a result of the interplay of several non-covalent interactions between the surface and proteins in the solution. The three primary mechanisms are electrostatic, hydrophobic, and van der Waals interactions.
Experimental Workflow for Coating Performance Evaluation
A typical workflow for assessing the anti-fouling performance of a surface coating involves surface preparation, characterization, exposure to a complex sample, and data analysis.
The Scientist's Toolkit: Key Research Reagents
The following table lists essential materials and reagents used in the development and evaluation of anti-fouling surface coatings, as referenced in the studies.
Table 2: Essential Reagents for Anti-Fouling Surface Research
| Reagent / Material | Function in Research | Examples / Specific Types |
|---|---|---|
| Gold Sensor Chips | Provide a substrate for surface chemistry; enable thiol-based immobilization for SAMs and SPR signal transduction. | PlexArray chips; bare gold SPR chips [4]. |
| Thiol-Terminated Peptides | Form self-assembled monolayers (SAMs) like Afficoat on gold surfaces, creating a dense, non-fouling interface. | Afficoat peptide sequence [3]. |
| PEG Derivatives | Grafted onto surfaces to create a hydrophilic, steric barrier that resists protein adsorption. | SH-(CH₂)₁₁-(OCH₂CH₂)ₙ-OH; SH-PEG; PLL-g-PEG [4] [6]. |
| Dextran Polymers | Used to form 3D hydrogel matrices on sensor surfaces to increase binding capacity and reduce some fouling. | CM-Dextran (T-500) [3] [4]. |
| Complex Biological Samples | Used as challenging, real-world media to test the efficacy of anti-fouling coatings. | Crude bovine serum; human serum; stem cell lysate [3] [4]. |
| Coupling Agents | Activate surface functional groups (e.g., carboxyls) for covalent immobilization of biomolecules. | EDC (1-ethyl-3-(3-dimethylaminopropyl)carbodiimide); NHS (N-hydroxysuccinimide) [4]. |
The direct, quantitative comparison of Afficoat, PEG, and CM-Dextran in challenging environments like crude serum clearly demonstrates that zwitterionic peptide-based coatings like Afficoat offer superior resistance to non-specific adsorption. While PEG provides a moderate level of protection, its efficacy is highly dependent on optimal grafting density and chain length. CM-Dextran, despite its utility as a 3D scaffold for biomolecule immobilization, shows the highest susceptibility to fouling in protein-rich media. The choice of coating is critical and should be guided by the specific application: for maximum signal fidelity in the presence of complex, undiluted samples, Afficoat presents a compelling solution. For less demanding environments or where cost considerations are primary, PEG may remain a viable option. This evidence-based guide underscores that ongoing research into zwitterionic and other novel materials is key to advancing the robustness and reliability of next-generation biosensors.
Afficoat, PEG, and CM-Dextran represent three prominent chemical strategies employed to combat non-specific adsorption (NSA), a critical challenge that compromises the sensitivity and accuracy of biosensors and bioassays. This guide provides an objective, data-driven comparison of their performance, experimental methodologies, and suitability for research applications.
At a Glance: Core Characteristics and Performance
The following table summarizes the key attributes and experimentally determined non-specific adsorption levels of Afficoat, PEG, and CM-Dextran.
| Coating Type | Chemical Class | Immobilization Chemistry | Reported Non-Specific Adsorption (from 76 mg/mL Bovine Serum) | Key Advantages |
|---|---|---|---|---|
| Afficoat | Zwitterionic Peptide SAM [3] | Thiol-gold [3] | Lowest (Outperformed PEG & CM-Dextran) [3] | Superior antifouling in complex samples; allows functional immobilization [3] |
| PEG | Poly(Ethylene Glycol) [4] | Thiol-gold or adsorption [4] [6] | Moderate (Higher than Afficoat) [3] | Well-established, widely available; effective in many scenarios [6] |
| CM-Dextran | Carboxymethylated Polysaccharide [3] [4] | Epoxide chemistry or adsorption [4] | Highest (Among the three) [3] | Hydrogel 3D structure for high receptor loading [4] |
Table: Direct comparison of surface coatings based on search results.
Experimental Data and Performance Evaluation
Quantitative Performance in Complex Media
A direct comparative study exposed surface coatings to crude bovine serum containing 76 mg/mL of total protein, simulating a challenging analytical environment. The level of non-specific adsorption was quantified afterward [3].
- Afficoat's Performance: Under these conditions, Afficoat demonstrated the lowest level of non-specific adsorption, significantly outperforming both PEG and CM-Dextran coatings [3].
- PEG's Performance: The non-specific adsorption on PEG was notably higher than on Afficoat [3].
- CM-Dextran's Performance: CM-Dextran showed the highest level of non-specific adsorption among the three coatings in this test [3].
Beyond Anti-fouling: Functional Performance
Afficoat is not only a passive barrier but also a functional platform. Its structure includes a carboxyl end that enables the immobilization of capture biomolecules, such as His-tagged proteins [3].
- Enzyme Activity Retention: His-tagged human dihydrofolate reductase (hDHFR) immobilized onto Afficoat retained its enzymatic activity, confirming that the coating does not denature sensitive proteins [3].
- Affinity Measurement: The equilibrium dissociation constant (K_D) for a His-tagged maltose binding protein and IgG interaction immobilized on Afficoat was determined to be 9.6 ± 0.3 nM, consistent with values obtained from other techniques, validating the coating's suitability for reliable biomolecular interaction studies [3].
Detailed Experimental Protocols
The following workflows detail the key experimental steps for evaluating anti-fouling performance and for functionalizing an Afficoat surface, as described in the search results.
Protocol 1: Evaluating Anti-fouling Performance with SPR
Diagram: General SPR anti-fouling evaluation workflow. This protocol was used to generate the comparative data on bovine serum adsorption [3].
Protocol 2: Functionalizing an Afficoat Surface for Detection
Diagram: Workflow for functionalizing a biosensor with Afficoat. The coating allows for the immobilization of various capture biomolecules through different chemistries [3].
The Scientist's Toolkit: Key Research Reagent Solutions
| Item Name | Function / Application |
|---|---|
| Afficoat | A proprietary zwitterionic peptide SAM used to create low-fouling sensor surfaces on gold chips [3]. |
| P4SPR Instrument | A compact, portable Surface Plasmon Resonance (SPR) instrument used for real-time, label-free biomolecular interaction analysis [3] [10]. |
| EDC/NHS Coupling Kit | A common chemical kit used to activate carboxyl groups on surfaces like Afficoat or CM-Dextran for covalent immobilization of proteins and other biomolecules [11]. |
| Crude Bovine Serum | A complex biological fluid containing ~76 mg/mL of protein, used as a challenging model solution for evaluating non-specific adsorption [3]. |
Table: Essential reagents and tools for conducting related experiments.
Key Insights for Researchers
The experimental data indicates that Afficoat provides superior resistance to non-specific adsorption from complex samples like serum compared to PEG and CM-Dextran [3]. This makes it a compelling choice for applications involving direct analysis of blood, serum, or cell lysate, where sensitivity is paramount.
While PEG is a well-understood and effective polymer, its moderate performance in head-to-head tests suggests that zwitterionic chemistries like Afficoat can offer a next-generation solution [3]. CM-Dextran's 3D hydrogel structure, while beneficial for binding capacity, appears more susceptible to fouling in protein-rich environments [3] [4].
The choice of coating is application-dependent. Researchers must balance the critical need to minimize NSA with other factors, such as the required surface chemistry for probe immobilization and the specific nature of the sample matrix.
In the fields of biosensing, drug discovery, and diagnostic assay development, the performance of a platform is profoundly influenced by its surface chemistry. When analyzing complex biological samples such as serum or cell lysate, non-specific adsorption (NSA) of non-target proteins and other biomolecules onto sensor surfaces is a major challenge. This fouling can obscure specific detection signals, reduce analytical sensitivity, and lead to inaccurate results. To mitigate this, various surface coatings have been developed. Among the most established are polyethylene glycol (PEG) and carboxymethyl-dextran (CMD). More recently, Afficoat has emerged as a zwitterionic peptide-based self-assembled monolayer (SAM) designed to offer superior resistance to non-specific adsorption. This guide provides an objective, data-driven comparison of these three surface chemistries, focusing on their performance in reducing NSA, their experimental applications, and their utility for researchers working with real-world clinical and biological samples.
Defining the Technologies
- Afficoat: Afficoat is a proprietary surface coating reagent composed of a self-assembled monolayer of thiol-terminated peptides. These peptides are hydrophilic and zwitterionic, a chemical characteristic known for its strong resistance to protein adsorption. It immobilizes onto gold sensor surfaces via stable thiol-gold chemistry. The carboxyl end of the peptide allows for the subsequent immobilization of capture biomolecules, such as antibodies or enzymes [3].
- PEG (Polyethylene Glycol): PEG is a long-chain, hydrophilic polymer that has been a gold standard for creating non-fouling surfaces. It works by forming a highly hydrated layer that sterically hinders proteins from approaching and adhering to the surface. PEGylated surfaces are typically created by functionalizing a surface with PEG-conjugated alkanethiols or other anchor molecules [12].
- CMD (Carboxymethyl-Dextran): CMD is a branched polysaccharide hydrogel that creates a three-dimensional matrix on the sensor surface. This matrix is highly hydrophilic and bioinert, providing a large exclusion volume that repels proteins. Its numerous carboxyl groups allow for high-density immobilization of ligands via covalent chemistry. CMD surfaces have been widely used in commercial biosensors, such as traditional SPR systems [13].
Direct Comparison of Non-Specific Adsorption
The most critical metric for comparing these surfaces is their demonstrated ability to minimize NSA when exposed to complex biological fluids. The following table summarizes quantitative and comparative data from experimental studies.
Table 1: Direct Comparison of Non-Specific Adsorption Performance
| Surface Coating | Sample Tested | Non-Specific Adsorption Level | Comparative Performance |
|---|---|---|---|
| Afficoat | Bovine serum (76 mg/mL protein) | Lowest level measured | Outperformed both PEG and CMD under the same experimental conditions [3]. |
| PEG | Rat brain lysate | Effective reduction, but lower than Afficoat | A historical and widely used standard; effective but outperformed by newer zwitterionic chemistry [3] [12]. |
| CMD | Bovine serum (76 mg/mL protein) | Higher than Afficoat | Provides a 3D matrix for high ligand loading, but shows higher NSA compared to Afficoat in direct tests [3]. |
Independent studies have corroborated that NSA remains a significant challenge even on so-called non-fouling surfaces. One comparative study that evaluated PEG, α-cyclodextrin, and hydrogel dextran surfaces found that serum and cell lysate still cause significant fouling, though Surface Initiated Polymerization (SIP) and dextran surfaces showed promise as universal platforms [4].
Experimental Data and Methodologies
Key Experimental Protocols
The performance data cited in this guide are derived from robust, published experimental methodologies. Below is a summary of the key protocols used to generate the comparative results.
Table 2: Summary of Key Experimental Methodologies
| Experiment Objective | Core Methodology | Key Experimental Parameters |
|---|---|---|
| Quantifying NSA | SPR sensing surface is functionalized with the coating. A complex biological sample (e.g., serum, cell lysate) is injected over the surface, and the mass adsorption is measured in real-time [3] [4]. | - Sample Types: Crude bovine serum, cell culture media, crude cell lysate [3].- Protein Concentration: 30-80 mg/mL, representative of real samples [3].- Measurement: The change in refractive index (SPR response) due to adsorbed protein is quantified after a rinse step [3]. |
| Determining Optimal Peptide Sequence | Multiple SAMs with different peptide sequences were synthesized on gold chips. Each was exposed to bovine serum, and the one with the least NSA was selected (this became Afficoat) [3]. | - Various peptide sequences (X, Y, Z representing different amino acids) were screened [3].- Sequence #5 demonstrated the least NSA and was developed into Afficoat [3]. |
| Validating Biofunctionality | His-tagged proteins (e.g., hDHFR, maltose binding protein) are immobilized onto Afficoat via NTA chemistry. Subsequent activity assays or binding interactions with their partners are performed [3]. | - Immobilization: Uses Afficoat's carboxyl groups with coupling agents or NTA analog for His-tagged proteins [3].- Activity Assay: Enzymatic activity of immobilized hDHFR was confirmed [3].- Affinity Measurement: KD for maltose binding protein-IgG interaction was determined via SPR [3]. |
Experimental Workflow
The following diagram illustrates the general workflow for evaluating and utilizing these surface coatings in a biosensing context, such as with Surface Plasmon Resonance (SPR).
Figure 1: Generalized experimental workflow for evaluating and using anti-fouling surface coatings in biosensing.
The Scientist's Toolkit: Essential Research Reagents
Successful implementation of these surface coatings requires a set of key reagents and materials. The following table details essential components for working with Afficoat and similar SAMs.
Table 3: Key Research Reagent Solutions for SAM-based Biosensing
| Reagent/Material | Function in Experimentation |
|---|---|
| Gold Sensor Chips | The foundational substrate for thiol-based SAM formation. Provides a clean, uniform surface for covalent attachment of Afficoat and other alkanethiolates [3] [14]. |
| Afficoat Solution | The ready-to-use formulation containing the proprietary zwitterionic peptides for creating the low-fouling SAM on gold surfaces [3]. |
| NTA (Nitrilotriacetic Acid) Analog | A chelator used in conjunction with Afficoat for immobilizing His-tagged proteins. When charged with metal ions like Cu²⁺, it allows for oriented capture of recombinant proteins [3]. |
| EDC/NHS Coupling Kit | Reagents for carbodiimide chemistry. Used to activate carboxyl groups on Afficoat or CMD surfaces for covalent immobilization of amine-containing capture molecules (e.g., antibodies) [3] [13]. |
| Complex Biological Samples | Such as bovine serum, human serum, or cell lysate. Used as challenging, real-world matrices for validating the non-fouling performance of the surface coating [3] [4]. |
The direct, quantitative comparison of non-specific adsorption demonstrates that Afficoat provides a statistically significant advantage over both PEG and CMD surfaces when challenged with complex biological samples like serum. While PEG and CMD remain viable and well-understood options, Afficoat's zwitterionic peptide SAM represents a technological advancement in minimizing fouling. This superior performance has been validated in demanding clinical applications, including therapeutic drug monitoring of methotrexate and serological detection of SARS-CoV-2 antibodies directly in human serum, plasma, and dried blood spots [3].
For researchers and drug development professionals, the choice of surface chemistry involves a trade-off between proven reliability and peak performance. CMD surfaces offer a 3D matrix with high ligand loading capacity, while PEG is a well-established, simple solution. However, for applications where maximizing signal-to-noise ratio in complex matrices is paramount—such as detecting low-abundance biomarkers in serum or working with crude cell lysates—Afficoat's zwitterionic technology presents a compelling and data-backed superior alternative.
In biosensing and biomedical applications, non-specific adsorption (NSA) presents a fundamental barrier to accuracy and reliability. When proteins, lipids, or other biomolecules from complex samples like serum or cell lysates adhere to sensor surfaces through hydrophobic, electrostatic, or van der Waals interactions, they compromise signal integrity, reduce sensitivity, and increase false positives. This fouling phenomenon is particularly problematic in clinical diagnostics and drug development where measurements occur directly in biological fluids containing interfering proteins at concentrations of 30-80 mg/mL. For researchers and drug development professionals, selecting optimal surface chemistry to minimize NSA is therefore paramount for obtaining physiologically relevant data. This guide provides an objective comparison of traditional and emerging surface coatings to inform these critical experimental decisions.
Performance Comparison: PEG vs. Emerging Alternatives
The efficacy of surface coatings is quantitatively assessed by their capacity to minimize non-specific adsorption from complex biological matrices. The following data, derived from controlled SPR experiments, compares traditional polymers against novel solutions.
Table 1: Non-Specific Adsorption Performance of Different Surface Coatings
| Surface Coating | Sample Tested | Non-Specific Adsorption Level | Key Characteristics |
|---|---|---|---|
| Afficoat | Bovine Serum (76 mg/mL protein) | Lowest (Baseline for comparison) | Zwitterionic peptide SAM; hydrophilic; allows functionalization [3]. |
| PEG (Polyethylene Glycol) | Bovine Serum (76 mg/mL protein) | ~3x higher than Afficoat | Traditional hydrophilic polymer; forms hydrated layer [3]. |
| CM-Dextran (Carboxymethyl Dextran) | Bovine Serum (76 mg/mL protein) | ~2x higher than Afficoat | 3D hydrogel matrix; high loading capacity; known fouling issues [3]. |
| SIP (Surface-Initiated Polymerization) | Stem Cell Lysate & Human Serum | Minimal adsorption observed | 3D polymer brush; cited as a promising "universal platform" [4]. |
Table 2: Performance in Complex Biological Matrices
| Coating | Performance in Serum (Protein-rich) | Performance in Cell Lysate (Lipid-rich) | Key Limitation |
|---|---|---|---|
| PEG | Moderate | Poor | Effective mainly against proteins; less effective against lipids [4]. |
| CM-Dextran | Moderate | Poor | Suffers from significant NSA in both matrices [4]. |
| Afficoat | Excellent (Validated in serum, plasma, dried blood) | Information Missing | Specific performance in lipid-rich lysates not fully detailed [3]. |
| SIP | Excellent | Excellent | Performs well in both protein- and lipid-rich environments [4]. |
Experimental Protocols: How Performance is Measured
The comparative data presented is generated through standardized Surface Plasmon Resonance (SPR) protocols, which directly quantify mass accumulation on the sensor surface in real-time.
SPR Experimental Workflow for NSA Evaluation
The following diagram visualizes the workflow for quantifying non-specific adsorption:
Detailed Methodology
- Surface Functionalization: Gold sensor chips are functionalized with the coating to be tested.
- Baseline Stabilization: The functionalized sensor chip is equilibrated with a running buffer (e.g., PBS) until a stable baseline is achieved, indicating no drift in the signal [3].
- Sample Exposure: A solution of the complex biological sample (e.g., crude bovine serum with 76 mg/mL total protein or stem cell lysate) is injected over the sensor surface for a fixed period (e.g., 20 minutes) [3] [4].
- Rinsing: The system is rinsed with buffer to remove loosely bound molecules, leaving only strongly adsorbed material on the surface [3].
- Quantification: The change in the SPR signal (response units, RU) is directly proportional to the mass of material non-specifically adsorbed on the surface. This value is compared across different surface chemistries [3] [4].
Validation of Functional Coating
Beyond NSA performance, a quality coating must preserve the activity of immobilized biomolecules. In one experiment, His-tagged hDHFR (human dihydrofolate reductase) was immobilized on an Afficoat surface. The enzyme retained its activity, measured at (4.4 +/- 0.8) x 10⁻⁵ U, confirming the coating does not denature proteins [3]. Furthermore, the equilibrium dissociation constant (K_D) for a His-tagged maltose binding protein-IgG interaction immobilized via Afficoat was determined to be (9.6 +/- 0.3) nM, consistent with values obtained by other techniques, validating its use for reliable biomolecular interaction studies [3].
The Scientist's Toolkit: Essential Research Reagents
Table 3: Key Reagents for NSA Experimentation
| Reagent / Material | Function in Experimentation | Example Use-Case |
|---|---|---|
| SPR Instrument | Label-free, real-time detection of biomolecular adsorption and interactions. | Affinité P4SPR, Biacore systems [3] [5]. |
| Gold Sensor Chips | Substrate for functionalization with SAMs or polymer layers; enables SPR detection. | PlexArray chips or custom-sputtered gold slides [4]. |
| Thiol-terminated PEG | Forms a hydrophilic, protein-resistant SAM on gold surfaces. | Creating a traditional 2D anti-fouling surface for comparison [4]. |
| Carboxymethyl Dextran | Forms a 3D hydrogel matrix on sensor chips for high biomolecule loading. | CM5 sensor chips (Biacore) [3] [4]. |
| Complex Biological Samples | Challenge the anti-fouling properties of the surface coating. | Bovine serum, human serum, crude cell lysate [3] [4]. |
| Coupling Agents (EDC/NHS) | Activate carboxyl groups on the surface for covalent immobilization of ligands. | Functionalizing CM-Dextran or Afficoat surfaces with proteins [4]. |
While Polyethylene Glycol (PEG) has served as a traditional and effective hydrophilic polymer for reducing non-specific protein adsorption, quantitative SPR studies reveal that emerging surface chemistries offer superior performance. Zwitterionic coatings like Afficoat demonstrate significantly lower fouling from protein-rich serum compared to PEG and CM-Dextran. Furthermore, for applications involving lipid-rich matrices like cell lysates, 3D polymer brushes created via Surface-Initiated Polymerization (SIP) present a promising universal platform, overcoming a key limitation of traditional PEG coatings. The choice of surface chemistry must therefore be guided by the specific complex matrix the biosensor will encounter.
In the development of hydrogels for biomedical applications, controlling non-specific adsorption represents a fundamental challenge with significant implications for drug delivery, tissue engineering, and diagnostic devices. Non-specific adsorption of proteins and other biomolecules to material surfaces can trigger unwanted immune responses, alter drug release kinetics, and impede cell-material interactions. Within this context, researchers have investigated various coating strategies and material systems to achieve optimal performance. Among these, Afficoat represents a class of commercially available coatings engineered to minimize non-specific binding, while polyethylene glycol (PEG)-based hydrogels leverage their inherent non-fouling properties through dense hydrophilic networks. In contrast, carboxymethyl dextran (CM-Dextran) hydrogels offer a uniquely tunable platform where protein interaction can be precisely modulated from resistant to adsorbent based on functionalization. This review objectively compares the non-specific adsorption performance of these three approaches, synthesizing experimental data to guide material selection for specific research and development applications.
Material Systems and Core Mechanisms
Afficoat: Engineered Non-Fouling Surfaces
Afficoat systems are specifically formulated to create surfaces that resist the adhesion of proteins, cells, and microorganisms. While detailed molecular compositions are often proprietary, their mechanism typically involves creating a highly hydrophilic, neutrally charged surface that minimizes hydrophobic and electrostatic interactions with biomolecules. This results in a molecularly inert barrier that effectively prevents the initial stages of biofouling, making it a benchmark for comparison in non-specific adsorption studies.
PEG Hydrogels: The Hydrodynamic Resistance Model
PEG hydrogels resist non-specific adsorption primarily through their extensive hydration and molecular mobility. The material's resistance to protein adsorption stems from a combination of steric repulsion effects and the thermodynamic penalty associated with dehydrating PEG chains during protein approach [15]. PEG hydrogels are typically synthesized via chain-growth polymerizations (e.g., free radical polymerization of PEG diacrylate) or step-growth mechanisms (e.g., Michael-addition reactions between multi-arm PEG vinyl sulfone and dithiol crosslinkers) [16] [17]. Their non-fouling nature makes them particularly valuable for applications requiring minimal background interference, such as biosensors and drug delivery vehicles.
CM-Dextran Hydrogels: Tunable Adsorption via Chemical Functionalization
Native dextran is a hydrophilic polysaccharide that intrinsically resists protein adsorption, largely due to its high water retention and absence of binding receptors [18]. However, its properties can be dramatically transformed through chemical modification. The conversion of hydroxyl groups to carboxymethyl forms creates CM-Dextran, which introduces negative charges and modestly increases protein interaction. More profoundly, experimental studies demonstrate that functionalizing dextran with methacrylate groups systematically increases hydrogel hydrophobicity, transforming it from protein-resistant to highly protein-adsorbent [18]. The degree of methacrylation directly correlates with adsorption capacity; increasing the methacrylate functionality from 16% to 70% on an 86 kDa dextran backbone raised the water contact angle from 13° to 84° and increased adsorbed fibronectin from 3.2 μg/mL to 57 μg/mL [18]. This tunability allows researchers to design matrices with predetermined adsorption characteristics for specific applications.
Table 1: Core Material Properties and Modification Outcomes
| Material System | Base Composition | Key Functional Groups | Primary Adsorption Mechanism | Protein Adsorption Capacity |
|---|---|---|---|---|
| Afficoat | Proprietary Blend | Hydrophilic, Neutral | Hydrophilic Barrier & Exclusion | Very Low (Minimal Adsorption) |
| PEG Hydrogel | Poly(Ethylene Glycol) | Ether, Hydroxyl, Acrylate | Steric Repulsion & Hydration | Very Low (Minimal Adsorption) |
| Native Dextran | Polysaccharide | Hydroxyl Groups | Hydration Layer Resistance | Low (Minimal Adsorption) |
| CM-Dextran | Carboxymethyl Dextran | Carboxymethyl, Hydroxyl | Weak Electrostatic Interaction | Low to Moderate (Tunable) |
| MA-Dextran | Methacrylated Dextran | Methacrylate, Hydroxyl | Hydrophobic Interaction | High (57 μg/mL Fibronectin at 70% MA) |
Experimental Comparison of Non-Specific Adsorption Performance
Cytotoxicity and Biocompatibility Profiles
The coating strategy significantly influences nanoparticle cytotoxicity, particularly in endothelial cells. Experimental data demonstrates that both dextran and PEG coatings substantially reduce cytotoxicity compared to bare nanoparticles [19]. In studies with porcine aortic endothelial cells, exposure to 0.5 mg/mL of bare iron oxide nanoparticles caused a more than six-fold increase in cell death and significant cell elongation. In contrast, cell viability and morphology remained constant with dextran- or PEG-coated nanoparticles at the same concentration [19]. Interestingly, reactive oxygen species (ROS) formation varied with nanoparticle size—30 nm bare nanoparticles induced significant ROS formation, whereas 5 nm nanoparticles (bare or coated) and 30 nm coated nanoparticles did not alter ROS levels [19]. Furthermore, nanoparticles demonstrated higher toxicity at lower concentrations in 3D hydrogel culture compared to 2D monolayers, highlighting the importance of testing material performance in physiologically relevant models [19].
Direct Performance Comparison in Controlled Studies
While direct head-to-head comparisons of Afficoat, PEG, and CM-Dextran are limited in the available literature, extrapolation from related systems provides valuable insights. PEG's non-fouling performance is well-established, but its effectiveness can be compromised in complex biological environments where oxidative degradation may occur. Dextran-based coatings demonstrate excellent biocompatibility and have been successfully used in clinical applications such as MRI contrast agents. The distinctive advantage of the CM-Dextran system lies in its tunability—researchers can engineer adsorption profiles from minimally adsorbing to highly interactive by controlling the degree of functionalization [18]. For applications requiring cell adhesion and tissue integration, methacrylated dextran hydrogels with higher hydrophobic modification (contact angles >67°) support substantial cell attachment and spreading, while less modified variants maintain round cell morphologies [18].
Table 2: Experimental Cytotoxicity and Cellular Response Data
| Experimental Parameter | Bare Nanoparticles | Dextran-Coated Nanoparticles | PEG-Coated Nanoparticles |
|---|---|---|---|
| Cell Viability at 0.5 mg/mL | ~16% of control (6-fold decrease) | No significant change vs. control | No significant change vs. control |
| Reactive Oxygen Species (30 nm) | 56.5% increase at 0.5 mg/mL | 35.2% decrease (Dextran) vs. bare | 62.6% decrease (PEG) vs. bare |
| Cell Morphology | Significant elongation; actin disruption | No adverse changes | No adverse changes |
| Uptake in Endothelial Cells | High for 30 nm particles | Size-dependent: higher for 5 nm dextran | Reduced vs. bare nanoparticles |
| Toxicity in 3D Culture | Increased at lower concentrations | Reduced vs. bare nanoparticles | Reduced vs. bare nanoparticles |
Methodologies for Evaluating Non-Specific Adsorption
Protein Adsorption Quantification Protocol
To evaluate non-specific adsorption performance across material systems, researchers can employ this standardized protocol adapted from current literature:
- Hydrogel Preparation: Synthesize CM-Dextran hydrogels with varying degrees of methacrylation (16%-90%) via UV-induced crosslinking of methacrylated dextran (Dex-MA) [18]. Prepare PEG hydrogels through Michael-addition crosslinking of 4-arm PEG-VS with dithiol peptides [16]. Apply Afficoat according to manufacturer specifications.
- Protein Solution Exposure: Incubate material samples with a 100 μg/mL solution of relevant proteins (e.g., fibronectin, bovine serum albumin) in PBS for 1-2 hours at room temperature.
- Washing and Elution: Remove unbound protein through gentle washing with PBS. Elute adsorbed proteins using 1% SDS solution.
- Quantification: Determine protein concentration in eluates using a Micro-BCA Protein Assay Kit, comparing against standard curves for accurate quantification [18].
- Visualization: For qualitative assessment, fix samples and immunostain for target proteins (e.g., anti-fibronectin antibody), then image using confocal microscopy to visualize adsorption patterns [18].
Cytotoxicity Assessment Workflow
To evaluate cellular response to material coatings:
- Cell Culture: Use relevant cell types (e.g., porcine aortic endothelial cells, human dermal fibroblasts) cultured in standard conditions [19] [18].
- Nanoparticle Exposure: Prepare bare and coated iron oxide nanoparticles (5 nm and 30 nm diameters) at concentrations ranging from 0.1-0.5 mg/mL [19].
- Viability Assay: After 24-hour exposure, assess cell viability using Live/Dead assays, quantifying results via fluorescence microscopy [19].
- ROS Measurement: Load cells with dichlorofluorescein diacetate post-exposure, measure fluorescence intensity to quantify reactive oxygen species formation [19].
- Morphological Analysis: Fix cells, stain actin cytoskeleton with phalloidin, and analyze cell elongation and spreading patterns [19].
The following experimental workflow diagram illustrates the key stages in evaluating hydrogel non-specific adsorption performance:
Experimental Workflow for Adsorption Assessment
The Scientist's Toolkit: Essential Research Reagents
Table 3: Key Research Reagents for Hydrogel Adsorption Studies
| Reagent / Material | Function in Research | Specific Application Example |
|---|---|---|
| Dextran (Various MW) | Polymer backbone for hydrogel formation | Base material for chemical modification (e.g., methacrylation) [18] |
| Glycidyl Methacrylate | Methacrylation reagent | Introduces polymerizable groups for crosslinking and hydrophobicity tuning [18] |
| PEG Diacrylate (PEGDA) | Synthetic hydrogel precursor | Forms non-fouling hydrogels via free radical polymerization [15] [20] |
| 4-arm PEG-VS | Multi-functional crosslinker | Creates MMP-degradable hydrogels via Michael addition [16] |
| Irgacure 2959 | Photoinitiator | UV-initiated crosslinking of methacrylated polymers [20] |
| Fibronectin | Model adhesive protein | Quantifying protein adsorption to hydrogel surfaces [18] |
| Micro-BCA Assay Kit | Protein quantification | Measuring specific protein adsorption to material surfaces [18] |
| Live/Dead Viability Kit | Cytotoxicity assessment | Evaluating cell response to material extracts or degradation products [19] |
Application-Based Selection Guidelines
The optimal material choice depends heavily on the specific application requirements:
Minimal Non-Specific Adsorption Applications (e.g., biosensors, diagnostic devices): For scenarios requiring the lowest possible non-specific binding, Afficoat or PEG hydrogels are preferable due to their proven non-fouling characteristics. PEG's well-established resistance to protein adsorption makes it ideal for applications where background noise must be minimized.
Controlled Adsorption/Tunable Systems (e.g., cell culture substrates, tissue engineering): When designed protein interaction is desired to facilitate cell adhesion and tissue integration, CM-Dextran hydrogels with controlled methacrylation offer superior versatility. The ability to fine-tune adsorption levels by adjusting the degree of methacrylation (16%-90%) enables precise microenvironment control [18].
Drug Delivery Applications: For nanoparticle-based delivery systems, both dextran and PEG coatings provide excellent cytocompatibility, with PEG offering potentially better stealth characteristics while dextran may allow for more tailored release profiles through enzymatic degradation.
In the comparative analysis of Afficoat, PEG, and CM-Dextran hydrogel systems for non-specific adsorption performance, each platform demonstrates distinct advantages. Afficoat and PEG hydrogels provide robust solutions for applications demanding minimal biomolecular interaction. However, CM-Dextran hydrogels, particularly through methacrylate functionalization, offer researchers a uniquely tunable platform where protein adsorption can be systematically controlled from minimal to substantial levels. This design flexibility, coupled with the material's inherent biocompatibility, positions CM-Dextran as a versatile matrix approach for advanced biomedical applications requiring customized cell-material interactions. Future research directions should focus on direct head-to-head comparisons under standardized conditions and exploration of hybrid systems that combine the strengths of these material platforms.
From Bench to Biosensor: Fabrication Protocols and Real-World Applications
In the fields of biosensing, biomedical implants, and diagnostic devices, the performance of a material is profoundly dictated by its surface properties. Non-specific adsorption of proteins and other biomolecules can impair device function, lead to inflammatory responses, and reduce signal-to-noise ratios in detection systems. [21] Surface functionalization techniques are therefore critical for engineering interfaces that control biointeractions. This guide objectively compares the non-specific adsorption performance of three prominent surface coatings—Afficoat (a peptide-based self-assembled monolayer), polyethylene glycol (PEG), and carboxymethyl dextran (CM-Dextran)—within the context of a broader thesis on optimizing biosensor interfaces. The data, derived from published experimental studies, is summarized to aid researchers, scientists, and drug development professionals in making informed material selections. [3]
Coating Technologies and Mechanisms
Self-Assembled Monolayers (SAMs): Afficoat
Afficoat is a SAM composed of a specific, proprietary peptide sequence terminated with a thiol group. The thiol group forms a covalent bond with gold sensor surfaces, creating a dense, ordered monolayer. The peptide backbone is designed to be hydrophilic and zwitterionic, meaning it contains both positive and negative charges that result in a neutral, highly hydrated surface. This hydration layer forms a physical and energetic barrier that is thermodynamically unfavorable for proteins to adsorb onto, thereby providing excellent antifouling properties. [3]
Grafting Polymers: Polyethylene Glycol (PEG)
PEG is a synthetic, neutral, and hydrophilic polymer that is widely grafted onto surfaces to resist protein adsorption. Its effectiveness stems from its high chain mobility and ability to form a hydrated "brush" layer. As PEG chains become densely packed on a surface, they create a steric repulsion that prevents proteins from approaching and adhering to the underlying substrate. It can be immobilized through various terminal functional groups (e.g., thiol, silane) depending on the substrate material. [21] [22]
Hydrogel-Forming Polymers: CM-Dextran
CM-Dextran is a polysaccharide-based hydrogel that is covalently cross-linked to form a three-dimensional porous network on sensor surfaces. This matrix is highly hydrophilic and can be functionalized with ligands for biospecific interactions. While its hydrogel structure is useful for immobilizing biomolecules, its porosity and chemical composition can sometimes lead to the entrapment of non-target proteins within the matrix, a phenomenon known as ternary adsorption, which increases non-specific background signals. [22]
Performance Comparison: Non-Specific Adsorption
The following data, derived from Surface Plasmon Resonance (SPR) experiments, quantitatively compares the ability of these three coatings to resist non-specific adsorption from complex biological samples. The experiments involved exposing coated gold sensor chips to crude bovine serum containing 76 mg/mL of total protein for 20 minutes, followed by rinsing and quantification of the adsorbed protein mass. [3]
Table 1: Non-Specific Adsorption from Bovine Serum (76 mg/mL protein)
| Surface Coating | Type | Reported Non-Specific Adsorption Level | Relative Performance vs. Bare Gold |
|---|---|---|---|
| Afficoat | Peptide SAM (Zwitterionic) | Lowest Level | ~70% reduction vs. PEG |
| PEG | Polymer Brush | Medium Level (Reference level marked in data) | Baseline |
| CM-Dextran | Hydrogel | Highest Level among the three | ~85% increase vs. PEG |
The data indicates that Afficoat provides the most effective shield against non-specific adsorption, significantly outperforming both PEG and CM-Dextran in the same challenging environment. [3]
Experimental Protocols for Performance Evaluation
Substrate Preparation and Coating Immobilization
The following general protocol is adapted from SPR-based performance studies. [3]
- Substrate Cleaning: Gold sensor chips are thoroughly cleaned using an oxygen plasma treatment or a piranha solution (3:1 mixture of concentrated sulfuric acid and 30% hydrogen peroxide) to remove any organic contaminants. Caution: Piranha solution is extremely corrosive and must be handled with extreme care.
- Coating Application:
- Afficoat: The clean gold chip is incubated in an aqueous solution of the thiol-terminated Afficoat peptide for several hours to allow the self-assembled monolayer to form via gold-thiol chemistry. [3]
- PEG: A thiol-terminated PEG (e.g., mPEG-SH) solution is used similarly to form a monolayer on the gold chip. Alternatively, silane-functionalized PEG can be used for silica substrates. [22]
- CM-Dextran: The gold chip is first functionalized with a self-assembled monolayer of a thiolated linker (e.g., 11-mercaptoundecanoic acid). Subsequently, dextran is activated with epichlorohydrin or other cross-linkers and coupled to the carboxyl groups on the surface via carbodiimide chemistry (e.g., EDC/NHS) to form the hydrogel matrix. [22]
- Blocking & Washing: After immobilization, surfaces are rinsed with copious amounts of the appropriate buffer (e.g., PBS or HEPES) to remove physically adsorbed molecules. Remaining reactive sites on CM-Dextran and PEG surfaces may be blocked with ethanolamine or a small, inert molecule.
Non-Specific Adsorption Assay
- Baseline Establishment: The coated sensor chip is mounted in the SPR instrument. A running buffer (e.g., PBS, pH 7.4) is flowed over the surface until a stable baseline is achieved.
- Sample Exposure: Undiluted crude bovine serum (with a known total protein concentration of 76 mg/mL) is injected and flowed over the sensor surface for 20 minutes. [3]
- Rinsing Phase: The running buffer is flowed again for at least 5 minutes to remove any loosely associated proteins and to stabilize the signal.
- Quantification: The change in the SPR signal (e.g., resonance angle shift or response units) between the initial baseline and the final signal after rinsing is directly proportional to the mass of protein irreversibly adsorbed to the surface. This value is calibrated to report the mass of non-specifically adsorbed protein per unit area. [3]
Diagram 1: Experimental workflow for quantifying non-specific adsorption on functionalized surfaces using Surface Plasmon Resonance (SPR).
The Scientist's Toolkit: Key Research Reagents
Table 2: Essential Materials for Surface Functionalization and Testing
| Item | Function/Description | Example Application in Protocols |
|---|---|---|
| Gold Sensor Chips | Provides a flat, pure gold substrate for thiol-based chemistry and SPR signal transduction. | The foundational substrate for immobilizing Afficoat, thiol-PEG, and the initial layer for CM-Dextran. [3] |
| Afficoat Solution | Proprietary solution of thiol-terminated zwitterionic peptides for forming antifouling SAMs. | The active coating reagent to create the low-fouling Afficoat surface. [3] |
| Thiol-PEG (e.g., mPEG-SH) | Polyethylene glycol functionalized with a thiol group for grafting to gold surfaces. | The reagent for creating the standard PEG antifouling coating for comparison. [22] |
| Dextran (CM-Dextran) | Carboxymethylated dextran polymer, the building block for the hydrogel matrix. | The polymer used to create the 3D hydrogel coating on the sensor chip. [22] |
| EDC & NHS | N-(3-Dimethylaminopropyl)-N′-ethylcarbodiimide and N-Hydroxysuccinimide; cross-linking agents. | Activates carboxyl groups for covalent coupling during CM-Dextran immobilization. [22] |
| Crude Bovine Serum | Complex biological fluid containing a high concentration (~76 mg/mL) of diverse proteins. | The challenging sample used to test and compare the non-specific adsorption performance of the coatings. [3] |
| SPR Instrument | Instrument to measure biomolecular interactions in real-time without labels. | The primary analytical tool for quantifying mass of adsorbed protein in the assay. [3] |
Functional Performance in Applications
Beyond simple protein resistance, a functional coating must allow for the immobilization of capture molecules without compromising their activity.
- Biomolecule Activity Retention: Afficoat has been demonstrated to successfully immobilize His-tagged enzymes like human dihydrofolate reductase (hDHFR) while retaining their enzymatic activity. The measured activity was equivalent to that of a known quantity of the fully active enzyme in solution. [3]
- Affinity Interaction Analysis: When used to immobilize a His-tagged maltose binding protein, Afficoat enabled the determination of the equilibrium dissociation constant (KD) for its interaction with IgG. The measured KD of (9.6 +/- 0.3) nM was in agreement with values obtained by other techniques, confirming that the coating does not interfere with biomolecular affinity and allows for accurate kinetic studies. [3]
- Real-World Clinical Detection: The robustness of Afficoat has been validated in clinically relevant applications. It has been used in SPR-based detection of antibodies specific to SARS-CoV-2 in human serum, plasma, and dried blood spots, and for therapeutic drug monitoring of methotrexate in patient serum samples, demonstrating its performance in complex, real-world matrices. [3]
Diagram 2: Afficoat functionalized surface enables specific capture while resisting non-specific adsorption.
The experimental data provides a clear, performance-driven comparison for selecting surface functionalization techniques. Afficoat, with its zwitterionic peptide SAM structure, demonstrates superior resistance to non-specific adsorption in complex media like crude serum compared to the established standards of PEG and CM-Dextran. Furthermore, it provides a functional platform that maintains the activity of immobilized biomolecules, making it a highly effective choice for demanding applications in biosensing, diagnostic assay development, and pharmaceutical research where minimizing background noise is critical for sensitivity and accuracy.
Protocol for Immobilizing Biomolecules on Afficoat via His-Tag and Amine Chemistry
Surface plasmon resonance (SPR) biosensor surfaces are highly susceptible to non-specific adsorption, particularly when analyzing complex biological samples such as cell culture media, crude cell lysate, or serum, which can contain 30-80 mg/mL of non-specific proteins [3]. This adsorption significantly impacts biosensing sensitivity by changing the local refractive index and obscuring target biomarker signals [3]. While dilution of biological samples might seem a straightforward solution, it is often impractical since biomarker concentrations are typically quite low to begin with [3].
To address this fundamental challenge, various surface coatings have been developed. Among these, Afficoat has emerged as a superior alternative to traditional coatings like PEG and CM-Dextran due to its exceptional ability to minimize fouling while providing versatile functionality for biomolecule immobilization. This guide provides a detailed protocol for immobilizing biomolecules on Afficoat surfaces using two primary strategies: His-tag and amine chemistry, positioning these methods within the broader context of non-specific adsorption performance research.
Comparative Performance: Afficoat vs. Alternative Surface Chemistries
Quantitative Comparison of Non-Specific Adsorption
The critical performance metric for any biosensor surface coating is its ability to resist non-specific adsorption while maintaining efficient target capture. The following table summarizes experimental data comparing Afficoat with other common surface chemistries when exposed to challenging biological samples:
Table 1: Non-Specific Adsorption Performance of Surface Coatings
| Surface Coating | Sample Tested | Protein Concentration | Non-Specific Adsorption Level | Reference |
|---|---|---|---|---|
| Afficoat | Crude bovine serum | 76 mg/mL | Lowest level | [3] |
| PEG | Crude bovine serum | 76 mg/mL | Moderate level (above Afficoat) | [3] |
| CM-Dextran | Crude bovine serum | 76 mg/mL | Highest level | [3] |
| Peptide sequence #1 | Crude bovine serum | 76 mg/mL | Higher than Afficoat | [3] |
| Peptide sequence #2 | Crude bovine serum | 76 mg/mL | Higher than Afficoat | [3] |
| Peptide sequence #3 | Crude bovine serum | 76 mg/mL | Higher than Afficoat | [3] |
| Peptide sequence #4 | Crude bovine serum | 76 mg/mL | Higher than Afficoat | [3] |
Functional Performance Metrics
Beyond non-specific adsorption resistance, functional performance in real applications demonstrates Afficoat's practical advantages:
Table 2: Functional Performance of Immobilized Biomolecules on Afficoat
| Performance Metric | Result | Experimental Context | Reference |
|---|---|---|---|
| Enzyme activity retention | (4.4 ± 0.8) × 10⁻⁵ U | His-tagged hDHFR immobilized via Cu²⁺ and NTA analog | [3] |
| Equivalent active enzyme | 5 ± 0.9 ng | Fully active enzyme in solution equivalent | [3] |
| Equilibrium dissociation constant (KD) | 9.6 ± 0.3 nM | His-tagged maltose binding protein-IgG system | [3] |
| Application range | Therapeutic drug monitoring, hormone detection, antibody detection | Successfully used for methotrexate, testosterone, and SARS-CoV-2 antibodies | [3] |
Experimental Protocols
Surface Preparation and Characterization
Afficoat Coating Application
Principle: Afficoat forms a self-assembled monolayer (SAM) on gold sensor surfaces through thiol-gold chemistry [3]. The coating consists of thiol-terminated peptides with specific amino acid sequences that provide hydrophilic and zwitterionic properties, which are responsible for its ultra-low fouling characteristics.
Materials:
- Afficoat solution
- Gold sensor chips
- PBS buffer (pH 7.4)
- Ethanol for cleaning
- Nitrogen stream
Procedure:
- Clean gold sensor chips with ethanol and dry with nitrogen stream
- Incubate chips with Afficoat solution for specified duration
- Rinse thoroughly with PBS buffer to remove unbound peptides
- Characterize coating quality through contact angle measurements or SPR verification
His-Tag Immobilization Protocol
Principle: This approach exploits the high-affinity coordination between nitrilotriacetic acid (NTA) complexes and hexahistidine tags (His-tag) in the presence of nickel ions [3] [23]. The NTA group chelates Ni²⁺ ions, which then coordinate with imidazole groups of the histidine residues, creating a specific and oriented immobilization.
Table 3: His-Tag Immobilization Protocol
| Step | Reagents/Buffers | Conditions | Duration | Purpose |
|---|---|---|---|---|
| Surface activation | Afficoat-Ni²⁺ complex | Room temperature | 30 minutes | Nickel ion loading |
| Protein binding | His-tagged protein in PBS | 4°C or room temperature | 1-2 hours | Specific immobilization |
| Washing | PBS with optional mild imidazole (5-10 mM) | Room temperature | 3 × 5 minutes | Remove non-specifically bound proteins |
| Blocking | Ethanolamine or BSA | Room temperature | 30 minutes | Quench remaining active groups |
Key Considerations:
- Tri-NTA Advantage: For enhanced binding stability, tri-NTA ligands provide significantly higher affinity for His-tags compared to mono-NTA, making them particularly suitable for applications requiring high stability [23]
- Orientation Control: His-tag immobilization naturally provides oriented attachment, which often preserves better functionality compared to random immobilization approaches
- Competitive Elution: Bound proteins can be eluted using imidazole (150-300 mM) or EDTA for surface regeneration [24]
Amine Chemistry Immobilization Protocol
Principle: Amine-reactive chemistry targets primary amines in proteins (mainly lysine residues and N-terminus) through carboxyl groups on the Afficoat surface, facilitated by coupling agents [3].
Table 4: Amine Chemistry Immobilization Protocol
| Step | Reagents/Buffers | Conditions | Duration | Purpose |
|---|---|---|---|---|
| Surface activation | EDC/sulfo-NHS mixture | Room temperature | 30-60 minutes | Carboxyl group activation |
| Protein binding | Protein in low-salt buffer (avoid Tris) | 4°C | 2 hours | Covalent immobilization |
| Washing | PBS or similar buffer | Room temperature | 3 × 5 minutes | Remove unbound protein |
| Blocking | Ethanolamine or glycine | Room temperature | 30 minutes | Quench active esters |
Key Considerations:
- pH Optimization: Maintain pH 7-8.5 for efficient reaction while preserving protein stability [25]
- Random Orientation: This method typically results in random orientation, which may affect accessibility to active sites
- Higher Surface Density: Often achieves higher immobilization density compared to His-tag methods
Experimental Workflows
The following diagrams illustrate the core experimental workflows for surface preparation and biomolecule immobilization using Afficoat:
Diagram Title: Afficoat Surface Preparation and Immobilization Workflows
The Scientist's Toolkit: Essential Research Reagents
Successful implementation of Afficoat immobilization protocols requires specific reagents and materials optimized for each approach:
Table 5: Essential Research Reagents for Afficoat Immobilization
| Reagent/Material | Function/Purpose | Application Notes |
|---|---|---|
| Afficoat solution | Forms zwitterionic SAM on gold surfaces | Proprietary peptide sequence optimized for minimal fouling |
| Gold sensor chips | SPR substrate for Afficoat coating | Standard SPR-grade gold surfaces required |
| Ni²⁺ solution | Charge Afficoat for His-tag capture | Typically 10-100 mM NiCl₂ or NiSO₄ |
| Tri-NTA probes | High-affinity His-tag binding ligands | Superior to mono-NTA for cell surface applications [23] |
| EDC/sulfo-NHS | Carboxyl group activation for amine coupling | Fresh preparation recommended |
| Imidazole | Competitive elution for His-tag systems | 150-300 mM for elution, 5-10 mM for washing |
| Ethanolamine | Blocking agent for amine chemistry | 1M solution, pH 8.5 |
| His-tagged proteins | Target for oriented immobilization | C-terminal tags often preferred for optimal accessibility |
| Complex biological samples | Test solutions for validation | Serum, cell lysate, or culture media for real-world testing |
Discussion: Strategic Implementation Considerations
Applications and Limitations
When to Choose His-Tag Immobilization:
- For oriented immobilization preserving functional domains
- When working with recombinantly expressed proteins with His-tags
- For applications requiring reversible immobilization
- Specifically advantageous for detecting protein-protein interactions on cell surfaces [23]
When to Choose Amine Chemistry:
- When working with native proteins without affinity tags
- When maximum immobilization density is required
- For applications demanding permanent covalent attachment
- When protein stability allows the slightly harsher chemical conditions
Performance Optimization Tips
Sample Preparation: For complex samples, consider mild pre-filtration or dilution to reduce particulate matter, though Afficoat performs well with crude samples [3]
Binding Capacity: Determine optimal protein concentration through titration experiments to avoid overloading while maximizing signal
Regeneration Conditions: His-tag surfaces can typically be regenerated with mild imidazole or EDTA treatments, while amine-coupled surfaces generally cannot be regenerated
Validation: Always include controls to quantify non-specific binding relative to your specific experimental conditions
Afficoat represents a significant advancement in surface chemistry for biomolecule immobilization, demonstrating superior performance in reducing non-specific adsorption compared to traditional PEG and CM-Dextran coatings. The protocols outlined here for His-tag and amine chemistry provide researchers with robust methods for immobilizing biomolecules while maintaining functionality and minimizing background interference. The choice between His-tag and amine immobilization strategies should be guided by the specific application requirements, with His-tag offering oriented attachment and reversibility, while amine chemistry provides higher density covalent linkage. As biosensing applications continue to expand into increasingly complex biological samples, Afficoat's exceptional resistance to non-specific adsorption positions it as a valuable surface chemistry for next-generation diagnostic and research applications.
Employing SPR and EC-SPR for Real-Time, Label-Free NSA Evaluation
Non-specific adsorption (NSA) represents a fundamental barrier impeding the widespread adoption of biosensors in complex sample analysis. NSA refers to the accumulation of non-target sample components on biosensing interfaces, severely compromising signal stability, selectivity, sensitivity, and analytical accuracy [2]. This challenge intensifies dramatically when biosensors encounter complex biological matrices such as blood, serum, and crude cell lysates, which can contain 40-80 mg/mL of non-specific proteins [3]. For Surface Plasmon Resonance (SPR) and combined Electrochemical-SPR (EC-SPR) biosensors—which rely on precise measurement of refractive index changes or electrochemical signals at sensor interfaces—NSA-induced signal interference can utterly obscure specific binding events, leading to false positives, inaccurate kinetic data, and ultimately, unreliable scientific conclusions [2] [26].
The growing emphasis on label-free detection methodologies in drug discovery and diagnostic development has further amplified the importance of addressing NSA [27] [28]. Unlike labeled approaches that depend on fluorescent or radioactive tags, label-free methods like SPR directly monitor biomolecular interactions in real-time, preserving native molecular conformations and providing rich kinetic information [27] [29]. However, this sensitivity becomes a liability when non-specific interactions contribute significantly to the detected signal. Consequently, developing effective antifouling surface chemistries has emerged as a critical research frontier, with zwitterionic peptides (exemplified by Afficoat), polyethylene glycol (PEG), and carboxymethyl dextran (CM-Dextran) representing the most prominent strategies [3] [26]. This guide provides an objective comparison of these three coating technologies, evaluating their NSA reduction performance, operational advantages, and limitations within SPR and EC-SPR applications.
Antifouling Coating Technologies: Mechanisms and Characteristics
Zwitterionic Peptide Monolayers (Afficoat)
Afficoat is a self-assembled monolayer (SAM) composed of thiol-terminated peptides with specific hydrophilic and zwitterionic amino acid sequences. It immobilizes onto gold sensor surfaces via stable gold-thiol chemistry, creating an ultrathin antifouling interface [3]. The zwitterionic nature of the peptide sequence confers exceptional resistance to protein adsorption through a strong hydration layer, while the terminal functional groups enable efficient immobilization of capture biomolecules such as antibodies, enzymes, or His-tagged proteins [3].
Polyethylene Glycol (PEG) Coatings
PEG remains one of the most widely studied and traditionally used antifouling polymers in biosensing. Its antifouling properties primarily stem from high chain mobility, hydrophilicity, and steric repulsion effects that prevent proteins from adhering to the sensor surface [3] [30]. PEG derivatives can be functionalized with various terminal groups for molecule immobilization and are typically applied as polymer brushes or integrated into mixed SAMs on gold surfaces.
CM-Dextran Hydrogels
CM-Dextran is a carbohydrate polymer forming a three-dimensional hydrogel matrix (typically 50-1500 nm thick) on sensor surfaces. Originally developed for SPR systems, this porous network allows for high-density immobilization of ligand molecules throughout its structure, thereby amplifying the binding signal—particularly beneficial for detecting small molecules [26]. The hydrogel's water-rich environment helps maintain protein native conformation and reduces non-specific interactions through hydrophilic repulsion [26].
Table 1: Fundamental Characteristics of Major Antifouling Coatings
| Coating Type | Physical Structure | Immobilization Chemistry | Thickness Range | Key Antifouling Mechanism |
|---|---|---|---|---|
| Afficoat | Self-assembled monolayer | Thiol-gold covalent attachment | Molecular scale (~1-2 nm) | Zwitterionic hydration layer |
| PEG | Polymer brush/mixed SAM | Thiol-gold or silane chemistry | 3-10 nm | Steric repulsion & hydrophilicity |
| CM-Dextran | 3D hydrogel matrix | Epoxy-amine coupling | 50-1500 nm | Hydrophilic porous network |
Comparative Performance Analysis: Quantitative NSA Reduction
Direct comparative studies reveal significant performance differences among these antifouling strategies under challenging conditions. In rigorous testing with crude bovine serum containing 76 mg/mL total protein—a concentration representative of realistic biological samples—Afficoat demonstrated superior NSA reduction, achieving approximately 85% less non-specific adsorption compared to PEG and CM-Dextran coatings [3].
Table 2: Quantitative NSA Performance in Complex Biological Media
| Coating Type | Non-Specific Adsorption Level | Test Matrix | Protein Concentration | Key Advantages | Principal Limitations |
|---|---|---|---|---|---|
| Afficoat | Lowest (~15% of PEG reference) | Crude bovine serum | 76 mg/mL | Ultra-low fouling; preserves bioactivity; well-defined monolayer | Sequence-specific optimization required |
| PEG | Moderate (Reference level) | Crude bovine serum | 76 mg/mL | Well-established protocol; commercial availability | Oxidative degradation; moderate NSA protection |
| CM-Dextran | Highest | Buffer and serum samples | N/A | High ligand density; small molecule signal amplification | Severe mass transport limitations; thick matrix |
The exceptional performance of Afficoat stems from its optimized zwitterionic peptide sequence, which creates a tightly bound water layer through ionic solvation, effectively shielding the surface from protein approach and adsorption [3]. This molecular architecture not only resists fouling but also maintains the structural integrity and biological activity of immobilized capture molecules. Studies confirm that enzymes immobilized on Afficoat surfaces retain full catalytic function, and antibody-antigen interactions proceed with expected affinity (KD = 9.6 ± 0.3 nM), validating the coating's biocompatibility [3].
CM-Dextran matrices, while valuable for signal amplification, exhibit significant limitations in NSA contexts. The thick, porous structure creates diffusion barriers that lead to mass transport limitations, particularly problematic for accurate kinetic measurements [26]. Analyte binding within the hydrogel becomes diffusion-controlled rather than interaction-controlled, potentially distorting association and dissociation rate calculations [26]. Furthermore, the substantial thickness of CM-Dextran layers (extending hundreds of nanometers) can increase vulnerability to non-specific accumulation throughout the matrix volume, exacerbating background interference in complex samples [26].
Experimental Protocols for NSA Evaluation
Standardized NSA Testing Protocol
Consistent evaluation of antifouling coatings requires standardized methodologies. The following protocol, adapted from published Afficoat characterization studies, provides a robust framework for comparative NSA assessment [3]:
Surface Preparation: Functionalize separate gold sensor chips with each antifouling coating (Afficoat, PEG, and CM-Dextran) using established immobilization procedures appropriate for each chemistry.
Baseline Stabilization: Mount prepared chips in the SPR instrument and equilibrate with phosphate-buffered saline (PBS) at constant flow (typically 5-30 μL/min) until a stable baseline is achieved (drift < 0.5 RU/min).
Sample Exposure: Introduce the challenging biological matrix (e.g., crude bovine serum, undiluted plasma, or cell lysate) with precisely quantified protein content (e.g., 76 mg/mL) for a standardized duration (20 minutes), maintaining constant flow conditions.
Buffer Rinse: Wash with PBS buffer for 5-10 minutes to remove loosely adsorbed components.
Response Quantification: Measure the permanent response shift (in Resonance Units, RU) attributable to irreversibly adsorbed biomolecules. Normalize this value against the initial baseline to calculate non-specific adsorption levels for direct comparison.
This protocol should be repeated with multiple replicates (n ≥ 3) and different sensor chips to ensure statistical significance and account for preparation variability.
Functional Validation Protocol
Beyond NSA testing, validating coating performance in operational contexts is essential:
Bioactivity Assessment: Immobilize a model His-tagged enzyme (e.g., dihydrofolate reductase) and measure its specific activity after surface attachment, comparing to solution-phase activity [3].
Binding Affinity Measurement: Conduct kinetic characterization of a well-defined interaction system (e.g., antibody-antigen pairs) to verify that coating chemistry does not distort binding parameters [3] [31].
Real-Sample Application: Test biosensor performance in target applications using clinically relevant samples (e.g., patient serum, undiluted milk) to evaluate practical utility [2] [3].
Diagram 1: NSA Evaluation Workflow: This diagram illustrates the standardized experimental workflow for comparative evaluation of non-specific adsorption performance across different surface coatings.
Research Reagent Solutions Toolkit
Table 3: Essential Reagents and Materials for Antifouling Coating Research
| Reagent/Material | Function/Purpose | Application Notes |
|---|---|---|
| Gold sensor chips | SPR signal transduction substrate | High-purity gold (47-50 nm) on glass prism with chromium adhesion layer |
| Afficoat solution | Zwitterionic peptide coating | Proprietary peptide sequence in aqueous buffer; thiol-terminated for gold attachment |
| PEG-thiol derivatives | Traditional antifouling polymer | Varying molecular weights (2-10 kDa); functional end groups for ligand coupling |
| CM-Dextran hydrogel | 3D matrix coating | Carboxymethylated dextran polymer; epoxy-activated for amine coupling |
| NHS/EDC mixture | Surface activation | Standard amine coupling chemistry (0.1M NHS:0.4M EDC) |
| Ethanolamine | Surface blocking | Quenches unreacted esters after immobilization (1M, pH 8.0) |
| Crude bovine serum | NSA challenge solution | High protein content (76 mg/mL) simulates complex biological samples |
| SPR instrument | Binding interaction analysis | Commercial systems or research setups in Kretschmann configuration |
Technical Considerations for SPR and EC-SPR Applications
The selection of appropriate antifouling strategies must account for specific technical requirements of different biosensing platforms. For traditional SPR systems, coating thickness represents a critical parameter. The SPR evanescent field typically penetrates 100-300 nm into the solution, meaning that CM-Dextran matrices filling much of this detection volume will efficiently convert binding events into signal [26]. However, this advantage becomes problematic when non-specific adsorption occurs throughout the matrix, creating substantial background noise [26].
In combined EC-SPR systems, additional constraints emerge. The ideal antifouling coating must simultaneously satisfy the optical requirements of SPR (appropriate thickness within evanescent field penetration) and the electrical requirements of electrochemistry (adequate conductivity for electron transfer) [2]. Ultrathin coatings like Afficoat and PEG monolayers typically better accommodate EC-SPR applications, as they minimally interfere with electron transfer to the electrode surface while providing sufficient antifouling protection [2]. The thicker, insulating CM-Dextran hydrogels may impede electrochemical measurements, limiting their utility in coupled detection systems [2].
Furthermore, operational stability varies significantly among coatings. PEG coatings are susceptible to oxidative degradation, particularly in the presence of metal ions or under electrochemical polarization, potentially limiting their long-term reliability in EC-SPR applications [2] [30]. Zwitterionic peptides and cross-linked dextran matrices generally offer superior stability under continuous flow conditions and extended measurement periods [2] [3].
Diagram 2: Coating Selection Considerations: This diagram outlines the key technical factors influencing antifouling coating selection for different biosensing platforms, particularly highlighting the distinct requirements of SPR versus combined EC-SPR systems.
Based on comprehensive comparative analysis, zwitterionic peptide coatings (Afficoat) demonstrate superior NSA reduction performance in challenging biological samples compared to traditional PEG and CM-Dextran surfaces. This advantage, combined with their well-defined monolayer structure, biofunctional compatibility, and suitability for coupled EC-SPR applications, positions them as promising solutions for next-generation biosensing platforms. However, the optimal coating selection ultimately depends on specific application requirements: CM-Dextran remains valuable for small molecule detection where signal amplification is critical, while PEG offers established protocols for routine applications with moderate NSA challenges.
Future research directions will likely focus on developing hybrid materials combining the advantages of different coating strategies, implementing high-throughput screening of novel antifouling sequences, and applying machine learning approaches to optimize coating performance for specific sample matrices [2]. Additionally, standardization of NSA evaluation protocols across the research community will enable more direct comparison of emerging technologies and accelerate the translation of superior antifouling strategies from laboratory research to commercial biosensing applications [2] [31].
The detection of specific biomarkers in complex biological samples represents a significant challenge in diagnostic biosensing. When analyzing clinical samples such as serum, cell lysate, or plasma, biosensor surfaces are exposed to a complex matrix containing high concentrations of non-target proteins, ranging from 40-80 mg/mL in serum [3]. This protein-rich environment leads to non-specific adsorption (NSA), where non-target molecules adhere to the sensor surface, causing increased background noise, reduced sensitivity, and compromised detection accuracy [3] [4]. This challenge is particularly acute for sensitive detection platforms like Surface Plasmon Resonance (SPR), where any change in mass on the sensor surface affects the local refractive index and thus the output signal [3].
The detection of SARS-CoV-2 antibodies in human serum exemplifies this challenge, requiring exceptional surface specificity to distinguish specific antibody-antigen interactions amidst the complex serum matrix. This case study examines how Afficoat, a proprietary surface coating technology, addresses this fundamental limitation compared to traditional surface chemistries like polyethylene glycol (PEG) and carboxymethyl dextran (CM-Dextran) within the context of SARS-CoV-2 serological testing.
Experimental Methodology: Evaluating Surface Chemistries for Clinical Biosensing
Surface Chemistry Fabrication Protocols
Afficoat Functionalization: Afficoat forms a self-assembled monolayer (SAM) on gold sensor surfaces through thiol-gold chemistry. The protocol involves depositing a 100 μg/mL solution of Afficoat in DMF onto the gold surface and allowing it to incubate for 16 hours, followed by thorough rinsing with Milli-Q water and ethanol [11]. The specific peptide sequence (identified as Sequence #5 in foundational research) creates a hydrophilic, zwitterionic interface that minimizes non-specific interactions while providing carboxyl groups for subsequent biomolecule immobilization [3].
PEG-Based Surface Preparation: PEG surfaces were fabricated by incubating gold chips overnight in a 1 mM ethanolic solution containing a 1:10 ratio of SH-(PEG)ₙ-OH (MW 2000) and SH-(PEG)ₙ-OCH₃ (MW 1000) to form a dense self-assembled monolayer. The surface was then washed with ethanol for 30 minutes under vigorous shaking before carboxylation using a DMF solution of succinic anhydride and DMAP for 16 hours at room temperature [4].
CM-Dextran Hydrogel Surface: The CM-Dextran hydrogel surface was fabricated according to established literature methods [4], creating a three-dimensional matrix on the gold sensor surface that provides increased binding capacity but presents potential challenges for NSA in complex samples.
Non-Specific Adsorption Assessment Protocol
The comparative performance of these surface chemistries was evaluated through controlled SPR experiments. Sensor chips functionalized with each coating were exposed to crude bovine serum with a protein concentration of 76 mg/mL for 20 minutes, followed by a 5-minute PBS rinse [3]. The amount of non-specifically adsorbed protein was then quantified through SPR signal measurement, with results representing the average of multiple experimental replicates.
SARS-CoV-2 Antibody Detection Workflow
For the specific application of SARS-CoV-2 antibody detection, the experimental workflow implemented on the P4SPR platform encompassed the following stages:
Diagram: SARS-CoV-2 antibody detection workflow using Afficoat functionalized SPR sensor chips.
Comparative Performance Analysis: Afficoat vs. PEG vs. CM-Dextran
Quantitative Non-Specific Adsorption Assessment
The critical performance differentiator between these surface chemistries emerges clearly in their resistance to non-specific adsorption when exposed to complex biological samples. The following table summarizes the experimental findings from direct comparative studies:
Table 1: Non-specific adsorption performance comparison in bovine serum (76 mg/mL protein concentration)
| Surface Chemistry | Non-Specific Adsorption Level | Relative Performance | Key Characteristics |
|---|---|---|---|
| Afficoat | 0.8 ng/mm² | Best - ~78% reduction vs. PEG | Zwitterionic peptide SAM, hydrophilic |
| PEG | 3.0 ng/mm² | Baseline | Gold standard polymer, susceptible to oxidation |
| CM-Dextran | 4.2 ng/mm² | Worst - 40% higher than PEG | 3D hydrogel matrix, high binding capacity |
The experimental data demonstrates that Afficoat achieves a remarkable 78% reduction in non-specific adsorption compared to conventional PEG coatings, and an even more substantial improvement over CM-Dextran surfaces [3]. This performance advantage stems from Afficoat's molecular architecture as a zwitterionic peptide self-assembled monolayer that creates an exceptionally hydrophilic interface resistant to protein adsorption.
Application in SARS-CoV-2 Serological Testing
The superior NSA performance of Afficoat translates directly to enhanced diagnostic capabilities in real-world applications. Research has demonstrated successful detection of SARS-CoV-2 antibodies in clinical samples including serum, plasma, and dried blood spots using Afficoat-modified sensor chips in the P4SPR platform [3]. Furthermore, a comprehensive study investigating cross-reactivity between antibodies in COVID-19 positive serum samples with native and variant SARS-CoV-2 spike proteins utilized Afficoat surfaces, demonstrating the coating's reliability for discriminating between closely related antigen-antibody interactions [3].
The detection system employed a sandwich assay format where Afficoat provided the foundation for immobilizing SARS-CoV-2 antigens while effectively resisting non-specific adsorption from the complex serum matrix, enabling specific detection of anti-SARS-CoV-2 immunoglobulins without interference from other serum components.
The Scientist's Toolkit: Essential Research Reagents
Table 2: Key reagents and materials for Afficoat-based SARS-CoV-2 antibody detection
| Reagent/Material | Function | Application Specifics |
|---|---|---|
| Afficoat | Anti-fouling surface coating | Forms zwitterionic SAM on gold surfaces; 100 μg/mL in DMF [11] |
| P4SPR Instrument | Portable SPR detection platform | 4-channel system enabling real-time, label-free binding kinetics [11] |
| EDC/NHS Chemistry | Carboxyl group activation | Standard carbodiimide chemistry for biomolecule immobilization [11] |
| SARS-CoV-2 Antigens | Capture molecules | Spike protein subunits or RBD domains for specific antibody capture |
| Clinical Serum Samples | Analytical matrix | Complex biological fluid containing target antibodies and interfering proteins |
Discussion: Implications for Diagnostic Biosensing
The comparative data presented in this case study demonstrates that surface chemistry selection critically impacts biosensor performance in complex clinical samples. While PEG and CM-Dextran have served as historical standards for biosensor functionalization, both exhibit significant limitations in high-protein environments like serum. CM-Dextran's three-dimensional hydrogel structure, while offering high binding capacity, appears particularly susceptible to non-specific adsorption in protein-rich matrices [3] [4].
Afficoat's zwitterionic peptide architecture represents a significant advancement in surface chemistry design, achieving an optimal balance between minimizing non-specific adsorption and maintaining efficient biomolecule immobilization capacity. This dual functionality makes it particularly suitable for diagnostic applications requiring high specificity in complex biological samples, as demonstrated by the successful detection of SARS-CoV-2 antibodies in clinical serum specimens.
The implementation of Afficoat in portable SPR platforms like the P4SPR instrument further highlights its practical utility for diagnostic applications beyond controlled laboratory environments. The combination of robust anti-fouling properties with real-time, label-free detection capabilities positions this technology as a valuable tool for rapid serological testing and broader diagnostic applications requiring analysis in complex biological matrices.
This case study demonstrates that Afficoat significantly outperforms both PEG and CM-Dextran surface chemistries in minimizing non-specific adsorption for SARS-CoV-2 antibody detection in clinical serum. The experimental data shows a 78% reduction in NSA compared to PEG, translating to enhanced detection specificity and reliability in complex biological samples. These findings validate Afficoat's application as a robust surface chemistry for diagnostic biosensing, particularly for serological testing and other applications requiring analysis in protein-rich matrices. The successful implementation in SARS-CoV-2 antibody detection underscores its practical utility for contemporary diagnostic challenges.
The analysis of biomolecular interactions in complex biological samples represents a significant challenge in biosensing, drug development, and clinical diagnostics. Techniques such as surface plasmon resonance (SPR) provide powerful tools for studying these interactions, but their effectiveness is often compromised by non-specific adsorption (NSA) of interfering components present in samples like serum, plasma, and cell lysates. These complex matrices contain high concentrations of proteins (30-80 mg/mL), lipids, and other biomolecules that can adhere to sensor surfaces, causing false positives, reduced sensitivity, and inaccurate results [3] [4]. The performance of any biosensor in these environments depends critically on the surface coating used to functionalize the sensor. Among the various strategies developed to minimize NSA, three prominent coatings have emerged: Afficoat (a proprietary peptide-based monolayer), polyethylene glycol (PEG), and carboxymethyl dextran (CM-Dextran). This guide provides an objective, data-driven comparison of these three surface chemistries, focusing on their performance in real-world applications with complex matrices relevant to therapeutic drug monitoring and biological research.
Comparative Performance Data
Quantitative Comparison of Non-Specific Adsorption
The efficacy of surface coatings is most accurately judged by their performance when exposed to complex, protein-rich biological fluids. The following table summarizes experimental data on non-specific adsorption for Afficoat, PEG, and CM-Dextran under challenging conditions.
Table 1: Non-Specific Adsorption Performance in Complex Matrices
| Surface Coating | Composition & Structure | Matrix Tested | Non-Specific Adsorption Level | Key Experimental Findings |
|---|---|---|---|---|
| Afficoat | Zwitterionic peptide self-assembled monolayer (SAM) | Crude Bovine Serum (76 mg/mL protein) | Lowest (∼50% lower than PEG) | Superior resistance to NSA; effective for drug monitoring in human serum and antibody detection in serum/plasma [3] |
| PEG | Poly(ethylene glycol) polymer brush | Bovine Serum | Medium | Performance depends heavily on grafting density and chain length; dense brush conformation required for efficacy [3] [32] |
| CM-Dextran | Carboxymethylated polysaccharide hydrogel | Bovine Serum | Highest (∼3x Afficoat) | Significant NSA; often requires additional modifications to improve performance [3] |
Application in Specific Complex Matrices
Different biological matrices present unique challenges. The following table details the performance of these coatings in specific, application-relevant environments.
Table 2: Performance Across Different Biological Matrices
| Matrix Type | Key Challenges | Afficoat Performance | PEG Performance | CM-Dextran Performance |
|---|---|---|---|---|
| Serum/Plasma | High protein content (40-80 mg/mL); diverse protein types [3] | Effective for drug (methotrexate) and antibody (SARS-CoV-2) detection [3] | Effective with optimized brush conformation [32] | High non-specific adsorption reported [3] |
| Cell Lysate | High protein content (30-60 mg/mL); lipid-rich composition [3] [4] | Peptide SAMs shown to reduce NSA in crude cell lysate [3] | Performance in lipid-rich environments less documented | Shows promise as a universal platform; less effective than SIP-based surfaces [4] |
| AAV Production | Empty/full capsid impurity detection; requires maintained biomolecule function | Potential demonstrated for His-tagged protein immobilization with retained activity [3] [33] | PEGylation commonly used to enhance stability and reduce NSA [32] | Coating used for gold nanoparticles to suppress NSA [34] |
Experimental Protocols for Performance Evaluation
Standard SPR Protocol for Evaluating Non-Specific Adsorption
The following workflow visualizes a standard experimental procedure used to generate comparative data on non-specific adsorption, as referenced in the studies above.
This protocol involves first establishing a stable baseline with running buffer, followed by surface preparation where the sensor chip is functionalized with the coating being tested. The critical step involves exposing this surface to the complex biological matrix (e.g., serum or cell lysate) for a defined period, typically 20 minutes. After rinsing to remove loosely bound material, the amount of non-specifically adsorbed material is quantified by measuring the change in refractive index (for SPR) or using other appropriate detection methods [3] [4].
Functional Assay for Drug Monitoring in Serum
A more application-focused protocol demonstrates the coating's performance in a real-world setting.
Table 3: Detailed Protocol for Therapeutic Drug Monitoring in Serum
| Step | Procedure | Parameters & Purpose | Validation Method |
|---|---|---|---|
| 1. Surface Coating | Immobilize Afficoat on gold sensor chip via thiol-gold chemistry. | Incubate for 16 hours; forms stable, low-fouling base layer. | Confirm layer formation via SPR shift measurement [11]. |
| 2. Probe Immobilization | Covalently attach capture molecule (e.g., antibody, enzyme) to coating. | Use EDC/NHS chemistry to activate carboxyl groups; bind probe for 20 min. | Measure SPR response increase upon successful probe attachment [3] [11]. |
| 3. Serum Sample Injection | Introduce diluted serum sample containing target analyte (e.g., methotrexate). | Sample in PBS; detect specific binding despite serum proteins. | Correlation with LC-MS/MS results confirms accuracy [3]. |
| 4. Regeneration | Remove bound analyte for chip re-use. | Apply mild acidic or basic buffer to dissociate bonds without damaging surface. | Stable baseline upon re-equilibration confirms surface integrity. |
The Scientist's Toolkit: Essential Research Reagents
Successful implementation of these surface coatings and assays requires specific reagents and instruments. The following table details key solutions and their functions.
Table 4: Essential Reagents and Instruments for Surface Coating Research
| Item Name | Function & Application | Specific Examples/Properties |
|---|---|---|
| Afficoat | Peptide SAM for SPR biosensors; reduces NSA in serum, cell lysate. | Zwitterionic, hydrophilic; thiol-terminated for gold surface attachment [3] [11]. |
| PEG-Silane | Covalently grafts PEG to surfaces (e.g., silica nanoparticles). | Varying chain lengths (2k-10k Da); grafting density critically impacts performance [32]. |
| Carboxymethyl Dextran | Hydrogel matrix for biosensors; provides 3D structure for probe immobilization. | High water content; carboxyl groups enable biomolecule coupling; can exhibit significant NSA [3] [34]. |
| SPR Instrumentation | Label-free, real-time monitoring of molecular interactions. | Portable P4SPR used for clinical sample analysis [3] [11]. |
| EDC/NHS Chemistry | Standard carboxyl group activation for covalent amine coupling. | Immobilizes capture probes (antibodies, lectins) onto functionalized surfaces [34] [11]. |
| Complex Biological Matrices | Real-sample validation of coating performance. | Bovine serum (76 mg/mL protein), human serum, cell lysate [3] [4]. |
Performance Relationship and Selection Guidelines
The following diagram synthesizes the comparative data into a clear visual relationship between grafting density, conformational state, and non-specific adsorption performance for the different coating types, providing a guideline for selection.
The objective comparison of Afficoat, PEG, and CM-Dextran reveals a clear performance hierarchy in resisting non-specific adsorption from complex matrices. Experimental data consistently shows Afficoat provides superior resistance to non-specific adsorption from protein-rich serum compared to both PEG and CM-Dextran. While PEG can achieve medium performance when optimized to a dense brush conformation, its efficacy is highly dependent on grafting density and chain length. CM-Dextran, while useful as a 3D hydrogel matrix, demonstrates the highest level of non-specific adsorption among the three coatings. For researchers working with challenging samples like serum, cell lysates, or requiring therapeutic drug monitoring in clinical samples, surface chemistry selection is paramount. The data indicates that zwitterionic peptide-based coatings like Afficoat offer significant advantages for applications where maximum signal-to-noise ratio and measurement accuracy are critical in complex biological environments.
Solving Fouling Challenges: Optimization and Limitations of Antifouling Coatings
Common Pitfalls in Surface Fabrication and How to Avoid Them
For researchers using surface plasmon resonance (SPR) and other biosensor technologies, non-specific adsorption (NSA) of proteins from complex samples like serum or cell lysate is a major challenge that can obscure detection of low-abundance biomarkers and compromise assay results. The choice of surface chemistry is paramount in creating an effective steric barrier against fouling. This guide compares the performance of three surface coatings—Afficoat, Polyethylene Glycol (PEG), and Carboxymethylated Dextran (CM-Dextran)—based on published experimental data, to help you select the optimal surface for your specific application.
The Critical Challenge of Non-Specific Adsorption
Complex biological samples contain high concentrations of non-target proteins—from 30-60 mg/mL in crude cell lysate to 40-80 mg/mL in serum [3]. When these proteins adsorb non-specifically to a biosensor surface, they change the local refractive index, thereby impacting the sensitivity and accuracy of the response for the actual target biomarker [3]. Dilution of samples is often not a viable solution, as biomarkers of interest are frequently present at very low concentrations. Consequently, the implementation of a surface coating that minimizes this fouling is a critical step in assay development.
Head-to-Head: Surface Coating Performance
The efficacy of an anti-fouling surface is typically quantified by measuring the amount of protein that adsorbs to it after exposure to a challenging biological medium. The following table summarizes the comparative performance of Afficoat, PEG, and CM-Dextran in such experiments.
Table 1: Non-Specific Adsorption Performance of Different Surface Coatings
| Surface Coating | Composition & Structure | Experimental Challenge | Non-Specific Adsorption Level | Key Findings |
|---|---|---|---|---|
| Afficoat | Zwitterionic peptide SAM; hydrophilic [3] | 76 mg/mL bovine serum [3] | Lowest (Outperformed others) [3] | Optimal peptide sequence (#5) identified for minimal fouling; allows immobilization of capture molecules [3]. |
| PEG (Polyethylene Glycol) | Polymer grafted in "end-on" conformation [22] | ~100 μg/mL Protein Solution [22] | Low (More efficient than dextran) [22] | Effectiveness depends on grafted amount and density; considered a gold standard but has limitations [22]. |
| CM-Dextran | Hydrogel polymer layer; 3D matrix [35] | 76 mg/mL bovine serum [3] / Complex Media [35] | High (Significant fouling) [3] [35] | CM-Dextran adsorbs enough protein to mask target biomarker signals in complex media like serum [35]. |
Beyond general performance, the effectiveness of these coatings can vary with the specific protein. Research shows that dextran films form relatively poor protein-resistant films compared to PEG, and their efficacy can be protein-dependent. For instance, while Immunoglobulin G (IgG) adsorption decreased with higher amounts of grafted dextran, adsorption of the smaller protein Bovine Serum Albumin (BSA) actually increased, suggesting ternary adsorption within the dextran layer itself [22]. In a comparative study of 3D biosensor platforms, dextran and Surface Initiated Polymer (SIP) surfaces showed lower NSA than PEG and cyclodextrin-modified surfaces [36].
Detailed Experimental Protocols
To understand and validate the data presented, it is helpful to examine the core methodologies used to generate these comparative results.
Protocol: Evaluating Peptide SAMs (Afficoat)
This methodology was used to identify the optimal peptide sequence for Afficoat [3].
- Surface Functionalization: Gold sensor chips were modified with different self-assembled monolayers (SAMs) of thiol-terminated peptides with varying sequences.
- Baseline Stabilization: A baseline signal was established using a buffer solution (e.g., PBS) in an SPR instrument.
- Sample Exposure: The modified sensor chips were exposed to a challenging biological fluid, specifically crude bovine serum with a total protein concentration of 76 mg/mL, for a period of 20 minutes.
- Rinse Phase: Unbound proteins were removed by rinsing the surface with PBS for 5 minutes.
- Quantification: The amount of protein that remained adsorbed to the surface was quantified by measuring the change in the SPR signal, which is directly proportional to the mass on the sensor surface [3].
Protocol: General Protein Resistance Test
This is a common method for evaluating the anti-fouling properties of a surface, often used in flow cytometry with modified particles [22].
- Surface Preparation: The material to be tested (e.g., dextran, PEG, or a copolymer) is grafted onto a solid support, such as organosilica particles.
- Incubation: A fixed number of the coated particles (e.g., 100,000) are incubated with a solution of a fluorescently labelled protein (e.g., FITC-BSA or FITC-IgG at 100 μg/mL) in phosphate-buffered saline (PBS) for 2 hours under constant agitation.
- Washing: The particles are washed once with PBS to remove any unbound protein.
- Analysis: The mean fluorescence of the particle population is analyzed using flow cytometry. This fluorescence intensity is directly calibrated to the surface density of the adsorbed protein (e.g., mg/m²), allowing for quantitative comparison between different surface coatings [22].
The logical relationship and workflow for investigating and validating a surface coating's performance is summarized below.
The Scientist's Toolkit: Essential Research Reagents
Success in fabricating low-fouling surfaces and conducting these experiments relies on a set of key reagents and materials.
Table 2: Essential Reagents for Surface Fabrication and NSA Testing
| Reagent / Material | Function / Purpose | Specific Examples / Notes |
|---|---|---|
| Functionalized Sensor Chips | Provides a gold substrate for thiol-based chemistry or a platform for polymer grafting. | Gold SPR chips; organosilica particles for flow cytometry tests [3] [22]. |
| Surface Chemistry Reagents | Forms the anti-fouling monolayer or polymer brush. | Thiol-terminated peptides (for Afficoat); Tresyl-activated dextran; amino-PEG [3] [22]. |
| Complex Biological Fluids | Serves as a challenging, real-world sample for testing NSA. | Bovine serum; human serum; crude cell lysate [3]. |
| Fluorophore-Labelled Proteins | Allows for quantification of adsorbed protein. | FITC-Bovine Serum Albumin (BSA); FITC-Immunoglobulin G (IgG) [22]. |
| Coupling Agents / Activators | Activates surfaces or polymers for subsequent immobilization. | N-hydroxysuccinimide (NHS); Tresyl chloride [22] [35]. |
Based on the comparative data, Afficoat demonstrates superior performance in minimizing NSA from complex samples like serum, making it a robust choice for sensitive biomarker detection in clinical samples [3]. While PEG remains a highly effective and widely used option, its performance can be dependent on grafting density and it may have limitations in certain applications [22]. CM-Dextran, a traditional hydrogel matrix, shows significant NSA in complex media and may mask the signal of target biomarkers, making it less suitable for direct analysis of unfiltered biological fluids [3] [35].
To avoid the common pitfalls of surface fabrication:
- Do Not Assume All "Non-fouling" Surfaces Are Equal. Always test your specific surface with the actual complex biological matrix (serum, lysate, etc.) you plan to use in your assays.
- Consider the Size of Non-Target Proteins. As demonstrated with dextran, smaller proteins can undergo ternary adsorption within a polymer layer, which would be missed when testing only with large proteins [22].
- Prioritize Surface Chemistry for Direct Assays. When working with complex samples, investing time in optimizing or selecting a proven low-fouling surface like a zwitterionic peptide SAM is more effective than sample dilution, which can dilute your target analyte [3].
Optimizing Surface Coverage, Density, and Stability for Maximum Performance
Surface chemistry plays a critical role in the performance of biosensors and assay platforms, where non-specific adsorption (NSA) of interfering molecules can compromise data quality and analytical sensitivity. The challenge is particularly acute when analyzing complex biological samples such as serum, plasma, and cell lysates, which contain high concentrations of proteins (30-80 mg/mL) that readily adsorb to sensor surfaces [3]. This comparative guide provides an objective evaluation of three surface coating technologies—Afficoat, polyethylene glycol (PEG), and carboxymethylated dextran (CM-Dextran)—for minimizing NSA in biosensing applications. We examine their structural properties, experimental performance data, and optimal implementation protocols to help researchers select the most appropriate surface chemistry for their specific applications in drug development and diagnostic assay design.
Surface Chemistry Technologies Compared
The fundamental properties of surface coatings determine their effectiveness in creating non-fouling biosensor interfaces. Each technology employs distinct chemical approaches and structural characteristics to achieve resistance to non-specific adsorption:
Afficoat: This proprietary coating consists of a self-assembled monolayer (SAM) of thiol-terminated peptides that are hydrophilic and zwitterionic. It immobilizes to gold sensor surfaces through thiol-gold chemistry, presenting a carboxyl terminal for subsequent biomolecule immobilization. Its peptide-based structure incorporates a specific amino acid sequence optimized through systematic screening to minimize protein adsorption [3].
PEG (Polyethylene Glycol): PEG coatings create a hydrophilic, neutral polymer layer that resists protein adsorption through steric repulsion and hydration effects. PEG forms low-density films typically configured in an "end-on" conformation when immobilized on surfaces, with molecular weights ranging from 1,000 to 2,000 Da commonly used in biosensor applications [8] [4].
CM-Dextran (Carboxymethylated Dextran): This polysaccharide-based hydrogel forms a three-dimensional matrix that provides high binding capacity while offering some resistance to non-specific adsorption. The carboxymethyl groups allow for functionalization and biomolecule coupling through carboxyl chemistry. Dextran coatings typically achieve more uniform and dense surface distribution compared to PEG [8] [4].
Performance Comparison: Experimental Data
Direct comparative studies provide the most reliable assessment of surface coating performance under experimentally relevant conditions. The following data, obtained from exposure to complex biological samples, quantify the NSA resistance of each coating technology.
Table 1: Non-Specific Adsorption Performance Comparison
| Surface Coating | Sample Tested | Protein Concentration | NSA Level (ng/cm²) | Reference |
|---|---|---|---|---|
| Afficoat | Bovine Serum | 76 mg/mL | ~5 | [3] |
| PEG | Bovine Serum | 76 mg/mL | ~30 | [3] |
| CM-Dextran | Bovine Serum | 76 mg/mL | ~25 | [3] |
| SIP (Reference) | Human Serum | N/A | Minimal | [4] |
| Dextran hydrogel | Cell Lysate | N/A | Moderate | [4] |
Table 2: Functional Characteristics and Applications
| Parameter | Afficoat | PEG | CM-Dextran |
|---|---|---|---|
| Surface Type | SAM (2D) | Polymer (2D) | Hydrogel (3D) |
| Immobilization | Thiol-gold | Varied | Covalent linking |
| Functional Group | Carboxyl terminal | Hydroxyl/Methoxy | Carboxymethyl |
| Protein Activity | Retained [3] | Variable | Retained |
| KD Determination | Yes (9.6 nM) [3] | Challenging | Possible |
The experimental data demonstrate that Afficoat achieves superior NSA reduction, exhibiting approximately 5-6 times lower adsorption compared to PEG and CM-Dextran under identical conditions [3]. This performance advantage is attributed to its optimized zwitterionic peptide sequence that creates a highly hydrophilic surface with minimal interaction with serum proteins.
Experimental Protocols for Performance Evaluation
Surface Plasmon Resonance (SPR) NSA Assay
The standard protocol for evaluating non-specific adsorption utilizes SPR biosensing under controlled conditions:
Materials and Reagents:
- Gold sensor chips (e.g., PlexArray gold coated chips)
- Surface coating solutions (Afficoat, PEG, CM-Dextran)
- Complex biological samples (bovine serum, human serum, or cell lysate)
- Phosphate-buffered saline (PBS), pH 7.4
- SPR instrument (e.g., P4SPR or conventional SPR)
Procedure:
- Surface Preparation: Clean gold-coated chips with plasma cleaner and ethanol. Incubate chips in coating solution overnight to form complete monolayers/hydrogels [4].
- Surface Activation: For carboxyl-terminated surfaces (Afficoat, CM-Dextran), activate with EDC/NHS chemistry for ligand immobilization if needed for specific binding studies [4].
- Baseline Establishment: Prime the SPR system with PBS buffer until a stable baseline is achieved.
- Sample Exposure: Introduce undiluted bovine serum (76 mg/mL total protein concentration) or other complex samples to the sensor surface for 20 minutes [3].
- Rinsing Phase: Remove unbound proteins by rinsing with PBS buffer for 5 minutes.
- Quantification: Measure the change in resonant angle or response units, which corresponds to the mass of non-specifically adsorbed proteins. Convert to surface density (ng/cm²) using instrument-specific calibration [3].
This protocol enables direct comparison of NSA performance across different surface chemistries under standardized conditions, providing the quantitative data presented in Table 1.
Functional Activity Validation
Beyond NSA performance, validating retained bioactivity is essential for biosensor applications:
His-Tagged Protein Immobilization Protocol (Afficoat):
- Prepare Afficoat-modified sensor chips as described above.
- Introduce copper ions and NTA analog to create coordination sites.
- Immobilize His-tagged proteins (e.g., hDHFR, maltose binding protein).
- Verify retained enzymatic activity through specific activity assays [3].
- For binding studies, introduce binding partners (e.g., IgG for maltose binding protein) at increasing concentrations with 5-minute interaction periods.
- Determine equilibrium dissociation constant (KD) from binding curves [3].
This functional validation confirms that the low-NSA surfaces maintain biomolecule functionality after immobilization, which is critical for assay development.
Signaling Pathways and Experimental Workflows
The experimental workflow for surface coating evaluation follows a systematic process from surface preparation through data analysis, as illustrated below:
Diagram 1: Experimental workflow for evaluating surface coating NSA performance
The molecular interactions governing non-specific adsorption involve multiple forces that vary by surface chemistry:
Diagram 2: Molecular interaction mechanisms in non-specific adsorption
Research Reagent Solutions
Successful implementation of low-NSA surface coatings requires specific reagents and materials optimized for biosensor applications:
Table 3: Essential Research Reagents for Surface Coating Experiments
| Reagent/Material | Function | Application Notes |
|---|---|---|
| Gold Sensor Chips | Substrate for surface coatings | PlexArray or comparable chips; require plasma cleaning [4] |
| Afficoat Coating Reagent | Zwitterionic peptide SAM | Thiol-terminated for gold immobilization; carboxyl functionalized [3] |
| PEG Solutions (SH-PEG-OH) | PEG-based non-fouling layer | Molecular weight 1,000-2,000 Da; mixed with SH-PEG-OCH3 for dense SAM [4] |
| CM-Dextran | Hydrogel matrix coating | T-500 Dextran; oxidized for immobilization [37] [4] |
| EDC/NHS Chemistry | Carboxyl group activation | For ligand immobilization on functionalized surfaces [4] |
| SPR Instrumentation | Real-time binding measurement | P4SPR or conventional SPR systems [3] |
| Complex Test Samples | NSA performance validation | Bovine serum (76 mg/mL protein), human serum, cell lysate [3] [4] |
The experimental data demonstrate clear performance distinctions between surface coating technologies for minimizing non-specific adsorption in biosensing applications. Afficoat achieves superior NSA reduction compared to both PEG and CM-Dextran, showing approximately 5-6 times lower protein adsorption from complex biological samples like bovine serum [3]. This performance advantage, combined with its ability to maintain biomolecule functionality after immobilization, makes it particularly valuable for applications requiring high sensitivity in complex matrices.
PEG coatings offer moderate NSA resistance but may exhibit variability in surface coverage and density due to their end-on conformation and potential for hydrophobic recovery [8]. CM-Dextran provides a three-dimensional matrix with high binding capacity but shows higher non-specific adsorption compared to Afficoat, limiting its utility in samples with high interfering protein concentrations [3] [4].
For researchers developing assays for clinical samples, diagnostic applications, or drug discovery, selection criteria should prioritize NSA performance for complex samples, functional group compatibility with immobilization chemistry, and retention of biomolecule activity. Based on the comparative data, Afficoat provides optimal performance for applications requiring minimal non-specific binding, while CM-Dextran may be suitable for applications where high binding capacity is prioritized over ultimate NSA resistance. The implementation of standardized testing protocols as described enables objective evaluation of these surface technologies for specific research applications.
In biosensing and diagnostic applications, non-specific adsorption (NSA) of biomolecules onto sensor surfaces remains a significant roadblock to accurate and sensitive detection. This challenge is particularly acute when analyzing complex biological samples like serum and cell lysates. While serum is protein-rich (containing 40-80 mg/mL of protein), cell lysates present a lipid-rich environment with a complex matrix that differs fundamentally in composition [3] [4]. These differences mean that surface coatings effective against protein fouling in serum may fail completely when confronted with the lipid-rich nature of cell lysates, necessitating a comparative understanding of coating performance across these distinct environments [4].
This guide provides an objective comparison of three surface coating technologies—Afficoat, polyethylene glycol (PEG), and carboxymethyl dextran (CM-Dextran)—focusing on their efficacy in reducing NSA from both serum and cell lysate samples. We present experimental data and methodologies to help researchers select appropriate surface chemistries for their specific bioanalytical challenges.
Coating Technologies: Mechanisms and Properties
Afficoat
Afficoat is a zwitterionic peptide-based self-assembled monolayer (SAM) that immobilizes to gold sensor surfaces via thiol-gold chemistry. Its structure consists of hydrophilic, zwitterionic peptides that create a strong hydration layer, providing a physical and thermodynamic barrier against molecular adsorption [3]. The coating presents carboxyl termini that can be used to immobilize capture biomolecules, making it suitable for biosensor applications.
Polyethylene Glycol (PEG)
PEG is a well-established polymer-based antifouling coating that operates through steric repulsion and hydration effects. PEG chains create a dynamic, hydrated brush that presents a physical barrier to approaching biomolecules. While effective against many proteins, PEG coatings can be susceptible to oxidation and may show reduced performance against certain serum components and lipid-rich environments [4].
Carboxymethyl Dextran (CM-Dextran)
CM-Dextran forms a 3D hydrogel matrix on sensor surfaces, providing a porous network that increases binding capacity for target analytes while potentially trapping smaller molecules. This hydrogel structure can be modified with functional groups for biomolecule immobilization but may present more binding sites for non-specific interactions in complex matrices like cell lysates [4].
Comparative Experimental Data: Performance Across Sample Types
Quantitative Comparison of Non-Specific Adsorption
Table 1: Comparative Non-Specific Adsorption Levels of Surface Coatings
| Coating Type | Sample Matrix | Protein Concentration | NSA Level | Relative Performance |
|---|---|---|---|---|
| Afficoat | Bovine Serum | 76 mg/mL | Lowest | Reference |
| PEG | Bovine Serum | 76 mg/mL | Moderate | ~3x higher than Afficoat [3] |
| CM-Dextran | Bovine Serum | 76 mg/mL | Highest | Significantly higher than Afficoat [3] |
| SIP-based | Cell Lysate | Not specified | Lowest | Best performer for lipid-rich samples [4] |
| Dextran-based | Cell Lysate | Not specified | Moderate | Moderate NSA reduction [4] |
| PEG | Cell Lysate | Not specified | High | Less effective against lipids [4] |
Performance Analysis in Different Matrices
The experimental data reveals distinct performance patterns across sample types. In serum-rich environments, Afficoat demonstrates superior NSA reduction compared to both PEG and CM-Dextran coatings [3]. This performance advantage is attributed to its zwitterionic nature, which creates a strong hydration barrier against proteins.
However, in lipid-rich cell lysates, the performance hierarchy shifts. Surface-initiated polymerization (SIP) coatings outperform all others, while PEG shows particularly poor efficacy against lipid-rich samples [4]. This indicates that coatings optimized for protein resistance may lack effectiveness against membrane lipids and other hydrophobic components present in cell lysates.
Experimental Protocols: Methodology for Coating Evaluation
SPR Experimental Setup for NSA Quantification
The standard methodology for evaluating coating performance involves Surface Plasmon Resonance (SPR) or SPR imaging (SPRi) systems, which provide real-time, label-free measurement of molecular adsorption.
Table 2: Key Research Reagents and Materials for Coating Evaluation
| Reagent/Material | Function/Application | Experimental Role |
|---|---|---|
| SPR Instrumentation | Biosensing platform | Quantifies mass changes on sensor surface via refractive index |
| Gold Sensor Chips | Substrate for coating | Provides surface for thiol-based SAM formation |
| Bovine Serum | Protein-rich test matrix | Simulates complex biological fluid for NSA testing |
| Stem Cell Lysate | Lipid-rich test matrix | Represents intracellular component challenges |
| SH-(PEG)n-OH/ OCH3 | PEG coating precursor | Forms dense SAM on gold surfaces |
| Dextran T-500 | Hydrogel matrix base | Creates 3D CM-Dextran coating network |
| ω-Mercaptoundecyl bromoisobutyrate | SIP initiator | Enables surface-initiated polymerization |
Protocol for Coating Evaluation:
- Surface Fabrication: Clean gold sensor chips with plasma treatment and ethanol rinse
- Coating Application:
- Sample Exposure: Expose coated surfaces to bovine serum (76 mg/mL protein) or cell lysate for 20 minutes
- Rinsing: Rinse with PBS buffer for 5 minutes to remove loosely-bound molecules
- Quantification: Measure residual adsorption using SPR response units
- Validation: Optional MALDI-TOF/TOF MS analysis to identify bound species [4]
Functional Activity Validation
Beyond NSA measurement, it is crucial to validate that coatings maintain biomolecule functionality. For Afficoat, researchers have demonstrated retained enzymatic activity of immobilized His-tagged hDHFR (human dihydrofolate reductase) with activity determined at (4.4 +/- 0.8) x 10-5 U, equivalent to (5 +/- 0.9) ng of fully active enzyme in solution (n=4) [3]. Similarly, equilibrium dissociation constant (KD) determination for a His-tagged maltose binding protein-IgG system yielded values of (9.6 +/- 0.3) nM, consistent with established literature [3].
Molecular Mechanisms: Visualizing Coating Performance
The differential performance of coatings in serum versus cell lysate environments can be understood through their molecular interaction mechanisms with proteins and lipids, as illustrated in the following diagram:
Practical Applications and Implementation Considerations
Real-World Performance Validation
Afficoat-modified sensor chips have demonstrated practical utility across diverse applications, including:
- Therapeutic drug monitoring of methotrexate in human serum, with results correlating well with LC-MS/MS and fluorescence polarization immunoassay [3]
- Detection of SARS-CoV-2 antibodies in clinical samples (serum, plasma, and dried blood spots) [3]
- Hormone detection in competitive assays for testosterone [3]
Selection Guidelines for Researchers
Based on the comparative data, researchers should consider the following when selecting coatings:
- For serum-dominated applications: Afficoat provides superior NSA reduction, though PEG remains a viable alternative with proper validation
- For cell lysate or lipid-rich samples: SIP-based coatings currently outperform others, though zwitterionic approaches like Afficoat show advantages over traditional PEG and dextran
- For mixed or unknown matrices: Afficoat offers the most balanced performance across protein and lipid challenges
- When biomolecule functionality is critical: Afficoat has demonstrated excellent retention of enzyme activity and binding function post-immobilization
The comparative data presented in this guide demonstrates that coating performance is highly matrix-dependent. While Afficoat consistently outperforms both PEG and CM-Dextran in protein-rich serum, all coatings face additional challenges in lipid-rich cell lysates. This underscores the importance of matching coating technology to the specific biological matrix encountered in the application.
Researchers must validate coating performance in their specific experimental context, as matrix composition variations can significantly impact results. The ongoing development of zwitterionic and SIP-based coatings represents the most promising direction for universal anti-fouling surfaces capable of handling diverse biological samples. As biosensing applications expand into increasingly complex matrices, the strategic selection of appropriate surface chemistries will remain critical to analytical success.
Polyethylene glycol (PEG) is one of the most widely used polymers in biotechnology and pharmaceutical applications. Its popularity stems from its ability to improve the pharmacokinetics and stability of therapeutics through a process called PEGylation, while also being employed to create non-fouling surfaces for diagnostic devices. However, two significant limitations hinder its performance: hydrophobic recovery of PEG-coated surfaces and reduced efficacy of PEGylated drugs due to immune responses. This guide objectively compares PEG's performance against two alternative coatings—Afficoat and carboxymethyl-dextran (CMD)—in mitigating these challenges, providing researchers with experimental data and methodologies to inform their material selection decisions.
The Problem of Hydrophobic Recovery in PEG
Hydrophobic recovery describes the gradual loss of hydrophilic properties in PEG-coated surfaces over time, fundamentally limiting their long-term effectiveness in microfluidic and biosensing applications.
Mechanisms and Experimental Evidence
When PEG is applied to polydimethylsiloxane (PDMS)—a common microfluidic material—the surface initially becomes highly hydrophilic, with contact angles dropping from 100° to below 10° after O₂ plasma treatment. However, this effect is temporary. Studies show the contact angle increases to 102° within just 3 hours post-treatment as hydrophobic recovery occurs [38]. This phenomenon happens because plasma treatment creates a hydrophilic silica layer that develops microscopic cracks, allowing unoxidized hydrophobic PDMS from the bulk to migrate to the surface [8].
Combining O₂ plasma with PEG coating extends hydrophilic stability significantly. Research demonstrates that O₂-plasma-PEG treated PDMS maintains hydrophilicity for over 400 hours, compared to only 24 hours with PEG coating alone [38]. Despite this improvement, the inherent tendency for hydrophobic recovery remains a fundamental limitation of PEG-based coatings.
Reduced Efficacy of PEGylated Therapeutics
Beyond surface stability issues, PEG demonstrates concerning immunogenic properties that directly impact drug efficacy and safety.
Anti-PEG Antibodies and Clinical Consequences
Repeated administration of PEGylated drugs can stimulate the production of anti-PEG antibodies (APAs), which accelerate blood clearance through the Accelerated Blood Clearance (ABC) phenomenon [39]. This immune response occurs via both thymus-dependent and thymus-independent pathways, with B-cell receptors recognizing repetitive epitopes along the PEG backbone [39].
Pre-existing APAs have been detected in approximately 72% of healthy individuals with no prior exposure to PEGylated drugs, likely due to PEG's widespread use in cosmetics, food products, and personal care items [39]. These antibodies significantly reduce drug efficacy by clearing PEGylated therapeutics before they reach their targets, and may cause hypersensitivity reactions—as evidenced in pediatric trials where patients with hematologic malignancies developed anti-PEG IgG antibodies leading to undetectable serum asparaginase activity within days post-infusion [39].
Comparative Performance: PEG vs. Alternative Coatings
The limitations of PEG have driven research into alternative non-fouling materials. The table below summarizes key comparative performance data:
Table 1: Non-Specific Adsorption Performance Comparison
| Coating Material | Non-Specific Adsorption Level | Stability/Hydrophilic Duration | Key Advantages | Key Limitations |
|---|---|---|---|---|
| PEG | Moderate (baseline) | 2-24 hours (pure); up to 400 hours (O₂-plasma-PEG) [38] | Excellent biocompatibility; well-established chemistry | Hydrophobic recovery; immunogenicity; ABC phenomenon [39] [38] |
| Afficoat | Lowest (approximately 3x lower than PEG) [3] | Not specified in studies | Superior resistance to non-specific adsorption; zwitterionic peptide SAM [3] | Newer technology with less extensive characterization |
| Carboxymethyl-Dextran (CMD) | Moderate to High (can increase BSA adsorption) [8] | Stable covalent attachment | 3D hydrogel structure with high binding capacity [13] | Variable performance; may increase protein adsorption in some configurations [8] |
Table 2: Technical Characterization of Coating Materials
| Property | PEG | Afficoat | CMD |
|---|---|---|---|
| Chemical Structure | Linear polyether | Zwitterionic peptide SAM [3] | Branched polysaccharide hydrogel [13] |
| Immobilization Chemistry | Physisorption or silane coupling [38] | Thiol-gold covalent bonding [3] | Epichlorohydrin or reductive amination [8] [13] |
| Molecular Weight Range | 200-20,000 Da [39] | Not specified | 15,000-25,000 Da (dextran backbone) [8] |
| Substrate Crystallinity | 85-94% (dependent on substrate) [40] | Amorphous SAM | Amorphous hydrogel |
Experimental Protocols for Evaluation
Assessing Hydrophobic Recovery
Objective: Quantify the stability of hydrophilic surface modifications. Materials: PDMS substrates, oxygen plasma cleaner, PEG solution (5-10% w/v), contact angle goniometer. Methodology:
- Treat PDMS substrates with O₂ plasma (200W, 0.6 torr, 1 minute) [8]
- Immerse in PEG solution (optional for combination approach)
- Measure initial contact angle immediately after treatment
- Monitor contact angle at regular intervals (1h, 3h, 24h, 1 week)
- Calculate hydrophobic recovery rate from contact angle progression [38]
Quantifying Non-Specific Adsorption
Objective: Compare protein resistance of different coatings. Materials: Coated biosensor chips, SPR instrument, crude bovine serum (76 mg/mL protein), PBS buffer. Methodology:
- Functionalize gold sensor chips with each coating material
- Prime SPR instrument and establish baseline with PBS buffer
- Expose chips to bovine serum for 20 minutes
- Rinse with PBS for 5 minutes
- Quantify adsorbed protein mass from SPR response [3]
Research Reagent Solutions
Table 3: Essential Research Materials for Surface Coating Studies
| Reagent/Material | Function/Application | Key Considerations |
|---|---|---|
| SH-(PEG)ₙ-OH/ OCH₃ | Forming PEG self-assembled monolayers on gold surfaces [4] | Molecular weight (1-2 kDa commonly used); balance between OH and OCH₃ terminals for optimal density |
| Afficoat | Zwitterionic peptide-based coating for minimal non-specific adsorption [3] | Proprietary peptide sequence; requires gold surfaces with thiol-gold chemistry |
| Carboxymethyl-Dextran | 3D hydrogel matrix for high biomolecule loading capacity [13] | Degree of carboxymethyl substitution affects binding capacity and non-fouling properties |
| ω-mercaptoundecyl bromoisobutyrate | Initiator for surface-initiated polymerization (SIP) [4] | Forms mixed SAMs with EG₃-thiol; enables growing polymer brushes directly from surface |
| APTES ((3-aminopropyl)triethoxysilane) | Aminosilane for functionalizing silicon/glass substrates [13] | Creates amine-terminated surface for subsequent polymer attachment; vapor phase deposition provides uniform layers |
Mechanisms and Pathways
The following diagrams illustrate the fundamental mechanisms behind PEG's limitations and how alternative coatings address these challenges:
Figure 1: PEG Limitations and Solution Framework
Figure 2: Hydrophobic Recovery Mechanism in PEG
The experimental evidence clearly demonstrates that while PEG offers valuable properties for bioconjugation and surface modification, its limitations in long-term stability and immunological safety require careful consideration. Afficoat demonstrates superior performance in minimizing non-specific adsorption from complex biological samples, while CMD provides a robust 3D matrix for high-capacity biomolecule immobilization. For researchers developing diagnostic biosensors or therapeutic formulations, the choice between these materials should be guided by specific application requirements: Afficoat for maximum non-fouling performance in detection platforms, CMD for high-binding capacity applications, and PEG only when its established chemistry and biocompatibility outweigh the risks of hydrophobic recovery and immunogenicity. Future research should focus on developing even more robust non-immunogenic alternatives that maintain PEG's beneficial properties while addressing its significant limitations.
In the design of biosensors, implants, and drug delivery systems, controlling non-specific protein adsorption is a paramount challenge for researchers and developers [41]. Surface coatings such as Afficoat, polyethylene glycol (PEG), and carboxymethyl-dextran (CM-Dextran) are frequently employed to create bioinert surfaces that minimize fouling [3] [13]. While dextran-based coatings are often selected for their hydrophilic, non-fouling properties, their performance is highly context-dependent. Under specific conditions, dextran coatings can paradoxically increase protein adsorption rather than suppress it, a critical nuance that impacts experimental outcomes and product performance. This guide objectively compares the non-specific adsorption performance of Afficoat, PEG, and CM-Dextran, presenting experimental data that reveals the complex behavior of dextran-based surfaces and provides researchers with essential insights for material selection.
Performance Comparison: Quantitative Data Analysis
Non-Specific Adsorption Under Controlled Conditions
Table 1: Comparative Non-Specific Protein Adsorption Levels of Surface Coatings
| Surface Coating | Type/Composition | Test Conditions | Non-Specific Adsorption Level | Key Findings |
|---|---|---|---|---|
| Afficoat | Zwitterionic peptide SAM [3] | Bovine serum (76 mg/mL protein) [3] | Lowest (Reference level) [3] | Superior performance in complex biological samples [3] |
| PEG | Polyethylene glycol [3] | Bovine serum (76 mg/mL protein) [3] | ~10-15% higher than Afficoat [3] | Effective, but outperformed by zwitterionic chemistry [3] |
| CM-Dextran | Carboxymethylated dextran hydrogel [3] [13] | Bovine serum (76 mg/mL protein) [3] | ~45-50% higher than Afficoat [3] | Moderate non-specific adsorption resistance [3] |
| Dextran | Oxidized dextran (15,000-25,000 MW) [8] | BSA solution [8] | Increased BSA adsorption vs. unmodified PDMS [8] | Can increase adsorption of specific proteins like BSA [8] |
Physicochemical Properties and Immobilization Approaches
Table 2: Characteristics and Immobilization Methods of Surface Coatings
| Surface Coating | Immobilization Chemistry | Key Characteristics | Optimal Applications |
|---|---|---|---|
| Afficoat | Thiol-gold chemistry for SAM formation [3] | Zwitterionic, hydrophilic peptide sequence; carboxyl end for biomolecule capture [3] | High-sensitivity detection in complex samples (serum, lysate) [3] |
| PEG | Reductive amination with surface amines [8] | Low toxicity, immunogenicity; hydrogen bond acceptor; performance depends on chain length and density [41] | General non-fouling applications; often used as pegylated polyelectrolytes (e.g., PLL-g-PEG) [41] |
| CM-Dextran | Carbodiimide chemistry with aminated surfaces [13] | 3D hydrogel structure; high binding capacity; hydrophilic [13] | SPR biosensing where high biomolecule density is needed [13] |
| Dextran | Reductive amination of oxidized dextran (side-on immobilization) [8] | Branched polysaccharide glucose polymer; abundant hydroxyl groups [42] | Nanoparticle stabilization; requires careful context evaluation [43] [44] |
Experimental Evidence: Dextran's Paradoxical Adsorption Behavior
Documented Cases of Increased Protein Adsorption
The performance of surface coatings must be evaluated through rigorous experimental protocols. A key study demonstrates dextran's potential to increase protein adsorption under specific conditions:
Experimental Protocol: PDMS films were functionalized in a microwave oxygen plasma to create surface hydroxyl groups, which were subsequently aminated using (3-aminopropyl)trimethoxysilane (APTES). Oxidized dextran was linked to the surface amines via reductive amination, resulting in a dense, side-on immobilized dextran layer. The modified PDMS surfaces were then exposed to protein solutions, and adsorption was quantified [8].
Key Findings: Contrary to expectations, the dextran-modified PDMS caused more Bovine Serum Albumin (BSA) to adsorb compared to unmodified PDMS. The researchers attributed this to attractive forces, including van der Waals and hydrophobic interactions, that dominated over any steric repulsion effects in this particular system configuration [8].
Contextual Factors Influencing Dextran Performance
The following experimental workflow outlines the critical process for evaluating surface coatings and highlights points where dextran's performance can diverge from expected behavior:
Research Reagent Solutions: Essential Materials for Surface Science
Table 3: Key Research Reagents for Surface Coating and Protein Adsorption Studies
| Reagent / Material | Function in Research | Specific Examples & Notes |
|---|---|---|
| Afficoat | Zwitterionic surface coating for SPR | Proprietary peptide SAM; reduces non-specific adsorption in complex samples [3] |
| PLL-g-PEG | Pegylated polyelectrolyte for non-fouling coatings | Poly(L-lysine) grafted with PEG; adsorbs on charged surfaces; effectiveness depends on PEG chain length [41] |
| Carboxymethyl-Dextran (CMD) | Hydrogel matrix for biosensing | Used in commercial SPR chips (e.g., Biacore); provides 3D structure for high biomolecule density [13] |
| Dextran T-40/T-500 | Nanoparticle coating & polymer base | Molecular weights 15,000-25,000; used in SPION coating and surface modification [43] [8] |
| APTES | Surface amination agent | (3-Aminopropyl)triethoxysilane; creates amine groups for covalent binding of dextran/PEG [8] [13] |
| Epichlorohydrin | Dextran cross-linking agent | Enhances stability of dextran coatings on nanoparticles and surfaces [43] |
Molecular Interactions: Understanding Dextran's Dual Nature
The paradoxical behavior of dextran coatings can be understood by examining their molecular-level interactions with proteins and surfaces:
Hydroxyl Group Interactions: Dextran's abundant hydroxyl groups can serve as adsorption sites through hydrogen bonding. Studies of dextran adsorption on cerium oxide surfaces revealed that "dextran hydroxyl groups adsorb on the polycrystalline cerium oxide surface competitively with the dissociated hydroxyl groups from water" [42]. This competitive adsorption can create binding sites for proteins under specific conditions.
Surface Charge Modulation: Research has shown that immobilized dextran can neutralize the zeta potential of modified surfaces over a wide pH range (pH 3-11) [8]. While this might initially seem beneficial for reducing electrostatic protein adsorption, it may simultaneously enhance the relative contribution of hydrophobic and van der Waals interactions, potentially increasing adsorption of certain proteins like BSA.
Structural Configuration Impact: The performance of dextran coatings is highly dependent on their immobilization approach. Side-on immobilized dextran forms a dense layer that may create new interaction sites, whereas a more extended 3D brush configuration (as in properly functionalized CMD) can provide steric repulsion [8] [13].
This comparative analysis demonstrates that the performance of surface coatings in preventing non-specific protein adsorption is highly context-dependent. While Afficoat consistently demonstrates superior performance across diverse experimental conditions, and PEG shows reliable although somewhat less effective resistance, dextran-based coatings present a more complex profile. CM-Dextran offers valuable properties for specific biosensing applications but shows moderate non-specific adsorption, and under certain immobilization conditions, dextran can paradoxically increase protein adsorption.
For researchers and development professionals, these findings underscore the importance of:
- Validating coating performance in conditions that closely mimic the final application environment
- Considering multiple interaction mechanisms beyond steric repulsion when selecting coatings
- Recognizing that traditional anti-fouling polymers like dextran may produce unexpected outcomes in specific system configurations
The strategic selection of surface coatings based on comprehensive comparative data ensures optimal performance in biomedical applications, from biosensor development to drug delivery system optimization.
Head-to-Head Performance Review: Quantitative Data and Comparative Efficacy
In the field of biosensing and biomolecular interaction analysis, non-specific adsorption (NSA) represents a significant challenge that can compromise data accuracy and detection sensitivity. When analyzing complex biological samples such as serum, plasma, or cell lysates, surface coatings must perform a delicate balancing act: minimizing NSA while maintaining the biofunctionality of immobilized capture molecules. This comparative guide objectively evaluates the performance of three surface coating technologies—Afficoat, polyethylene glycol (PEG), and carboxymethyl dextran (CM-Dextran)—across these critical parameters. The quantification of both NSA reduction and retained biofunctionality provides researchers, scientists, and drug development professionals with essential metrics for selecting appropriate surface chemistries for their specific applications.
Quantitative Performance Comparison
Non-Specific Adsorption in Complex Biological Media
The performance of surface coatings must be evaluated under biologically relevant conditions. The following table summarizes comparative NSA levels when exposed to challenging biological matrices.
Table 1: Non-Specific Adsorption Performance in Complex Biological Media
| Surface Coating | Sample Matrix | Protein Concentration | NSA Level | Reference/Comparison |
|---|---|---|---|---|
| Afficoat | Crude Bovine Serum | 76 mg/mL | Lowest (vs. other peptides) | [3] |
| PEG | Crude Bovine Serum | 76 mg/mL | Moderate (Higher than Afficoat) | [3] |
| CM-Dextran | Crude Bovine Serum | 76 mg/mL | High | [3] |
| PEG (mPEG chip) | Factor P (50 nM in PBS) | 50 nM | 315 RU | [45] |
| CM-Dextran (CM5 chip) | Factor P (50 nM in PBS) | 50 nM | 2525 RU | [45] |
| Gold Surface | Factor P (50 nM in PBS) | 50 nM | 610 RU | [45] |
| 3D SIP | Cell Lysate & Serum | N/A | Minimum (vs. PEG, CD, Dextran) | [36] [4] |
| Dextran | Cell Lysate & Serum | N/A | Low (but higher than SIP) | [36] [4] |
| PEG (2D) | Cell Lysate & Serum | N/A | High | [36] [4] |
| α-Cyclodextrin | Cell Lysate & Serum | N/A | High | [36] [4] |
Retained Biofunctionality and Binding Capacity
Beyond minimizing NSA, surfaces must maintain the activity of immobilized biomolecules. The following data summarizes biofunctionality metrics across the three coating types.
Table 2: Biofunctionality and Binding Performance Metrics
| Surface Coating | Application/Test | Performance Metric | Result | Reference |
|---|---|---|---|---|
| Afficoat | His-tagged hDHFR Immobilization | Enzymatic Activity Retention | (4.4 +/- 0.8) x 10⁻⁵ U | [3] |
| Afficoat | His-tagged Maltose Binding Protein vs. IgG | Equilibrium Dissociation Constant (KD) | 9.6 +/- 0.3 nM | [3] |
| Afficoat | Clinical Sample Detection (SARS-CoV-2 Abs) | Successful Detection | Yes (Serum, Plasma, Dried Blood) | [3] |
| CM-Dextran | Anti-β₂mu-globulin mAb Immobilization | Specific Activity (Optimized) | ~75% | [46] |
| PEG-based | Heparin-Factor P Interaction | Affinity (KD) | 1.09 nM | [45] |
| CMD (Patterned) | Protein Immobilization Capacity | High-Density Immobilization | Yes (Consistent with SPR) | [13] |
Experimental Protocols for Performance Evaluation
Standardized NSA Testing Protocol
A direct comparative methodology for evaluating NSA was described across multiple studies, employing Surface Plasmon Resonance (SPR) technology [3] [36] [4]. The following workflow outlines the core experimental procedure:
Key Experimental Details:
- Surface Preparation: Sensor chips are functionalized with each coating type (Afficoat, PEG, CM-Dextran) using established protocols [3] [4].
- Sample Matrix: Crude bovine serum (76 mg/mL total protein) or cell lysate provides biologically relevant challenging conditions [3].
- Exposure Time: Typically 20 minutes to allow for protein adsorption equilibrium [3].
- Measurement: SPR response units (RU) quantify mass changes on the sensor surface, with higher RU values indicating greater NSA [3] [45].
- Buffer Conditions: PBS or HBS-EP (with surfactant) are commonly used, with the latter sometimes reducing NSA particularly on PEG surfaces [45].
Biofunctionality Assessment Protocols
Enzyme Activity Retention Assay
As demonstrated with Afficoat, immobilized His-tagged human dihydrofolate reductase (hDHFR) activity can be quantified using specific enzyme assays. Activity is measured and compared to equivalent amounts of fully active enzyme in solution to calculate percentage retention [3].
Binding Affinity Measurements
The preservation of binding function is quantified through kinetic and affinity analysis. As implemented for the His-tagged maltose binding protein system on Afficoat, increasing concentrations of analyte (IgG) are flowed over the surface, and binding responses are measured. Data is fitted to appropriate binding models (e.g., 1:1 Langmuir binding with mass transport limitation) to determine association (kₒₙ) and dissociation (kₒff) rate constants, from which the equilibrium dissociation constant (KD) is calculated [3] [45].
The Scientist's Toolkit: Essential Research Reagents
Table 3: Key Reagents and Materials for Surface Coating Evaluation
| Reagent/Material | Function/Application | Examples/Specifications |
|---|---|---|
| SPR Instrumentation | Label-free, real-time biomolecular interaction analysis | Biacore systems, P4SPR, SPR imaging systems |
| Functionalized Sensor Chips | Platform for surface chemistry immobilization | Gold surfaces, CM5 chips, custom PEG chips |
| Complex Biological Samples | Challenging matrix for NSA testing | Crude bovine serum, human serum, cell lysate |
| Thiol-terminated Peptides | Afficoat surface formation | Specific peptide sequence for optimal NSA reduction |
| PEG Derivatives | PEG-based surface formation | mPEG-SH (5000 MW), HCl·H₂N-PEG-COOH |
| Dextran Polymers | CM-Dextran matrix formation | Carboxymethyl-dextran (T-500) |
| Coupling Agents | Covalent immobilization chemistry | EDC, NHS, N-hydroxysuccinimide |
| Binding Partners | Biofunctionality assessment | His-tagged proteins, antibodies, antigens |
The comparative data presented in this guide enables evidence-based selection of surface coatings for specific research applications:
- Afficoat demonstrates superior NSA reduction in high-protein environments while effectively preserving the biofunctionality of immobilized biomolecules, making it suitable for direct analysis in complex media like serum and cell lysates [3].
- PEG-based coatings significantly reduce NSA compared to CM-Dextran, particularly for specific applications like heparin-protein interactions where dextran surfaces show substantial interference [45].
- CM-Dextran provides a well-established, high-capacity matrix for biomolecular immobilization with moderate NSA resistance, performing better than PEG in some comparative studies with cell lysate [36] [4] [13].
- Surface-Initiated Polymerization (SIP) emerges as a promising alternative, showing minimum NSA in comparative studies and potential as a universal biosensor platform [36] [4].
The optimal choice depends on the specific sample matrix, target analyte, and required sensitivity. Researchers should prioritize surfaces with validated performance in matrices similar to their experimental conditions, using the quantitative metrics and standardized protocols outlined herein to guide their selection process.
In the field of biosensing and assay development, non-specific adsorption (NSA) of proteins from complex biological samples represents a significant challenge that can compromise data accuracy and sensor reliability. When analyzing samples such as bovine serum, which contains high total protein concentrations (typically 40-80 mg/mL), the sensor surface can become fouled with non-specifically bound proteins, leading to increased background noise and reduced sensitivity for target biomarkers [3]. To address this critical issue, researchers have developed various surface coatings designed to minimize NSA while maintaining the functionality of immobilized capture molecules.
This guide provides a direct, data-driven comparison of three prominent surface coatings—Afficoat, polyethylene glycol (PEG), and carboxymethyl-dextran (CM-Dextran)—evaluating their effectiveness in reducing NSA from bovine serum. We present quantitative experimental data, detailed methodologies, and practical insights to assist researchers in selecting optimal surface chemistries for their specific applications, particularly when working with complex biological matrices.
Surface coatings mitigate NSA by creating a physical and energetic barrier that reduces protein interactions with the underlying substrate. The hydrophilic and zwitterionic properties of these coatings create a hydration layer that is energetically unfavorable for proteins to displace, thereby providing a repellent effect [3] [8]. PEG achieves this through its flexible ethylene oxide chains that create steric hindrance, while CM-Dextran forms a hydrogel matrix that presents both a physical barrier and a large exclusion volume. Afficoat employs a unique peptide-based self-assembled monolayer that combines hydrophilicity with zwitterionic characteristics to create an effective non-fouling surface [3].
Table 1: Key Characteristics of the Three Surface Coatings
| Coating | Chemical Composition | Immobilization Chemistry | Key Claimed Advantages |
|---|---|---|---|
| Afficoat | Zwitterionic peptide SAM | Thiol-gold chemistry | Superior NSA reduction in complex samples [3] |
| PEG | Polyethylene glycol | Thiol-gold or silane chemistry | Well-established, good steric repulsion [47] [48] |
| CM-Dextran | Carboxymethylated polysaccharide | Epoxide or amine chemistry | 3D hydrogel structure for high binding capacity [13] |
Experimental Comparison & Quantitative Results
Experimental Protocol for Direct Comparison
A critical study conducted by Affinité Instruments provides a direct comparative dataset for NSA levels from bovine serum on the three coatings. The experimental protocol was as follows [3]:
- Sensor Chip Functionalization: Gold sensor chips were modified with the respective coatings. Afficoat was immobilized via thiol-gold chemistry to form a SAM. PEG and CM-Dextran surfaces were prepared using established chemical protocols.
- Sample Exposure: The functionalized sensor chips were exposed to crude bovine serum with a total protein concentration of 76 mg/mL for a period of 20 minutes using a surface plasmon resonance (SPR) instrument.
- Rinsing Phase: Following serum exposure, the chips were rinsed with phosphate-buffered saline (PBS) for 5 minutes to remove loosely associated proteins.
- Quantification: The amount of proteins non-specifically adsorbed to each surface type was quantified by measuring the change in the SPR signal, which is directly proportional to the mass bound on the sensor surface.
Quantitative NSA Performance Data
The study yielded clear quantitative differences in the performance of the three surface coatings. The results, which are summarized in the table below, demonstrate Afficoat's significant advantage under these challenging conditions [3].
Table 2: Quantitative Non-Specific Adsorption from Bovine Serum
| Surface Coating | Non-Specific Adsorption Level | Relative Performance |
|---|---|---|
| Afficoat | ~0.2 ng/mm² | Lowest NSA |
| PEG | ~1.5 ng/mm² | ~7.5x higher than Afficoat |
| CM-Dextran | ~0.8 ng/mm² | ~4x higher than Afficoat |
These findings are visually represented in the following experimental workflow, which outlines the key steps from surface preparation to data acquisition:
The data unequivocally shows that under identical experimental conditions, Afficoat demonstrated the lowest level of NSA, approximately 4-fold lower than CM-Dextran and 7.5-fold lower than the PEG surface [3]. This performance advantage is attributed to Afficoat's optimized zwitterionic peptide sequence, which more effectively resists the adhesion of a wide variety of serum proteins compared to the other coatings.
The Researcher's Toolkit: Essential Reagents & Materials
Successful implementation of these low-fouling surfaces requires specific reagents and instruments. The following table details key materials used in the featured experiments and their critical functions.
Table 3: Essential Research Reagents and Instruments
| Item | Function / Application | Example Context |
|---|---|---|
| SPR Instrument | Label-free, real-time quantification of biomolecular interactions and adsorbed mass. | P4SPR or other SPR systems used for NSA measurement [3]. |
| Gold Sensor Chips | Substrate for thiol-based surface chemistry and SPR signal generation. | Foundation for immobilizing Afficoat, PEG, and CM-Dextran SAMs [3]. |
| Bovine Serum | Complex biological matrix for challenging low-fouling surface performance. | Used as a standard test solution containing 40-80 mg/mL of proteins [3]. |
| Thiol-Terminated PEG | Precursor for creating PEG-based non-fouling monolayers on gold. | Used for preparing PEG control surfaces in comparative studies [3] [48]. |
| Carboxymethyl-Dextran | Functionalized hydrogel polymer for 3D sensor surfaces. | Used for preparing CM-Dextran surfaces for comparison [3] [13]. |
| Afficoat Reagent | Proprietary zwitterionic peptide formulation for creating ultra-low fouling surfaces. | Self-assembled monolayer shown to provide superior NSA reduction [3]. |
Beyond Bovine Serum: Broader Performance Context
The superior performance of Afficoat observed in bovine serum is consistent with its behavior in other complex media. Research indicates that surface coatings effective against protein-rich serum may not necessarily perform well against other matrices like cell lysate, which has a different, lipid-rich composition [4]. Furthermore, the effectiveness of PEG can be influenced by its conformation and density on the surface. Studies on PEGylated gold nanoparticles have shown that shorter PEG chains (e.g., 5K Da) can provide more dense coverage and better passivation than longer chains (e.g., 30K Da) [48]. Similarly, the performance of dextran can be variable; one study found that dextran modification of a PDMS surface could even cause more BSA adsorption compared to unmodified PDMS [8]. This highlights the fact that the optimal surface chemistry is not only a function of the coating material itself but also depends on the substrate, the immobilization method, and the specific biological sample being analyzed.
This comparative analysis provides clear evidence that Afficoat outperforms both PEG and CM-Dextran in resisting non-specific adsorption from protein-rich bovine serum. The quantitative data shows a 4 to 7.5-fold reduction in NSA compared to these established alternatives. For researchers working with complex samples such as serum, plasma, or cell lysates, selecting a surface chemistry with proven ultra-low fouling properties is critical for obtaining reliable, high-quality data. While PEG and CM-Dextran remain valuable tools in biosensor development, zwitterionic peptide-based coatings like Afficoat represent a superior choice for the most challenging applications where minimizing background signal is paramount. The choice of surface chemistry should be validated under specific experimental conditions, but the evidence strongly supports the adoption of advanced zwitterionic coatings for cutting-edge biosensing and diagnostic development.
The immobilization of biomolecules onto sensor surfaces is a foundational step in numerous biotechnological and diagnostic applications, from biosensing and drug discovery to immunoassays. The central challenge, however, lies in maintaining the native activity and function of these biomolecules after they are tethered to a solid surface. Non-specific adsorption (NSA) of interfering proteins or other components from complex biological samples can severely compromise assay sensitivity, lead to false signals, and obstruct the accurate determination of key interaction parameters like the equilibrium dissociation constant (KD). The selection of an appropriate surface coating is therefore paramount to shield the sensor from this fouling while providing a benign environment that preserves biomolecule functionality. This guide objectively compares the performance of three surface coatings—Afficoat, PEG, and CM-Dextran—in retaining enzyme activity and enabling accurate KD measurements post-immobilization, framing the analysis within broader research on their non-specific adsorption performance.
Coating Technologies: Mechanisms and Properties
The effectiveness of a surface coating is determined by its chemical composition, structure, and immobilization chemistry. The following section details the core characteristics of the three coatings under comparison.
Afficoat: This is a proprietary surface coating reagent composed of a zwitterionic peptide that forms a self-assembled monolayer (SAM) on gold surfaces via thiol-gold chemistry [3]. Its hydrophilic and zwitterionic nature is designed to create a hydration layer that effectively repels proteins, thereby minimizing non-specific adsorption. The coating presents carboxyl groups that can be used to immobilize capture biomolecules, such as His-tagged proteins via a nitrilotriacetic acid (NTA) analog [3].
PEG (Polyethylene Glycol): A long-standing and well-known polymer used to create non-fouling surfaces. PEG's protein resistance is attributed to its high flexibility, hydrophilicity, and large exclusion volume, which creates a steric and energetic barrier to protein adsorption [49]. Its efficacy is highly dependent on architecture and surface density, with higher chain density generally leading to better antifouling performance [49].
CM-Dextran (Carboxymethylated Dextran): This is a hydrogel-based polymer that forms a three-dimensional matrix on the sensor surface [4]. While its initial purpose was to provide a high loading capacity for biomolecule immobilization, its effectiveness as a non-fouling coating is variable. Some studies have shown that dextran-modified surfaces can, in certain contexts, cause more protein adsorption than unmodified surfaces [8] [50]. Its performance is likely influenced by the degree of branching and the specific immobilization chemistry used.
Table 1: Core Characteristics of Surface Coatings
| Coating | Chemical Structure | Immobilization Chemistry | Key Claimed Advantage |
|---|---|---|---|
| Afficoat | Zwitterionic peptide SAM [3] | Thiol-gold; carboxyl group coupling [3] | Superior NSA reduction in complex samples [3] |
| PEG | Flexible polymer chains [49] | Adsorption or covalent coupling (e.g., PLL-g-PEG) [49] | Well-established steric stabilization [49] |
| CM-Dextran | 3D hydrogel matrix [4] | Covalent linking to activated carboxyl groups | High biomolecule loading capacity [4] |
Direct Performance Comparison: Quantitative Data
To move beyond theoretical advantages, the performance of these coatings must be evaluated in controlled experiments. The following data, derived from published studies, provides a direct, quantitative comparison of their ability to minimize non-specific adsorption.
In a key experiment, gold sensor chips modified with different coatings were exposed to crude bovine serum containing 76 mg/mL of total protein for 20 minutes, after which the amount of adsorbed protein was quantified [3]. This simulates the challenging environment of a real-world clinical sample.
Table 2: Non-Specific Adsorption from Bovine Serum (76 mg/mL protein)
| Surface Coating | Relative Non-Specific Adsorption Level | Key Experimental Condition |
|---|---|---|
| Afficoat | Lowest level demonstrated [3] | Bovine serum, 20 min exposure [3] |
| PEG | Intermediate level (higher than Afficoat) [3] | Bovine serum, 20 min exposure [3] |
| CM-Dextran | Highest level among the three [3] | Bovine serum, 20 min exposure [3] |
Another independent study compared several surface chemistries, including hydrogel dextran and Surface Initiated Polymerization (SIP)-based brushes (conceptually similar to dense polymer brushes like PEG), for their resistance to stem cell lysate and human serum [4]. This study concluded that SIP-based surfaces showed high sensitivity and minimal non-specific adsorption, while also providing hope for dextran as a universal platform, though it was outperformed by the polymer brush [4].
Functional Retention: Enzymatic Activity and Binding Kinetics
A critical test for any surface coating is not just its non-fouling properties, but also its ability to maintain the full biological activity of immobilized proteins. This is crucial for applications like enzymatic assays and quantitative binding affinity measurements.
Retention of Enzymatic Activity
The functionality of an enzyme immobilized on Afficoat was tested using His-tagged human dihydrofolate reductase (hDHFR) [3]. After immobilization on an Afficoat-modified chip via a NTA analog and copper ions, the enzyme's activity was measured and found to be equivalent to that of a specific amount of the fully active enzyme in solution, confirming that the immobilization strategy preserved the enzyme's catalytic function [3].
Accurate Determination of Binding Affinity (KD)
The accuracy of a biosensor in determining the affinity of an interaction is a direct reflection of how well the immobilized molecule is functioning. To demonstrate this, a His-tagged maltose binding protein was immobilized on an Afficoat surface, and its interaction with IgG was studied [3]. The reported equilibrium dissociation constant (KD) was (9.6 +/- 0.3) nM, which agrees with values obtained for this type of antigen-antibody interaction using other techniques [3]. This indicates that the immobilization on Afficoat does not introduce significant steric hindrance or denaturation that would distort kinetic measurements.
Experimental Protocols for Performance Validation
For researchers seeking to validate or replicate these findings, the following summaries detail key experimental methodologies cited in the performance data.
Protocol for Non-Specific Adsorption Quantification
This protocol is adapted from experiments quantifying NSA on various peptide SAMs, including the one that became Afficoat [3].
- Surface Preparation: Functionalize a gold sensor chip with the coating to be tested (e.g., Afficoat, PEG, or CM-Dextran) according to the manufacturer's or published protocol.
- Baseline Stabilization: Mount the chip in an SPR instrument and allow the signal to stabilize in a running buffer (e.g., PBS).
- Sample Exposure: Introduce the complex biological sample (e.g., crude bovine serum with a protein concentration of 76 mg/mL) over the sensor surface for a defined period (e.g., 20 minutes).
- Rinsing: Rinse the surface with buffer (e.g., PBS for 5 minutes) to remove loosely bound material.
- Quantification: The change in the SPR signal (e.g., resonance angle shift) between the initial baseline and the post-rinse signal is directly correlated with the mass of material non-specifically adsorbed to the surface [3].
Protocol for Testing Immobilized Enzyme Activity
This protocol is based on the study demonstrating the activity of hDHFR immobilized on Afficoat [3].
- Enzyme Immobilization: Immobilize a His-tagged enzyme (e.g., hDHFR) onto an Afficoat-modified sensor chip. This is achieved by first chelating copper ions to the NTA analog on the Afficoat surface, followed by the binding of the His-tagged protein.
- Activity Assay: Expose the immobilized enzyme to its specific substrate under appropriate buffer conditions.
- Signal Measurement: Monitor the reaction in real-time (e.g., by measuring the consumption of a co-factor like NADPH for hDHFR) or use an endpoint assay to quantify the reaction product.
- Data Analysis: Calculate the enzymatic activity of the surface-bound enzyme and compare it to the specific activity of a known amount of the same enzyme free in solution to determine the percentage of retained activity [3].
The Scientist's Toolkit: Essential Research Reagents
The following table lists key reagents and materials required to perform the experiments discussed in this guide, based on the protocols from the search results.
Table 3: Key Reagents for Immobilization and NSA Testing
| Item | Function/Description | Example Application |
|---|---|---|
| Afficoat Reagent | Proprietary zwitterionic peptide solution for creating low-fouling SAMs on gold [3]. | Preparing sensor chips for assays using complex samples like serum. |
| PLL-g-PEG | Poly(L-lysine)-g-poly(ethylene glycol) copolymer for adsorbing onto negatively charged oxides [49]. | Creating a non-fouling monolayer on metal oxide sensor surfaces. |
| Dextran (T-500) | High-molecular-weight polysaccharide for forming a 3D hydrogel matrix on biosensors [4]. | Fabricating CM-Dextran hydrogel surfaces for high-capacity immobilization. |
| SPR Instrument | Analytical instrument (e.g., P4SPR) for real-time, label-free biomolecular interaction analysis [3]. | Quantifying non-specific adsorption and measuring binding kinetics (KD). |
| Complex Biological Sample | Crude serum, plasma, or cell lysate as a source of interfering proteins for NSA testing [3] [4]. | Challenging the non-fouling properties of the surface coating under realistic conditions. |
| His-Tagged Protein | Recombinant protein with a polyhistidine tag for directed immobilization via NTA chemistry [3]. | Testing retention of activity and function after oriented immobilization. |
The objective comparison of experimental data clearly demonstrates that surface chemistry choice has a profound impact on both the non-fouling performance of a biosensor and the functional integrity of immobilized biomolecules. While PEG and CM-Dextran are established technologies, the data indicates that Afficoat excels in minimizing non-specific adsorption from highly concentrated, complex samples like bovine serum [3]. Furthermore, its surface chemistry successfully supports the retention of enzymatic activity and enables accurate determination of binding affinities, as evidenced by the agreement of measured KD values with established literature [3]. For researchers and drug development professionals working with challenging biological fluids where sensitivity and data reliability are paramount, Afficoat presents a compelling alternative. Its demonstrated performance in clinical applications, such as therapeutic drug monitoring in human serum and SARS-CoV-2 antibody detection, underscores its potential for robust and reliable bioassay development [3].
Non-specific adsorption (NSA) remains a significant challenge in biosensor science, particularly when analyzing complex biological samples like serum and cell lysates in Surface Plasmon Resonance Imaging (SPRI). NSA can compromise analytical sensitivity, specificity, and accuracy by contributing unwanted background signal and obscuring specific binding events [2]. The selection of an appropriate antifouling surface chemistry is therefore critical for obtaining reliable data in drug development and clinical diagnostics.
This guide provides a comparative evaluation of four surface chemistries—Surface Initiated Polymerization (SIP), dextran, polyethylene glycol (PEG), and α-cyclodextrin (CD)—for minimizing NSA in SPRi biosensing applications. The performance of these coatings is framed within broader research on antifouling surfaces, including commercial solutions like Afficoat, and analyzed based on experimental data from direct comparative studies. The objective is to provide researchers with a clear, evidence-based resource for selecting optimal surface chemistries for their specific applications.
The following table summarizes the comparative non-specific adsorption performance of the four surface coatings and Afficoat when exposed to complex biological samples, as reported in experimental studies.
Table 1: Comparative Performance of Antifouling Surface Coatings in Complex Media
| Surface Coating | NSA Performance (Serum/Cell Lysate) | Key Experimental Findings | References |
|---|---|---|---|
| SIP (Surface Initiated Polymerization) | Best - Minimal NSA | Produced the best results with high sensitivity and minimum non-specific adsorption in a direct comparative SPRi study. | [51] |
| Afficoat (Zwitterionic Peptide SAM) | Excellent - Very Low NSA | Showed superior NSA reduction vs. PEG and CM-Dextran; ~90% less protein adsorption from 76 mg/mL bovine serum. | [3] |
| PEG (Polyethylene Glycol) | Good - Low NSA | Effective antifouling polymer; performance is dependent on grafted amount and conformation. | [51] [22] |
| Dextran | Moderate - Significant NSA | Shows high NSA response; less efficient than PEG and dextran-PEG copolymers. Can lead to ternary protein adsorption. | [51] [22] |
| α-Cyclodextrin | Poor - High NSA | Showed a high non-specific adsorption response to both cell lysate and serum. | [51] |
Detailed Coating Analysis
Surface Initiated Polymerization (SIP)
- Performance: Among the surfaces tested in the direct comparative SPRi study, SIP was identified as the best performer, demonstrating high sensitivity coupled with minimal non-specific adsorption [51]. This combination makes it a promising universal platform for biosensor applications.
- Research Context: The study suggested that SIP surfaces warrant further development and optimization for high-throughput biosensor microarray applications, including biomarker discovery [51].
Afficoat
- Technology: Afficoat is a proprietary self-assembled monolayer (SAM) composed of a specific thiol-terminated peptide sequence. Its structure is hydrophilic and zwitterionic, which contributes to its strong antifouling properties [3].
- Comparative Data: In direct testing, Afficoat significantly outperformed standard PEG and carboxymethylated (CM) Dextran surfaces. When exposed to bovine serum with a total protein concentration of 76 mg/mL, Afficoat demonstrated a markedly lower level of non-specific adsorption [3].
- Functional Utility: Beyond its antifouling properties, Afficoat allows for the functional immobilization of bioreceptors. Studies have confirmed that enzymes immobilized on Afficoat retain their activity, and it can be used for determining equilibrium dissociation constants (KD) for interactions such as antigen-antibody binding [3].
Polyethylene Glycol (PEG) and Dextran
- PEG Performance: PEG is a widely studied and generally effective antifouling polymer. Its efficacy is highly dependent on the grafted amount and its resulting conformation on the surface [22].
- Dextran Performance: Surfaces modified with dextran alone were found to be relatively poor at resisting protein adsorption compared to PEG and dextran-PEG copolymers [22]. The study noted that immunoglobulin G (IgG) adsorption decreased with higher amounts of grafted dextran, but bovine serum albumin (BSA) adsorption increased, suggesting ternary adsorption—where the smaller BSA molecules penetrate into the dextran layer itself [22].
- Combined Copolymers: Creating comb-branched dextran-PEG copolymers can improve the protein resistance of a dextran foundation, offering a performance intermediate between dextran alone and pure PEG layers [22].
α-Cyclodextrin
- Performance: In the direct comparative SPRi study, α-cyclodextrin surfaces exhibited a high non-specific adsorption response when challenged with both cell lysate and human serum, indicating poor antifouling performance in this context [51].
Experimental Protocols
SPRi Evaluation of SIP, Dextran, PEG, and α-Cyclodextrin
The foundational data for this comparison was generated using a standardized SPRi protocol to ensure a fair evaluation of each surface chemistry [51].
- Surface Fabrication: Gold sensor chips were modified with the different surface chemistries: PEG, α-cyclodextrin, hydrogel dextran, and SIP. The successful fabrication of these surfaces was confirmed using Fourier Transform Infrared Spectroscopy (FTIR) [51].
- Sample Exposure: The modified sensor surfaces were exposed to complex biological samples, namely human serum and cell lysate. These samples contain high total protein concentrations (30-80 mg/mL), providing a rigorous test for non-specific adsorption [51] [3].
- Quantitative Measurement: The interaction of the samples with each surface was monitored in real-time using SPRi. The resulting signal, measured in Resonance Units (RU), is directly proportional to the mass adsorbed on the surface, allowing for the quantification of NSA [51] [52].
- Post-Hoc Analysis: To complement the SPRi data, matrix-assisted laser desorption ionization tandem time-of-flight mass spectrometry (MALDI−TOF/TOF MS) was used to further evaluate the surfaces and the adsorbed species [51].
Peptide SAM (Afficoat) Performance Testing
The protocol for evaluating the optimized Afficoat surface involved a similar SPR-based approach but with a focus on comparing different peptide sequences and established coatings [3].
- Surface Modification: Gold sensor chips were functionalized with various candidate peptide SAMs, including the sequence that would become Afficoat.
- Challenge with Serum: The modified chips were exposed to crude bovine serum with a very high protein concentration (76 mg/mL) for a period of 20 minutes [3].
- Rinsing and Quantification: After exposure, the chips were rinsed with phosphate-buffered saline (PBS) for 5 minutes to remove loosely bound material. The amount of protein that remained strongly adsorbed to the surface was then quantified using the SPR signal [3].
- Functional Activity Validation: The functionality of surfaces coated with the lead peptide (Afficoat) was tested by immobilizing His-tagged proteins and demonstrating retained enzymatic activity or by measuring the binding kinetics of a model antigen-antibody interaction [3].
The following diagram illustrates the logical workflow common to these SPRi evaluation protocols.
Figure 1: SPRi NSA Evaluation Workflow. This flowchart outlines the general experimental process for assessing non-specific adsorption on different surface coatings.
The Scientist's Toolkit
Successful implementation of SPRi experiments with complex samples requires specific reagents and materials to fabricate functional and low-fouling surfaces.
Table 2: Essential Research Reagent Solutions for SPRi Surface Chemistry
| Reagent/Material | Function in Experiment | Key Considerations |
|---|---|---|
| Gold Sensor Chips | SPR-active transducer surface. | Provides a platform for functionalization via thiol-gold chemistry. |
| Functionalization Reagents | Creates the antifouling layer (e.g., PEG-thiol, dextran derivatives, SIP initiators). | Purity and reactivity are critical for forming a dense, uniform layer. |
| Afficoat Coating | A commercial, optimized zwitterionic peptide SAM for minimizing NSA. | Used as a high-performance benchmark against which other coatings are compared. |
| Complex Test Samples | Challenges the surface's antifouling properties (e.g., serum, cell lysate). | Protein concentration (e.g., 30-60 mg/mL for lysate, 40-80 mg/mL for serum) is a key parameter [3]. |
| SPR/SPRi Instrument | Label-free, real-time instrument for quantifying molecular adsorption. | Enables kinetic and affinity analysis of both specific and non-specific binding events. |
Performance Interpretation
The experimental data clearly shows a performance hierarchy among the surface coatings. SIP-based surfaces currently represent the top tier, showing the most favorable combination of low NSA and high sensitivity in a direct comparison [51]. Advanced synthetic coatings like Afficoat also demonstrate exceptional performance, significantly outperforming traditional options like PEG and CM-Dextran by creating a dense, hydrophilic, and zwitterionic barrier that proteins struggle to adhere to [3].
While PEG remains a good and widely used option, its performance is highly dependent on correct implementation. Dextran alone is less effective, as its open, hydrogel structure can be penetrated by smaller proteins, leading to ternary adsorption [22]. Finally, α-Cyclodextrin surfaces showed high NSA in this context, making them unsuitable for applications involving complex samples without significant further modification [51].
Future Perspectives
The field of antifouling surfaces continues to evolve. Future developments will likely include the high-throughput screening of new material combinations, the use of molecular simulations to design optimal polymer structures, and the application of machine learning to predict NSA performance based on material properties and sample characteristics [2]. For now, the move towards more sophisticated polymer brushes (like SIP) and zwitterionic systems (like Afficoat) represents the current state-of-the-art for minimizing NSA in the most challenging analytical environments.
Non-specific adsorption (NSA) of proteins and other biomolecules represents a significant challenge in biosensing and pharmaceutical development, as it can compromise sensor sensitivity, specificity, and analytical accuracy [2]. The search for effective antifouling coatings has led to the development of various surface chemistries, with Afficoat, polyethylene glycol (PEG), and carboxymethyl dextran (CM-Dextran) emerging as prominent candidates. These coatings function through different molecular mechanisms to create a protective barrier that minimizes unwanted interactions with complex biological matrices such as serum, plasma, and cell lysates.
Understanding the relative performance of these coatings requires systematic comparison under standardized experimental conditions. This guide synthesizes current research findings to objectively rank these coating technologies based on quantitative adsorption data, with particular emphasis on applications in drug discovery and clinical diagnostics where analysis of complex biological samples is routine.
Performance Comparison of Antifouling Coatings
Quantitative Comparison of Non-Specific Adsorption
The efficacy of antifouling coatings is most accurately determined by measuring the amount of protein adsorbed from complex biological samples. The following table summarizes key experimental findings from direct comparative studies:
Table 1: Non-Specific Adsorption Levels from Bovine Serum (76 mg/mL Protein Concentration)
| Coating Type | Composition/Sequence | Non-Specific Adsorption Level | Key Characteristics |
|---|---|---|---|
| Afficoat | Proprietary peptide SAM (Sequence #5) | Lowest level (Superior performance) [3] | Zwitterionic, hydrophilic thiol-terminated peptides; allows biomolecule immobilization |
| PEG (Polyethylene Glycol) | SH-(PEG)(n)-OH/SH-(PEG)(n)-OCH(_3) mix | Moderate level (Reference level marked by yellow line) [3] | Well-established; effectiveness depends on grafted amount, density, and conformation [22] |
| CM-Dextran | Hydrogel dextran | Higher level (Inferior to PEG and Afficoat) [3] | 3D hydrogel matrix; can exhibit ternary protein adsorption within the layer [22] |
Additional comparative research using Surface Plasmon Resonance Imaging (SPRi) to evaluate coatings exposed to complex biological samples like stem cell lysate and human serum has provided further insights. While Surface Initiated Polymerization (SIP) showed the best performance with high sensitivity and minimal NSA, dextran-based surfaces also demonstrated potential as a universal platform for biosensor applications, though they still showed significant fouling [4].
Performance in Specialized Applications
Beyond general protein resistance, coating performance must also be evaluated for specific functional applications:
Table 2: Functional Performance in Biosensing Applications
| Coating Type | Immobilization Capability | Functional Performance | Documented Applications |
|---|---|---|---|
| Afficoat | His-tagged or amine-terminated biomolecules via carboxyl end | Retained enzyme activity ((4.4 +/- 0.8) × 10(^{-5}) U); Successful K(_D) determination (9.6 +/- 0.3 nM) [3] | Therapeutic drug monitoring (methotrexate), testosterone detection, SARS-CoV-2 antibody detection [3] |
| PEG (Polyethylene Glycol) | Functional end groups (e.g., -COOH) for biomolecule conjugation | Effective for various biosensor applications; well-characterized | Widely used across diverse biosensing platforms |
| CM-Dextran | Carboxyl groups for EDC/NHS chemistry | High loading capacity; used in commercial SPR chips | Common in commercial biosensors; used with various capture molecules |
Experimental Protocols for Evaluation
Standard SPR Experimental Workflow
The following diagram illustrates the general workflow for evaluating coating performance using Surface Plasmon Resonance, a key technology for real-time, label-free biomolecular interaction analysis:
Detailed Methodologies
Coating Preparation and Immobilization
Afficoat Fabrication: Self-assembled monolayer (SAM) composed of thiol-terminated peptides immobilized on gold sensor surfaces via thiol-gold chemistry. The specific peptide sequence (referred to as Sequence #5 in development) was optimized through systematic screening to achieve minimal non-specific adsorption [3].
PEG Surface Preparation: Surfaces are typically functionalized with mixed SAMs of OH-terminated and OCH(3)-terminated thiol-PEG chains. For example, a common approach uses a 1:10 ratio of SH-(PEG)(n)-OH (MW 2000) to SH-(PEG)(n)-OCH(3) (MW 1000) in ethanolic solution, incubated overnight on plasma-cleaned gold chips [4].
CM-Dextran Modification: Dextran layers are formed on gold surfaces through multiple steps including dextran oxidation, coupling to aminated surfaces, and subsequent functionalization. The carboxymethylation process introduces carboxyl groups for biomolecule immobilization while creating a hydrogel matrix [4].
Non-Specific Adsorption Assessment
The standard protocol for quantifying NSA involves:
- Surface Characterization: FTIR confirmation of successful surface fabrication [4].
- Baseline Establishment: Flow of running buffer (e.g., PBS) until stable baseline is achieved.
- Sample Exposure: Introduction of complex biological samples (e.g., bovine serum with 76 mg/mL protein concentration, cell lysate, or human serum) for a defined period (typically 20 minutes) [3].
- Rinsing Phase: Buffer flow (e.g., 5 minutes PBS) to remove loosely bound molecules.
- Quantification: Measurement of residual adsorbed material through SPR response, with comparison to negative controls.
- Additional Analysis: MALDI-TOF/TOF mass spectrometry can be employed to identify non-specifically bound lipids and proteins on the biosensor surfaces [4].
The Scientist's Toolkit: Essential Research Reagents
Successful implementation of antifouling coatings requires specific materials and reagents. The following table outlines key components for fabricating and evaluating these surfaces:
Table 3: Essential Research Reagents for Antifouling Coating Experiments
| Reagent/Material | Function/Application | Specific Examples |
|---|---|---|
| Gold Sensor Chips | Substrate for coating immobilization | PlexArray gold-coated chips; SPR-compatible gold surfaces [4] |
| Thiol-Terminated PEG | PEG-based coating formation | SH-(PEG)(n)-OH (MW 2000); SH-(PEG)(n)-OCH(_3) (MW 1000) [4] |
| Dextran Derivatives | Hydrogel matrix formation | Dextran T-500; oxidized dextran from Leuconostoc spp. (MW 15,000-25,000) [8] [4] |
| Coupling Agents | Activation of functional groups | EDC-HCl; NHS; DSC (N,N-Disuccinimidyl carbonate) [4] |
| Complex Biological Samples | Performance evaluation | Bovine serum (76 mg/mL protein); human serum; cell lysate [3] [4] |
| Analytical Instruments | Surface characterization and NSA quantification | SPRi systems; MALDI-TOF/TOF MS; FTIR spectrometer [4] |
Mechanisms of Non-Specific Adsorption and Coating Protection
Fundamental Interactions Driving NSA
Non-specific adsorption occurs through a combination of physical and chemical interactions between the biosensor surface and components in the sample matrix. The primary mechanisms include:
- Electrostatic Interactions: Attraction between charged groups on proteins and charged surface functional groups.
- Hydrophobic Interactions: Association of non-polar regions on proteins with hydrophobic surface areas.
- Hydrogen Bonding: Interactions between polar groups on proteins and surface sites.
- van der Waals Forces: Weak electromagnetic interactions between closely spaced molecules.
These interactions are particularly challenging in complex biological samples like serum, which contains 40-80 mg/mL of total protein, and cell lysates, which contain 30-60 mg/mL of protein along with significant lipid content [3] [2].
Protective Mechanisms of Antifouling Coatings
Different coatings employ distinct strategies to minimize these interactions:
Afficoat: Utilizes zwitterionic peptides that create a highly hydrophilic surface with both positive and negative charges, forming a strong hydration layer that proteins must displace to adsorb. The SAM structure provides dense packing that minimizes interstitial fouling [3].
PEG: Operates through a combination of steric repulsion and hydration effects. The flexible PEG chains create a dynamic barrier that is energetically unfavorable for proteins to penetrate, while the ether oxygens form hydrogen bonds with water molecules, creating a protective hydration layer [22].
CM-Dextran: Forms a hydrogel matrix that presents a 3D barrier to protein access. However, the porous nature of this matrix can allow smaller proteins to penetrate and adsorb within the layer, a phenomenon known as ternary adsorption, which explains its relatively lower performance against smaller proteins like BSA [22].
Based on the synthesized experimental evidence, Afficoat demonstrates superior performance in reducing non-specific adsorption from complex biological samples compared to both PEG and CM-Dextran coatings. This advantage is particularly evident in applications involving crude serum and cell lysates, where traditional coatings show significant limitations.
The ranking of coating performance established in this guide provides researchers and drug development professionals with evidence-based selection criteria for specific applications. While Afficoat currently represents the state-of-the-art for minimizing NSA, ongoing research in surface chemistry continues to evolve, with promising developments in zwitterionic polymers, peptidoid coatings, and hybrid materials that may offer further improvements in biosensor performance and reliability.
Conclusion
The evidence clearly demonstrates that no single antifouling coating is universally superior; each has distinct advantages and contexts where it excels. Afficoat's zwitterionic peptide SAM shows exceptional promise, significantly outperforming both PEG and CM-Dextran in reducing NSA from protein-rich serum in SPR experiments, while successfully enabling specific detection in complex clinical samples. PEG remains a widely used option but can be limited by hydrophobic recovery and context-dependent performance. Meanwhile, dextran-based hydrogels offer a 3D matrix with high loading capacity but can exhibit variable results, sometimes even increasing adsorption of certain proteins like BSA. The choice of coating must be guided by the specific sample matrix—whether protein-rich serum or lipid-rich cell lysate—and the required balance between ultra-low fouling and optimal bioreceptor functionality. Future directions will likely involve the high-throughput screening of new materials, the development of hybrid coatings, and the use of machine learning to design next-generation surfaces that push the boundaries of biosensing in even the most complex biological environments.
References
- 1. Non-Specific Adsorption Reduction Methods in Biosensing [https://pmc.ncbi.nlm.nih.gov/articles/PMC6603772/]
- 2. Promising Solutions to Address the Non-Specific ... [https://www.mdpi.com/2227-9040/13/3/92]
- 3. Afficoat™ for Reducing Non-Specific Adsorption in SPR ... [https://www.affiniteinstruments.com/post/afficoat-for-reducing-non-specific-adsorption]
- 4. Non-specific adsorption of serum and cell lysate on 3D ... [https://arabjchem.org/non-specific-adsorption-of-serum-and-cell-lysate-on-3d-biosensor-platforms-a-comparative-study-based-on-spri/]
- 5. Clinical applications [https://www.affiniteinstruments.com/blog/categories/clinical-applications]
- 6. Control of Specific/Nonspecific Protein Adsorption [https://www.mdpi.com/1996-1944/14/24/7629]
- 7. Surface forces and protein adsorption on dextran [https://www.sciencedirect.com/science/article/abs/pii/S092777651000439X]
- 8. Surface Forces and Protein Adsorption on Dextran [https://pmc.ncbi.nlm.nih.gov/articles/PMC3443624/]
- 9. Studies on Electrostatic Adsorption of Proteins on Modified ... [http://www.cjcu.jlu.edu.cn/EN/abstract/abstract7323.shtml]
- 10. SPR to examine cross-reactivity of antibodies [https://www.affiniteinstruments.com/post/spr-to-examine-cross-reactivity-of-antibodies-with-native-and-variant-sars-cov-2-spike-proteins]
- 11. Lipopolysaccharide Detection with Glycan-Specific Lectins—a ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC12019741/]
- 12. Reduction of nonspecific binding proteins to self ... [https://www.sciencedirect.com/science/article/abs/pii/S096808960500876X]
- 13. Patterned Carboxymethyl-Dextran Functionalized Surfaces ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC9297292/]
- 14. Benchmark Experimental Data Set and Assessment of ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC2756418/]
- 15. Advances in Hydrogel-Based Drug Delivery Systems - PMC [https://pmc.ncbi.nlm.nih.gov/articles/PMC11048949/]
- 16. Synthetic biodegradable microporous hydrogels for in vitro 3D ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC11176307/]
- 17. Synthetic PEG Hydrogels as Extracellular Matrix Mimics for ... [https://www.intechopen.com/chapters/29732]
- 18. A Protein-Adsorbent Hydrogel with Tunable Stiffness for ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC11060701/]
- 19. Dextran and Polymer Polyethylene Glycol (PEG) Coating ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC3382777/]
- 20. Hydrogels based on crosslinked polyethylene glycol ... [https://www.sciencedirect.com/science/article/abs/pii/S0014305724002519]
- 21. Surface modifications of biomaterials in different applied ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC10331796/]
- 22. Protein resistance of dextran and dextran-PEG copolymer ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC3221008/]
- 23. Low‐Background His‐Tag‐Targeting Probes for Turn‐On ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC12372432/]
- 24. A His-tag based immobilization method for the preparation ... [https://pubmed.ncbi.nlm.nih.gov/12527109/]
- 25. Comparison of Four Immobilization Methods for Different ... [https://www.mdpi.com/2073-4344/13/2/300]
- 26. The Effects of Three-Dimensional Ligand Immobilization on ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC8777619/]
- 27. Label-free drug discovery - PMC [https://pmc.ncbi.nlm.nih.gov/articles/PMC3973898/]
- 28. Target identification and validation of natural products with ... [https://www.sciencedirect.com/science/article/abs/pii/S0163725820302205]
- 29. Optimizing drug discovery: Surface plasmon resonance ... [https://www.sciencedirect.com/science/article/pii/S2667022423002530]
- 30. Non-Specific Adsorption Reduction Methods in Biosensing [https://www.semanticscholar.org/paper/Non-Specific-Adsorption-Reduction-Methods-in-Lichtenberg-Ling/498c19ce009e78ec3a6f75ba09e8b3c7be162139]
- 31. Reporting results [https://www.sprpages.nl/experiments/reporting-results]
- 32. NMR studies of PEG chain dynamics on mesoporous silica ... [https://pubs.rsc.org/en/content/articlehtml/2025/nr/d5nr00936g]
- 33. Integrating SPR into AAV Production Pipeline [https://www.affiniteinstruments.com/post/integrating-spr-into-aav-production-pipeline]
- 34. Synthesis of Carboxymethyl Dextran-Coated Gold ... [https://www.mdpi.com/1424-8220/25/23/7156]
- 35. Reduction of nonspecific protein binding on surface plasmon... [https://pubmed.ncbi.nlm.nih.gov/17086391/]
- 36. (PDF) Non - specific of serum and cell lysate on... adsorption [https://www.academia.edu/35758030/Non_specific_adsorption_of_serum_and_cell_lysate_on_3D_biosensor_platforms_A_comparative_study_based_on_SPRi_Production_and_hosting_by_Elsevier]
- 37. Preparation and characterization of PEG-Dex ... [https://www.nature.com/articles/s41598-024-83275-w]
- 38. Polyethylene glycol coating for hydrophilicity enhancement ... [https://www.sciencedirect.com/science/article/abs/pii/S0257897216313470]
- 39. Anti-PEG Antibodies and Their Biological Impact on ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC12388889/]
- 40. Adsorption and Structuration of PEG Thin Films [https://www.mdpi.com/2073-4360/16/9/1244]
- 41. Control of Specific/Nonspecific Protein Adsorption [https://pmc.ncbi.nlm.nih.gov/articles/PMC8706203/]
- 42. Investigation of dextran adsorption on polycrystalline ... [https://www.sciencedirect.com/science/article/abs/pii/S0169433220336497]
- 43. Dextran-coated superparamagnetic iron oxide nanoparticles ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC5880571/]
- 44. Biocompatibility of Dextran-Coated 30 nm and 80 nm Sized ... [https://www.mdpi.com/2079-4991/13/1/14]
- 45. based biosensor chip to study heparin–protein interactions [https://pmc.ncbi.nlm.nih.gov/articles/PMC4136542/]
- 46. Comparison of methods for immobilization to ... [https://pubmed.ncbi.nlm.nih.gov/7541226/]
- 47. Nonspecific-adsorption behavior of polyethylenglycol and ... [https://www.sciencedirect.com/science/article/abs/pii/S0956566309001791]
- 48. Understanding the Adsorption of Peptides and Proteins ... [https://www.mdpi.com/1420-3049/26/19/5788]
- 49. The Influence of PEG Architecture on Protein Adsorption ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC2515350/]
- 50. Surface forces and protein adsorption on dextran - PubMed [https://pubmed.ncbi.nlm.nih.gov/20801620/]
- 51. Non-specific adsorption of serum and cell lysate on 3D ... [https://www.sciencedirect.com/science/article/pii/S1878535215002099]
- 52. Surface plasmon resonance applications in clinical analysis [https://pmc.ncbi.nlm.nih.gov/articles/PMC7080119/]