Optical Biosensors in Fermentation Monitoring: A Comprehensive Guide for Biopharmaceutical Research
This article provides a comprehensive overview of the application of optical biosensors for advanced fermentation monitoring, a critical process in biopharmaceutical development.
Optical Biosensors in Fermentation Monitoring: A Comprehensive Guide for Biopharmaceutical Research
Abstract
This article provides a comprehensive overview of the application of optical biosensors for advanced fermentation monitoring, a critical process in biopharmaceutical development. Tailored for researchers, scientists, and drug development professionals, it explores the foundational principles of optical biosensing, details specific methodologies like Reflectometric Interference Spectroscopy (RIfS) and SPR, and presents real-world case studies, such as the monitoring of glycopeptide antibiotic production. The content further addresses practical challenges, including matrix interference and sensor drift, and offers optimization strategies. Finally, it delivers a critical comparative analysis of optical against other sensing technologies, supported by performance metrics and data on the growing market and integration of AI-driven soft sensors, providing a holistic resource for enhancing bioprocess control and accelerating therapeutic development.
Core Principles and Market Landscape of Optical Biosensing in Bioprocessing
Core Principles of Optical Biosensors
An optical biosensor is a compact analytical device that integrates a biorecognition element with an optical transducer system [1] [2]. The fundamental objective is to produce a quantifiable signal proportionate to the concentration of a specific analyte by converting a biological recognition event into a measurable optical signal [1] [3]. These sensors function on the principle of direct, real-time, and often label-free detection, offering high specificity, sensitivity, small size, and cost-effectiveness [1] [2].
The operational framework involves two core components:
- Biorecognition Element: This biological entity provides analyte specificity. Key types include enzymes, antibodies, nucleic acids, aptamers, receptors, and whole cells [1] [3] [4]. This element is immobilized on a sensor surface and interacts specifically with the target analyte.
- Optical Transducer: This component converts the biorecognition event into a quantifiable optical signal. Transduction can be based on changes in absorbance, fluorescence, luminescence, refractive index, or reflectance [3] [4].
Many advanced optical biosensors, such as those based on surface plasmon resonance (SPR) or evanescent wave fluorescence, utilize the evanescent field in close proximity to the biosensor surface to detect interactions [1]. This allows for sensitive, label-free, and real-time monitoring of biochemical interactions, which is paramount for dynamic processes like fermentation.
Application in Fermentation Monitoring: A Practical Focus
In fermentation research and industrial bioprocessing, optical biosensors enable real-time monitoring of key analytes without extensive sample pretreatment [5] [4]. This capability is crucial for maintaining optimal process conditions, maximizing yield, and ensuring product quality.
Table 1: Key Analytes in Fermentation Monitoring Detectable by Optical Biosensors
| Analyte Category | Specific Examples | Relevance in Fermentation |
|---|---|---|
| Substrates & Metabolites | Glucose, Lactate, Sucrose, Amino Acids | Carbon source tracking; metabolic activity indicator [5] [4] |
| Gases | Oxygen (O₂), Carbon Dioxide (CO₂) | Critical for aerobic/anaerobic process control; cell respiration rate [3] |
| Biomass | Microbial cells (e.g., Yeast, E. coli) | Cell density and growth rate monitoring [4] |
| Products | Ethanol, Antibiotics, Organic Acids | Primary product titer and yield quantification [5] [4] |
| Contaminants | Microbial toxins, Endotoxins | Ensuring product safety and purity [4] |
Table 2: Performance Metrics of Optical Biosensors for Fermentation Analytes
| Transducer Technology | Detectable Analytic (Example) | Reported Detection Limit | Key Advantage for Fermentation |
|---|---|---|---|
| Surface Plasmon Resonance (SPR) | Antibiotics, Toxins, Proteins | ~0.1 nM (e.g., Mycotoxin Patulin) [1] | Real-time, label-free kinetics |
| Localized SPR (LSPR) | Pesticides, Microbial Contaminants | ~1.0 nM (e.g., As(III)) [1] | Enhanced sensitivity; adaptable fabrication |
| Fluorescence | Oxygen, pH, Intracellular metabolites | Single-molecule sensitivity (in research) [3] | Extreme sensitivity for low-abundance analytes |
| Fiber-Optic SPR (FO-SPR) | Pathogens in liquid media (e.g., milk) | High sensitivity in complex matrices [4] | Suitable for in-situ probes in bioreactors |
| Chemiluminescence | Pathogens (e.g., Listeria) | Low background noise [4] | Simple, effective platform |
Experimental Protocols
Protocol: Immobilization of a Biorecognition Element on an SPR Chip
This protocol details the covalent immobilization of an enzyme (e.g., Glucose Oxidase) onto a carboxymethylated dextran SPR chip surface for real-time glucose monitoring in a fermentation broth [1].
Principle: The carboxymethylated dextran matrix on a gold sensor chip is activated using N-hydroxysuccinimide (NHS) and N-ethyl-N'-(3-dimethylaminopropyl)carbodiimide (EDC) chemistry, enabling covalent bond formation with primary amines in the enzyme [1].
Materials:
- SPR instrument (e.g., Biacore series)
- Carboxymethylated dextran sensor chip (e.g., CM5)
- 0.1 M NHS (N-hydroxysuccinimide)
- 0.4 M EDC (N-ethyl-N'-(3-dimethylaminopropyl)carbodiimide)
- 1.0 mM HCl (for surface conditioning)
- Biorecognition element (e.g., 10-100 μg/mL Glucose Oxidase in 10 mM sodium acetate buffer, pH 5.0)
- 1 M Ethanolamine-HCl, pH 8.5 (deactivation solution)
- Running buffer (e.g., HBS-EP: 10 mM HEPES, 150 mM NaCl, 3 mM EDTA, 0.005% v/v Surfactant P20, pH 7.4)
Procedure:
- System Priming: Prime the SPR instrument and fluidic system with degassed running buffer.
- Surface Conditioning: Inject 1.0 mM HCl over the target flow cell for 60 seconds to clean and condition the dextran matrix.
- Surface Activation: Inject a 1:1 mixture of 0.1 M NHS and 0.4 M EDC over the flow cell for 7-20 minutes to activate the carboxyl groups to NHS esters.
- Ligand Immobilization: Immediately inject the solution of Glucose Oxidase (in sodium acetate buffer, pH 5.0) for 7-20 minutes. The low pH ensures the ligand is positively charged and attracted to the negatively charged dextran surface, promoting efficient coupling.
- Surface Deactivation: Inject 1 M Ethanolamine-HCl (pH 8.5) for 7 minutes to block any remaining activated ester groups.
- Surface Washing: Wash the surface with running buffer for at least 30 minutes to establish a stable baseline and remove any non-covalently bound material.
Validation: A successful immobilization is confirmed by a stable increase in the SPR signal (Response Units, RU) after the deactivation and washing steps, indicating the covalent attachment of the enzyme to the chip surface.
Protocol: Real-Time Analyte Detection and Kinetic Analysis
This protocol describes the procedure for monitoring analyte binding and determining kinetic parameters using an SPR biosensor.
Materials:
- SPR system with immobilized biorecognition element
- Analyte samples (in running buffer, serially diluted)
- Running buffer (as above)
Procedure:
- Baseline Stabilization: Allow the running buffer to flow over the sensor surface until a stable baseline is achieved.
- Sample Injection (Association Phase): Inject the analyte sample over the immobilized surface for a defined contact time (e.g., 2-5 minutes). The binding event causes an increase in the local refractive index, recorded as a rising signal in the sensorgram.
- Dissociation Phase: Switch the flow back to running buffer. The decrease in signal as the analyte dissociates from the ligand is monitored.
- Surface Regeneration: Inject a brief pulse (15-60 seconds) of a regeneration solution (e.g., 10 mM Glycine-HCl, pH 2.0-3.0) to completely remove bound analyte without damaging the immobilized ligand.
- Replication: Repeat steps 2-4 for all analyte concentrations and include a blank (buffer) injection for double-referencing.
Data Analysis:
The resulting sensorgrams (response vs. time) are fitted to a suitable interaction model, such as the 1:1 Langmuir binding model [1]. Software provided with the instrument (e.g., Biacore Evaluation Software) is used to calculate the association rate constant (k_on), the dissociation rate constant (k_off), and the equilibrium dissociation constant (K_D = k_off / k_on).
Signaling Pathways and Workflow Visualization
Diagram 1: Biosensor Integration in Fermentation Monitoring
Diagram 2: Molecular Interaction and Transduction Pathways
The Scientist's Toolkit: Key Research Reagent Solutions
Table 3: Essential Materials for Optical Biosensor-Based Fermentation Monitoring
| Item | Function/Description | Application Example |
|---|---|---|
| Carboxymethylated Dextran Sensor Chip | Gold sensor surface with a hydrogel layer for high-capacity, low non-specific binding immobilization of ligands via amine coupling [1]. | Standard platform for SPR-based kinetic and concentration analysis. |
| NHS/EDC Crosslinker Kit | N-hydroxysuccinimide (NHS) and N-ethyl-N'-(3-dimethylaminopropyl)carbodiimide (EDC) for activating carboxylated surfaces to enable covalent immobilization of proteins and other ligands containing primary amines [1]. | Covalent attachment of antibodies or enzymes to sensor chips. |
| Ethanolamine-HCl | Used to deactivate and block remaining activated ester groups on the sensor surface after ligand immobilization, minimizing non-specific binding [1]. | Final step in NHS/EDC immobilization protocol. |
| HEPES Buffered Saline with EDTA & Surfactant (HBS-EP) | A standard running buffer (10 mM HEPES, 150 mM NaCl, 3 mM EDTA, 0.005% P20). HEPES maintains pH, NaCl provides ionic strength, EDTA chelates metal ions, and surfactant P20 reduces non-specific binding [1]. | Standard running and dilution buffer for SPR experiments. |
| Glycine-HCl (pH 2.0-3.0) | A low-pH regeneration solution used to break the binding interaction between the ligand and analyte, restoring the sensor surface for the next analysis cycle without damaging the immobilized ligand [1]. | Regeneration of antibody-coated surfaces after analyte binding. |
| Gold Nanoparticles (AuNPs) & Quantum Dots (QDs) | Nanomaterials used to enhance signal transduction. AuNPs amplify LSPR and SERS signals, while QDs provide bright, photostable fluorescence labels [4]. | Signal amplification in LSPR or fluorescence-based assays. |
Optical biosensors have emerged as transformative tools for real-time, label-free monitoring of bioprocesses, offering significant advantages for fermentation research and development. These technologies enable scientists to track critical parameters such as metabolite concentrations, biomass, and substrate consumption directly within bioreactors, providing invaluable data for process optimization and control [6] [7]. This document details the application notes and experimental protocols for four key optical transduction mechanisms—Surface Plasmon Resonance (SPR), Reflectometric Interference Spectroscopy (RIfS), Fluorescence, and Fiber Optics—within the specific context of fermentation monitoring. The content is structured to provide researchers, scientists, and drug development professionals with practical methodologies for implementing these biosensing technologies in microbial fermentation and cell culture processes, supporting the advancement of intelligent bio-manufacturing [6].
Performance Comparison of Optical Biosensing Mechanisms
The selection of an appropriate transduction mechanism depends on the specific requirements of the fermentation monitoring application. The table below summarizes the key characteristics, advantages, and limitations of each technology for easy comparison.
Table 1: Performance Comparison of Optical Biosensing Mechanisms for Fermentation Monitoring
| Transduction Mechanism | Typical Detection Limit | Key Advantages | Primary Limitations | Example Fermentation Application |
|---|---|---|---|---|
| Surface Plasmon Resonance (SPR) | High sensitivity (fM level for 3rd gen.) [6] | Label-free, real-time kinetics, highly sensitive [8] [9] | Sensitive to bulk RI changes, complex data analysis | Real-time monitoring of metabolite (e.g., lactic acid) production [7] |
| Reflectometric Interference Spectroscopy (RIfS) | Not specified in results | Label-free, measures layer thickness and RI, simple optical setup | Lower sensitivity vs. SPR, limited multiplexing | Monitoring biofilm formation or enzyme immobilization [4] |
| Fluorescence | Exceptional sensitivity for low-level toxins [4] | Very high sensitivity, multiplexing capability, wide dynamic range | Requires labeling, photobleaching potential, autofluorescence | Tracking specific protein expression or intracellular pH using fluorescent probes [10] |
| Fiber Optics | Varies with core mechanism | Miniaturization, in situ placement, immune to EMI, remote sensing [11] | Coupling losses, fragility, potential for biofouling | In-situ monitoring of glucose or ethanol in harsh bioreactor environments [11] |
Surface Plasmon Resonance (SPR)
Application Note
SPR biosensors are powerful tools for label-free, real-time analysis of biomolecular interactions. In fermentation monitoring, they are particularly valuable for tracking specific metabolites (e.g., antibiotics, organic acids) or biomarkers of cell stress without the need for fluorescent or radioactive labels [8] [6]. The technology operates by detecting minute changes in the refractive index (RI) at the surface of a thin metal film (typically gold), which occur when target analytes bind to immobilized biorecognition elements (e.g., antibodies, aptamers) [9]. This allows for the continuous monitoring of fermentation broths, providing kinetic data on metabolic pathways and enabling rapid process interventions.
Experimental Protocol for Metabolite Monitoring
Objective: To monitor the concentration of a specific metabolite (e.g., lactic acid) in a fermentation broth in real-time using an SPR biosensor.
Principle: A specific biorecognition element (e.g., an aptamer or antibody against the target metabolite) is immobilized on the SPR sensor chip. The binding of the metabolite to this receptor layer causes a local increase in the refractive index, leading to a shift in the SPR angle or wavelength, which is measured in real-time [9].
Materials:
- SPR instrument (e.g., commercial system like Biacore or custom Kretschmann-configuration setup)
- Gold-coated sensor chips
- Carboxymethylated dextran or similar surface for immobilization
- Biorecognition element (aptamer or monoclonal antibody)
- Coupling reagents: N-ethyl-N'-(3-dimethylaminopropyl)carbodiimide (EDC) and N-hydroxysuccinimide (NHS)
- Ethanolamine HCl (for blocking)
- Running buffer (e.g., HEPES-buffered saline, HBS)
- Fermentation broth samples (clarified by centrifugation/filtration)
Procedure:
- Sensor Chip Preparation: Dock a fresh gold sensor chip in the instrument. Prime the system with running buffer.
- Surface Functionalization:
- Inject a mixture of EDC and NHS to activate the carboxymethylated dextran surface.
- Dilute the biorecognition element (e.g., antibody) in sodium acetate buffer (pH 4.5-5.5) and inject it over the activated surface to achieve covalent immobilization.
- Inject ethanolamine HCl to deactivate any remaining ester groups and block the surface.
- Baseline Establishment: Flow running buffer over the sensor surface at a constant rate (e.g., 10-30 µL/min) until a stable baseline is achieved.
- Sample Analysis:
- Clarify fermentation broth samples by centrifugation (e.g., 10,000 × g for 10 min) and filtration (0.22 µm filter) to remove cells and debris.
- Inject the clarified sample over the sensor surface for a defined contact time (e.g., 3-5 minutes).
- Monitor the SPR signal in Resonance Units (RU) in real-time. The binding response is proportional to the mass concentration of the analyte bound.
- Switch back to running buffer to initiate dissociation.
- Surface Regeneration: After each cycle, regenerate the sensor surface by injecting a mild regeneration solution (e.g., 10 mM glycine-HCl, pH 2.0) to remove bound analyte without damaging the immobilized receptor. Re-equilibrate with running buffer.
- Data Analysis: Plot the maximum binding response (RU) for each sample against known standard concentrations to generate a calibration curve. Use this curve to determine the concentration of the target metabolite in unknown fermentation samples.
Diagram 1: SPR experimental protocol workflow
Reflectometric Interference Spectroscopy (RIfS)
Application Note
RIfS is a label-free optical technique that transduces binding events on a sensor surface into a measurable interference signal. It is based on the white-light interference pattern reflected from a thin, transparent layer, typically a SiO₂ or TiO₂ waveguide [4]. Changes in the optical thickness of this layer (a product of physical thickness and refractive index) caused by the adsorption of biomolecules lead to a shift in the interference pattern. In fermentation, RIfS is well-suited for monitoring non-specific parameters such as biomass accumulation, biofilm formation on surfaces, or the deposition of macromolecules, providing insights into process consistency and reactor fouling [7].
Experimental Protocol for Biomass Deposition Monitoring
Objective: To monitor the deposition of biomass or biofilm formation on a sensor surface in a fermentation environment using RIfS.
Principle: A white light source is directed onto a thin film transducer. The reflected light from the top and bottom interfaces of the film creates an interference pattern, which is detected by a spectrometer. The adsorption of biomass (cells, proteins) onto the transducer surface alters its optical thickness, causing a measurable shift in the interference spectrum [4].
Materials:
- RIfS instrument with a white light source and spectrometer
- SiO₂ or TiO₂ transducer chips
- Cleaning solution (e.g., Hellmanex, piranha solution [handle with extreme caution])
- Ethanol and water for rinsing
- Sterilized buffer (e.g., phosphate-buffered saline)
- Fermentation broth or cell suspension
Procedure:
- Transducer Cleaning: Clean the transducer chip thoroughly with appropriate solvents (e.g., Hellmanex, followed by ethanol and water). For SiO₂ chips, oxygen plasma treatment can be used to ensure a clean, hydrophilic surface.
- Instrument Calibration: Place the clean, dry transducer chip in the RIfS flow cell. Record the reference interference spectrum in air or buffer.
- Baseline Acquisition: Flow sterilized buffer through the system at a constant rate until a stable spectral baseline is achieved.
- Sample Exposure:
- Introduce the fermentation broth or a defined cell suspension into the flow cell.
- Continuously monitor the interference spectrum over time. The shift in the wavelength of the interference pattern (e.g., of a specific peak or the entire pattern) is recorded.
- Data Analysis: The shift in the interference pattern (Δλ) is directly related to the change in optical thickness (Δn·d), which is proportional to the mass density of the adsorbed biomass layer. Convert spectral shifts to mass deposition using the de Feijter formula or a pre-established calibration.
Table 2: Research Reagent Solutions for RIfS Biomass Monitoring
| Item Name | Function/Description | Critical Notes |
|---|---|---|
| SiO₂/TiO₂ Transducer Chips | Creates the interference phenomenon; biomolecules adsorb to its surface. | SiO₂ offers a biocompatible, silica-like surface. Thickness is precisely controlled. |
| Hellmanex Solution | Alkaline detergent for rigorous cleaning of optical surfaces. | Removes organic contaminants. Must be thoroughly rinsed with water. |
| Oxygen Plasma System | Creates a clean, hydrophilic surface on SiO₂ chips, enhancing biomolecule adhesion. | Optional but recommended step for maximum reproducibility. |
| Phosphate-Buffered Saline (PBS) | Provides a stable, isotonic, and biocompatible running buffer for baseline establishment. | Prevents cell lysis and non-specific binding due to ionic strength. |
Fluorescence Biosensing
Application Note
Fluorescence-based biosensors are among the most sensitive optical detection methods, capable of detecting targets at ultralow concentrations, which is ideal for tracking low-abundance metabolites or early indicators of stress in fermentations [10] [4]. These biosensors rely on the detection of light emitted by a fluorophore when it returns from an excited state to its ground state. The signal can be generated by the intrinsic fluorescence of a target (autofluorescence), a fluorescent label, or an environment-sensitive probe whose fluorescence properties change upon binding (e.g., molecular beacons). Their high sensitivity makes them suitable for monitoring specific protein expression, intracellular metabolite levels, or the presence of contaminating pathogens in a production line [10] [12].
Experimental Protocol for Intracellular Metabolite Sensing
Objective: To monitor the concentration of an intracellular metabolite (e.g., NADH) in microbial cells during fermentation using fluorescence.
Principle: The coenzyme NADH is intrinsically fluorescent, while its oxidized form NAD⁺ is not. By measuring the fluorescence intensity of NADH (excitation ~340 nm, emission ~460 nm), one can infer the metabolic state and energy balance of the cells in real-time, a key parameter in fermentation control [12].
Materials:
- Spectrofluorometer or fluorescence-capable bioreactor probe
- Microorganism culture (e.g., Saccharomyces cerevisiae)
- Fermentation medium
- Standard solutions of NADH for calibration
- Centrifuge and filters for sample preparation (if using offline method)
Procedure:
- System Setup:
- Online: Install a fluorescence probe directly into the bioreactor, ensuring proper sterilization (e.g., autoclaving).
- Offline: Set up a spectrofluorometer with a cuvette and temperature control.
- Calibration: Prepare a series of NADH standards in a buffer that mimics the fermentation broth. Measure the fluorescence intensity of each standard to create a calibration curve, accounting for potential quenching from broth components.
- Measurement:
- Online: During fermentation, continuously monitor the fluorescence signal from the probe. The signal correlates with the intracellular NADH pool.
- Offline: At regular intervals, aseptically withdraw samples from the bioreactor. Centrifuge immediately to separate cells. Resuspend the cell pellet in a buffer and measure fluorescence in the spectrofluorometer.
- Data Analysis: Correlate the fluorescence intensity measurements with the NADH concentration using the calibration curve. Plot the NADH levels over time to observe metabolic shifts (e.g., between oxidative and reductive metabolism) during different fermentation phases.
Diagram 2: Jablonski diagram illustrating fluorescence principle
Fiber Optic Biosensing
Application Note
Fiber optic biosensors integrate the sensing function into an optical fiber, allowing for miniaturization, remote sensing, and operation in electrically harsh environments due to their immunity to electromagnetic interference (EMI) [11]. They are ideal for in-situ placement directly into bioreactors for continuous monitoring. The optical fiber can simply act as a light guide to and from a remote sensing region, or the fiber itself can be modified to function as the transducer (e.g., by removing the cladding to create an evanescent wave sensor). In fermentation, they are widely used for monitoring critical parameters like pH, dissolved oxygen (DO), glucose, and ethanol in real-time, enabling precise feedback control for optimized product yield [6] [11].
Experimental Protocol for In-situ Bioprocess Monitoring
Objective: To continuously monitor a key biochemical parameter (e.g., glucose concentration) directly within a bioreactor using a fiber optic biosensor.
Principle: An enzyme (e.g., Glucose Oxidase, GOD) is immobilized on the tip or side of an optical fiber. The enzymatic reaction (glucose + O₂ → gluconic acid + H₂O₂) consumes oxygen, which can be transduced into an optical signal via an oxygen-sensitive fluorescent dye (e.g., a ruthenium complex) co-immobilized with the enzyme. The fluorescence intensity or lifetime of the dye is quenched by oxygen, providing an inverse measure of glucose concentration [11].
Materials:
- Fiber optic spectrometer or fluorescence lifetime analyzer
- Optical fibers (e.g., silica multimode)
- Oxygen-sensitive fluorescent dye (e.g., Tris(2,2'-bipyridyl)dichlororuthenium(II) hexahydrate)
- Enzyme (e.g., Glucose Oxidase)
- Immobilization matrix (e.g., sol-gel, hydrogel like polyacrylamide)
- Coupling reagents (e.g., glutaraldehyde)
- Bioreactor with standard ports for probe insertion
- Glucose standards for calibration
Procedure:
- Probe Fabrication:
- Prepare a solution containing the oxygen-sensitive dye and Glucose Oxidase in a sol-gel precursor solution or hydrogel monomer solution.
- Dip the tip of the optical fiber into this solution and allow the matrix to polymerize or gelate, forming a thin sensing layer at the fiber tip.
- Alternatively, bind the enzyme to the fiber surface via covalent chemistry using cross-linkers like glutaraldehyde.
- Sterilization: Sterilize the fabricated probe following manufacturer guidelines (e.g., chemical sterilization with ethanol, or gamma radiation if compatible with the sensing chemistry). Autoclaving may degrade the biological component.
- Calibration: Calibrate the probe before insertion into the bioreactor. Immerse the probe in a sterile buffer and measure the fluorescence response at different known glucose concentrations under controlled temperature and oxygen levels.
- In-situ Monitoring: Aseptically insert the probe into a dedicated port on the bioreactor. Connect the other end of the fiber to the readout instrument (spectrometer or lifetime analyzer). Continuously monitor the fluorescence signal throughout the fermentation process.
- Data Analysis: Relate the measured fluorescence intensity or lifetime to the glucose concentration using the calibration model, accounting for any cross-sensitivity to dissolved oxygen. The signal provides a real-time profile of glucose consumption.
Table 3: Research Reagent Solutions for Fiber Optic Glucose Sensing
| Item Name | Function/Description | Critical Notes |
|---|---|---|
| Silica Multimode Fiber | Transmits light to and from the sensing region. | Core/cladding structure guides light via total internal reflection. |
| Ruthenium Complex Dye | Oxygen-sensitive indicator; fluorescence is quenched by O₂. | Enables transduction of enzymatic O₂ consumption into optical signal. |
| Glucose Oxidase (GOD) | Biorecognition element; catalyzes oxidation of glucose. | Specificity for glucose is critical. Stability at fermentation T° is key. |
| Sol-Gel Matrix | Porous silica glass used to entrap the dye and enzyme. | Provides mechanical stability and biocompatibility for the sensing layer. |
The Critical Role of Real-Time Monitoring in Modern Fermentation Processes
In the realm of modern bioprocessing, real-time monitoring has emerged as a cornerstone technology, transforming fermentation from a largely empirical practice into a data-driven science. The ability to track critical process parameters (CPPs) and key performance indicators (KPIs) in real-time is indispensable for ensuring product quality, optimizing yield, and maintaining process consistency across scales [6]. This is particularly crucial in regulated industries like pharmaceutical drug development, where the Quality by Design (QbD) framework mandates a thorough understanding of processes to ensure the quality of biologics, vaccines, and other therapeutic agents [13].
Optical biosensors represent a revolutionary tool in this context. These devices combine a biological recognition element (such as an enzyme, antibody, or nucleic acid) with an optical transducer that converts a biological binding event into a quantifiable optical signal [4] [3]. This capability allows for the specific, sensitive, and non-invasive monitoring of a wide array of analytes directly within the complex matrix of a fermentation broth, providing unprecedented insight into cellular physiology and process performance [14]. This document details the application of advanced optical biosensors for real-time monitoring, providing structured protocols and data analysis frameworks to enhance fermentation process development and control.
Key Analytes and Optical Biosensor Modalities
The effective monitoring of a fermentation process hinges on the precise measurement of specific biochemical and physical parameters. The table below summarizes the key analytes targeted by optical biosensors and the primary sensing modalities employed.
Table 1: Key Analytes for Real-Time Fermentation Monitoring and Corresponding Optical Biosensor Modalities
| Analyte Category | Specific Examples | Optical Biosensor Modality | Significance in Fermentation |
|---|---|---|---|
| Gases | Dissolved Oxygen (DO), Carbon Dioxide (CO₂) | Fluorescence-based sensors (e.g., oxygen quenching), Fiber-optic sensors [14] [15] | Indicators of metabolic activity and cell health; critical for aerobic and anaerobic process control. |
| Metabolites | Glucose, Lactate, Glutamine, Ethanol | Enzyme-based optical biosensors (fluorescence, chemiluminescence) [6] [16] | Track substrate consumption and product formation; enable fed-batch strategy optimization. |
| Biomass | Cell Density, Cell Viability | Optical density (OD) probes, In-situ microscopy, NAD(P)H fluorescence [14] | Direct measure of growth; basis for calculating specific rates. |
| Ions & pH | H⁺ (pH), NH₄⁺ | Fiber-optic sensors with pH-sensitive dyes (e.g., HPTS) [15] | Impact cellular metabolism and product stability; require sterilisable probes. |
| Metabolic Intermediates | Specific intracellular metabolites | Genetically encoded biosensors (e.g., FRET-based) [6] | Provide insight into flux through metabolic pathways; used for strain engineering. |
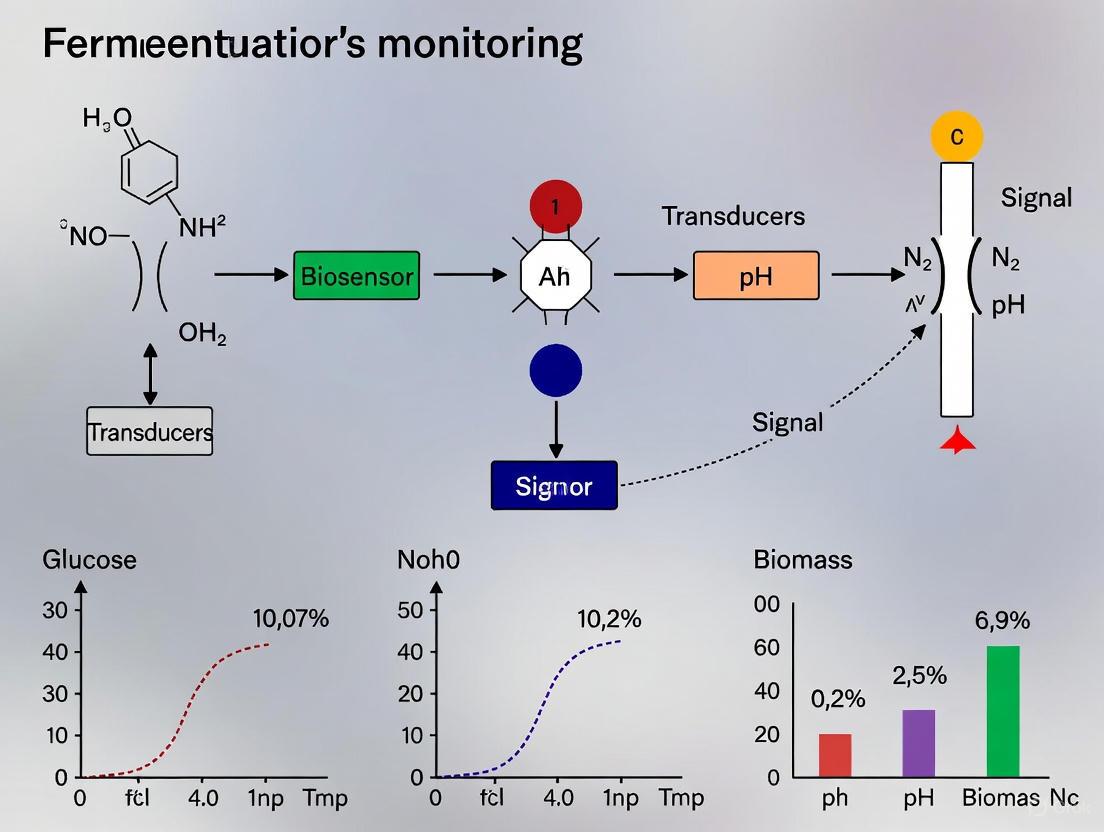
The working principle of an optical biosensor involves a cascade of events from recognition to signal output. The following diagram illustrates the generic workflow and signal transduction pathway.
Experimental Protocols for Real-Time Monitoring
Protocol: Real-Time Monitoring of Dissolved Oxygen and Carbon Dioxide Using Fiber-Optic Sensors
Principle: This protocol utilizes fiber-optic sensors for dissolved oxygen (DO) and carbon dioxide (pCO₂). The DO sensor is based on the fluorescence quenching of a dye by oxygen molecules [14]. The pCO₂ sensor operates on the Severinghaus principle, where CO₂ diffuses across a permeable membrane, causing a pH change in a bicarbonate buffer that is detected by a pH-sensitive fluorescent dye (e.g., Hydroxypyrenetrisulfonic acid, HPTS) [15].
Materials:
- Bioreactor system (e.g., Sartorius Biostat, Eppendorf BioFlo)
- Fiber-optic DO sensor (e.g., PreSens Fibox 4, Ocean Optics NeoFox)
- Fiber-optic pCO₂ sensor (e.g., based on HPTS dye [15])
- Calibration standards for DO (0% and 100% air saturation) and pCO₂ (known CO₂/N₂ gas mixtures)
- Data acquisition software
Procedure:
- Pre-sterilization Calibration: Calibrate the fiber-optic DO sensor in a sterile vessel using a two-point calibration (0% DO via nitrogen sparging and 100% DO via air saturation). Calibrate the pCO₂ sensor using at least two gas mixtures with known pCO₂ (e.g., 0.05 atm and 0.20 atm) [15].
- Sensor Installation and Sterilization: Aseptically install the sensor probes into the bioreactor ports designated for in-situ probes. Many modern fiber-optic sensors are designed to withstand in-situ steam sterilization (e.g., 121°C, 20 minutes). Verify sensor stability and calibration post-sterilization.
- On-line Monitoring: Initiate the fermentation process. The sensor control unit will continuously excite the fluorescent dyes and measure the emitted light intensity or lifetime. Data is recorded in real-time by the bioreactor control system.
- Data Acquisition: Record DO (%) and pCO₂ (atm or %) at a frequency of ≥1 Hz. Correlate these values with other process parameters like agitation, aeration, and substrate feed rates.
- Post-run Validation: After the run, validate sensor performance by checking the calibration against a reference method if available.
Protocol: Monitoring Metabolites Using Enzyme-Based Optical Biosensors in a Flow-Injection Analysis (FIA) System
Principle: This protocol describes the off-line/at-line monitoring of key metabolites (e.g., glucose, lactate) using an enzyme-based optical biosensor integrated into a Flow-Injection Analysis (FIA) system [16]. The analyte in a sampled broth reacts with an immobilized enzyme (e.g., Glucose Oxidase), producing a product (e.g., H₂O₂) that is detected optically via a chemiluminescence or fluorescence reaction.
Materials:
- Automated sample collector or autosampler
- Flow-Injection Analysis (FIA) system with peristaltic pump and injection valve
- Enzyme reactor column (immobilized oxidase enzymes)
- Chemiluminescence or fluorescence detector (e.g., photomultiplier tube)
- Reagents: Buffer, luminol/peroxidase mix (for chemiluminescence)
Procedure:
- System Setup: Configure the FIA system with the enzyme reactor column placed upstream of the optical detector. Establish a stable buffer flow rate (e.g., 0.5 - 1.0 mL/min).
- Calibration: Create a standard curve by injecting a series of known analyte concentrations (e.g., 0.1, 0.5, 1.0, 5.0 g/L glucose) into the FIA stream. Record the peak height or area of the optical signal.
- Sample Collection and Preparation: Automatically or manually collect samples from the bioreactor at defined intervals (e.g., every 30 minutes). Immediately centrifuge samples to remove cells and dilute the supernatant if necessary to fit within the calibration range.
- Automated Analysis: Inject the prepared sample into the FIA stream. The analyte reacts in the enzyme column, and the resulting product generates an optical signal proportional to its concentration.
- Data Processing: The control software converts the signal peak into a concentration value based on the calibration curve. Data can be fed back to the bioreactor control system to adjust nutrient feeds in a feedback loop.
The Scientist's Toolkit: Research Reagent Solutions
The following table lists essential reagents and materials critical for implementing optical biosensing in fermentation monitoring.
Table 2: Essential Research Reagents and Materials for Optical Biosensing in Fermentation
| Item Name | Function/Brief Explanation | Example Application |
|---|---|---|
| Hydroxypyrenetrisulfonic Acid (HPTS) | A pH-sensitive fluorescent dye entrapped behind a gas-permeable membrane for pCO₂ sensing [15]. | Fiber-optic carbon dioxide sensor. |
| Ruthenium Complexes | Oxygen-sensitive fluorescent dyes whose fluorescence is quenched in the presence of O₂ [14]. | Dissolved oxygen sensing via phase-fluorometry. |
| Immobilized Oxidase Enzymes | Biological recognition element (e.g., Glucose Oxidase) that provides specificity to the target metabolite [16] [3]. | Enzyme-based optical biosensors in FIA systems. |
| Gold Nanoparticles | Nanomaterial used to enhance optical signals via Localized Surface Plasmon Resonance (LSPR) [4]. | Increasing sensitivity of SPR-based pathogen sensors. |
| Quantum Dots | Semiconductor nanocrystals with superior brightness and photostability used as fluorescent labels [4]. | High-sensitivity fluorescence-based immunoassays. |
| Gas-Permeable Membranes (e.g., PTFE) | Allows selective diffusion of gaseous analytes (O₂, CO₂) while excluding ions and macromolecules [15]. | Protecting the sensing chemistry in DO and pCO₂ probes. |
Data Management, Analysis, and Integration with Control Systems
The high-frequency, multi-parameter data generated by optical biosensors requires robust management and analysis to be actionable. The following workflow outlines the path from raw data to process control.
Data Pre-processing: Raw optical signals often require preprocessing to remove noise and correct for baseline drift. Techniques include Savitzky-Golay filtering and moving average smoothing [3].
Multivariate Data Analysis (MVDA): Advanced chemometric methods like Principal Component Analysis (PCA) and Partial Least Squares (PLS) regression are used to extract meaningful information from complex datasets. For instance, PLS models can correlate real-time optical data (e.g., NIR spectra) with critical quality attributes (CQAs) of the product, enabling soft-sensor approaches [14] [13].
Integration with AI and Control Systems: The integration of optical biosensor data with Internet of Things (IoT) platforms and Artificial Intelligence (AI) is a key trend. Machine learning algorithms can analyze historical and real-time data to predict fermentation trajectories, identify deviations, and recommend adjustments. This enables adaptive feedback control for feeding strategies or environmental parameters, moving towards fully autonomous, self-optimizing bioreactors [6] [13].
The adoption of optical biosensors for real-time monitoring marks a significant leap forward in the control and optimization of fermentation processes. The protocols and frameworks outlined in this document provide a practical roadmap for researchers and drug development professionals to implement these powerful technologies. By enabling non-invasive, specific, and continuous measurement of CPPs and KPIs, optical biosensors facilitate a deeper process understanding, which is the foundation of QbD. The ongoing convergence of biosensor technology with AI and advanced data analytics promises to further revolutionize bioprocessing, paving the way for more efficient, robust, and intelligent manufacturing of next-generation therapeutics.
The global biologics market is experiencing unprecedented growth, driven by the rising prevalence of chronic diseases and the superior efficacy of targeted therapies [17]. This expansion is concurrently propelling advances in biomanufacturing, with precision fermentation emerging as a critical production technology for a new generation of complex biomolecules [18] [19]. Monitoring and controlling these sophisticated biological processes requires equally advanced analytical tools. Optical biosensors have thereby become indispensable in fermentation monitoring research, providing the real-time, non-invasive data necessary to ensure product quality, optimize yields, and accelerate the commercialization of novel biologics [4] [19] [20].
This application note details the integration of optical biosensors within precision fermentation platforms, framed by the powerful market forces driving the biologics sector. It provides structured quantitative market data, detailed experimental protocols for biosensor implementation, and visualizations of core workflows to support researchers and drug development professionals in this rapidly evolving field.
Market Analysis: The Biologics and Precision Fermentation Landscape
Global Biologics Market Projections
The biologics market is on a strong growth trajectory, fueled by demand for monoclonal antibodies, vaccines, and personalized therapies for complex conditions like cancer and autoimmune diseases [21] [17].
Table 1: Global Biologics Market Size and Growth Forecasts
| Market Size Year | Market Size Value | Projected Year | Projected Value | CAGR | Source/Region Highlights |
|---|---|---|---|---|---|
| 2025 | USD 487 Billion | 2034 | USD 1,144.20 Billion | 9.96% | Precedence Research [21] |
| 2025 | USD 450.2 Billion | 2035 | USD 1,077.2 Billion | 9.1% | Future Market Insights [22] |
| 2024 | USD 445.2 Billion | North America held 44.53% share [21] |
Key Market Drivers and Segment Analysis
Several interrelated factors are creating a powerful push for biologics and the advanced manufacturing platforms that produce them.
- Therapeutic Efficacy and Demand: Biologics, particularly monoclonal antibodies (mAbs), offer targeted action with superior efficacy and safety profiles compared to conventional small-molecule drugs. This drives their adoption for chronic diseases, a burden exacerbated by an aging global population [17] [22].
- R&D Investment and Innovation: Sustained investment in biopharmaceutical R&D is accelerating the development of next-generation biologics, including antibody-drug conjugates (ADCs) and cell and gene therapies, which often require advanced production platforms like precision fermentation [21] [17].
- The Rise of Precision Fermentation: This technology uses engineered microbes for efficient, scalable, and cost-effective production of complex biomolecules. The biopharmaceutical fermentation market is expected to grow from USD 10.17 billion in 2023 to USD 16.03 billion by 2031 [19]. Its advantages over traditional mammalian cell culture include shorter production cycles and lower costs, making it ideal for smaller biologics like peptides and antibody fragments [18] [19].
Table 2: Key Biologics Market Drivers and Segment Analysis
| Driver/Segment | Key Findings | Quantitative Data |
|---|---|---|
| Leading Drug Class | Monoclonal Antibodies | 65.1% revenue share [22] |
| Leading Application | Oncology | 32.4% revenue share [22] |
| Fastest Growing Region | Asia Pacific | CAGR of 11.05% (2025-2034) [21] |
| Precision Fermentation Market | Rapid Growth | CAGR of 38.73% (from USD 293.75M in 2021) [18] |
Application in Fermentation Monitoring: Optical Biosensor Technology
Principles and Relevance
Optical biosensors are analytical devices that combine a biorecognition element with an optical transducer, converting a biological interaction into a quantifiable optical signal [4] [23]. Their relevance in precision fermentation is paramount: they enable real-time, in-situ monitoring of Critical Process Parameters (CPPs), which is essential for maintaining product consistency and quality in a highly variable biological system [19] [14].
Biosensor Types and Applications
The choice of biosensor depends on the target analyte and required sensitivity.
- Surface Plasmon Resonance (SPR): Effective for label-free, real-time analysis of biomolecular interactions (e.g., antigen-antibody binding). Applications include monitoring product titers or detecting contaminants [4] [20].
- Fluorescence and Chemiluminescence-Based Sensors: Offer high sensitivity for detecting low-abundance analytes like specific proteins or toxins. They function by measuring changes in fluorescence intensity or luminescence upon binding [4] [20].
- Fiber-Optic Sensors: Utilize optical fibers to guide light, making them ideal for in-line monitoring within bioreactors. Variants like Fiber-Optic SPR (FO-SPR) enhance sensitivity in complex media like fermentation broth [4] [20] [14].
Experimental Protocols
This section provides a detailed methodology for implementing an optical biosensor system for real-time monitoring in a precision fermentation process.
Protocol 1: SPR-Based Monitoring of a Recombinant Protein Titer
1. Objective: To continuously monitor the concentration of a target recombinant protein during a microbial fermentation process using an SPR biosensor.
2. Materials
- Bioreactor system (e.g., 5 L benchtop fermenter)
- In-line or at-line SPR biosensor system (e.g., with a flow cell)
- Specific antibody or binding protein for the target recombinant product
- Immobilization reagents (e.g., CMS chip, EDC/NHS coupling reagents)
- Running buffer (e.g., HBS-EP, pH 7.4)
- Regeneration solution (e.g., 10 mM Glycine-HCl, pH 2.0)
- Sterile sampling kit
3. Experimental Workflow
4. Procedure
- Sensor Preparation: Immobilize the specific capture antibody onto the SPR chip surface using standard amine-coupling chemistry.
- Calibration: Create a calibration curve by injecting purified target protein at known concentrations and measuring the Response Units (RU) at equilibrium.
- Fermentation & Monitoring: Initiate the fermentation. Continuously divert a small, sterile stream of broth (centrifuged if necessary to remove cells) through the SPR flow cell.
- Binding Cycle:
- Sample Injection: Pass the conditioned broth over the sensor surface for 3-5 minutes.
- Buffer Wash: Switch to running buffer to remove unbound molecules and non-specifically adsorbed material.
- Regeneration: Inject a short pulse (30-60 seconds) of regeneration solution to dissociate the bound target and regenerate the antibody surface for the next cycle.
- Data Analysis: The SPR software will generate a sensorgram. The shift in RU is proportional to the mass bound, allowing for real-time calculation of product titer.
Protocol 2: Fluorescence-Based Monitoring of Cellular Metabolites
1. Objective: To use a fluorescent biosensor to track the concentration of a key metabolite (e.g., NAD(P)H) as an indicator of cellular metabolic state.
2. Materials
- Bioreactor with integrated fluorescence probe port
- In-situ fluorescence sensor (capable of ~360 nm excitation / ~450 nm emission)
- Calibration standards
- E. coli or S. cerevisiae strain in defined media
3. Procedure
- Sensor Installation and Calibration: Install the fluorescence probe according to the manufacturer's guidelines. Perform a baseline calibration.
- Process Initiation: Inoculate the bioreactor and begin the fermentation process under defined conditions.
- Real-Time Monitoring: The sensor continuously excites the broth at 360 nm and detects the fluorescent emission at 450 nm, which is primarily associated with intracellular NAD(P)H.
- Data Correlation: Correlate the fluorescence signal with offline measurements (e.g., cell density, substrate concentration, product titer) to build a model for predicting the metabolic state of the culture. A sudden drop in NAD(P)H fluorescence may indicate substrate depletion or a shift in metabolic pathway.
The Scientist's Toolkit: Essential Research Reagents and Materials
Table 3: Key Research Reagent Solutions for Optical Biosensing in Fermentation
| Item | Function & Application | Example |
|---|---|---|
| SPR Sensor Chips | Solid support for immobilizing biorecognition elements (antibodies, aptamers). | Carboxymethyl dextran (CM5) gold chips |
| Bio-Specific Ligands | Provides selectivity by binding the target analyte with high affinity. | Monoclonal Antibodies, Engineered Nanobodies, DNA Aptamers |
| Fluorescent Dyes & Probes | Act as transducers for fluorescence-based sensors, changing optical properties upon analyte binding. | NAD(P)H (native fluorescence), Oxygen-sensitive dyes (e.g., Ruthenium complexes), pH-sensitive dyes (e.g., SNARF) |
| Immobilization Chemistry Kits | Enables covalent attachment of ligands to the sensor surface. | Amine-coupling kits (EDC/NHS), Thiol-coupling kits |
| Optical Fiber Probes | Enable in-situ light delivery and collection directly within the bioreactor. | Fiber-optic probes for DO/pH monitoring |
| Nanoparticles for Signal Enhancement | Used in SERS and LSPR biosensors to amplify the optical signal. | Gold Nanoparticles (AuNPs), Silver Nanoparticles (AgNPs) |
Technology Integration Pathway
The successful application of optical biosensors relies on their integration within a holistic framework that includes process analytical technology (PAT) and data management systems [19].
The diagram illustrates the synergistic relationship between a Quality-by-Design (QbD) framework, which defines Critical Quality Attributes (CQAs) and the Target Product Profile (TPP), and the deployment of PAT tools like optical biosensors to monitor CPPs [19]. The real-time data from these sensors feeds into advanced modeling and digital tools (e.g., digital twins), enabling deeper process understanding and facilitating automated process control for consistent, high-yield production [19].
Optical biosensors have emerged as a transformative technology in fermentation monitoring, offering a suite of analytical advantages that address critical limitations of conventional methods. These advantages are particularly defined by three core capabilities: label-free detection, product-specific analysis, and real-time monitoring [24] [25]. Unlike traditional analytical techniques such as HPLC or ELISA, which often require extensive sample preparation, labeling, and result in destructive endpoint analysis, optical biosensors enable direct, non-invasive observation of the fermentation process [24]. This allows researchers to obtain immediate, quantitative data on critical quality attributes, such as the concentration of a target product like glycopeptide antibiotics, without interrupting the process [25]. The intrinsic versatility of optical biosensing platforms facilitates their integration into bioreactors, providing a promising route toward sophisticated, automated process control for advanced biomanufacturing [26] [27].
Theoretical Advantages & Comparative Analysis
The operational benefits of optical biosensors stem from their fundamental principles of detection. The table below summarizes these key advantages and contrasts them with the limitations of traditional methods.
Table 1: Comparative Analysis of Optical Biosensors vs. Traditional Methods in Fermentation Monitoring
| Analytical Feature | Optical Biosensors (e.g., RIfS, SPR) | Traditional Methods (e.g., HPLC, ELISA) |
|---|---|---|
| Detection Mode | Label-free, direct measurement of binding events [24] [28] | Often requires fluorescent or enzymatic labels |
| Temporal Resolution | Real-time, continuous monitoring [24] [25] | Discrete, off-line sampling; long turnaround times [26] |
| Specificity | Product-specific, via immobilized biorecognition elements (e.g., antibodies) [25] | Can be specific, but often requires separation steps |
| Sample Preparation | Minimal; can often handle complex matrices like fermentation broth [4] | Extensive; often requires purification, leading to sample destruction |
| Automation Potential | High; suitable for inline integration and process control [25] | Low; primarily manual, off-line operations |
| Throughput | High for continuous monitoring | Low to medium due to manual and sequential processing |
Signaling Pathway and Detection Logic
The following diagram illustrates the fundamental signaling pathway and logical relationships in a label-free optical biosensor, such as one based on Reflectometric Interference Spectroscopy (RIfS).
Experimental Protocol: Monitoring Glycopeptide Antibiotic Fermentation
This protocol details the application of a Reflectometric Interference Spectroscopy (RIfS) optical biosensor for the qualitative and quantitative monitoring of vancomycin-type glycopeptide antibiotics during a microbial fermentation process, as adapted from a foundational study [25].
Experimental Workflow
The integrated process of fermentation and biosensor analysis involves the following key steps:
Detailed Methodology
Phase 1: Biosensor Preparation and Functionalization
- Sensor Surface Activation: Clean the RIfS sensor chip (typically a SiO₂-coated substrate) using an oxygen plasma cleaner for 5 minutes to generate reactive hydroxyl groups [24].
- Immobilization of Biorecognition Element: Employ a silane-based covalent chemistry (e.g., (3-Aminopropyl)triethoxysilane, APTES) to create an amine-functionalized surface. Subsequently, immobilize an anti-vancomycin monoclonal antibody (~100 µg/mL in 10 mM acetate buffer, pH 5.0) onto the activated surface using standard amine-coupling reagents (e.g., EDC/NHS). Aim for an immobilization density that yields a robust signal without causing steric hindrance [25].
- Surface Blocking: Passivate any remaining active esters on the sensor surface by injecting a 1.0 M ethanolamine hydrochloride solution (pH 8.5) for 10 minutes to minimize non-specific binding in subsequent steps [4].
Phase 2: Fermentation Monitoring Setup
- Integration with Bioreactor: Connect the functionalized RIfS biosensor to the fermentation bioreactor via an at-line or inline flow system. This typically involves a peristaltic pump and tubing that draws samples from the bioreactor into a flow cell housing the sensor chip [25].
- System Priming: Prime the entire fluidic path with a sterile running buffer (e.g., 10 mM PBS, pH 7.4) to establish a stable optical baseline before introducing the fermentation broth.
Phase 3: Quantitative Analysis and Calibration
- Calibration Curve Generation: Prior to fermentation monitoring, perform a calibration by injecting a series of known concentrations of purified vancomycin standard (e.g., 0.1, 1, 10, 100 µg/mL) in running buffer over the sensor surface.
- Data Recording: Record the RIfS signal (e.g., interference pattern shift in nm or response units) for each concentration. The signal is proportional to the mass of analyte bound to the sensor surface [25].
- Regression Analysis: Plot the maximum response for each standard against its concentration and fit with a suitable model (e.g., 4-parameter logistic fit) to create the quantitative calibration curve.
Phase 4: Real-Time Fermentation Monitoring
- Initiating Monitoring: Once the microbial fermentation has progressed to the expected production phase (e.g., after 24 hours), initiate the continuous or periodic sampling from the bioreactor through the biosensor flow cell.
- Signal Acquisition: Monitor the RIfS signal in real-time. The binding of vancomycin from the broth to the immobilized antibody will cause a shift in the interference pattern, which is directly recorded [25].
- Data Processing: Convert the acquired signal into product concentration values using the pre-established calibration curve. This provides a continuous, quantitative profile of antibiotic production throughout the fermentation run.
Phase 5: Post-Run Sensor Regeneration
- Surface Regeneration: After each measurement cycle, regenerate the sensor surface for reuse by injecting a mild regeneration solution (e.g., 10 mM glycine-HCl, pH 2.5) for 60 seconds to dissociate the bound vancomycin without damaging the immobilized antibody. Re-equilibrate with running buffer before the next sample injection [24].
Quantitative Performance Data
The performance of optical biosensors in fermentation and other applications can be quantified through key metrics. The data below, synthesized from the provided sources, highlights their capabilities and growing market impact.
Table 2: Quantitative Performance Metrics of Optical Biosensors
| Performance / Market Metric | Reported Value or Capability | Context & Application |
|---|---|---|
| Detection Limit | Down to 26.6 fg/mL (for VEGF) [29] | Demonstrates potential for extreme sensitivity in biomarker detection. |
| Detection Limit (Other Targets) | 5.98 ng/mL (for Lipocalin 1) [29] | Shows robust performance for different protein classes. |
| Market Growth (CAGR) | 8.1% (2023-2032) [30] | Robust growth of the overall optical biosensor market. |
| Fiber Optic Biosensor Market Growth (CAGR) | 10.2% (2024-2033) [31] | Specific segment showing even faster adoption. |
| Key Advantage | Real-time, label-free analysis [25] | Enables direct monitoring of fermentation products. |
The Scientist's Toolkit: Essential Research Reagent Solutions
Successful implementation of the described protocol relies on a set of key reagents and materials. The following table details these essential components and their functions.
Table 3: Key Research Reagent Solutions for Optical Biosensor-Based Fermentation Monitoring
| Reagent / Material | Function / Role in the Experiment | Critical Specifications |
|---|---|---|
| RIfS or SPR Biosensor Chip | The solid support and optical transducer that converts binding events into a measurable signal [24]. | SiO₂ coating for RIfS; gold film for SPR; functional groups for immobilization. |
| Specific Biorecognition Element | Provides product-specificity by binding exclusively to the target analyte (e.g., vancomycin) [25]. | High-affinity and specificity monoclonal antibody, DNA aptamer, or molecularly imprinted polymer. |
| Silane Coupling Agent (e.g., APTES) | Creates a functionalized monolayer on the sensor chip for covalent attachment of the biorecognition element [24]. | High purity (>95%), appropriate reactive groups (e.g., amine, epoxy). |
| Amine-Coupling Kit (EDC/NHS) | Activates carboxyl groups or enables cross-linking for stable immobilization of proteins like antibodies [4]. | Freshly prepared solutions for optimal activation efficiency. |
| Analyte Standard (Pure) | Used for generating the calibration curve for quantitative analysis [25]. | High-purity (>98%) reference standard of the target molecule (e.g., vancomycin). |
| Regeneration Buffer | Dissociates the bound analyte from the immobilized receptor, allowing for sensor surface reuse [24]. | Must effectively break bonds without denaturing the bioreceptor (e.g., low pH, high ionic strength). |
Optical Biosensing Methods and Real-World Biopharmaceutical Applications
Reflectometric Interference Spectroscopy (RIfS) for Glycopeptide Antibiotic Monitoring
Reflectometric Interference Spectroscopy (RIfS) is a label-free optical biosensor technology that enables the real-time monitoring of biomolecular interactions by detecting changes in the optical thickness of a sensor surface [32]. This application note details the use of RIfS for qualitative and quantitative monitoring of glycopeptide antibiotic fermentation processes, providing researchers with detailed protocols for implementing this technology in bioprocess control and optimization [25]. The non-destructive, real-time capabilities of RIfS make it particularly valuable for monitoring critical quality attributes in pharmaceutical fermentation, supporting the advancement of intelligent bio-manufacturing through precise biochemical sensing [6].
Fundamental Principles of RIfS Technology
RIfS operates based on the interference patterns of white light reflected from thin transparent films. When light encounters the phase boundaries of a thin sensor layer, partial beams are reflected and superimposed, creating a characteristic interference spectrum [33]. As biological binding events occur on the sensor surface, they alter the optical thickness (refractive index × physical thickness), resulting in a quantifiable shift in the interference pattern that can be monitored in real-time [33] [32].
This detection mechanism enables RIfS to track binding kinetics without requiring fluorescent or radioactive labels, preserving the native state of the interacting molecules [32]. The technology is particularly suited for fermentation monitoring as it accommodates complex sample matrices and provides continuous data on product formation and quality attributes throughout the bioprocess [25].
Application to Glycopeptide Antibiotic Fermentation
Vancomycin Monitoring Case Study
RIfS has been successfully implemented for monitoring vancomycin-type glycopeptide antibiotics during microbial fermentation processes [25]. In this application, the biosensor enables both qualitative identification and quantitative measurement of target antibiotics directly from fermentation broths, facilitating real-time process control decisions.
Table 1: Performance Characteristics of RIfS for Glycopeptide Antibiotic Monitoring
| Parameter | Specification | Experimental Conditions |
|---|---|---|
| Detection Principle | Label-free, based on optical thickness changes | White light interference at thin films [33] |
| Target Analytes | Vancomycin-type glycopeptide antibiotics | Microbial fermentation broths [25] |
| Monitoring Capability | Qualitative and quantitative analysis | Real-time process monitoring [25] |
| Key Advantage | Product-specific monitoring without labels | Enables on-line process control [25] |
| Technology Platform | Reflectometric Interference Spectroscopy (RIfS) | Suitable for bioprocess control [25] |
Quantitative Analysis of Binding Kinetics
The quantitative assessment of binding interactions is fundamental to RIfS applications. For glycopeptide antibiotic analysis, evaluation of binding curves following pseudo-first-order kinetics enables determination of critical kinetic parameters [34] [33]. Research demonstrates that multiple mathematical approaches can be applied to evaluate these binding curves, including linear transformation of primary data using derivatives or integrals, and the integrated rate equation yielding exponential functions [33].
Diagram 1: RIfS Working Principle. The diagram illustrates the sequential process from illumination to detection in RIfS biosensing.
Experimental Protocols
RIfS Transducer Functionalization
Objective: Prepare RIfS transducers with appropriate surface chemistry for glycopeptide antibiotic monitoring [33].
Materials:
- RIfS glass transducers (1 cm × 1 cm) with 10 nm Ta₂O₅ and 330 nm SiO₂ layers [33]
- 3-glycidyloxypropyl-trimethoxysilane (GOPTS)
- Poly(ethylene glycol) diamine (PEG-DA, MW 2000 Da) and ɑ-methoxy-ω-amino PEG (PEG-MA, MW 2000 Da)
- Glutaric acid (GA)
- N,N'-diisopropyl-carbodiimide (DIC) and N-hydroxysuccinimide (NHS)
- Target ligand (e.g., glycopeptide antibiotic or binding partner)
- Organic solvents: dichloromethane (DCM), DMF, acetone
Procedure:
- Transducer Cleaning: Clean transducers for 30 seconds in 6 M KOH solution, followed by thorough washing with H₂O [33].
- Surface Activation: Immerse transducers in freshly prepared piranha solution (3:2 concentrated H₂SO₄:H₂O₂ 30%) for 15 minutes [33].
- Silanzation: After washing with H₂O and drying under nitrogen, modify transducers with GOPTS for 1 hour [33].
- PEG Modification: Clean GOPTS-functionalized transducers with acetone and dry under nitrogen. Apply polymer mixture of PEG-DA and PEG-MA (1:1000 ratio) in dichloromethane (4 mg/mL) and covalently bind to the GOPTS layer by reacting overnight at 70°C [33].
- Carboxyl Group Formation: Transfer amino functions of PEG-DA to carboxyl functions using glutaric acid solution (0.67 mg/μL in DMF) in a DMF vapor-saturated chamber for at least 6 hours [33].
- Ligand Immobilization: Activate carboxyl groups using NHS (150 mg/mL) and DIC (302 μL/mL) in DMF for 4 hours in DMF vapor-saturated chamber. After cleaning with DMF and acetone, incubate with ligand solution (2 mg/mL in H₂O) in water vapor-saturated chamber overnight [33].
RIfS Measurement for Fermentation Monitoring
Objective: Perform real-time monitoring of glycopeptide antibiotics during fermentation processes [25] [33].
Materials:
- Functionalized RIfS transducers
- RIfS instrument system with flow cell
- Phosphate-buffered saline (PBS: 150 mM sodium chloride, 10 mM potassium phosphate, pH 7.4)
- Fermentation broth samples or standard solutions
- Regeneration solution (guanidine hydrochloride, 6 M, pH 1.5) [33]
- Air supply for bubble separation
Procedure:
- System Setup: Install functionalized transducer in RIfS flow cell (typical dimensions: 50 μm depth × 1 mm width × 4 mm length) [33].
- Baseline Establishment: Flush transducer surface with PBS buffer until stable baseline is achieved [33].
- Sample Injection: Inject fermentation broth samples or standard solutions (concentration range: 33-500 nM for antibody analytes) at constant flow rate (0.5 μL/s) at room temperature [33].
- Association Phase: Monitor binding for 600 seconds while maintaining continuous flow [33].
- Dissociation Phase: Initiate dissociation by switching to buffer flow and monitor for 900 seconds [33].
- Surface Regeneration: Regenerate surface with 400-second injection of guanidine hydrochloride solution [33].
- Re-equilibration: Re-establish baseline with buffer flow before subsequent measurements [33].
Table 2: Research Reagent Solutions for RIfS Experimental Setup
| Reagent/Chemical | Function | Specifications/Notes |
|---|---|---|
| RIfS Transducers | Signal transduction platform | 1 cm × 1 cm glass with 10 nm Ta₂O₅ + 330 nm SiO₂ layers [33] |
| PEG Polymers | Bio-compatible surface matrix | PEG-DA & PEG-MA (MW 2000 Da), 1:1000 ratio [33] |
| GOPTS | Silane coupling agent | Enables covalent attachment of polymer matrix [33] |
| NHS/DIC | Activation reagents | For carboxyl group activation prior to ligand immobilization [33] |
| PBS Buffer | Running buffer | 150 mM sodium chloride, 10 mM potassium phosphate, pH 7.4 [33] |
| Guanidine HCl | Regeneration solution | 6 M, pH 1.5 for removing bound analytes [33] |
Data Analysis and Kinetic Parameter Calculation
Objective: Extract kinetic parameters from RIfS binding data for glycopeptide antibiotic quantification [34] [33].
Procedure:
- Data Preprocessing: Normalize binding curves and subtract reference signals.
- Model Selection: Apply pseudo-first-order kinetic model for 1:1 binding interactions [33].
- Parameter Calculation: Use multiple mathematical approaches for evaluation:
- Quality Assessment: Verify model consistency by comparing results from different evaluation approaches [33].
- Kinetic Constants: Determine association (kₐ) and dissociation (kd) rate constants, then calculate equilibrium constant (KD = k_d/kₐ) [33].
Diagram 2: RIfS Experimental Workflow. The cyclic process from surface preparation to data analysis enables continuous monitoring capabilities.
Critical Experimental Considerations
Optimization for Fermentation Monitoring
Successful implementation of RIfS for glycopeptide antibiotic fermentation monitoring requires careful attention to several critical parameters:
- Mass Transport Limitations: Use fast flow rates (≥0.5 μL/s) and minimize ligand immobilization level to reduce mass transport effects [33]. Verify absence of mass transport limitation by varying flow rate [33].
- Matrix Effects: Account for complex fermentation broth composition through appropriate controls and reference channels [4].
- Regeneration Conditions: Optimize regeneration conditions to completely remove bound analytes while maintaining ligand activity over multiple cycles [33].
- Temperature Control: Maintain constant temperature during measurements to prevent drift in interference signals [33].
Data Quality Assessment
Ensure data quality through the following verification steps:
- Perform triplicate injections of each sample to assess reproducibility [33].
- Evaluate consistency of results across different mathematical approaches [33].
- Verify that binding curves follow pseudo-first-order kinetics under the experimental conditions [33].
- Check for non-specific binding using appropriate control surfaces [32].
RIfS technology provides a powerful platform for monitoring glycopeptide antibiotic fermentation processes, enabling real-time, label-free, and product-specific quantification [25]. The experimental protocols outlined in this application note offer researchers a comprehensive framework for implementing RIfS in bioprocess monitoring and optimization. With proper surface chemistry, experimental design, and data analysis, RIfS can significantly enhance process control in pharmaceutical fermentation, contributing to the advancement of intelligent bio-manufacturing systems [6]. The technology's ability to provide kinetic information alongside concentration measurements makes it particularly valuable for quality assurance in antibiotic production.
The monitoring of fermentation processes is critical in the pharmaceutical, biotechnology, and food industries for ensuring product quality, yield, and process efficiency. Traditional monitoring methods often involve off-line sampling, which is discontinuous, time-consuming, and risks contamination. Fiber optic biosensors represent a transformative technology for in-situ fermentation monitoring, offering real-time, continuous, and non-invasive measurement of key analytes within the bioreactor environment [35] [36]. These sensors leverage the unique properties of optical fibers to guide light to and from a sensing region functionalized with a biorecognition element, translating a biological interaction into a quantifiable optical signal.
Framed within the broader thesis on the application of optical biosensors in fermentation monitoring research, this document details the specific design advantages of fiber optic configurations. Their miniaturization, flexibility, and immunity to electromagnetic interference make them ideally suited for integration into sterile, complex fermentation setups, enabling advanced process control and paving the way for more consistent and efficient biomanufacturing [35].
Key Design Advantages for Fermentation Monitoring
The intrinsic properties of optical fibers confer several distinct advantages for in-situ sensing in the challenging environment of a fermenter.
- Miniaturization and Lab-on-Fiber Technology: Optical fibers can be fabricated with extremely small diameters (microns), allowing for the development of miniaturized "labs-on-fiber." This enables multi-parameter sensing from a single, compact probe that causes minimal disruption to the fermentation broth and can be sterilized in-situ [35].
- Flexibility and Remote Sensing Capability: The flexible nature of optical fibers allows the sensing tip to be placed directly in the vessel, while the spectrometer and light source can be located remotely, sometimes hundreds of meters away. This protects sensitive instrumentation from the harsh conditions (e.g., high temperature, pressure, humidity) inside a fermenter and facilitates monitoring in multiple, separate bioreactors with a single analytical device [35].
- Immunity to Electromagnetic Interference: Unlike electrochemical sensors, fiber optic biosensors are based on light propagation and are immune to electromagnetic interference from stirrers, pumps, and other electrical equipment typically found in fermentation facilities. This results in a stable signal with a high signal-to-noise ratio [35] [36].
- Real-time and Continuous Monitoring: Fiber optic sensors enable real-time, continuous tracking of critical process parameters (CPPs) and critical quality attributes (CQAs). This provides a dynamic view of the fermentation process, allowing for proactive interventions and advanced control strategies based on live data trends, a significant improvement over the lag time associated with offline sampling [36].
- Multiplexing Potential: Different sensing regions on a single optical fiber can be functionalized to detect multiple analytes simultaneously (e.g., glucose, ethanol, lactate, and pH). This multiplexing capability provides a comprehensive view of the metabolic state of the culture from a single probe, simplifying the sensor architecture and reducing costs [35].
Sensing Mechanisms and Quantitative Performance
Fiber optic biosensors for fermentation monitoring employ various optical transduction mechanisms. The table below summarizes the primary types, their principles, and documented performance metrics for key fermentation analytes.
Table 1: Optical Transduction Mechanisms and Performance in Fermentation Monitoring
| Transduction Mechanism | Principle of Operation | Target Analyte (Example) | Reported Performance | Key Advantages |
|---|---|---|---|---|
| Surface-Enhanced Raman Scattering (SERS) | Enhancement of Raman signal by molecules adsorbed on nanostructured metal surfaces on the fiber [35]. | Unknown organics, proteins, metabolites | Extremely high sensitivity; capable of single-molecule detection [35]. | Provides unique molecular "fingerprint"; multiplexing capability. |
| Fluorescence | Measurement of changes in fluorescence intensity, lifetime, or wavelength of a fluorophore in response to analyte binding [37] [38]. | Ethanol, ions, metabolites | Continuous monitoring over >6 days; good reversibility [37]. | High sensitivity; wide range of available fluorescent indicators. |
| Colorimetric / Absorbance | Detection of changes in light absorption at specific wavelengths due to a color change in a reagent phase [38]. | pH, dissolved O₂, ethanol, glucose | Detection in 90-120 min demonstrated for bacterial metabolism [38]. | Simplicity; can be visually interpreted (qualitatively). |
| Refractometry | Measurement of changes in the refractive index at the fiber surface upon analyte binding [37]. | Biomass, sugars, alcohols | Sensitive to bulk property changes in the broth. | Label-free; direct detection. |
The following workflow diagram illustrates the general process of developing and deploying a fiber optic biosensor for in-situ fermentation monitoring.
Sensor Deployment Workflow
Experimental Protocol: Monitoring Ethanol with a Fiber Optic Optode
This protocol details the use of a fiber optic optode for the continuous monitoring of ethanol in the vapour phase of a bioreactor, based on a validated methodology [37].
Principle
The sensor utilizes a reagent-infused polymer membrane (optode) attached to the tip of an optical fiber. Ethanol diffuses into the membrane and interacts with a chromoionophore, causing a measurable change in absorbance at a specific wavelength (e.g., 305 nm), which is correlated to ethanol concentration.
Materials and Equipment
Table 2: Research Reagent Solutions and Essential Materials
| Item | Function / Description |
|---|---|
| Silica Optical Fiber (e.g., 400 µm core diameter) | Light transmission to and from the sensing region. |
| Ethanol-Selective Optode Membrane | Polymer matrix containing a selective ionophore and chromoionophore. |
| UV-Vis Spectrometer or LED-PD System | Light source and detector for measuring absorbance changes. |
| Data Acquisition Software | Records and processes the optical signal over time. |
| Lab-Scale Bioreactor | Controlled environment for the fermentation process. |
| Sterilization Equipment (e.g., autoclave) | Ensures aseptic sensor introduction. |
Step-by-Step Procedure
- Sensor Preparation: A short portion of the optical fiber's cladding is removed, and the core is coated with the ethanol-selective optode membrane following established thin-film deposition techniques [37].
- Calibration: Prior to sterilization, calibrate the sensor by immersing it in standard solutions with known ethanol concentrations (e.g., 0.1% to 5% v/v) and recording the absorbance at 305 nm. Generate a calibration curve of absorbance versus concentration.
- Sterilization and Integration: Sterilize the sensor probe (e.g., using chemical sterilants compatible with the membrane, or autoclave if the membrane is stable). Aseptically insert the sensor probe into a dedicated port on the bioreactor, ensuring the sensing tip is exposed to the vapour phase (for vapour-phase monitoring) or the liquid broth.
- Continuous Monitoring:
- Initiate the fermentation process.
- The light source (e.g., a 305 nm LED) sends light through the optical fiber to the sensing membrane.
- The reflected or transmitted light is guided back through the same or a separate fiber to the spectrometer/detector.
- Absorbance is calculated and recorded at set intervals (e.g., every minute).
- Data Analysis: Convert the recorded absorbance values to ethanol concentration using the pre-established calibration curve. Plot the ethanol concentration against time to monitor the fermentation profile.
Data Interpretation and Validation
The resulting real-time ethanol profile should be compared with a reference method (e.g., GC or offline distillation/density measurements) to validate sensor accuracy [37]. The data can be used to identify key fermentation phases (lag, exponential, stationary) and calculate specific production rates.
The Scientist's Toolkit
The following table catalogs key resources essential for research and development in fiber optic biosensing for fermentation.
Table 3: Key Research Reagent Solutions and Materials
| Category / Item | Specific Function in Research |
|---|---|
| Optical Components | |
| Hollow-Core Microstructured Fibers | Confines light and analyte interaction in a tiny space, intensifying the signal over long distances [35]. |
| Plasmonic Nanoparticles (Au, Ag) | Used to functionalize fiber tips for SERS-based sensors, providing massive signal enhancement [35]. |
| Biorecognition Elements | |
| Ion-Selective Chromoionophores | Changes optical properties upon binding specific ions (H⁺, Na⁺) for pH and metabolite sensing [37]. |
| Enzymes (e.g., Alcohol Oxidase) | Provides high specificity for target analytes like ethanol in enzyme-based optical biosensors [37]. |
| Membrane & Immobilization | |
| Solvent Polymeric Membranes | Houses the sensing chemistry (ionophore, chromoionophore) for optode-based sensors [37]. |
| Polymer Matrices (e.g., PVC, PDMS) | Serves as a solid support for immobilizing biorecognition elements onto the fiber surface. |
The relationships between these components in a typical sensing architecture are shown below.
Biosensing Component Relationships
Surface Plasmon Resonance (SPR) for Label-Free Pathogen and Metabolite Detection
Surface Plasmon Resonance (SPR) has emerged as a powerful, label-free technology for the real-time monitoring of biomolecular interactions, making it exceptionally valuable for applications in fermentation monitoring and bioprocess control. Within the context of optical biosensors, SPR's unique capability to provide quantitative data on binding kinetics, concentration, and specificity without the need for fluorescent or enzymatic labels enables researchers to track microbial contaminants and key metabolites with minimal process disruption [39] [6]. This is particularly critical in fermentation research, where the timely detection of pathogenic contamination and the precise monitoring of metabolic intermediates can dictate the success of a biomanufacturing campaign. The technology converts biological recognition events, such as antigen-antibody binding, into measurable changes in the refractive index at a sensor surface, allowing for the direct observation of interactions as they occur [40]. This application note details the use of SPR for detecting foodborne pathogens and fermentation-relevant metabolites, providing structured experimental protocols, key performance data, and a curated list of essential research reagents to facilitate its adoption in bioprocessing and drug development workflows.
Application in Pathogen Detection
Foodborne and waterborne pathogenic bacteria, such as Escherichia coli, Salmonella, and Listeria, represent a serious threat to public health and can lead to significant economic losses [39]. In fermentation processes, their inadvertent introduction can compromise product quality, safety, and yield. SPR biosensors address this challenge through direct, label-free detection, often achieving results in minutes to hours—a significant advantage over traditional culture methods that can take several days [39] [7].
The foundational principle involves immobilizing a specific biorecognition element (e.g., an antibody) onto a gold sensor chip. When a solution containing the target bacteria flows over this surface, binding occurs, leading to an increase in the local refractive index. This change is detected as a shift in the resonance angle or wavelength of the reflected light, which is recorded in real-time as a sensorgram [39] [40]. The magnitude of this signal is proportional to the mass bound and, consequently, to the bacterial concentration [39].
Recent advancements have significantly enhanced the capabilities of SPR for pathogen detection. The development of SPR imaging (SPRi) allows for the high-throughput, simultaneous analysis of multiple regions of interest on a single sensor chip, enabling the detection and analysis of single bacterial cells [41]. Furthermore, sophisticated optimization algorithms have been employed to push detection limits to unprecedented levels. For instance, one study utilizing a multi-objective particle swarm optimization strategy reported a limit of detection (LOD) for mouse IgG as low as 54 ag/mL (0.36 aM), demonstrating the potential for detecting low-abundance analytes [42].
Table 1: Summary of SPR-based Pathogen Detection Performance
| Target Pathogen | Immobilization Method | LOD / Detection Range | Key Sensor Features | Reference |
|---|---|---|---|---|
| Salmonella spp. | Antibody, covalent binding | Low pH and high antibody concentration | Planar SPR, antibody-coated chip | [39] |
| Pseudomonas & other bacteria | Physical adsorption with graphene coating | Improved adhesion | Graphene-coated gold surface | [39] |
| Five E. coli strains | Physical adsorption to carbohydrates | 10² CFU/mL | SPR imaging, pyrrole-electropolymerized surface | [39] |
| Acidovorax avenae (Aac) | Antibody on mixed SAMs | Specific and reliable | SPR imaging, functionalized chip | [41] |
| Mouse IgG (Model Analyte) | Optimized design parameters | 54 ag/mL (0.36 aM) | Algorithm-optimized SPR sensor | [42] |
Detailed Protocol: Direct Detection ofSalmonellavia Antibody Immobilization
This protocol outlines the steps for configuring an SPR biosensor to detect Salmonella species through the covalent immobilization of a specific capture antibody [39] [43].
Materials Required:
- SPR instrument (e.g., in Kretschmann configuration)
- Gold sensor chips (~50 nm Au on ~2 nm Cr adhesion layer)
- Mouse monoclonal antibodies against Salmonella
- 11-mercaptoundecanoic acid (11MUA) and 3-Mercaptopropionic acid (3MPA)
- Coupling agents: EDC and NHSS
- Ethanolamine hydrochloride (EA)
- Phosphate Buffered Saline (PBS), pH 7.4
- Salmonella standards at known concentrations
Procedure:
- Sensor Chip Cleaning: Clean the bare gold sensor chip in a piranha solution (NH₃ aq./H₂O₂ aqueous solution, 1:1:5 v/v) at 80–90 °C for 10 minutes. Rinse thoroughly with deionized water and dry under a stream of nitrogen. A final 10-minute treatment in a UV-ozone cleaner is recommended to ensure a clean, hydrophilic surface [43].
- Formation of Self-Assembled Monolayer (SAM): Incubate the cleaned gold chip with a solution of carboxyl-terminated alkanethiols (e.g., 11MUA) to form a SAM. This layer provides functional groups for subsequent antibody attachment [43].
- Antibody Immobilization:
- Activate the carboxyl groups on the SAM by injecting a fresh mixture of EDC and NHSS over the sensor surface for 7-10 minutes.
- Dilute the anti-Salmonella antibody in a low-pH buffer (e.g., sodium acetate, pH 4.5-5.0) to promote electrostatic pre-concentration onto the sensor surface. Inject the antibody solution until the desired immobilization level is achieved (e.g., ~2.4 × 10¹¹ molecules/cm²) [43].
- Deactivate any remaining activated esters by injecting a 1 M ethanolamine solution (pH ~8.5) for 5-7 minutes to block unreacted sites.
- Binding Assay:
- Establish a stable baseline by flowing running buffer (PBS) over the functionalized sensor surface.
- Inject the sample or standard solution containing Salmonella over the surface for a defined contact time (e.g., 10-20 minutes). The binding event will be observed as an increase in the SPR signal.
- Switch back to running buffer to initiate the dissociation phase. The sensor surface can be regenerated for subsequent analyses by injecting a mild acidic or basic solution (e.g., 10 mM Glycine-HCl, pH 2.0) to break the antigen-antibody complex without denaturing the immobilized antibody.
- Data Analysis: The sensorgram, a plot of response (Resonance Units, RU) versus time, is analyzed to extract kinetic parameters (association rate, kₐ; dissociation rate, kₑ) or to generate a calibration curve of response versus bacterial concentration for quantitative purposes [40].
Application in Metabolite and Fermentation Monitoring
Beyond pathogen detection, SPR is increasingly applied in fermentation monitoring to track metabolites, sugars, amino acids, and indicators of cell viability in real-time [7] [6]. This provides invaluable insights for optimizing bioreactor conditions and controlling bioprocesses. For example, SPR can be used to monitor the production of lactic acid by Lactobacillus species in yogurt fermentation or to track the consumption of sugars and production of alcohols during microbial fermentations [7].
In these applications, the biorecognition element is tailored to the target metabolite. This can include:
- Enzymes that specifically convert the metabolite, resulting in a local change in refractive index.
- Aptamers (single-stranded DNA or RNA oligonucleotides) that undergo a conformational change upon binding the target molecule.
- Whole cells as biorecognition elements to study cell-surface interactions and secreted factors.
The real-time, label-free nature of SPR allows for continuous monitoring without the need for sampling and complex sample preparation, making it an ideal tool for integration into intelligent bio-manufacturing systems [6].
Table 2: SPR for Monitoring Microbial Interactions and Metabolites in Food & Fermentation
| Target / Process | Biosensor Type | Detected Signal / Analyte | Application Context |
|---|---|---|---|
| Competition (e.g., in Cheese) | Electrochemical (Reference) | Bacteriocins | LAB outcompeting pathogens like Listeria [7] |
| Cooperation (e.g., in Yogurt) | Optical (SPR) | Volatile compounds / Lactic acid | Synergy between S. thermophilus and L. bulgaricus [7] |
| Quorum Sensing (e.g., in Meat) | QCM / SPR | Biofilm mass / Signaling molecules | Early warning of spoilage or contamination [7] |
| Sugar and Alcohol Levels | SPR / Other Optical Biosensors | Refractive index change | Fermentation process control [4] |
Detailed Protocol: Monitoring Lactic Acid via an Enzyme-Based SPR Assay
This protocol describes a generic approach for detecting a key fermentation metabolite, such as lactic acid, using an enzyme-modified SPR chip.
Materials Required:
- SPR instrument and sensor chips (e.g., CM5 with carboxymethylated dextran)
- Lactate Oxidase enzyme
- Coupling agents: EDC and NHSS
- Ethanolamine hydrochloride (EA)
- HEPES or Phosphate buffer
- Lactic acid standards
Procedure:
- Sensor Chip Functionalization: Following the cleaning steps outlined in Protocol 2.1, activate the carboxymethylated dextran matrix on a CM5 sensor chip with a pulse of EDC/NHSS.
- Enzyme Immobilization: Dilute the Lactate Oxidase enzyme in a suitable coupling buffer (e.g., sodium acetate, pH 5.0). Inject the enzyme solution over the activated sensor surface to achieve covalent immobilization via primary amine groups. Block any remaining active esters with ethanolamine.
- Calibration and Measurement:
- Flush the system with running buffer to establish a stable baseline.
- Inject standard solutions of lactic acid at known concentrations. As lactic acid interacts with the immobilized lactate oxidase, the catalytic reaction and subsequent local changes are detected as an SPR signal shift.
- The response is recorded, and a calibration curve is constructed by plotting the maximum response or the initial binding rate against the concentration of lactic acid.
- Sample Analysis: Fermentation broth samples, potentially diluted and filtered to remove cells and debris, are injected over the sensor surface. The concentration of lactic acid in the unknown samples is determined by interpolating the sensor response from the calibration curve.
The Scientist's Toolkit: Essential Research Reagents
Successful implementation of SPR assays relies on a carefully selected suite of reagents and materials. The table below catalogs key solutions for developing SPR-based detection methods.
Table 3: Key Research Reagent Solutions for SPR Biosensing
| Reagent / Material | Function in SPR Assay | Specific Examples & Notes |
|---|---|---|
| Sensor Chips | Provides the physical substrate for the gold film and chemical matrix for ligand immobilization. | CM5: Carboxymethylated dextran; general-purpose [40].C1: Flat carboxymethylated surface; ideal for cells and viruses [40].SA: Pre-immobilized streptavidin for capturing biotinylated ligands [40]. |
| Capture Ligands | Acts as the biorecognition element that confers specificity to the assay. | Antibodies: For specific pathogen (e.g., Salmonella) or protein detection [39] [43].Aptamers: Nucleic acid-based receptors for small molecules, proteins, or cells [7].Enzymes: For metabolite detection (e.g., Lactate Oxidase) [4]. |
| Chemicals for Immobilization | Enables stable and oriented attachment of the ligand to the sensor surface. | Alkanethiols (11MUA, 3MPA): Form a self-assembled monolayer (SAM) on gold for functionalization [43].EDC/NHSS: Activates carboxyl groups for covalent coupling to amine-containing ligands [43]. |
| Regeneration Solutions | Removes bound analyte without damaging the immobilized ligand, allowing for chip re-use. | Low pH Buffer: e.g., 10-100 mM Glycine-HCl, pH 2.0-3.0 [40].High pH Buffer: e.g., 10-50 mM NaOH or Glycine-NaOH, pH 9.0-11.0.Other: SDS, high salt, or chelating agents (for His-tagged ligands). |
Workflow and Signaling Visualization
The following diagrams illustrate the core principle of SPR and a generalized experimental workflow for an SPR binding assay.
Diagram 1: Principle of Surface Plasmon Resonance (SPR)
Diagram 2: SPR Experimental Workflow for a Binding Assay
In the field of fermentation monitoring research, the accurate, real-time determination of optical density (OD) is a critical requirement for assessing cell density and microbial growth, thereby providing valuable insights into the state of the bioprocess [44]. However, direct physical measurement of OD, particularly in high-throughput microbioreactor (HT-MBR) systems, presents significant challenges. These systems require robotic liquid-handling systems for essential process control tasks, and frequent physical sampling occupies valuable resources, potentially leading to intermittent carbon source limitations that adversely affect the microorganism's metabolic state [44]. The conflict between the need for process information and the practical limitations of automated systems represents a major bottleneck in bioprocess development, especially in the production of biopharmaceuticals like antibody fragments [44].
AI-powered soft sensors present a transformative solution to this challenge. These model-based systems are designed to estimate critical process variables in real-time where physical sensors are impractical [44]. Leveraging Artificial Neural Networks (ANNs), these soft sensors can accurately predict OD using readily available online process parameters, minimizing the need for physical sampling without sacrificing process information [44]. This advancement is particularly valuable for accelerating biopharmaceutical process development, contributing to more scalable and predictive fermentation processes in MBR systems [44]. This protocol details the implementation of an ANN-based soft sensor for OD estimation, framed within the broader application of optical biosensors in fermentation monitoring.
Key Principles and Technologies
Optical Density in Fermentation Monitoring
Optical density (OD) is a critical process parameter directly correlated with cell density during fermentation. Traditional measurement requires sampling of the fermentation broth and measuring light scattering with a spectrophotometer, a process that is difficult to automate and can disrupt the fermentation, especially in systems with limited volume [44] [45]. In industrial practice, monitoring often relies on parameters like pH, temperature, and dissolved oxygen (DO), while key biological indicators such as biomass, substrates, and products are typically measured offline, delaying process control interventions [46].
Soft Sensor Paradigms
Soft sensors can be categorized into three main types [44]:
- Mechanistic Models: Based on first-principle equations describing the direct coherence between accessible process variables and estimated key parameters. These require in-depth process knowledge.
- Statistical Models: Fitted to historical data representing past process behavior. Techniques include decision trees, multiple linear regression, and ANNs. They can detect complex behaviors but require substantial historical data.
- Hybrid Models: Combine mechanistic and statistical approaches, either sequentially or in parallel.
For high-throughput MBR systems that rapidly generate large experimental datasets, data-driven statistical models, particularly ANNs, are an attractive choice as they balance the data requirement with powerful modeling capability [44].
Artificial Neural Networks for Regression
ANNs are powerful machine learning models that estimate a function linking a set of specified inputs to a desired output by minimizing error via gradient descent optimization [44]. Their architecture, consisting of interconnected layers of nodes, allows them to approximate complex linear and nonlinear relationships, making them exceptionally well-suited for modeling the dynamic and interconnected parameters of a fermentation process [47] [48]. In the context of OD estimation, ANNs use available online parameters to predict the target OD value, effectively learning the underlying biological relationships from data.
ANN-Based OD Soft Sensor Implementation Protocol
This protocol outlines the development and implementation of an ANN-based soft sensor for real-time OD estimation in a high-throughput microbioreactor system, based on the methodology validated by Boehringer Ingelheim and University of Natural Resources and Life Sciences, Vienna [44].
Research Reagent and Material Solutions
Table 1: Essential Research Reagents and Materials
| Item | Specification / Function |
|---|---|
| Microbioreactor (MBR) System | Automated system based on temperature-controlled fermentation blocks (e.g., bioREACTOR 8 from 2mag AG) [44]. |
| Microorganism | Recombinant production strains (e.g., Escherichia coli for single-chain variable fragment (scFv) expression) [44]. |
| Fermentation Media | Contains carbon source (e.g., glucose), nitrogen source, and salts (e.g., KH2PO4, MgSO4, Yeast Extract, CaCl2, (NH4)2SO4) [44] [46]. |
| Process Control Solutions | Acid, base, and carbon feed solutions for pH regulation and nutrient supplementation [44]. |
| Data Acquisition Software | Software for collecting and storing time-series data from the bioreactor's physical sensors. |
| Computing Environment | Platform for ANN model development and deployment (e.g., MATLAB) [48]. |
Software and Computational Requirements
- Programming Environment: MATLAB or Python with relevant deep learning libraries (e.g., TensorFlow, PyTorch).
- ANN Training: Use a Multilayer Perceptron (MLP) architecture. For spectral data, consider Backpropagation ANN (BP-ANN), Radial Basis Function ANN (RBF-ANN), or Extreme Learning Machine ANN (ELM-ANN) [47].
- Performance Metrics: Evaluate the model using Coefficient of Determination (R²), Root Mean Squared Error (RMSE), and Mean Squared Error (MSE) [48].
- Optimization: Employ meta-heuristic algorithms (e.g., Chameleon Swarm Algorithm, White Shark Optimizer) to optimize the random parameters of the ANN and prevent convergence to local optima [47].
Step-by-Step Experimental Procedure
Step 1: Data Collection and Preprocessing
- Conduct Fermentation Runs: Execute multiple fermentation experiments using your HT-MBR system, ensuring a range of operating conditions to build a robust dataset.
- Record Online Process Parameters: Continuously log data from standard bioreactor sensors. The critical input parameters for the soft sensor are:
- Acquire Reference OD Measurements: Periodically take at-line samples and measure OD using a spectrophotometer to create the target values for model training [44]. This ground truth data is essential.
Step 2: ANN Model Development and Training
- Structure the Dataset: Organize the collected data into a time-synchronized table where each row contains the input parameters and the corresponding reference OD value.
- Partition the Data: Split the dataset into three subsets:
- Training Set (~70%): Used to train the ANN model.
- Validation Set (~15%): Used to tune hyperparameters and prevent overfitting during training.
- Testing Set (~15%): Used for the final, unbiased evaluation of model performance.
- Design and Train the ANN:
- Initialize a feedforward MLP network.
- The input layer should have nodes corresponding to each of the selected online parameters.
- Experiment with the number of hidden layers and neurons to find the optimal architecture.
- The output layer has a single node for the predicted OD value.
- Train the network using a backpropagation algorithm (e.g., Levenberg-Marquardt) to minimize the error between predictions and reference OD values.
Step 3: Model Validation and Performance Evaluation
- Validate the Model: Use the validation dataset to assess the model's performance during training. A successful model should achieve a high degree of accuracy (>95%) in estimating OD [44].
- Conduct Final Testing: Evaluate the final model on the held-out test set. Report standard performance metrics:
- Perform Sensitivity Analysis: Identify which input parameters most significantly drive the OD prediction. Research indicates that temperature (47.2%) and dissolved oxygen (40.2%) are often the largest contributors, while aeration period, chemical oxygen demand, and oxygen uptake rate may have smaller effects [48].
Step 4: Deployment for Real-Time Monitoring
- Integrate with Bioreactor Control System: Deploy the trained ANN model onto the system controlling the MBR.
- Enable Real-Time Estimation: The soft sensor should now continuously read the live stream of online process data and output a real-time estimate of the OD.
- Implement Process Control: Use the real-time OD estimates for advanced process control strategies, such as triggering glucose feeding or induction at specific growth phases, thereby optimizing the fermentation process [46].
Data Analysis and Interpretation
The following table summarizes the expected performance outcomes based on published studies employing ANN-based soft sensors in bioprocesses.
Table 2: Performance Metrics from ANN-Based Soft Sensor Studies
| Application Context | Model Type | Key Performance Metrics | Reference |
|---|---|---|---|
| OD Estimation in MBR | ANN (MLP) | >95% estimation accuracy achieved without informative variables from off-gas analysis. | [44] |
| Bacterial Concentration in Kombucha | POA-RBF-ANN | RPD (Ratio of SD to prediction set) of 6.7878 in internal validation. | [47] |
| Bacterial Concentration in Kombucha | CSA-BP-ANN | Maximum error of only 0.0169 Au in external verification. | [47] |
| Biomass Growth in Wastewater | ANN (MLP) | R²: 0.844 (train), 0.853 (validation), 0.823 (test). RMSE: 0.7476 (train), 1.1641 (validation), 0.7798 (test). | [48] |
Workflow and System Architecture Visualization
Alternative and Advanced Methodologies
Integration with Optical Spectroscopy
For systems equipped with advanced optical sensors, ANNs can be combined with spectroscopic data for enhanced prediction. One study established an on-line monitoring system for bacterial concentration during kombucha fermentation using visible/near-infrared spectroscopy (vis-NIR) combined with ANN models (BP-ANN, ELM-ANN, RBF-ANN) [47]. The optimized models, particularly the RBF-ANN, achieved excellent results with a Ratio of the Standard Deviation of the verification set to the prediction set (RPD) of 6.7878, indicating a robust predictive ability [47].
Multi-Sensor Data Fusion
Beyond basic parameters, fusing data from multiple advanced sensors can create a more comprehensive process digital twin. This can include:
- Viable Cell Sensors: Measure capacitance to specifically characterize the number of living cells [46].
- Electronic Noses: Analyze volatile components in off-gas (e.g., ethanol, CO2) to monitor product formation and metabolic activity in real-time [46].
- Raman or NIR Spectroscopy: Provide real-time, non-invasive data on multiple process variables like cell concentration, product formation, and substrate depletion [45] [47].
ANNs are capable of integrating these diverse data streams to build a more accurate and reliable soft sensor for overall process state prediction.
Troubleshooting and Best Practices
Table 3: Troubleshooting Guide for ANN OD Soft Sensor Implementation
| Problem | Potential Cause | Solution |
|---|---|---|
| Poor Model Generalization | Insufficient or low-quality training data. | Conduct more fermentation runs to expand the dataset, ensuring it covers a wide range of process conditions and variations. |
| High Prediction Error | Non-informative input variables or incorrect model architecture. | Perform sensitivity analysis to select the most relevant inputs (prioritize temperature and DO). Tune ANN architecture and hyperparameters. |
| Model Performance Drift Over Time | Natural process drift or changes in microbial strain characteristics. | Periodically retrain the model with new data incorporating the latest process behavior. Implement a model update schedule. |
| Failure in Real-Time Deployment | Integration issues between the model and the bioreactor's data stream. | Verify data formatting, sampling rates, and communication protocols between the soft sensor and the process control system. |
The implementation of an AI-powered soft sensor for estimating optical density represents a significant advancement in fermentation monitoring research. By leveraging Artificial Neural Networks and standard online process parameters, this protocol enables real-time, accurate monitoring of cell density with minimal physical sampling. This approach directly addresses the resource conflicts in high-throughput systems and paves the way for more efficient, data-driven, and automated control of fermentation processes, ultimately accelerating biopharmaceutical development and optimization.
Therapeutic drug monitoring (TDM) is crucial for vancomycin due to its narrow therapeutic window and complex pharmacokinetics [49]. Traditional methods for vancomycin analysis, including chromatographic techniques and immunoassays, present limitations in cost, time, and sometimes sensitivity, creating a need for advanced biosensing alternatives [49] [50]. Reflectometric Interference Spectroscopy (RIfS) is a label-free optical technique that enables real-time biomolecular interaction analysis by monitoring changes in optical thickness at a functionalized transducer surface [51] [52]. This case study explores the application of RIfS biosensors for monitoring vancomycin-type antibiotic production, providing detailed protocols and analytical performance data to support its implementation in fermentation process control and pharmaceutical development.
Theoretical Background
Vancomycin Specificity and Production Challenges
Vancomycin is a glycopeptide antibiotic critical for treating life-threatening infections caused by resistant Gram-positive bacteria such as methicillin-resistant Staphylococcus aureus (MRSA) [49]. Its mechanism of action involves forming a five-point hydrogen bond with the terminal moieties D-alanyl-D-alanine (D-Ala-D-Ala) of peptidoglycans in bacterial cell walls, thereby inhibiting cell wall synthesis [53] [54]. This specific binding mechanism can be leveraged in biosensor design for both antibiotic detection and production monitoring.
The recommended therapeutic trough concentration ranges between 10-20 μg mL⁻¹ (approximately 6.9-13.8 μM), with levels exceeding 15 μg mL⁻¹ associated with significantly increased risk of nephrotoxicity [49]. Maintaining concentrations within this narrow range presents substantial challenges during both production and clinical administration, necessitating precise monitoring methodologies.
Principles of Reflectometric Interference Spectroscopy (RIfS)
RIfS belongs to the direct optical detection methods that measure changes in optical thickness (nd) – the product of refractive index (n) and physical thickness (d) – at a functionalized transducer surface [51]. When white light is directed at the sensor surface, interference occurs between beams reflected from different phase boundaries, generating a characteristic interference spectrum. Biomolecular binding events on the sensor surface increase the optical thickness, causing a measurable shift in the interference pattern that can be monitored in real-time without requiring labeled molecules [51] [52].
The RIfS principle for vancomycin monitoring can be visualized as follows:
Experimental Protocols
RIfS Transducer Functionalization
Objective: To immobilize vancomycin-binding ligands on the RIfS transducer surface for specific antibiotic detection.
Materials and Reagents:
- RIfS glass transducers (1 cm × 1 cm) with 10 nm Ta₂O₅ layer covered with 330 nm SiO₂ [51]
- 3-glycidyloxypropyl-trimethoxysilane (GOPTS) [51]
- Poly(ethylene glycol) diamine (PEG-DA, MW 2000 Da) and ɑ-methoxy-ω-amino PEG (PEG-MA, MW 2000 Da) [51]
- Glutaric acid (GA) in dimethylformamide (DMF) [51]
- N,N′-diisopropyl-carbodiimide (DIC) and N-hydroxysuccinimide (NHS) [51]
- Vancomycin-binding peptide (e.g., AcKAA or D-Ala-D-Ala derivatives) [55]
Procedure:
- Surface Cleaning and Activation:
Silane Modification:
PEG Layer Formation:
Carboxyl Group Activation:
Ligand Immobilization:
- Activate carboxyl groups with NHS (150 mg) and DIC (302 μL) in 1 mL DMF for 4 hours in DMF vapour-saturated chamber [51].
- Clean with DMF and acetone, then dry under nitrogen [51].
- Incubate with vancomycin-binding peptide (2 mg/mL in H₂O) in water vapour-saturated chamber overnight [51].
- Wash and dry transducers before use [51].
RIfS Measurement Setup and Biosensor Operation
Materials and Equipment:
- RIfS instrument with flow cell system [51]
- Peristaltic or syringe pump for precise flow control [51]
- Temperature control system (if available) [51]
- Phosphate-buffered saline (PBS: 150 mM sodium chloride, 10 mM potassium phosphate, pH 7.4) as running buffer [51]
- Vancomycin standards in concentration range 0.1-100 μM [49] [55]
Measurement Procedure:
- System Initialization:
Sample Analysis:
- Inject vancomycin samples or fermentation broth samples (appropriately diluted in PBS) [51].
- Allow association phase for 600 seconds while continuously recording interference signal [51].
- Switch back to pure buffer for dissociation phase monitoring (900 seconds) [51].
- Regenerate surface if necessary using appropriate regeneration solution (e.g., 6 M guanidine hydrochloride, pH 1.5) [51].
Data Collection:
The complete experimental workflow for RIfS-based vancomycin monitoring is systematically outlined below:
Results and Data Analysis
Analytical Performance of RIfS Biosensor
Table 1: Performance comparison of vancomycin monitoring biosensors
| Technology | Detection Limit | Linear Range | Sample Volume | Analysis Time | Reference |
|---|---|---|---|---|---|
| RIfS Biosensor | ~1 nM (theoretical) | Not specified | Microfluidic scale | Real-time monitoring | [51] |
| Electrochemical (GR-GC) | 0.2 μM | Up to 20 μM | 3 μL | Minutes | [49] |
| MOF-based Electrochemical | 1 nM | Not specified | Diluted blood samples | Minutes | [49] |
| Microneedle-Optical | <100 nM | 0.1-72.6 μM | 0.6 nL | ~10 minutes | [55] |
| Fluorescence Biosensor | Not specified | Not specified | Microdialysis samples | Continuous monitoring | [56] |
| Traditional HPLC | 0.1 μg/mL (~0.07 μM) | Wide range | 50-100 μL | Hours | [49] |
| Immunoassays | ~5 μg/mL (~3.4 μM) | Limited | 50-100 μL | Hours | [49] |
Binding Kinetics Analysis
For quantitative analysis of vancomycin binding, sensorgrams obtained from RIfS measurements are fitted to appropriate kinetic models. The one-to-one binding model following pseudo-first-order kinetics is commonly applied [51]:
Data Fitting Approach:
- Linear Transformation Method: Plotting ln(dR/dt) versus time for association phase [51]
- Integrated Rate Equation: Fitting to exponential function R(t) = Rₑₓ(1 - e^(-kₒᵦₛt)) [51]
- Global Fitting: Simultaneous analysis of multiple concentration curves [51]
Table 2: Key parameters for RIfS biosensor validation in vancomycin monitoring
| Parameter | Target Specification | Experimental Validation Method |
|---|---|---|
| Sensitivity | Sufficient to detect <100 nM | Calibration with vancomycin standards [49] [55] |
| Selectivity | Specific to vancomycin | Cross-reactivity testing with analogs [49] |
| Reproducibility | <5% RSD | Repeated measurements of same sample [49] |
| Stability | Maintain performance over 6+ weeks | Long-term stability testing [49] |
| Matrix Effects | Minimal interference from fermentation media | Spiked recovery experiments [49] [53] |
The Scientist's Toolkit: Essential Research Reagents and Materials
Table 3: Key research reagents and materials for RIfS-based vancomycin monitoring
| Item | Function/Application | Specifications/Notes |
|---|---|---|
| RIfS Transducers | Optical sensing element | 1 cm × 1 cm glass with 10 nm Ta₂O₅ + 330 nm SiO₂ [51] |
| GOPTS | Surface silanization | Creates epoxy-functionalized surface for immobilization [51] |
| PEG-DA/PEG-MA | Anti-fouling layer | Forms hydrogel matrix reducing non-specific binding [51] |
| DIC/NHS | Carboxyl group activation | En covalent ligand immobilization [51] |
| Vancomycin-Binding Peptides | Molecular recognition | AcKAA or D-Ala-D-Ala derivatives [55] |
| PBS Buffer | Running buffer | 150 mM sodium chloride, 10 mM potassium phosphate, pH 7.4 [51] |
| Guanidine HCl | Surface regeneration | 6 M, pH 1.5 for removing bound vancomycin [51] |
| Microfluidic Flow Cell | Sample delivery | Controlled flow environment for binding studies [51] |
Discussion and Implementation Notes
Advantages of RIfS for Fermentation Monitoring
The implementation of RIfS technology for vancomycin production monitoring offers several significant advantages over conventional analytical methods. Unlike endpoint assays such as ELISA or HPLC, RIfS provides real-time kinetic data enabling continuous process monitoring [51] [52]. This capability is particularly valuable in fermentation processes where subtle changes in production kinetics can indicate process optimization opportunities or early signs of production issues.
The label-free nature of RIfS eliminates the need for complex sample preparation or labeling procedures, reducing analysis time and potential artifacts introduced by labeling chemistries [51] [52]. Furthermore, the microfluidic format minimizes sample consumption, making the technology suitable for monitoring small-scale fermentation trials where sample volume may be limited.
Technical Considerations and Limitations
Several technical considerations must be addressed for successful implementation of RIfS biosensors in vancomycin production environments. Matrix effects from fermentation media components can potentially interfere with binding signals, necessitating appropriate control experiments and possibly sample dilution or purification [49] [51]. The stability of the functionalized transducer surface under prolonged operation conditions should be validated, as some studies report performance degradation after approximately six weeks [49].
Sensor calibration and regular performance verification are essential for maintaining data quality. Implementation of reference channels and internal standards can help compensate for potential baseline drift or environmental fluctuations [51]. For integration into fermentation bioreactors, compatibility with sterilization procedures and long-term stability in the production environment must be thoroughly evaluated.
Integration with Fermentation Process Control
The real-time monitoring capabilities of RIfS biosensors align with the emerging paradigm of smart fermentation technologies, which leverage real-time data acquisition, IoT connectivity, and machine learning algorithms for enhanced process control [57] [58]. Integration of RIfS data with multiparameter fermentation monitoring systems (e.g., pH, dissolved oxygen, biomass) can provide comprehensive insights into production kinetics and enable automated feedback control strategies.
Future developments in miniaturized RIfS systems and high-throughput array formats could further enhance their utility in fermentation optimization and scale-up studies [57] [58]. The ability to monitor multiple analytes simultaneously would be particularly valuable for understanding metabolic pathways and optimizing production yields in vancomycin biosynthesis.
This case study demonstrates that RIfS biosensor technology represents a promising approach for real-time monitoring of vancomycin-type antibiotic production. The detailed protocols provided herein enable researchers to implement this methodology for fermentation process development and optimization. The label-free, real-time monitoring capabilities of RIfS offer significant advantages over traditional analytical methods, potentially accelerating process development and enhancing product quality control in pharmaceutical manufacturing.
Overcoming Implementation Challenges in Complex Fermentation Matrices
Addressing Matrix Interference and Non-Specific Binding in Biological Broths
The real-time monitoring of fermentation processes using optical biosensors is transformative for the biotechnology and pharmaceutical industries, enabling precise control over product quality and yield [7]. However, the complex and dynamic nature of biological broths presents a significant challenge for analytical techniques. These matrices contain a diverse mixture of cells, nutrients, metabolites, and proteins, which can interfere with sensor measurements through two primary mechanisms: matrix interference, where the background composition affects the physicochemical signal transduction, and non-specific binding (NSB), where unintended molecules adhere to the sensor surface [59]. These phenomena can obscure the true concentration of the target analyte, leading to inaccurate readings and potentially compromising process control. This application note details the sources of these challenges and provides validated protocols and material solutions to mitigate them, facilitating the robust application of optical biosensors in fermentation research and development.
Technical Background
Mechanisms of Interference in Complex Matrices
The efficacy of a biosensor is contingent upon its specificity and sensitivity within the operational environment. In fermentation broths, several factors contribute to signal distortion:
- Ionic Strength Variations: Fluctuations in salt concentration during fermentation can cause severe signal distortion in charge-based transducers, such as nanowires and electrochemical sensors, due to Debye screening effects. This screening can shield the sensor from detecting the target protein binding events [59].
- pH and Temperature Fluctuations: Microbial metabolism can cause significant shifts in broth pH and temperature. These changes can alter the charge and conformation of both the immobilized biorecognition elements and the target analytes, affecting binding kinetics and signal output. While some sensors are highly sensitive to such changes, others, like magnetic nanosensors, are notably robust against them [59].
- Autofluorescence and Optical Interference: The intrinsic fluorescence of certain broth components, such as vitamins or media ingredients, can create a high background signal in fluorescence-based optical biosensors, drastically reducing the signal-to-noise ratio [59] [1].
- Fouling and Non-Specific Binding: Proteins, polysaccharides, and other macromolecules in the broth can physically adsorb onto the sensor surface. This biofouling can block binding sites, alter the refractive index at the sensor surface (critical for label-free techniques like SPR), and lead to false-positive signals [7] [4].
Comparison of Biosensing Platforms and Their Matrix Susceptibility
Different optical biosensor platforms exhibit varying degrees of susceptibility to matrix effects. The following table summarizes the key characteristics of several prominent technologies.
Table 1: Comparison of Optical Biosensor Platforms for Fermentation Monitoring
| Biosensor Platform | Transduction Principle | Key Advantages | Susceptibility to Matrix Effects | Suitable for Broth Analysis? |
|---|---|---|---|---|
| Surface Plasmon Resonance (SPR) | Refractive index change at metal surface [1] | Label-free, real-time, kinetic data [7] [1] | High susceptibility to NSB and refractive index shifts from bulk solution [4] | Yes, with stringent surface chemistry and reference channels [7] |
| Localized SPR (LSPR) | Light interaction with metallic nanoparticles [1] | Label-free, highly sensitive, more compact than SPR [1] | Less sensitive to bulk effects than SPR, but still requires careful surface functionalization [1] | Yes, promising for point-of-use applications [1] |
| Fluorescence / Luminescence | Emission of light from labeled probes [4] | High sensitivity, multiplexing capability [4] | High susceptibility to autofluorescence and light scattering from broth components [59] | Challenging; requires sample cleanup or advanced quenching-resistant probes |
| Colorimetric | Visible color change [4] | Simple, low-cost, visual readout [4] | Susceptible to interference from colored broth components [4] | Limited, best for clear solutions or with separation steps |
| Magnetic Nanosensors | Magnetic field detection via GMR sensors [59] | Matrix-insensitive; immune to pH, ionic strength, and autofluorescence [59] | Very low; biological matrices lack a magnetic background [59] | Excellent for direct analysis in complex fluids [59] |
| Surface-Enhanced Raman Scattering (SERS) | Enhanced Raman signal on nanostructured metal [4] [60] | Ultra-sensitive, fingerprinting capability [4] | Susceptible to fouling, but signal is highly specific [4] | Yes, with appropriate surface passivation |
Experimental Protocols
This section provides detailed methodologies for mitigating interference, focusing on surface functionalization for optical biosensors and a protocol for a matrix-insensitive magnetic assay.
Protocol 1: Surface Functionalization for SPR-Based Monitoring of a Model Metabolite
This protocol describes the development of an SPR biosensor for detecting a specific fermentation metabolite (e.g., lactate), incorporating a carboxymethylated dextran matrix and a blocking regimen to minimize NSB.
Workflow Overview:
Materials:
- SPR instrument (e.g., Biacore series)
- Sensor chip with carboxymethylated dextran surface (e.g., CM5)
- N-hydroxysuccinimide (NHS)
- 1-Ethyl-3-(3-dimethylaminopropyl)carbodiimide (EDC)
- Ethanolamine-HCl, pH 8.5
- Capture molecule (e.g., monoclonal antibody or specific aptamer against the target)
- Blocking solution (e.g., 1% BSA in PBS or 0.1% PEG-2000)
- Running buffer (e.g., HBS-EP, pH 7.4)
- Regeneration solution (e.g., 10 mM Glycine-HCl, pH 2.0)
- Filtered (0.22 µm) and centrifuged fermentation broth sample
Step-by-Step Procedure:
- System Setup: Prime the SPR instrument with the designated running buffer according to the manufacturer's instructions.
- Surface Activation: Inject a 1:1 mixture of NHS and EDC over the sensor chip surface for 7-10 minutes. This activates the carboxyl groups on the dextran matrix to form reactive NHS esters.
- Ligand Immobilization: Dilute the capture molecule (antibody or aptamer) in a low-salt sodium acetate buffer (pH 4.0-5.5, optimized for the specific molecule). Inject the solution over the activated surface for a sufficient time to achieve the desired immobilization level (typically 5-15 minutes).
- Surface Deactivation: Inject 1M ethanolamine-HCl (pH 8.5) for 5-7 minutes to block any remaining activated ester groups.
- Blocking for NSB Reduction: Inject the blocking solution (1% BSA or 0.1% PEG-2000) for 10-15 minutes. This step is critical for passivating the surface against adsorption of non-target proteins from the broth.
- Kinetic Analysis & Calibration: Create a calibration curve by injecting a series of known concentrations of the purified target analyte in running buffer. This allows for the determination of the binding kinetics (kon, koff) and the equilibrium dissociation constant (Kd).
- Sample Analysis: Dilute the processed fermentation broth sample (1:10 to 1:100) in running buffer to reduce the overall matrix load. Inject the sample over the functionalized sensor surface. Always use a reference flow cell (functionalized with a non-specific antibody or BSA) for simultaneous measurement. The signal from the reference cell is automatically subtracted from the sample cell signal to correct for bulk refractive index shifts and non-specific binding.
- Surface Regeneration: After each sample analysis cycle, inject the regeneration solution for 30-60 seconds to remove bound analyte and regenerate the surface for the next injection.
Protocol 2: Magnetic Nanosensor-Based Detection of a Intracellular Enzyme
This protocol leverages the inherent matrix insensitivity of magnetic nanosensors for the direct detection of a target (e.g., a recombinant enzyme) in clarified fermentation lysate.
Workflow Overview:
Materials:
- Giant Magnetoresistive (GMR) sensor array
- Capture antibody specific to the target protein
- Biotinylated detection antibody specific to a different epitope of the target
- Streptavidin-coated superparamagnetic nanoparticles (e.g., 50 nm diameter)
- Phosphate Buffered Saline (PBS) with 0.1% BSA
- Clarified cell lysate in lysis buffer (pH 8.0) or fermentation broth
Step-by-Step Procedure:
- Sensor Functionalization: Immobilize the capture antibody onto the surface of the GMR sensors in the array. This can be done via passive adsorption or covalent chemistry tailored to the sensor chip.
- Sample Incubation: Apply the clarified fermentation broth or cell lysate directly to the sensor array. Incubate for 15-30 minutes to allow the target antigen to bind to the capture antibody in a "sandwich" assay format.
- Magnetic Tagging: Wash the sensor array with buffer to remove unbound material. Introduce the biotinylated detection antibody and incubate. Follow with a wash step. Then, introduce the streptavidin-coated magnetic nanoparticles, which will bind to the biotin on the detection antibody.
- Signal Detection and Quantification: Apply a controlled external magnetic field to magnetize the nanoparticles bound to the sensor surface. The GMR sensors directly detect the fringe fields from these nanoparticles, producing a change in electrical resistance proportional to the number of bound tags, and thus, the target concentration [59]. The signal is quantified against a pre-established calibration curve. As demonstrated in the literature, this method allows for quantitative protein detection over a linear dynamic range of over six orders of magnitude, with attomolar sensitivity, directly in complex matrices like serum, urine, and lysis buffer without any desalting or dilution steps [59].
The Scientist's Toolkit: Key Research Reagent Solutions
The successful implementation of the above protocols relies on a suite of critical reagents designed to ensure specificity and minimize interference.
Table 2: Essential Reagents for Mitigating Matrix Effects
| Reagent / Material | Function / Principle of Action | Example Use Case |
|---|---|---|
| Carboxymethylated Dextran Matrix | A hydrogel that provides a hydrophilic environment for ligand immobilization, reducing non-specific protein adsorption through steric repulsion [1]. | Standard surface chemistry for SPR biosensors (e.g., Biacore CM5 chips) [1]. |
| Poly(ethylene glycol) (PEG)-based Blockers | Forms a dense, hydrophilic brush layer on surfaces, creating a steric and energetic barrier that prevents fouling by biomolecules [4]. | Passivating sensor surfaces (SPR, QCM) and magnetic nanoparticles to increase specificity in crude broths. |
| Bovine Serum Albumin (BSA) | A common blocking protein that adsorbs to uncovered surface sites, preventing non-specific binding of other proteins from the sample. | Used as a cost-effective blocking agent in ELISA, SPR, and other biosensor formats (Protocol 1, Step 5). |
| Magnetic Nanoparticles | Superparamagnetic tags for GMR-based detection. Biological matrices have no magnetic background, making this a uniquely matrix-insensitive label [59]. | Acting as the detection tag in magnetic nanosensor assays for direct detection in serum, urine, or lysates (Protocol 2). |
| Ethanolamine-HCl | A small molecule amine used to deactivate (block) reactive NHS esters remaining on the sensor surface after ligand immobilization, preventing subsequent random coupling of sample proteins [1]. | Standard deactivation step in covalent immobilization protocols on carboxylated surfaces (Protocol 1, Step 4). |
Data Presentation and Analysis
The performance of interference mitigation strategies is quantitatively assessed through key validation parameters.
Table 3: Quantitative Performance Metrics for Matrix Insensitivity
| Assessment Metric | Description and Methodology | Exemplary Data from Literature |
|---|---|---|
| Limit of Detection (LOD) in Buffer vs. Broth | Comparison of the lowest detectable analyte concentration in a clean buffer versus in a spiked fermentation broth sample. | Magnetic nanosensors detected CEA down to 50 attomolar in both PBS and mouse serum, showing no loss of sensitivity [59]. |
| Signal Recovery in Spiked Broth | Measures the accuracy of detection by spiking a known amount of analyte into the broth. Calculated as (Measured Concentration / Spiked Concentration) × 100%. | Ideal recovery is 90-110%. SPR biosensors showed similar signals for VEGF and CEA in PBS, mouse serum, and lysis buffer, indicating high recovery [59]. |
| Linear Dynamic Range | The range of analyte concentration over which the sensor response is linear. A wide range in complex matrices indicates robustness. | Magnetic nanosensors exhibited a linear dynamic range of over six orders of magnitude in diverse biological fluids [59]. |
| Response to pH/Ionic Strength | Measures the signal fluctuation of the sensor platform itself in response to changes in pH or salt concentration. | Magnetic nanosensors showed no signal change across pH 4-10 or with varying ionic strength, unlike nanowire sensors [59]. |
Strategies for Robust Biomolecule Immobilization and Sensor Surface Regeneration
Within the rapidly advancing field of intelligent bio-manufacturing, optical biosensors have established themselves as indispensable tools for the real-time monitoring and precise control of fermentation processes [6]. These analytical devices combine a biological recognition element with a transducer that converts a biological event into a measurable signal [3]. The core of an optical biosensor's functionality lies in its biorecognition interface, which is principally responsible for the reliability and accuracy of the assay [61]. In fermentation applications, this interface must selectively detect target analytes—such as metabolites, substrates, or product concentrations—amidst the complex mixture of a fermentation broth, which contains thousands of proteins, nucleic acids, cells, and ions [62] [6].
The performance and commercial viability of these biosensors in industrial fermentation settings are fundamentally governed by two critical aspects: the initial creation of a robust, sensitive, and specific biorecognition layer through effective biomolecule immobilization, and the subsequent sensor surface regeneration to allow multiple reuses while maintaining analytical performance [61] [63]. The immobilization strategy must ensure uniform bioreceptor coverage, proper orientation to maximize target accessibility, and stability under flow-through conditions, while the regeneration protocol must efficiently remove bound analyte without damaging the immobilized bioreceptor [61]. This Application Note provides a comprehensive overview of current strategies for achieving robust biomolecule immobilization and successful sensor surface regeneration, framed within the context of optical biosensors for fermentation monitoring research.
Biomolecule Immobilization Strategies
The methodology for immobilizing bioreceptors is a decisive factor influencing biosensor performance. An ideal immobilization protocol must preserve the biological activity of the recognition element, provide the correct orientation for optimal target binding, ensure stability under operational conditions, and minimize non-specific adsorption from the complex fermentation matrix [61] [64].
Selection of Biorecognition Elements
The choice of bioreceptor is dictated by the target analyte and the specific requirements of the fermentation monitoring application.
Table 1: Common Biorecognition Elements for Optical Biosensors in Fermentation
| Receptor Type | Advantages | Limitations | Example Fermentation Targets |
|---|---|---|---|
| Antibodies | High affinity & specificity; well-established production [61] | Long/production; high cost; low reusability [61] | Microbial contaminants; specific protein products |
| Bioengineered Antibodies | High affinity; scalable production; no animal immunization [61] | Requires sequence knowledge; storage similar to antibodies [61] | Recombinant protein products |
| Enzymes | High catalytic activity; reagentless detection possible [65] | Stability can be limited; susceptible to microenvironment [64] | Glucose, lactate, glutamate, alcohol [65] |
| Aptamers | Chemical synthesis; good stability; reusability [61] | Complex selection (SELEX); subject to structural changes [61] | Small molecule metabolites, toxins |
| DNA strands | High stability & reusability; versatile synthesis [61] | Limited target range (nucleic acids) [61] | Specific genetic markers or microbial contamination |
Immobilization Techniques
Various immobilization methods are employed, each with distinct advantages, drawbacks, and protocols.
Table 2: Comparison of Common Biomolecule Immobilization Techniques
| Immobilization Method | Mechanism | Advantages | Disadvantages | Impact on Biosensor Performance |
|---|---|---|---|---|
| Physical Adsorption | Van der Waals, electrostatic, hydrophobic forces [64] | Simple and rapid; no chemical modification [64] | Poor reproducibility; enzyme leaching/denaturation [61] [64] | Low stability; often unsuitable for long-term fermentation monitoring |
| Covalent Binding | Stable covalent bonds between functional groups on the sensor surface and the bioreceptor [64] | Strong, stable attachment; high reproducibility [64] | Risk of bioactivity loss; complex procedure [64] | High stability and reusability; ideal for robust sensor fabrication |
| Affinity Binding | Specific, non-covalent interactions (e.g., avidin-biotin, His-tag/Ni-NTA) [64] [66] | Controlled, oriented immobilization; preserves bioactivity [64] | Requires genetic/chemical modification of the bioreceptor [64] | High sensitivity due to proper orientation; good stability |
| Entrapment | Bioreceptor confined within a polymeric matrix (e.g., silica gel, polymer) [64] | Mild conditions; protects the bioreceptor [64] | Diffusion limitations; can lead to increased response time [64] | Good stability; useful for whole-cell or tissue-based sensors |
| Cross-linking | Bioreceptors linked to each other or an inert protein using bifunctional agents (e.g., glutaraldehyde) [64] | Strong binding; high bioreceptor loading [64] | Can cause significant activity loss and conformational changes [64] | High stability but potentially lower sensitivity |
Detailed Protocol: Covalent Immobilization of an Enzyme for Metabolite Monitoring
This protocol details the covalent immobilization of a typical enzyme, such as Glucose Oxidase or Glutamate Oxidase, onto a silanized optical sensor surface (e.g., optical fiber, SPR chip) for monitoring metabolite levels during fermentation [65].
Research Reagent Solutions & Essential Materials:
- Sensor Chip: Silicon photonic or plasmonic transducer chip.
- Bioreceptor: Purified enzyme (e.g., Glucose Oxidase, Glutamate Oxidase).
- (3-Aminopropyl)triethoxysilane (APTES): Used to create an amine-functionalized surface.
- Glutaraldehyde: A bifunctional crosslinker that reacts with amine groups.
- 1-Ethyl-3-(3-dimethylaminopropyl)carbodiimide (EDC) & N-Hydroxysuccinimide (NHS): A common coupling system for activating carboxyl groups.
- Ethanolamine: Used to block unreacted active sites.
- Phosphate Buffered Saline (PBS), pH 7.4: Standard buffer for preparation and washing steps.
- Bovine Serum Albumin (BSA): Used as a blocking agent to minimize non-specific adsorption.
Procedure:
- Surface Cleaning: Clean the sensor chip thoroughly in an oxygen plasma cleaner or piranha solution (Caution: highly corrosive) to create reactive hydroxyl groups. Rinse extensively with deionized water and dry under a stream of nitrogen.
- Silanization: Incubate the clean sensor chip in a 2% (v/v) solution of APTES in anhydrous toluene for 2 hours at room temperature to form an amine-terminated self-assembled monolayer. Rinse sequentially with toluene and ethanol to remove physisorbed silane.
- Crosslinker Activation: React the aminated surface with a 2.5% (v/v) solution of glutaraldehyde in PBS for 1 hour. Rinse thoroughly with PBS to remove excess glutaraldehyde.
- Enzyme Immobilization: Prepare a solution of the target enzyme (e.g., 1 mg/mL in PBS). Pipette the enzyme solution onto the activated sensor surface and incubate in a humidified chamber for 2 hours at room temperature. The amine groups on the enzyme will react with the aldehyde groups on the surface, forming stable covalent bonds.
- Quenching and Blocking: To passivate the remaining aldehyde groups, incubate the functionalized sensor with a 1M ethanolamine solution (pH 8.5) for 30 minutes. Following this, incubate with a 1% (w/v) BSA solution in PBS for 1 hour to block any remaining non-specific binding sites.
- Storage: Rinse the prepared biosensor with PBS and store at 4°C in PBS until use.
Diagram 1: Covalent enzyme immobilization workflow.
Sensor Surface Regeneration Strategies
Regeneration is the process of dissociating the bound target analyte from the immobilized bioreceptor without permanently damaging the sensing interface, thereby allowing the biosensor to be reused multiple times [63]. This is crucial for reducing the cost-per-test and enabling continuous, real-time monitoring in long-term fermentation processes [63] [6].
Regeneration Techniques and Their Applications
Different regeneration strategies exploit various mechanisms to disrupt the binding affinity between the bioreceptor and the analyte.
Table 3: Common Biosensor Regeneration Techniques and Performance
| Regeneration Method | Working Principle | Conditions | Efficiency / Cycles Reported | Suitable Bioreceptors |
|---|---|---|---|---|
| Chemical Regeneration | Alters pH or ionic strength to disrupt electrostatic/H-bond interactions [63] | Low/high pH buffers (e.g., Glycine-HCl pH 2.0, NaOH); high salt; chaotropic agents (e.g., urea) [63] | High efficiency; widely used; cycles depend on bioreceptor robustness [63] | Antibodies, some enzymes, aptamers |
| Thermal Regeneration | Applies localized heat to denature the analyte or weaken binding interactions [63] | Controlled temperature pulses | Moderate efficiency; can damage sensitive bioreceptors [63] | Thermally stable aptamers or proteins |
| Re-functionalization | Complete removal of the bioreceptor layer and re-application of a new layer [63] | Harsh chemicals (e.g., H₂SO₄) to strip surface, followed by re-immobilization [63] | Very high (e.g., >80 cycles reported [63]); but time-consuming | Aptamers, antibodies on robust surfaces |
| Affinity-based Reversible Systems | Uses reversible capture systems (e.g., Switchavidin, His-tag/Ni-NTA) [66] | Competitive elution (e.g., imidazole for His-tag, biotin for Switchavidin) [66] | Good for specific setups; can suffer from baseline drift [66] | His-tagged or biotinylated recombinant proteins |
| Electrochemical Regeneration | Applies electric potential to induce oxidation/reduction, disrupting bonds [63] | Specific voltage/current pulses in an electrochemical cell | High efficiency for electrochemical sensors; less common for optical | Redox-active bioreceptor systems |
Detailed Protocol: Chemical Regeneration of an Aptamer-Based Sensor
This protocol describes the regeneration of an optical biosensor functionalized with a DNA or RNA aptamer for detecting a small-molecule metabolite (e.g., ATP, caffeine) in a fermentation broth sample. Aptamers are particularly suitable for regeneration due to the reversible nature of their non-covalent binding interactions [63].
Research Reagent Solutions & Essential Materials:
- Functionalized Biosensor: Optical biosensor with the appropriate aptamer immobilized via a chosen strategy (e.g., thiol-gold or amide coupling).
- Regeneration Buffer: 10 mM Glycine-HCl, pH 2.0. Alternatively, 10 mM NaOH (pH ~12) or 4-6 M urea can be tested.
- Assay Buffer: Phosphate Buffered Saline (PBS) or HEPES, pH 7.4.
- Sample Solution: Fermentation broth sample containing the target analyte.
- Microfluidic Flow System (Recommended): For controlled and automated delivery of samples and regeneration buffers across the sensor surface.
Procedure:
- Baseline Establishment: Flush the sensor flow cell with assay buffer at a constant flow rate (e.g., 20-50 µL/min) until a stable baseline signal is achieved.
- Sample Binding: Introduce the fermentation broth sample (diluted or filtered if necessary) for a defined period (e.g., 10-15 minutes) to allow the target analyte to bind to the immobilized aptamer, resulting in a measurable signal increase.
- Rinsing: Flush with assay buffer to wash away unbound or weakly bound matrix components from the sample. The signal should stabilize at a higher level, corresponding to the bound analyte.
- Regeneration: Switch the flow to the pre-optimized regeneration buffer (e.g., Glycine-HCl, pH 2.0). Inject for a short, precise duration (e.g., 30-60 seconds). A sharp drop in the sensor signal should be observed as the low pH disrupts the binding interactions, releasing the analyte.
- Re-equilibration: Immediately switch back to the assay buffer and flush until the signal returns to the original baseline, indicating the sensor is ready for the next measurement cycle.
- Validation: The regeneration efficiency can be validated by comparing the sensor's response to a standard analyte concentration before and after multiple regeneration cycles.
Diagram 2: Sensor surface regeneration cycle.
The successful application of optical biosensors in fermentation monitoring research is intrinsically linked to the robustness of the biomolecule immobilization strategy and the feasibility of sensor surface regeneration. Covalent and affinity-based immobilization methods provide the stable, oriented, and functional interfaces necessary for reliable detection in complex fermentation broths. Concurrently, chemical regeneration and surface re-functionalization strategies offer pathways to reusable sensors, which is a key economic and practical driver for their adoption in industrial bioprocessing. As the field progresses towards more intelligent and integrated bio-manufacturing, the continued development of novel, stable bioreceptors and gentle yet efficient regeneration protocols will be paramount in unlocking the full potential of optical biosensors for real-time, continuous monitoring and control of fermentation processes.
Managing Sensor Calibration Drift and Ensuring Long-Term Stability
For researchers and scientists in drug development, maintaining the accuracy and reliability of optical biosensors during fermentation processes is paramount. Calibration drift—the gradual deviation of a sensor's output from its reference value over time—poses a significant challenge to achieving precise, real-time monitoring of critical process parameters. In the context of fermentation monitoring, where processes can extend from several days to several weeks, undetected drift can compromise data integrity, leading to suboptimal process control and potentially affecting final product quality [67] [68]. This document outlines the primary sources of calibration drift and provides detailed protocols for its management and validation, ensuring the long-term stability of your optical biosensing platforms.
Understanding Drift Mechanisms and Stability Assessment
The complex fermentation environment presents multiple factors that can influence biosensor performance. Understanding these is the first step toward effective drift mitigation.
Key Mechanisms of Calibration Drift:
- Biofouling: The accumulation of cells, proteins, or other biological materials on the sensor's optical surface can alter the signal by physically interfering with the light path or by non-specifically binding to the bioreceptor elements [7] [68].
- Bioreceptor Degradation: The biological recognition elements (e.g., enzymes, antibodies, aptamers) immobilized on the sensor can denature or lose activity over time, especially when exposed to fluctuating pH, temperature, or harsh chemical conditions prevalent in fermenters [6] [69].
- Matrix Effects: Changes in the fermentation broth's composition—such as ionic strength, viscosity, or the concentration of interfering substances—can affect the optical signal independently of the target analyte concentration [67] [70].
- Physical Instability: Minor changes in the alignment of optical components or the integrity of the sensor's flow cell can lead to signal drift, particularly in online monitoring setups subject to vibration or pressure changes [6].
Evidence of remarkable long-term stability exists for certain biosensor designs. For instance, one study on a bioelectrochemical sensor reported a stable calibration for over 800 days without requiring recalibration, demonstrating the potential for highly robust systems [71]. Another study focusing on a commercial electrochemical glucose biosensor platform showed stable performance throughout a yeast fed-batch fermentation, quantifying glucose in a complex broth with minimal resource consumption compared to traditional HPLC [67] [72]. These examples highlight that with appropriate design and management, long-term stability is an achievable goal.
Table 1: Stability Performance of Representative Biosensors
| Sensor Type / Application | Reported Stability / Duration | Key Factors for Stability | Citation |
|---|---|---|---|
| Bioelectrochemical Sensor (BOD) | Stable calibration >800 days | Stable biofilm acclimatization; controlled external resistance | [71] |
| Electrochemical Glucose Biosensor (Fermentation) | Stable activity over ~600 h fermentation | Flow-through-cell design; multi-array sensor | [67] [72] |
| Microbial Biosensor (Food Safety) | Operational stability for up to a week | Bioreceptor selection and immobilization method | [7] |
Experimental Protocol for Drift Assessment and Management
This protocol provides a systematic methodology for quantifying sensor drift and validating long-term performance under simulated or real fermentation conditions.
Reagent and Sensor Preparation
Research Reagent Solutions:
- Standard Calibrants: Prepare a series of standard solutions of the target analyte (e.g., glucose, lactate) in a matrix that closely mimics the fermentation broth. For example, use a phosphate buffer at pH 5–9 for general testing, or a defined fermentation medium for more specific applications [67] [70].
- Validation Samples: Independent samples with known analyte concentrations, verified by a reference method (e.g., HPLC, enzymatic assay). These should span the sensor's operational range [68] [70].
- Cleaning and Regeneration Solutions: Depending on the sensor type, prepare mild cleaning solutions (e.g., 0.1 M NaOH, diluted ethanol) or regeneration buffers (e.g., glycine-HCl) to remove fouling agents without damaging the bioreceptor [6].
- Storage Buffer: For sensors not in continuous use, a recommended storage buffer (often provided by the manufacturer) to maintain bioreceptor stability.
Step-by-Step Drift Monitoring Protocol
Initial Calibration Curve Generation:
- Condition the biosensor according to the manufacturer's instructions.
- Measure the sensor's response to the series of standard calibrants, covering the entire expected dynamic range (e.g., 0-150 mM for glucose) [67] [72].
- Perform triplicate measurements for each standard and plot the response (e.g., fluorescence intensity, shift in resonant wavelength) against concentration.
- Fit an appropriate model (e.g., linear, sigmoidal, Hill model) to the data to establish the initial calibration curve. Record the model parameters (slope, intercept, R²) [71] [70].
Long-Term Stability Assay:
- Integrate the biosensor into the fermentation monitoring setup (at-line or on-line).
- At predefined intervals (e.g., every 24 hours), measure the response to a single, mid-range calibrant solution.
- Simultaneously, measure the response for the independent validation samples to assess predictive accuracy.
- Plot the sensor's response to the mid-range calibrant over time to visualize signal drift.
Data Analysis and Drift Quantification:
- Signal Drift: Calculate the percentage change in the response to the mid-range calibrant relative to the initial value (Day 0).
- Accuracy Drift: For each validation sample, calculate the prediction error:
(Predicted Concentration - Known Concentration) / Known Concentration * 100%. Track this error over time. - A stable sensor will show minimal change in both signal and accuracy over the operational period. A predefined threshold (e.g., ±10% deviation from the initial value) should be established to trigger corrective action or recalibration.
Corrective Actions and Recalibration Strategy
- Recalibration: If drift exceeds the acceptable threshold, a full recalibration with the series of standard calibrants must be performed.
- Sensor Regeneration/Cleaning: For biofouling or reversible contamination, implement a cleaning cycle using the prepared solutions and re-validate performance with the validation samples [6].
- Model Adjustment: In some advanced systems, where drift is consistent and predictable, it may be possible to adjust the calibration model algorithmically, though this requires robust validation [6] [70].
The Scientist's Toolkit: Essential Research Reagents
Table 2: Key Research Reagent Solutions for Drift Management
| Reagent Solution | Function | Example Application / Note | |
|---|---|---|---|
| Matrix-Matched Calibrants | Provides the primary standard for establishing the sensor's calibration curve. | Use a synthetic medium that mimics the chemical and physical properties of the actual fermentation broth to minimize matrix-effect errors. | [67] [70] |
| Independent Validation Samples | Used to verify the sensor's predictive accuracy and quantify drift without influencing the calibration model. | Concentrations should be confirmed by a reference method (e.g., HPLC). | [68] [70] |
| Sensor Cleaning Solution | Removes biofouling and other contaminants from the optical and flow-path surfaces to restore baseline performance. | Composition must be compatible with the bioreceptor; e.g., 0.1 M NaOH is common, but harsh solvents may denature proteins. | [6] |
| Storage Buffer | Preserves the activity and integrity of the immobilized bioreceptor during periods of non-use. | Typically provided by the sensor manufacturer; often contains stabilizers like BSA or glycerol. | [73] |
Workflow and Signaling Pathways for Drift Mitigation
The following diagram illustrates a logical workflow for the proactive management of calibration drift, from initial setup to data interpretation and corrective action.
Drift Management Workflow
Effective management of calibration drift is not merely a technical exercise but a fundamental requirement for generating reliable, high-quality data in fermentation research and development. By understanding the underlying mechanisms, implementing a rigorous and scheduled protocol for stability assessment, and maintaining a toolkit of essential reagents, scientists can ensure the long-term stability of their optical biosensors. This proactive approach safeguards the integrity of research outcomes and supports the advancement of robust, controlled fermentation processes in drug development.
The Role of Nanomaterials and Advanced Surface Chemistries in Enhancing Performance
Optical biosensors have emerged as a transformative technology for real-time, non-invasive monitoring within fermentation research, a field critical to pharmaceutical and bio-industrial processes. The integration of optically active nanomaterials and advanced surface chemistries has been pivotal in overcoming the limitations of traditional analytical methods, which are often offline, time-consuming, and incapable of providing the kinetic data essential for understanding dynamic bioprocesses [4] [74]. These enhancements allow biosensors to achieve exceptional sensitivity, specificity, and stability in the complex and often harsh environment of a fermenter.
The fundamental principle of these sensors involves converting a biological recognition event into a quantifiable optical signal. Nanomaterials elevate this process by providing a high-surface-area scaffold for immobilizing biorecognition elements (e.g., enzymes, antibodies), enhancing light-matter interactions via phenomena such as localized surface plasmon resonance (LSPR), and facilitating efficient signal transduction [75]. When combined with tailored surface chemistries that ensure robust and oriented immobilization, the result is a biosensor platform with significantly improved performance metrics, including lower detection limits, faster response times, and greater resilience to fouling [60] [74]. This application note details the protocols and methodologies for leveraging these advanced materials to develop high-performance optical biosensors for fermentation monitoring.
Nanomaterial Architectures and Functionalization Strategies
The selection and functionalization of nanomaterials are critical steps in biosensor design. These materials can be categorized based on their chemical composition, each offering distinct advantages for optical biosensing applications in fermentation.
Table 1: Categories of Optically Active Nanomaterials for Biosensing
| Nanomaterial Category | Key Examples | Relevant Optical Properties | Role in Biosensor Enhancement |
|---|---|---|---|
| Carbon-Based Nanomaterials | Graphene Oxide (GO), Carbon Nanotubes (CNTs), Graphene Quantum Dots (GQDs) | Strong ionic contact, sturdy π-stacking, fluorescence quenching, LSPR sensitivity enhancement [75]. | Increases enzyme binding sites, improves biocompatibility, and enhances the sensitivity of transducers like optical fibers [75]. |
| Inorganic-Based Nanomaterials | Gold Nanoparticles (AuNPs), Silver Nanoparticles (AgNPs), Quantum Dots (QDs) | Surface Plasmon Resonance (SPR), Localized SPR (LSPR), strong fluorescence, Surface-Enhanced Raman Scattering (SERS) [75] [4]. | Provides strong signal amplification, enables label-free detection, and improves the signal-to-noise ratio [75] [60]. |
| Organic-Based Nanomaterials | Molecularly Imprinted Polymers (MIPs), Lipidic and Polymeric Nanoparticles | Tailorable refractive index, biocompatibility, selective binding cavities [75]. | Offers highly specific recognition layers, protects the transducer interface, and improves sensor stability in complex matrices. |
| Composite-Based Nanomaterials | GO-AuNPs, CNT-MoS2, MOF-Polymer composites | Combines and synergizes properties of individual components [75] [74]. | Achieves multi-functional performance, such as combined signal amplification and high biomolecule loading capacity. |
The functionalization of these nanomaterials is achieved through various immobilization techniques, which are paramount to maintaining the activity of the biological recognition element and ensuring the sensor's longevity. Key methods include:
- Covalent Binding: Provides stable, irreversible attachment of biomolecules via functional groups (e.g., amine, carboxyl), minimizing leaching during long-term fermentation runs [74].
- Physical Adsorption: A simpler method based on van der Waals forces, electrostatic, or hydrophobic interactions, though it may be susceptible to desorption in changing environmental conditions [74].
- Entrapment within Polymers: Biomolecules are encapsulated within a porous polymeric matrix (e.g., hydrogel), which protects them from the fermentation broth while allowing analyte diffusion [4].
Experimental Protocols for Biosensor Development and Validation
This section provides a detailed, step-by-step methodology for fabricating, functionalizing, and validating a nanomaterial-enhanced optical biosensor, using a model system for glucose monitoring in fermentation.
Protocol: Fabrication of an LSPR-Based Optical Fiber Glucose Biosensor
Principle: This protocol describes the development of a biosensor where the surface of an optical fiber is modified with a nanocomposite of Gold Nanoparticles (AuNPs) and Graphene Oxide (GO) to enhance LSPR signals. The enzyme Glucose Oxidase (GOx) is then immobilized on this surface. The catalytic reaction of glucose produces a local change in the refractive index, causing a shift in the LSPR wavelength that is correlated with glucose concentration [75].
Materials & Equipment:
- Optical fiber (e.g., silica multimode)
- Gold (III) chloride trihydrate, Trisodium citrate, Graphene Oxide dispersion
- Glucose Oxidase (GOx) enzyme
- (3-Aminopropyl)triethoxysilane (APTES), Glutaraldehyde solution
- Phosphate Buffered Saline (PBS, 0.1 M, pH 7.4)
- UV-Vis-NIR Spectrometer, Fiber optic spectrometer
- Microfluidic flow cell or dip probe housing
Procedure:
- Fiber Probe Preparation:
- Cleave the optical fiber to ensure a clean, flat end-face. Clean sequentially with acetone, ethanol, and deionized water in an ultrasonic bath for 15 minutes each. Dry under a stream of nitrogen gas.
- Silanization: Immerse the fiber tip in a 10% (v/v) solution of APTES in ethanol for 1 hour. Rinse thoroughly with ethanol to remove unbound silane and cure at 110°C for 30 minutes. This introduces amine (-NH₂) groups to the fiber surface.
Nanomaterial Synthesis and Immobilization:
- AuNP Synthesis: Prepare a 1 mM solution of Gold (III) chloride trihydrate in deionized water. Bring to a boil under reflux with vigorous stirring. Rapidly add 10 mL of a 38.8 mM trisodium citrate solution. Continue heating and stirring until the solution turns deep red. Cool to room temperature. Characterize the AuNPs by UV-Vis spectroscopy (λmax ~520 nm) [75].
- Nanocomposite Formation: Mix the AuNP colloid with a GO dispersion (1 mg/mL) in a 1:1 volume ratio and allow it to incubate for 1 hour with mild shaking.
- Immobilization: Immerse the aminated fiber probe into the AuNP-GO nanocomposite solution for 12 hours at 4°C. The nanocomposite will adsorb to the surface via electrostatic and other physical interactions. Rinse gently with PBS to remove loosely bound material.
Enzyme Immobilization:
- Activation: Submerge the nanomaterial-coated fiber probe in a 2.5% (v/v) glutaraldehyde solution in PBS for 1 hour at room temperature. The glutaraldehyde reacts with the amine groups on the nanomaterial and fiber surface, forming aldehyde terminals.
- Binding: Rinse the probe with PBS to remove excess glutaraldehyde. Immediately incubate the probe in a solution of GOx (2 mg/mL in PBS) for 2 hours at 4°C. The enzyme's amine groups will form Schiff base linkages with the aldehyde groups.
- Quenching & Storage: To block unreacted aldehyde groups, immerse the probe in a 1 M ethanolamine solution (pH 8.5) for 30 minutes. Rinse thoroughly with PBS. The biosensor can be stored in PBS at 4°C until use.
Protocol: Calibration and Validation in a Simulated Fermentation Environment
Procedure:
- Experimental Setup: Integrate the biosensor probe into a flow cell or a dip probe assembly connected to a light source (e.g., white LED) and a spectrometer. Use software to record the transmission spectrum in real-time.
- Calibration:
- Prepare a series of glucose standards in PBS (e.g., 0 mM, 1 mM, 2 mM, 5 mM, 10 mM).
- Pump each standard past the sensor surface while monitoring the LSPR peak wavelength.
- Plot the steady-state wavelength shift (Δλ) against glucose concentration to generate a calibration curve. Fit the data with a linear or Michaelis-Menten model.
- Performance Validation:
- Specificity Test: Challenge the sensor with potential interferents found in fermentation broths (e.g., fructose, galactose, lactate) at physiologically relevant concentrations to assess cross-reactivity.
- Stability & Reusability Test: Continuously monitor a single glucose standard over 8-24 hours to determine signal drift. Perform repeated calibration cycles to assess the sensor's operational lifetime.
- Real-sample Analysis: Spike a clarified fermentation broth sample with known concentrations of glucose and measure the recovery rate to validate accuracy in a complex matrix.
The following workflow diagram illustrates the complete biosensor fabrication and testing process.
Diagram 1: Workflow for fabricating and validating a nanomaterial-enhanced optical biosensor.
The Scientist's Toolkit: Essential Reagent Solutions
The successful implementation of the protocols above relies on a suite of key reagents and materials. The table below outlines these essential components and their functions.
Table 2: Key Research Reagent Solutions for Nanomaterial-Enhanced Optical Biosensors
| Reagent / Material | Function / Application | Key Characteristics & Notes |
|---|---|---|
| Gold Nanoparticles (AuNPs) | LSPR signal amplification; core component of the transducer interface [75]. | Tunable optical properties based on size and shape; functionalizable with thiol chemistry. |
| Graphene Oxide (GO) | Increases biocompatibility and surface area for biomolecule immobilization [75]. | Provides abundant oxygen-containing functional groups for covalent attachment. |
| (3-Aminopropyl)triethoxysilane (APTES) | Silane coupling agent for introducing amine groups onto transducer surfaces (e.g., optical fibers, chips) [74]. | Enables subsequent covalent cross-linking; critical for creating a stable base layer. |
| Glutaraldehyde | Homobifunctional crosslinker for covalently linking amine-rich surfaces (from APTES) to amine-containing biomolecules (e.g., enzymes) [74]. | Creates a stable Schiff base; use should be optimized to avoid over-crosslinking and loss of activity. |
| Molecularly Imprinted Polymers (MIPs) | Synthetic antibody mimics for detecting specific metabolites (e.g., antibiotics, organic acids) [75]. | High stability and customizable for targets where biological receptors are unstable or unavailable. |
| Quantum Dots (QDs) | Fluorescent nanocrystals for highly sensitive fluorescence-based detection schemes [4]. | Size-tunable emission, high photostability; can be used as FRET donors in conjunction with quenchers. |
Quantitative Performance of Nanomaterial-Enhanced Biosensors
The integration of nanomaterials directly translates into quantifiable improvements in biosensor performance. The following table summarizes reported data from the literature, demonstrating the enhanced sensitivity and lower detection limits achievable with various nanomaterial platforms.
Table 3: Performance Metrics of Selected Nanomaterial-Enhanced Optical Biosensors
| Sensor Platform / Technology | Target Analyte | Reported Detection Limit | Reported Sensitivity | Key Nanomaterial(s) Used |
|---|---|---|---|---|
| LSPR-Optical Fiber [75] | Creatinine | 128.4 μM | 0.0025 nm/μM | Functionalized Graphene Oxide (GO), AuNPs |
| LSPR-Optical Fiber [75] | Glucose | Not Specified | 0.93 nm/mM (enhanced with GO) | Graphene Oxide (GO) |
| Serial Tapered Fiber Structure [75] | Glucose | Not Specified | Significantly improved vs. non-nano | Multi-Walled Carbon Nanotubes (MWCNTs), GO |
| Whispering Gallery Mode (WGM) [74] | Vascular Endothelial Growth Factor (VEGF) | 17.8 fg/mL | Not Specified | Not Specified |
| Photonic Crystal Nanocavity [74] | Human IgG | 1.5 fg | 2.3 ± 0.24 × 10⁵ nm/M | Silicon-on-Insulator |
The signaling pathway of the example LSPR biosensor, from analyte binding to signal generation, is depicted below.
Diagram 2: The signaling pathway from analyte binding to measurable signal output in an LSPR biosensor.
The strategic incorporation of nanomaterials and advanced surface chemistries is no longer an option but a necessity for developing next-generation optical biosensors capable of meeting the rigorous demands of fermentation monitoring. The protocols and data presented herein provide a framework for researchers to construct sensors that offer the rapid, real-time, and highly sensitive analysis required for optimizing yields, controlling quality, and ensuring the economic viability of fermentation-based processes in pharmaceutical and industrial biotechnology. As synthesis and functionalization techniques continue to advance, these nanomaterial-enhanced platforms are poised to become indispensable tools in the rational design and control of bioprocesses.
Integration with IoT and Cloud Platforms for Real-Time Data and Predictive Control
The integration of optical biosensors with the Internet of Things (IoT) and cloud platforms is revolutionizing fermentation monitoring by enabling real-time, data-driven control over critical process parameters. This synergy provides a powerful framework for the precision management of microbial processes, which is essential for achieving consistent product quality, enhancing yield, and reducing production losses in pharmaceutical and biotechnology applications [36] [57]. These technologies facilitate a shift from traditional, static endpoint measurements to a dynamic, proactive approach to bioprocess control.
Within fermentation research and industrial bioprocessing, this integration addresses longstanding challenges such as microbial variability and batch-to-batch inconsistencies [57]. Optical biosensors act as the critical data acquisition point, continuously monitoring key biomarkers. IoT architecture wirelessly transmits this data to cloud computing resources, where artificial intelligence (AI) and machine learning (ML) models perform predictive analytics, enabling preemptive interventions and optimizing the entire fermentation lifecycle [36] [76].
System Architecture and Workflow
The operational framework for IoT-enabled optical biosensing is structured in distinct layers, each performing a specific function to create a closed-loop control system.
The system is typically organized into three primary layers:
- Perception/Physical Layer: This layer comprises the physical hardware, including optical biosensors (e.g., SPR, fluorescence-based) and conventional sensors (e.g., pH, temperature, dissolved oxygen) that are in direct contact with the fermentation broth [36] [1].
- Network Layer: This layer is responsible for data communication and connectivity. It uses protocols like Message Queuing Telemetry Transport (MQTT) or Wi-Fi to transmit sensor data from the fermentation vessel to the cloud platform securely and reliably [36].
- Application Layer: This is the data processing and user interface layer. It involves cloud-based data analytics platforms that leverage AI/ML models for predictive control. The results are visualized through custom dashboards accessible via PCs, tablets, or smartphones, providing operators with actionable insights and enabling remote control [36] [77] [76].
Logical Workflow Diagram
The following diagram illustrates the logical flow of data and control actions within the integrated system.
Key Optical Biosensing Technologies
The core of this integrated system relies on advanced optical biosensors that provide label-free, real-time analysis of biochemical interactions.
Table 1: Key Optical Biosensor Technologies for Fermentation Monitoring
| Technology | Transduction Principle | Key Measurands | Advantages | Reported Sensitivity (LOD) |
|---|---|---|---|---|
| Surface Plasmon Resonance (SPR) | Measures refractive index change at a metal-dielectric interface [1]. | Biomolecular interactions, concentration of specific proteins, antibodies [1]. | Label-free, real-time kinetic analysis. | ~0.5 nM for drug-protein interaction [1]. |
| Localized SPR (LSPR) | Light interaction with metallic nanoparticles causing localized plasmon oscillations [1]. | Environmental dielectric changes, binding events [1]. | simpler instrumentation, adaptable fabrication. | Capable of femtomolar sensitivity for DNA [2]. |
| Evanescent Wave Fluorescence | Detects fluorescence from excited molecules within a thin surface layer [1]. | pH, oxygen, ions, metabolites via fluorescent labels or dyes [1]. | High specificity, reduced background interference. | Varies with fluorescent probe used. |
| Optical Fiber Biosensors | Uses optical fibers to guide light; biorecognition element immobilized on fiber tip/cladding [1]. | Metabolites like glucose, lactate, process biomarkers [3]. | Small size, remote sensing capability, electrical passiveness. | Dependent on immobilized bioreceptor. |
Experimental Protocols for System Validation
To ensure the reliability and accuracy of the integrated monitoring system, rigorous experimental protocols must be followed. The following section provides a detailed methodology for establishing and validating a sensor-based system for a model fermentation process, such as yoghurt production, which serves as an excellent proxy for more complex pharmaceutical fermentations.
Protocol: AI-Driven Real-Time Monitoring of a Model Fermentation
Aim: To implement a real-time monitoring and predictive control system for a fermentation process using sensor-integrated hardware and AI models [76].
Materials:
- Fermentation Substrate: Defined growth medium or milk of specified composition (e.g., skim, full-cream) [76].
- Microbial Strain: Specific lactic acid bacteria (e.g., Lactobacillus delbrueckii subsp. bulgaricus, Streptococcus thermophilus) or other relevant production microorganism [76].
- Sensor Array: In-line probes for pH, Electrical Conductivity (EC), Temperature, and Total Dissolved Solids (TDS) [76].
- Data Acquisition Hardware: Microcontroller (e.g., ESP32) or PLC to interface with sensors [76] [78].
- Computing Infrastructure: Cloud platform or local server with software for data storage, ML model training, and visualization [76].
Methodology:
- System Calibration: Calibrate all sensors using standard buffer solutions and reference instruments prior to fermentation runs.
- Inoculation and Data Logging:
- Model Training and Validation:
- Collect data from multiple fermentation batches to build a comprehensive training dataset.
- Train AI/ML models (e.g., Feedforward Neural Networks - FFNN, LSTM, GRU) to predict future pH trajectories based on real-time inputs of EC, TDS, and temperature [76].
- Validate model performance on a separate test dataset using metrics like R², RMSE, and MAE.
- Real-Time Prediction and Control:
- Deploy the trained model for live fermentation tracking.
- The software should provide real-time visualization of sensor data and model predictions.
- Implement a decision-support system that triggers alerts or automated control actions (e.g., temperature adjustment) if the predicted pH trajectory deviates from the predefined optimal path [76].
Experimental Workflow Diagram
The procedural steps for the protocol are summarized in the workflow below.
Performance Data and AI Model Analysis
Quantitative assessment of the system's performance and the predictive accuracy of its AI models is critical for validation.
Table 2: Performance of AI Models in Predicting Fermentation Endpoints (e.g., pH) [76]
| AI/ML Model | R² Score | RMSE | MAE | Key Strengths |
|---|---|---|---|---|
| Feedforward Neural Network (FFNN) | 0.999 | 0.079 | 0.073 | Superior accuracy, simple architecture, fast inference [76]. |
| Long Short-Term Memory (LSTM) | >0.990 | <0.100 | <0.100 | Excels at modeling temporal dependencies in time-series data [76]. |
| Gated Recurrent Unit (GRU) | >0.990 | <0.100 | <0.100 | Similar to LSTM but with a simpler structure, computationally efficient [76]. |
| Support Vector Regression (SVR) | >0.980 | ~0.150 | ~0.140 | Effective in high-dimensional spaces [76]. |
| Gaussian Process Regression (GPR) | >0.980 | ~0.150 | ~0.140 | Provides uncertainty estimates with predictions [76]. |
The implementation of such intelligent systems has demonstrated significant process improvements. In yoghurt fermentation, an FFNN-based system achieved remarkably high predictive accuracy for pH, enabling dynamic control that surpasses conventional fixed time-temperature protocols [76]. Commercially available IoT fermentation systems report operational benefits, including up to 90% reduction in manual checks, 85% faster detection of process deviations such as stalled fermentations, and a 60% reduction in batch inconsistencies [79].
The Scientist's Toolkit: Research Reagent Solutions
The successful implementation of these advanced monitoring systems relies on a suite of essential reagents and materials.
Table 3: Essential Research Reagents and Materials for IoT-Integrated Biosensing
| Item | Function/Application | Technical Notes |
|---|---|---|
| Biorecognition Elements | Provide analytical specificity by binding to the target analyte [1] [3]. | Choice depends on target: enzymes for metabolites, antibodies for proteins, aptamers for small molecules [3]. |
| Immobilization Chemistry | Anchors the biorecognition element to the transducer surface [1]. | NHS/EDC chemistry for covalent bonding to carboxymethylated dextran chips is common in SPR [1]. |
| Sensor Chips (e.g., SPR) | Solid support with a functional layer for biomolecule immobilization [1]. | Gold film with a self-assembled monolayer (SAM) and carboxymethylated dextran is a standard configuration [1]. |
| Calibration Standards | Essential for sensor calibration and quantifying analyte concentration [3] [76]. | Use analyte solutions of known concentration in a matrix matching the fermentation broth to account for matrix effects [3]. |
| Blocking Agents (e.g., BSA) | Reduce non-specific binding to the sensor surface, minimizing false positives [3]. | Critical when analyzing complex samples like fermentation broth or cell lysates [3]. |
| Microcontroller & DAQ Hardware | The electronic interface for sensor data acquisition and transmission to the cloud [76] [78]. | Platforms like ESP32 are popular for their integrated Wi-Fi and processing capabilities [78]. |
Performance Validation and Comparative Analysis of Sensing Technologies
Optical biosensors have emerged as transformative analytical tools for fermentation monitoring, offering the potential for real-time, sensitive, and specific detection of critical biochemical parameters. These devices combine a biological recognition element with an optical transducer to convert molecular interactions into quantifiable signals [4] [80]. Within the complex and dynamic environment of fermentation processes, the ability to rapidly monitor substrates, products, and biomarkers is essential for process optimization, quality control, and metabolic engineering [65] [23]. This document establishes application notes and experimental protocols for the rigorous benchmarking of optical biosensor performance, with specific emphasis on the key metrics of sensitivity, specificity, and limit of detection (LOD) relevant to fermentation research and development.
The fundamental advantage of optical biosensing in fermentation lies in its capability for label-free analysis, real-time monitoring, and minimal sample preparation [4]. Techniques such as Surface Plasmon Resonance (SPR), fluorescence, and interferometry enable researchers to track metabolic fluxes without disrupting the fermentation process. The integration of artificial intelligence (AI) and machine learning algorithms further enhances analytical performance by improving signal processing, pattern recognition, and automated decision-making capabilities [27] [26]. For drug development professionals, these advancements translate to more reliable process analytical technology (PAT) tools for ensuring product quality and regulatory compliance in biopharmaceutical fermentation.
Key Performance Metrics in Biosensor Evaluation
The performance of optical biosensors for fermentation monitoring is quantitatively assessed through several critical metrics that determine their analytical utility and reliability in complex biological matrices.
- Sensitivity: In optical biosensing, sensitivity reflects the magnitude of signal change per unit change in analyte concentration. For SPR biosensors, this is typically expressed as the angular shift per refractive index unit (RIU) (e.g., °/RIU) [81]. In concentration-dependent measurements, sensitivity may be reported as signal change per unit concentration (e.g., µA mM⁻¹ cm⁻² for electrochemical signals adapted to optical formats) [82]. Higher sensitivity enables detection of minor metabolic fluctuations in fermentation broths.
- Specificity: This metric defines a biosensor's ability to exclusively detect the target analyte amidst complex fermentation media containing nutrients, cells, proteins, and other interferents. Specificity is primarily conferred by the biorecognition element (e.g., enzymes, antibodies, nucleic acids) immobilized on the sensor surface [4] [23]. For instance, enzyme-based biosensors leverage substrate-specific enzymes like glucose oxidase or glutamate oxidase to ensure selective detection of their respective analytes [65].
- Limit of Detection (LOD): The LOD represents the lowest analyte concentration that can be reliably distinguished from background noise. It is a crucial parameter for monitoring low-abundance metabolites or early process indicators. LOD is typically calculated as 3.3 × σ/S, where σ is the standard deviation of the blank signal and S is the sensitivity of the calibration curve [82] [81]. In fermentation applications, low LOD values are essential for tracking substrate depletion or product formation kinetics.
- Dynamic Range: The concentration interval over which the biosensor provides a quantifiable response, bounded by the LOD at the lower end and signal saturation at the upper end. This determines the biosensor's applicability across different stages of fermentation with varying analyte concentrations.
- Response Time: The time required for the biosensor to reach a stable signal output following analyte exposure. Rapid response times are critical for real-time process monitoring and control in dynamic fermentation environments [4].
Table 1: Key Performance Metrics for Optical Biosensor Evaluation
| Metric | Definition | Typical Units | Importance in Fermentation Monitoring |
|---|---|---|---|
| Sensitivity | Signal change per unit analyte concentration | °/RIU, nm/M, µA mM⁻¹ cm⁻² | Detects subtle metabolic changes |
| Specificity | Ability to distinguish target from interferents | Dimensionless (often reported as % cross-reactivity) | Ensures accurate measurement in complex broth |
| Limit of Detection (LOD) | Lowest detectable analyte concentration | M, mg/L, ng/mL | Enables tracking of low-abundance metabolites |
| Dynamic Range | Concentration interval with quantifiable response | Orders of magnitude (e.g., 10⁻⁶ to 10⁻² M) | Covers varying substrate/product levels |
| Response Time | Time to reach 90% of final signal | Seconds to minutes | Supports real-time process control |
Performance Benchmarking of Optical Biosensing Techniques
Various optical biosensing platforms have been developed, each with distinct operating principles and performance characteristics suited to different fermentation monitoring applications. The benchmarking data presented below provides a comparative analysis of these technologies.
Table 2: Performance Benchmarking of Optical Biosensing Techniques for Fermentation Analytics
| Biosensor Type | Principle | Sensitivity | LOD | Specificity Mechanism | Fermentation Application Examples |
|---|---|---|---|---|---|
| Surface Plasmon Resonance (SPR) | Refractive index change at metal interface | 197.70°/RIU [81] | 16.73 ng/mL (for α-fetoprotein) [82] | Antibody-antigen recognition | Biomass monitoring, metabolite detection |
| Localized SPR (LSPR) | Enhanced EM fields at nanostructures | ~2-5x better than conventional SPR [4] | Sub-nM range [4] | Enzyme-substrate interaction | Trace contaminant detection |
| Fluorescence-Based | Fluorescence emission changes | N/A | ~10³ CFU/mL (for pathogens) [83] | Nucleic acid hybridization | Microbial contamination detection |
| SERS | Enhanced Raman scattering | N/A | Single molecule level [4] | Molecular vibration fingerprints | Product quality verification |
| Interferometric | Phase shift of light waves | High mass sensitivity [4] | pg/mm² level [4] | Biomolecular binding | Multiplexed analyte detection |
| Colorimetric | Visible color changes | Visual detection limit | µM-mM range [4] | Enzyme-catalyzed reactions | Process endpoint determination |
The data reveals that SPR and LSPR biosensors offer excellent sensitivity and low LOD values, making them suitable for monitoring low-concentration analytes in fermentation processes. Fluorescence-based biosensors provide robust detection for microbial contaminants, a critical application in sterile fermentation operations. SERS platforms deliver exceptional specificity through molecular fingerprinting, enabling precise product quality verification. The choice of biosensor technology depends heavily on the specific fermentation parameter being measured and the required performance specifications.
Recent advancements in nanomaterial integration have significantly enhanced biosensor performance. The incorporation of two-dimensional materials like MoSe₂ in SPR biosensors has demonstrated improved sensitivity and lower LOD compared to conventional designs [81]. Similarly, the use of highly porous gold structures with polyaniline and platinum nanoparticles in electrochemical sensors adapted for optical detection has achieved sensitivities of 95.12 ± 2.54 µA mM⁻¹ cm⁻² for glucose monitoring [82]. For fermentation monitoring where glucose is a key substrate, such enhancements enable more precise control of feeding strategies.
Experimental Protocols for Biosensor Benchmarking
Protocol: SPR Biosensor Performance Evaluation for Metabolite Detection
4.1.1 Principle Surface Plasmon Resonance biosensors detect analyte binding through changes in the refractive index at the interface between a metal film (typically gold) and the fermentation medium. This protocol outlines the procedure for evaluating SPR biosensor performance specifically for monitoring metabolites relevant to fermentation processes.
4.1.2 Materials and Reagents
- SPR instrument with flow cell system
- Sensor chips (e.g., carboxymethyl dextran-coated gold films)
- Coupling reagents: N-hydroxysuccinimide (NHS), 1-ethyl-3-(3-dimethylaminopropyl) carbodiimide (EDC)
- Ethanolamine hydrochloride (1.0 M, pH 8.5)
- Analytes of interest (e.g., glucose, glutamate, lactate)
- Fermentation broth samples (centrifuged and filtered)
- Running buffer (e.g., HEPES buffered saline, pH 7.4)
4.1.3 Experimental Workflow
4.1.4 Step-by-Step Procedure
Sensor Surface Preparation
- Mount the sensor chip in the SPR instrument according to manufacturer instructions.
- Prime the system with running buffer at a flow rate of 5-10 µL/min until a stable baseline is established (±1 RU for 5 minutes).
Biorecognition Element Immobilization
- Activate the carboxyl groups on the sensor surface by injecting a 1:1 mixture of 0.4 M EDC and 0.1 M NHS for 7 minutes.
- Dilute the biorecognition element (e.g., antibody, enzyme) to 10-50 µg/mL in sodium acetate buffer (pH 4.5-5.5).
- Inject the biorecognition element solution for 10-15 minutes to achieve immobilization levels of 5,000-15,000 RU.
- Block remaining activated groups by injecting 1.0 M ethanolamine (pH 8.5) for 7 minutes.
Reference Channel Setup
- Use one flow cell as an active sensor and another as a reference surface treated similarly but without biorecognition element immobilization.
- This configuration corrects for bulk refractive index changes and non-specific binding in complex fermentation samples.
Calibration Curve Generation
- Prepare standard solutions of the target analyte in running buffer across a concentration range spanning 3-5 orders of magnitude.
- Inject each standard for 2-3 minutes at a flow rate of 20-30 µL/min, followed by a dissociation phase.
- Regenerate the surface with a mild regeneration solution (e.g., 10 mM glycine-HCl, pH 2.0-3.0) for 30-60 seconds.
- Plot maximum response units (RU) versus analyte concentration and fit with an appropriate binding model (e.g., Langmuir isotherm).
Fermentation Sample Analysis
- Centrifuge fermentation broth samples at 10,000 × g for 10 minutes and filter through a 0.22 µm membrane.
- Dilute samples as necessary in running buffer to fall within the dynamic range of the calibration curve.
- Inject samples using the same parameters as for standards.
- Calculate analyte concentrations from the calibration curve, applying dilution factors.
Performance Calculation
- Sensitivity: Determine from the slope of the linear portion of the calibration curve (RU/M).
- LOD: Calculate as the mean response of the blank + 3 times the standard deviation of the blank response, converted to concentration units.
- Specificity: Assess by challenging the sensor with structurally similar compounds and potential interferents present in fermentation media.
Protocol: Fluorescence-Based Biosensor for Microbial Contamination Monitoring
4.2.1 Principle This protocol utilizes fluorescence detection combined with nucleic acid hybridization to identify specific microbial contaminants in fermentation processes, enabling rapid detection of spoilage organisms or pathogenic contaminants.
4.2.2 Materials and Reagents
- Fluorescence spectrometer or microplate reader
- Oligonucleotide probes specific to target microbial DNA/RNA
- Fluorescent dyes (e.g., SYBR Green, molecular beacons)
- Lysis buffer for nucleic acid extraction
- Hybridization buffer
- Positive and negative control DNA samples
- PCR reagents if amplification is required
4.2.3 Procedure
Sample Preparation
- Collect 1 mL samples from the fermentation broth at regular intervals.
- Centrifuge at 8,000 × g for 5 minutes to pellet microbial cells.
- Resuspend pellet in lysis buffer and extract nucleic acids using standard protocols.
Hybridization Reaction
- Dilute fluorescently-labeled DNA probes to 1 µM in hybridization buffer.
- Mix 10 µL of sample nucleic acid with 10 µL of probe solution.
- Incubate at hybridization temperature (typically 5-10°C below probe Tm) for 15-30 minutes.
Fluorescence Measurement
- Transfer reactions to a black-walled microplate to minimize background.
- Measure fluorescence at excitation/emission wavelengths appropriate for the fluorophore used.
- Include no-template controls and positive controls in each assay run.
Data Analysis
- Calculate ΔF/F = (Fsample - Fblank)/Fblank
- Establish a threshold fluorescence value significantly above background (typically 3× standard deviation of negative controls).
- Plot ΔF/F versus microbial concentration to generate a calibration curve.
The Scientist's Toolkit: Essential Research Reagent Solutions
Successful implementation of optical biosensors for fermentation monitoring requires carefully selected reagents and materials optimized for specific detection platforms and biological recognition elements.
Table 3: Essential Research Reagents for Optical Biosensor Development and Application
| Reagent/Material | Function | Application Example | Considerations for Fermentation Use |
|---|---|---|---|
| Carboxymethyl Dextran Sensor Chips | Provides hydrogel matrix for biomolecule immobilization | SPR-based metabolite monitoring | Compatibility with fermentation broth matrices |
| EDC/NHS Crosslinking Chemistry | Activates carboxyl groups for covalent coupling | Antibody/enzyme immobilization | Stability under continuous flow conditions |
| Nafion Polymer | Reduces interferent access to sensor surface | Amperometric biosensors adapted for optical detection [65] | Charge-based exclusion of media components |
| Gold Nanoparticles | Signal amplification through plasmonic enhancement | LSPR biosensors [4] | Tunable optical properties, surface functionalization |
| Quantum Dots | Fluorescent labels with high quantum yield | Fluorescence-based pathogen detection [4] | Superior photostability for continuous monitoring |
| Molybdenum Diselenide (MoSe₂) | 2D material enhancing SPR sensitivity [81] | Viral detection (adaptable to microbial monitoring) | Enhanced light-matter interaction |
| Molecularly Imprinted Polymers | Synthetic biorecognition elements | Detection of small molecule metabolites | Superior stability versus biological elements |
| Polyaniline-Platinum Nanocomposites | Electron transfer mediation in electrochemical sensors | Glucose monitoring in interstitial fluid [82] | High conductivity and catalytic activity |
Advanced Applications in Fermentation Monitoring
Optical biosensors are finding expanding applications throughout fermentation process development and control, enabled by their diverse sensing modalities and performance characteristics.
6.1 Substrate and Metabolite Monitoring The real-time tracking of key fermentation nutrients and metabolites represents a primary application for optical biosensors. Glucose biosensors have become particularly advanced, with commercial systems available since 1975 that have revolutionized saccharification process control in fermentation industries [23]. Modern approaches incorporate nanostructured composite electrodes with highly porous gold, polyaniline, and platinum nanoparticles, achieving sensitivities as high as 95.12 ± 2.54 µA mM⁻¹ cm⁻² [82]. For amino acid monitoring, enzyme-based biosensors utilizing L-amino acid oxidases and dehydrogenases provide specific detection of glutamate, arginine, and other critical amino acids in fermentation media [65]. These systems enable precise feeding strategies and metabolic engineering interventions.
6.2 Microbial Contamination Detection Rapid identification of contaminating microorganisms represents another critical application, particularly in pharmaceutical fermentations where product purity is paramount. SERS-based immunoassays using Au-Ag nanostar platforms have demonstrated detection capabilities for biomarkers like α-fetoprotein with LOD values of 16.73 ng/mL [82]. Similar approaches can be adapted for microbial antigen detection. For nucleic acid-based detection, rolling circle amplification (RCA) enables spatially resolved signal amplification for single molecule counting assays, providing ultra-sensitive detection of microbial pathogens without compartmentalization requirements [82].
6.3 Process Analytical Technology (PAT) Implementation The integration of optical biosensors into PAT frameworks for biopharmaceutical manufacturing represents a growing application area. SPR biosensors functionalized with specific biorecognition elements can monitor critical quality attributes in near real-time, enabling advanced control strategies. The combination of these sensing platforms with AI and machine learning algorithms enhances their analytical performance through intelligent signal processing, pattern recognition, and automated decision-making capabilities [27]. Furthermore, the integration of optical biosensors with IoT and cloud computing creates networked monitoring systems for distributed fermentation operations [27].
Visualization of Biosensor Signaling Pathways
The molecular and biochemical mechanisms underlying optical biosensor operation involve specific signaling pathways that translate molecular recognition into detectable optical signals.
The signaling pathway begins with analyte presence in the fermentation broth, followed by specific molecular recognition events including antigen-antibody binding, enzyme-substrate reactions, or nucleic acid hybridization. These recognition events trigger signal transduction pathways that vary by biosensor design, including refractive index changes in SPR, fluorescence emission in fluorescence-based biosensors, or colorimetric changes in visual detection systems. The final stages involve optical signal generation and processing to produce quantitative outputs correlating with analyte concentration.
Optical biosensors represent powerful analytical tools for advancing fermentation monitoring research and development. Through rigorous benchmarking of sensitivity, specificity, and LOD metrics, researchers can select appropriate biosensing platforms for specific fermentation applications. The experimental protocols and performance data presented herein provide a foundation for standardized evaluation of these critical analytical devices. As optical biosensor technology continues to evolve through integration with nanomaterials, AI algorithms, and IoT connectivity, their role in fermentation monitoring and control will expand, enabling more efficient bioprocess development and enhanced product quality assurance for drug development professionals. Future developments should focus on addressing current challenges related to long-term stability in complex fermentation matrices, standardization of performance metrics, and multiplexing capabilities for simultaneous monitoring of multiple critical process parameters.
Optical vs. Electrochemical Sensors: A Direct Comparison of Transduction Mechanisms
Biosensors have revolutionized analytical chemistry by integrating a biological recognition element with a physical transducer to create self-contained analytical devices. The core of any biosensor lies in its transduction mechanism, which converts a biological event into a quantifiable electrical or optical signal. Among the various transduction methods, optical and electrochemical techniques have emerged as the most prominent, particularly for applications requiring high sensitivity, portability, and real-time monitoring capabilities. In fermentation monitoring research, where parameters like glucose, lactate, ethanol, and biomass must be tracked in real-time to optimize yield and process control, the choice between optical and electrochemical biosensors represents a critical decision point for researchers and process engineers. This application note provides a direct comparison of these two dominant transduction mechanisms, offering structured quantitative data and detailed experimental protocols to guide sensor selection and implementation for fermentation monitoring applications.
The fundamental distinction between these sensing modalities lies in their signal generation and detection principles. Electrochemical biosensors measure electrical signals—current, potential, or impedance—resulting from biochemical reactions at electrode interfaces. In contrast, optical biosensors detect changes in light properties—wavelength, intensity, polarization, or phase—induced by analyte binding or catalytic reactions. Each approach offers distinct advantages and limitations in terms of sensitivity, detection limits, multiplexing capability, and compatibility with complex biological matrices like fermentation broths.
Fundamental Principles and Mechanisms
Electrochemical Transduction Mechanisms
Electrochemical biosensors operate by detecting electrical signals generated from biochemical reactions occurring at the interface between a biological recognition element and an electrode transducer. These sensors primarily function through three distinct mechanisms: amperometry, potentiometry, and impedimetry, each with unique operating principles and applications in fermentation monitoring.
Amperometric sensors represent the most common electrochemical approach, particularly for enzyme-based detection schemes relevant to fermentation. These devices apply a constant potential to an working electrode and measure the resulting current from the oxidation or reduction of electroactive species. In fermentation applications, this typically involves monitoring the enzymatic production or consumption of molecules like H₂O₂ or NADH. For example, glucose oxidase-based sensors detect glucose by measuring the current generated from H₂O₂ oxidation at a platinum electrode [5] [84]. The measured current is directly proportional to the analyte concentration, following the relationship i = nFAJ, where n is the number of electrons transferred, F is Faraday's constant, A is the electrode area, and J is the flux of the electroactive species. This direct proportionality enables quantitative analysis with excellent sensitivity and low detection limits, typically in the micromolar to nanomolar range.
Potentiometric sensors differ fundamentally by measuring the potential difference between working and reference electrodes under conditions of zero current flow. This potential develops according to the Nernst equation (E = E⁰ + (RT/nF)ln(a)), where E is the measured potential, E⁰ is the standard potential, R is the gas constant, T is temperature, n is the number of electrons transferred, F is Faraday's constant, and a is the activity of the target ion. In fermentation systems, potentiometric sensors commonly incorporate ion-selective membranes or field-effect transistors (FETs) to monitor pH changes resulting from enzymatic reactions, such as urease-catalyzed urea hydrolysis [5] [84]. While generally less sensitive than amperometric approaches, potentiometric sensors offer superior miniaturization potential and are less susceptible to fouling in turbid fermentation broths.
Impedimetric sensors represent a more recent development in electrochemical biosensing, measuring changes in the electrical impedance of the electrode-electrolyte interface resulting from biorecognition events. Unlike amperometric and potentiometric sensors that target specific reaction products, impedimetric sensors monitor the interfacial properties through techniques like electrochemical impedance spectroscopy (EIS). This approach is particularly valuable for label-free detection of binding events, such as antibody-antigen interactions or cellular adhesion, without requiring enzymatic amplification. In fermentation monitoring, impedimetric sensors can track microbial growth, biomass accumulation, or specific metabolite binding through changes in charge transfer resistance or double-layer capacitance [85] [86].
Optical Transduction Mechanisms
Optical biosensors detect and quantify biological interactions through changes in the properties of light, offering diverse signaling modalities that can be tailored to specific fermentation monitoring requirements. These sensors leverage various optical phenomena, including surface plasmon resonance, fluorescence, chemiluminescence, and reflectance, each with distinct mechanisms and implementation considerations.
Surface Plasmon Resonance (SPR) represents one of the most widely utilized optical transduction mechanisms for label-free, real-time monitoring of biomolecular interactions. SPR sensors operate by detecting changes in the refractive index at the interface between a metal surface (typically gold) and the sample medium. When target analytes bind to recognition elements immobilized on the sensor surface, the local refractive index increases, causing a shift in the resonance angle or wavelength of reflected light [85] [4]. This shift can be monitored in real-time, enabling kinetic analysis of binding events without requiring secondary labels or reagents. In fermentation monitoring, SPR biosensors have been successfully applied to track biomarkers, contaminants, and process indicators with high sensitivity. Recent advancements have led to the development of specialized SPR configurations with enhanced performance characteristics. Pedestal High-Contrast Gratings (PHCG) represent an innovative dielectric sensing platform that exhibits significantly improved sensitivity compared to conventional designs. Experimental studies demonstrate that PHCG structures achieve bulk refractive index sensitivity of 536 nm/RIU, representing an 11.2% improvement over conventional HCG designs, along with substantially better limits of detection for model proteins like avidin (2.1 ng/mL for PHCG vs. 3.2 ng/mL for conventional HCG) [87].
Fluorescence-based biosensors operate through the detection of changes in fluorescence intensity, lifetime, or polarization resulting from biorecognition events. These sensors typically employ fluorescent labels—such as organic dyes, quantum dots, or lanthanide complexes—that are directly attached to recognition elements or whose emission properties are modulated by analyte binding. Fluorescence detection offers exceptional sensitivity, often reaching picomolar to femtomolar detection limits, making it suitable for tracking low-abundance analytes in fermentation systems [85] [4]. Common implementations include fluorescence resonance energy transfer (FRET) assays, where analyte binding alters the distance between donor and acceptor fluorophores, and molecular beacons, where conformational changes upon target binding modulate fluorescence emission. Recent innovations have enhanced fluorescence detection through signal amplification strategies. Surface-Enhanced Fluorescence (SEF) leverages metallic nanostructures to amplify fluorescence signals by coupling fluorophores with localized surface plasmons, significantly improving detection sensitivity for targets like allergens and trace contaminants [4].
Chemiluminescence and colorimetric biosensors offer simplified detection schemes that are particularly well-suited for point-of-care and field-deployable fermentation monitoring systems. Chemiluminescence biosensors detect light emission generated as a byproduct of chemical reactions, typically catalyzed by enzyme labels such as horseradish peroxidase or alkaline phosphatase. This approach provides exceptionally low background signals since no external light source is required, resulting in high signal-to-noise ratios and improved detection limits [85]. Colorimetric biosensors, in contrast, produce visible color changes that can be detected with the naked eye or simple optical readers, making them extremely user-friendly and cost-effective. These sensors often utilize gold nanoparticles, enzyme-linked reactions, or pH-responsive dyes that generate color changes proportional to analyte concentration. The simplicity and portability of colorimetric detection have made it the foundation for numerous commercial lateral flow immunoassays, including pregnancy tests and SARS-CoV-2 rapid tests [85]. In fermentation applications, colorimetric sensors are valuable for on-line monitoring of key process parameters like pH, glucose, or contaminant levels.
Comparative Performance Analysis
Quantitative Comparison of Sensor Performance
Table 1: Direct Performance Comparison of Optical and Electrochemical Biosensors
| Performance Parameter | Electrochemical Sensors | Optical Sensors | Fermentation Monitoring Implications |
|---|---|---|---|
| Sensitivity | High (nA-μA range) [85] | Very High (SPR: 536 nm/RIU for PHCG) [87] | Optical preferred for trace metabolites; electrochemical suitable for major fermentation indicators |
| Detection Limit | nM-pM range [86] | pM-fM range (SPR: 2.1 ng/mL for avidin) [87] | Optical better for low-abundance contaminants; electrochemical adequate for primary metabolites |
| Response Time | Seconds to minutes [5] | Real-time to minutes (SPR: real-time) [4] | Both suitable for real-time process monitoring with optical offering faster kinetics |
| Multiplexing Capacity | Limited (requires multiple electrodes) [85] | High (wavelength, spatial encoding) [4] | Optical superior for multi-analyte panels in complex fermentation processes |
| Sample Volume | μL-mL range [84] | μL range (miniaturized systems) [85] | Optical better for microfermentation systems with volume constraints |
| Turbidity Tolerance | Moderate (fouling concerns) [86] | Low (light scattering interference) [4] | Electrochemical more robust for dense fermentation broths |
| Instrument Complexity | Low to moderate [85] | Moderate to high [85] | Electrochemical more suitable for field-deployable fermentation monitoring |
| Cost per Analysis | Low [85] | Moderate to high [85] | Electrochemical more economical for high-frequency routine monitoring |
Fermentation Monitoring Application Suitability
Table 2: Sensor Selection Guide for Specific Fermentation Monitoring Applications
| Fermentation Application | Recommended Transduction Mechanism | Rationale | Implementation Example |
|---|---|---|---|
| Glucose Monitoring | Amperometric electrochemical [5] | Robust, cost-effective, established technology | Glucose oxidase-modified electrode measuring H₂O₂ production |
| Protein Biomarker Detection | SPR optical [87] [4] | Label-free, real-time kinetics, high sensitivity | PHCG biosensor for avidin detection (LoD: 2.1 ng/mL) |
| Pathogen Contamination | Fluorescence optical [4] | Ultra-sensitive, multiplexing capability | Quantum dot-labeled immunoassay for bacterial detection |
| pH Monitoring | Potentiometric electrochemical [84] | Direct measurement, miniaturization potential | Ion-selective field-effect transistor (ISFET) |
| Biomass Estimation | Impedimetric electrochemical [86] | Label-free, correlates with cell density | Interdigitated electrode array measuring broth impedance |
| Toxin/Metabolite Screening | Colorimetric optical [4] | Rapid, visual readout, minimal instrumentation | Gold nanoparticle aggregation assay for mycotoxins |
| Oxygen Dissolved Levels | Amperometric electrochemical [5] | Direct measurement, robust performance | Clark-type oxygen electrode |
| Antibiotic Residues | SPR or LSPR optical [4] | Sensitive, label-free, real-time monitoring | SPR immunosensor for penicillin detection |
Experimental Protocols
Protocol: Electrochemical Glucose Biosensor for Fermentation Monitoring
This protocol describes the fabrication and implementation of a disposable amperometric biosensor for continuous glucose monitoring in fermentation processes, based on well-established enzyme electrode principles with enhancements for fermentation broth compatibility [5] [84].
Research Reagent Solutions:
Table 3: Essential Materials for Electrochemical Glucose Biosensor
| Reagent/Material | Function | Specifications/Alternatives |
|---|---|---|
| Glucose oxidase (GOx) | Biological recognition element | ≥100 U/mg, from Aspergillus niger |
| Carbon working electrode | Signal transducer | Screen-printed carbon, 3 mm diameter |
| Nafion perfluorinated resin | Interference rejection layer | 5% wt solution in lower aliphatic alcohols |
| Glutaraldehyde | Crosslinking agent | 25% aqueous solution |
| Bovine serum albumin (BSA) | Enzyme stabilizer | ≥98% purity |
| Phosphate buffer saline (PBS) | Electrolyte and dilution medium | 0.1 M, pH 7.4 |
| Fermentation broth sample | Test matrix | Filtered through 0.45 μm membrane |
Step-by-Step Procedure:
Electrode Pretreatment: Clean the screen-printed carbon electrode by cycling the potential between 0 V and +1.0 V (vs. Ag/AgCl reference) in 0.1 M PBS, pH 7.4, until a stable background current is achieved (typically 10-15 cycles at 100 mV/s).
Enzyme Immobilization: Prepare the enzyme cocktail by mixing 10 μL glucose oxidase (1000 U/mL), 5 μL BSA (10% w/v), and 2 μL glutaraldehyde (2.5% v/v). Apply 5 μL of this mixture to the working electrode surface and allow to crosslink for 1 hour at 4°C in a humidified chamber.
Membrane Application: After enzyme immobilization, apply 5 μL of Nafion solution (diluted 1:20 with ethanol) to form an interference-rejection layer. Dry for 30 minutes at room temperature.
Calibration Curve Generation: Prepare glucose standards in filtered fermentation medium at concentrations of 0, 2, 5, 10, and 20 mM. Measure the amperometric response at +0.6 V vs. Ag/AgCl after stabilization (approximately 30 seconds). Plot current versus concentration to generate the calibration curve.
Sample Measurement: Filter fermentation broth samples through a 0.45 μm membrane to remove particulate matter. Apply 50 μL of filtered sample to the sensor and measure the steady-state current after 30 seconds. Calculate glucose concentration using the established calibration curve.
Quality Control: Include a quality control standard (5 mM glucose in fermentation medium) with each analysis batch. Sensor-to-sensor reproducibility should be within ±10% coefficient of variation.
Troubleshooting Notes:
- If sensitivity declines, check enzyme activity and crosslinking time
- If linearity is poor at high concentrations, consider adding a diffusion-limiting membrane
- If interference is observed, increase Nafion concentration or layer thickness
Protocol: SPR-Based Biomarker Detection in Fermentation Broths
This protocol describes the implementation of a pedestal high-contrast grating (PHCG) SPR biosensor for sensitive, label-free detection of protein biomarkers in fermentation samples, based on advanced dielectric sensing platforms with enhanced sensitivity [87] [4].
Research Reagent Solutions:
Table 4: Essential Materials for SPR Biosensor
| Reagent/Material | Function | Specifications/Alternatives |
|---|---|---|
| PHCG sensor chip | Optical transducer | Silicon-based with pedestal structure [87] |
| Amino-propyltrimethoxysilane (APTMS) | Surface functionalization | ≥97% purity |
| Biotinylated recognition element | Capture probe | Biotinylated antibody or aptamer |
| Streptavidin | Linker molecule | Recombinant, >90% purity |
| Phosphate buffer saline (PBS) | Running buffer | 0.01 M, pH 7.4 |
| Regeneration solution | Surface regeneration | 10 mM glycine-HCl, pH 2.0 |
| Analytic standards | Calibration | Purified target protein in fermentation medium |
Step-by-Step Procedure:
Sensor Surface Functionalization: Clean the PHCG sensor surface with oxygen plasma treatment (100 W, 30 seconds). Incubate with 2% APTMS in ethanol for 2 hours at room temperature to create amine-functionalized surfaces. Rinse thoroughly with ethanol and dry under nitrogen.
Biorecognition Element Immobilization: Activate the amine surface with 2.5% glutaraldehyde in PBS for 1 hour. Incubate with 100 μg/mL streptavidin in PBS for 4 hours at 4°C. Wash with PBS and incubate with 50 μg/mL biotinylated antibody/aptamer for 1 hour at room temperature.
System Calibration: Establish a baseline resonance wavelength with PBS flow (10 μL/min) until stable (approximately 10 minutes). Inject analyte standards (0, 1, 5, 25, 100 ng/mL) in filtered fermentation medium and monitor resonance wavelength shifts until equilibrium is reached (typically 15-20 minutes).
Sample Analysis: Filter fermentation broth through 0.22 μm membrane and dilute 1:10 in running buffer if necessary. Inject samples and monitor resonance shifts. Between samples, regenerate the surface with two 1-minute injections of glycine-HCl regeneration solution followed by re-equilibration with running buffer.
Data Analysis: Plot resonance shift versus analyte concentration to generate a binding isotherm. Calculate sample concentrations using the standard curve. For kinetic analysis, fit association and dissociation phases to appropriate models (e.g., 1:1 Langmuir binding model).
Quality Control: Include a positive control (known concentration of analyte) and negative control (analyte-free medium) with each analysis batch. The surface should be regenerated successfully for at least 20 cycles without significant loss of binding capacity.
Troubleshooting Notes:
- If baseline drift occurs, check temperature stability and buffer degassing
- If non-specific binding is observed, include blocking steps with BSA or casein
- If sensitivity is inadequate, optimize biorecognition element density on the surface
Advanced Applications in Fermentation Monitoring
Integrated Sensor Systems for Bioprocess Control
Modern fermentation monitoring increasingly relies on integrated sensor arrays that combine multiple transduction mechanisms to provide comprehensive process analytics. These systems typically incorporate both electrochemical sensors for routine metabolite monitoring (glucose, lactate, ethanol) and optical sensors for specialized applications (protein expression, contaminant detection). The complementary nature of these transduction technologies enables real-time monitoring of critical process parameters that directly impact product yield, quality, and consistency in pharmaceutical fermentation processes [4] [86].
Electrochemical sensors serve as the workhorse for continuous monitoring of central carbon metabolism in fermentation systems. Amperometric enzyme electrodes provide robust, cost-effective monitoring of key substrates like glucose and products like lactate, with response times sufficient for dynamic control applications. Meanwhile, impedimetric sensors offer label-free monitoring of biomass accumulation and cellular physiology, providing early detection of process deviations or contamination events. The compatibility of electrochemical sensors with automated sampling systems and their relatively low maintenance requirements make them ideal for incorporation into distributed control architectures for large-scale fermentation processes [85] [86].
Optical sensors complement electrochemical systems by addressing analytical challenges that require higher sensitivity, specificity, or multiplexing capability. SPR-based sensors enable real-time monitoring of recombinant protein expression and secretion, providing valuable insights for optimizing induction timing and duration in heterologous expression systems. Fluorescence sensors equipped with near-infrared (NIR) fluorophores facilitate deep-tissue monitoring in dense fermentation broths, overcoming the traditional limitation of optical methods in turbid media. Additionally, multi-analyte optical arrays permit simultaneous monitoring of multiple product quality attributes, significantly enhancing process understanding and control capability for complex biopharmaceutical production [4].
Emerging Technologies and Future Directions
The convergence of biosensing with materials science, nanotechnology, and artificial intelligence is driving the development of increasingly sophisticated fermentation monitoring platforms. Several emerging technologies show particular promise for enhancing the capabilities of both optical and electrochemical sensors in bioprocess applications.
Nanomaterial-enhanced sensors represent a significant advancement in transduction technology for fermentation monitoring. The integration of nanomaterials—including metallic nanoparticles, quantum dots, carbon nanotubes, and graphene—dramatically improves sensor performance through enhanced signal transduction, increased surface area for biorecognition element immobilization, and novel sensing mechanisms. For electrochemical sensors, nanomaterials facilitate direct electron transfer between enzymes and electrodes, enabling third-generation biosensors that operate without diffusional mediators [5]. Gold nanoparticles and carbon nanotubes significantly increase electrode surface area, leading to improved sensitivity and lower detection limits. In optical sensors, nanomaterials enable enhanced detection modalities such as surface-enhanced Raman scattering (SERS) and localized surface plasmon resonance (LSPR), providing orders-of-magnitude improvement in detection sensitivity [4]. Quantum dots offer superior photostability and tunable emission profiles compared to traditional organic fluorophores, facilitating multiplexed detection schemes.
Wearable and implantable fermentation monitors represent another frontier in bioprocess analytics. Miniaturized sensors that can be directly integrated into fermentation vessels or sampling systems enable continuous, non-invasive monitoring of critical process parameters. Electrochemical sensors are particularly well-suited for this application due to their compatibility with miniaturization, low power requirements, and robustness in challenging environments [85]. Recent advances in flexible electronics and wireless communication have facilitated the development of autonomous sensor networks that provide comprehensive, spatially-resolved monitoring throughout fermentation systems. Optical sensors based on fiber-optic configurations offer similar advantages for distributed sensing applications, particularly for monitoring parameters like pH, dissolved oxygen, and culture density [4].
Artificial intelligence and machine learning algorithms are increasingly being integrated with both optical and electrochemical sensor systems to enhance data analysis, predictive modeling, and process control. These computational approaches can extract subtle patterns from complex sensor data that may not be apparent through conventional analysis, enabling early detection of process deviations, predictive maintenance of sensor systems, and optimization of control parameters [88]. Machine learning algorithms can also compensate for sensor drift and cross-sensitivity issues, improving measurement reliability over extended fermentation cycles. The integration of AI with multi-parameter sensor arrays represents a powerful approach for enhancing process understanding and control in complex fermentation systems for pharmaceutical production.
The comparative analysis presented in this application note demonstrates that both optical and electrochemical biosensors offer distinct advantages for fermentation monitoring applications, with the optimal choice depending on specific analytical requirements, operational constraints, and performance priorities. Electrochemical biosensors provide robust, cost-effective solutions for monitoring major fermentation metabolites and process parameters, with particular strengths in operational simplicity, miniaturization potential, and compatibility with turbid fermentation broths. Optical biosensors, in contrast, offer superior sensitivity, multiplexing capability, and label-free operation for monitoring low-abundance analytes, protein biomarkers, and contaminants.
The future of fermentation monitoring lies not in selecting one transduction mechanism over the other, but in the strategic integration of both technologies to create comprehensive analytical systems that leverage their complementary strengths. The ongoing convergence of biosensing with advancements in nanotechnology, materials science, and artificial intelligence will further enhance the capabilities of both optical and electrochemical platforms, enabling increasingly sophisticated monitoring and control of fermentation processes for pharmaceutical production. As these technologies continue to evolve, they will play an increasingly critical role in optimizing bioprocess efficiency, ensuring product quality, and accelerating the development of novel biopharmaceuticals.
The accurate and timely monitoring of biochemical parameters is a critical requirement in fermentation research and industrial bioprocessing. Optical sensing techniques have emerged as vital tools for this purpose, with spectrophotometry, imaging, and LED photometry being prominent methods. This application note presents a systematic comparative evaluation of these three optical sensing approaches, contextualized within the framework of fermentation monitoring. The findings demonstrate that the Paired Emitter–Detector Diode (PEDD) method of LED photometry delivers superior performance in key sensory metrics while offering significant advantages in cost-effectiveness and scalability, making it particularly suitable for deployment in fermentation monitoring systems and other bio/chemical sensing applications [89].
Comparative Performance Analysis
A rigorous comparative study was conducted to evaluate three optical sensing approaches for colorimetric bio/chemical detection, focusing on their applicability to fermentation monitoring research. The evaluated methods included laboratory-grade spectrophotometry, portable camera-based imaging, and low-cost LED photometry utilizing the PEDD charge-discharge methodology [89].
Table 1: Overall Performance Comparison of Optical Sensing Methods
| Performance Metric | Spectrophotometry | Imaging | LED Photometry (PEDD) |
|---|---|---|---|
| Measurement Range | 1.00 (Reference) | Not Specified | ×16.39 Improvement |
| Dynamic Range | 1.00 (Reference) | Not Specified | ×147.06 Improvement |
| Accuracy | 1.00 (Reference) | Not Specified | ×1.79 Improvement |
| Sensitivity | 1.00 (Reference) | Not Specified | ×107.53 Improvement |
| Limit of Detection | Lower Performance | Lower Performance | Superior |
| Cost & Scalability | High cost, complex | Moderate cost | Low-cost, highly scalable |
| Industrial Applicability | Limited by cost and scale | Moderate | High suitability |
Table 2: Key Advantages of PEDD for Fermentation Monitoring
| Feature | Benefit for Fermentation Research |
|---|---|
| Superior Resolution & Accuracy | Enables precise tracking of metabolite concentrations and nutrient levels. |
| Enhanced Sensitivity | Allows detection of low-abundance analytes and early process deviations. |
| Low-Cost & Scalability | Facilitates deployment of multiple, parallel monitoring systems. |
| Autonomous Operation Potential | Supports continuous, real-time monitoring in bioreactor systems. |
| Reduced Complex Instrumentation | Lowers barriers to adoption in resource-limited settings. |
The results indicated that the LED-based PEDD system significantly outperformed the other two methods across multiple key sensory metrics. Specifically, when compared to spectrophotometry, the PEDD approach demonstrated a 16.39-fold improvement in measurement range, a 147.06-fold improvement in dynamic range, a 1.79-fold improvement in accuracy, and a 107.53-fold improvement in sensitivity [89]. This performance advantage, combined with its inherent cost-effectiveness and scalability, positions LED photometry as a highly promising solution for widespread industrial and field applications, including fermentation process control [89].
Experimental Protocols
Protocol 1: PEDD Setup and Calration for pH Monitoring
This protocol describes the assembly and calibration of a PEDD sensor for monitoring pH changes, a critical parameter in fermentation processes, using bromocresol green (BCG) as a pH indicator [89].
Materials:
- Bromocresol green (BCG) powder
- Ultrapure water
- 0.1 M HCl and 0.1 M KOH solutions
- 3 mL cuvettes
- 5 mm LEDs (emitter and detector)
- Microcontroller (e.g., Arduino Nano)
- 3D-printed black cuvette holder
Procedure:
- Dye Solution Preparation: Prepare a 50 µM BCG stock solution by dissolving BCG powder in ultrapure water [89].
- Sample Preparation: Generate pH solutions (e.g., pH 2–8) via controlled titration of HCl and KOH. Add BCG stock to each solution for a consistent 25 µM dye concentration [89].
- PEDD Hardware Assembly:
- Software Configuration: Implement capacitive discharge timing code on the microcontroller. The detector LED is first reverse-biased (charged), then allowed to discharge through photocurrent. The discharge time, proportional to light intensity, is measured by the microcontroller [90].
- Data Collection & Analysis:
- Place each pH standard in the cuvette holder.
- Record the discharge time for each sample.
- Construct a calibration curve by plotting discharge time against known pH values.
Protocol 2: Turbidity Monitoring in Fermentation Broths
This protocol adapts the PEDD technique for monitoring turbidity, a key parameter for tracking microbial growth in fermenters [91].
Materials:
- Formazin turbidity standards (e.g., 0-1000 NTU range)
- High-intensity 860 nm emitter LED (e.g., OSRAM SFH 4550)
- Detector LED
- 3D-printed measurement chamber
- Microcontroller
Procedure:
- System Setup: Configure the PEDD setup according to ISO 7027 standards for turbidity measurement. Use an 860 nm infrared LED as the emitter to comply with the standard [91].
- Calibration: Measure a series of diluted Formazin standards across the desired range (e.g., 0-100 NTU and 0-1000 NTU) to generate a calibration curve of discharge time versus NTU [91].
- Sample Measurement: Introduce fermentation broth samples into the cuvette and record the discharge time measurements.
- Data Interpretation: Convert discharge times to turbidity values using the calibration curve to monitor biomass density throughout the fermentation process.
The Scientist's Toolkit: Research Reagent Solutions
Table 3: Essential Materials for Optical Biosensor Implementation
| Item | Function/Application | Example/Specification |
|---|---|---|
| Bromocresol Green (BCG) | pH indicator for colorimetric sensing | Large molar extinction coefficient; suitable for visible light detection [89]. |
| Formazin Standards | Calibration of turbidity sensors | Certified reference material for establishing NTU calibration curves [91]. |
| High-Intensity IR LEDs | Light source for specific sensing applications | OSRAM SFH 4550 (860 nm) for ISO 7027-compliant turbidity measurement [91]. |
| Microcontroller | Signal processing and data acquisition | Arduino boards for implementing PEDD charge-discharge timing logic [90]. |
| 3D-Printed Cuvette Holder | Sample presentation and optical alignment | Black ABS/PLA material to minimize specular reflections and external light interference [89]. |
Technological Workflows and Signaling Pathways
The following diagrams illustrate the core operational principles and experimental workflows for the PEDD sensing technique, which is critical for understanding its application in fermentation monitoring.
The comparative data unequivocally establishes LED photometry utilizing the PEDD methodology as a superior sensing platform for fermentation monitoring applications. Its exceptional performance in resolution, accuracy, sensitivity, and detection limits, combined with inherent advantages in cost-effectiveness and scalability, provides researchers and drug development professionals with a powerful tool for enhancing bioprocess control and optimization. The experimental protocols detailed herein offer a practical framework for implementing this technology in both laboratory and industrial fermentation settings.
The integration of advanced optical biosensors into biopharmaceutical fermentation processes represents a significant leap toward intelligent bio-manufacturing. These sensors provide real-time, quantitative data on critical process parameters (CPPs) and critical quality attributes (CQAs), enabling precise control over product quality and consistency [6]. However, the deployment of these systems in a regulated production environment necessitates robust validation frameworks to ensure they meet stringent regulatory standards for safety, efficacy, and data integrity. Effective validation demonstrates that the biosensor system is fit for its intended purpose and consistently delivers reliable data for critical decision-making throughout the biopharmaceutical production lifecycle [92]. This document outlines application notes and protocols for the validation of optical biosensor systems, framed within the context of fermentation monitoring research to support regulatory compliance.
Biosensor Fundamentals and Regulatory Landscape
Structure and Classification of Optical Biosensors
Optical biosensors are analytical devices that convert a biological response into a quantifiable optical signal [93]. Their core structure consists of:
- Biological Recognition Element: A biomolecule (e.g., enzyme, antibody, nucleic acid, or whole cell) that specifically interacts with the target analyte.
- Transducer: A physicochemical component that transforms the biological interaction into a measurable optical signal.
- Signal Processing Unit: Electronics and software that amplify, process, and display the signal [6].
In fermentation monitoring, optical biosensors are primarily classified based on their transduction mechanism, with key types including:
- Localized Surface Plasmon Resonance (LSPR): Utilizes noble metal nanoparticles to detect refractive index changes near the sensor surface.
- Surface-Enhanced Raman Scattering (SERS): Employs nanostructured substrates to significantly enhance Raman scattering signals for ultra-sensitive detection [82] [94].
- Photoluminescence (PL): Measures fluorescence or phosphorescence intensity, lifetime, or anisotropy of a reporter molecule.
- Chemiluminescence/Bioluminescence: Detects light emission from a chemical or biological reaction [94].
Regulatory Principles and Quality by Design (QbD)
Regulatory frameworks for biopharmaceutical production, such as the FDA's "Guideline on General Principles of Process Validation," emphasize a lifecycle approach integrating Quality by Design (QbD) principles [92]. This involves:
- Critical Process Parameters (CPPs): Identifying and controlling process variables that directly impact CQAs.
- Critical Quality Attributes (CQAs): Defining the physical, chemical, biological, and microbiological properties of the product within appropriate limits.
- Design Space: Establishing the multidimensional combination of input variables proven to assure quality.
For a biosensor system, validation must prove that the data it generates for monitoring CPPs and CQAs is accurate, precise, specific, and reliable under actual conditions of use. This aligns with regulatory requirements for process validation, which entails collecting and evaluating data to establish scientific evidence that a process can consistently deliver a quality product [92].
Table 1: Core Regulatory Requirements for Process Validation and Corresponding Biosensor Validation Activities
| Process Validation Stage (FDA) | Biosensor Validation Activity | Key Documentation |
|---|---|---|
| Stage 1: Process Design | - Define intended use and analytical target profile (ATP).- Select appropriate biosensor technology. | - User Requirements Specification (URS).- Risk Analysis Report. |
| Stage 2: Process Qualification | - Install and qualify the biosensor system.- Verify performance under simulated process conditions. | - Installation/Operational Qualification (IQ/OQ).- Performance Qualification (PQ). |
| Stage 3: Continued Process Verification | - Ongoing calibration and monitoring of biosensor performance.- Ensure data integrity over the system's lifecycle. | - Calibration records.- Audit trails and system security logs. |
Experimental Protocols for Biosensor Validation
This section provides a detailed methodology for validating a SERS-based optical biosensor for real-time monitoring of a key metabolite during microbial fermentation, using the principles outlined above.
Protocol: Validation of a SERS-Based Biosensor for Metabolite Monitoring
1. Objective: To establish and validate a SERS-based biosensor platform for the accurate, precise, and specific quantification of α-fetoprotein (AFP)-like metabolites in a fermentation broth.
2. Principle: The protocol uses Au-Ag nanostars as the SERS substrate. Their sharp-tipped morphology provides intense plasmonic enhancement, allowing for sensitive detection of the target analyte's intrinsic vibrational modes without the need for exogenous Raman reporters [82]. The biomarker is detected by functionalizing the nanostars with specific antibodies.
3. Materials and Reagents Table 2: Research Reagent Solutions for SERS-Based Biosensor Validation
| Item | Function / Role in Experiment |
|---|---|
| Au-Ag Nanostars | SERS substrate; provides intense signal enhancement due to sharp-tipped morphology and plasmonic properties [82]. |
| Monoclonal Anti-Analyte Antibodies | Biological recognition element; provides specificity by binding to the target metabolite. |
| Mercaptopropionic Acid (MPA) | Forms a self-assembled monolayer on nanostars; facilitates covalent antibody attachment via EDC/NHS chemistry [82]. |
| 1-Ethyl-3-(3-dimethylaminopropyl) carbodiimide (EDC) | Crosslinking agent; activates carboxyl groups for amine coupling. |
| N-Hydroxysuccinimide (NHS) | Stabilizes the EDC-activated intermediate; improves coupling efficiency. |
| Analyte Standard Solutions | Used for calibration curve generation; defines the dynamic range and sensitivity. |
| Fermentation Broth (Blank & Spiked) | Complex matrix for testing specificity and robustness. |
4. Equipment
- Raman Spectrometer with a 785 nm or 633 nm laser source.
- Microtiter plates or custom flow cell for SERS measurement.
- Centrifuge for nanostar concentration tuning [82].
- pH meter and analytical balance.
5. Procedure
Step 1: Biosensor Fabrication and Functionalization
- Synthesis and Concentration of Au-Ag Nanostars: Synthesize Au-Ag nanostars via a seed-mediated growth method. Tune the concentration and enhance SERS activity by centrifuging the nanostar solution at different durations (e.g., 10, 30, 60 minutes) and re-suspending in the desired buffer [82].
- Functionalization: Incubate the concentrated nanostars with 1 mM MPA in ethanol for 1 hour to form a self-assembled monolayer. Wash twice with buffer to remove excess MPA.
- Antibody Conjugation: Activate the carboxyl groups of MPA with a fresh mixture of EDC (400 mM) and NHS (100 mM) for 30 minutes. Wash the activated nanostars and incubate with a solution of monoclonal anti-analyte antibodies (e.g., 167 µg/mL) for 2 hours at room temperature. Block any remaining active sites with 1% BSA for 30 minutes [82].
Step 2: Biosensor Calibration and Analytical Validation
- Calibration Curve: Prepare a series of analyte standard solutions in a buffer covering the expected concentration range (e.g., 0-500 ng/mL). Incubate each standard with the functionalized SERS platform for a fixed time (e.g., 15 minutes). Acquire SERS spectra and plot the intensity of the characteristic analyte peak against concentration. Fit the data using a four-parameter logistic (4PL) model.
- Determination of Analytical Figures of Merit:
- Limit of Detection (LOD): Calculate as (3.3 × standard deviation of the blank) / slope of the calibration curve. The LOD for a similar SERS immunoassay was reported as 16.73 ng/mL [82].
- Linearity: Assess via the coefficient of determination (R²) of the calibration curve. It should be ≥ 0.99.
- Precision: Perform repeatability (intra-assay) and intermediate precision (inter-assay) tests by analyzing QC samples (low, mid, high concentration) multiple times. Calculate % Relative Standard Deviation (%RSD). Acceptable precision is typically ≤ 15-20% RSD.
- Specificity/Selectivity: Test against structurally similar molecules and common fermentation broth components (e.g., sugars, amino acids, salts) to ensure no significant cross-reactivity or matrix interference.
Step 3: In-Process Validation during Fermentation
- Integration: Integrate the validated SERS biosensor into a side-stream of the bioreactor, ensuring a sterile interface and continuous flow of sample.
- Real-time Monitoring: Acquire SERS spectra at regular intervals (e.g., every 5-10 minutes). Convert the spectral data to concentration values using the pre-established calibration model.
- Parallel Testing: Correlate biosensor readings with offline reference method results (e.g., HPLC) from samples taken at the same time points to confirm accuracy throughout the fermentation run.
Diagram 1: Biosensor validation workflow.
Application Note: Validating a Fermentation Monitoring System
Title: Implementation and Validation of a Custom-Built, Real-Time Fermentation Monitoring System (FerME) for CPP Monitoring.
Background: The high cost and limited flexibility of commercial fermentation monitoring systems can constrain their broader application in R&D and production. A low-cost, modular, and validated custom system can provide a viable alternative [95].
System Description: The Fermentation Monitoring Equipment (FerME) is a modular system assembled from commercial components for real-time monitoring of gas pressure, temperature, and pH in fermentation vessels. It is fully sealed, waterproof, and operates on a stable 110V power supply, avoiding battery-related signal instability common in some wireless systems [95].
Validation Study and Results: A comparative validation study was conducted between the FerME system and a commercial benchmark (Ankom RF system) under identical fermentation conditions using rumen fluid as a biological model.
Table 3: Performance Data from FerME System Validation Study [95]
| Performance Metric | FerME System | Ankom RF System | Validation Outcome |
|---|---|---|---|
| Cumulative Gas Production | No significant difference (p > 0.05) | No significant difference (p > 0.05) | Equivalent performance |
| Key Metabolites (VFA, NH₃-N) | No significant difference (p > 0.05) | No significant difference (p > 0.05) | Equivalent performance |
| System Precision (Coefficient of Variation) | 4.8% | 13.2% | Superior precision for FerME |
| Maximum Pressure Rating | 50 psi | 10 psi | Wider operational range for FerME |
| Approx. Cost (12-channel) | ~$6,000 USD | ~$12,297 USD | Significant cost reduction |
Conclusion: The FerME system was validated as a reliable, low-cost alternative for real-time fermentation monitoring, demonstrating performance equivalent or superior to the commercial system in key areas like precision and pressure range [95]. This validation framework is directly applicable to the qualification of custom optical biosensor systems in biopharmaceutical fermentation.
Data Integrity and Lifecycle Management
For biosensors to be acceptable in a regulated GMP environment, the data they generate must adhere to strict integrity principles. The FDA's ALCOA+ criteria define data attributes: Attributable, Legible, Contemporaneous, Original, and Accurate, plus Complete, Consistent, Enduring, and Available [92].
Key Implementation Strategies:
- Computer System Validation (CSV): The biosensor's software and data management system must be validated to ensure accuracy, reliability, and consistent intended performance.
- Audit Trails: Secure, timestamped, and traceable electronic audit trails must be enabled to record all data creation, modification, and deletion events. Regular audit trail reviews are mandatory [92].
- Electronic Record Management: Procedures must be in place to ensure the completeness, consistency, and accuracy of all electronic records throughout the records retention period.
- System Security: Robust access controls with unique user logins and role-based permissions prevent unauthorized access or data manipulation.
Diagram 2: Biosensor data lifecycle and management.
The successful application of optical biosensors in regulated biopharmaceutical fermentation hinges on a systematic and thorough validation framework. This framework, encompassing analytical performance qualification, system integration, and rigorous data integrity management, provides the scientific evidence required to meet regulatory standards. By adopting the QbD principles and validation protocols outlined in this document—from the functionalization of SERS substrates to the qualification of entire monitoring systems—researchers and drug development professionals can confidently deploy these powerful tools. This enables enhanced real-time process control, optimization of bioprocesses, and ultimately, the consistent production of high-quality biopharmaceuticals. Future trends will focus on the deeper integration of AI for data analysis and the continued miniaturization and automation of biosensor systems to further advance intelligent bio-manufacturing [6].
Market Trends and Leading Players in Optical Fermentation Monitoring Solutions
The global market for fermentation monitoring sensors is experiencing robust growth, propelled by the critical need for process control in biopharmaceutical, food and beverage, and biofuel production. Optical sensing technologies are at the forefront of this expansion, enabling non-invasive, real-time monitoring of critical process parameters (CPPs) [13].
Table 1: Global Fermentation Monitoring Sensor Market Overview
| Metric | Value | Source & Year |
|---|---|---|
| Overall Market Size (2023) | ~USD 2.5 Billion | [96] |
| Projected Market Size (2025) | USD 1,250 Million | [97] |
| Projected Market Size (2032) | ~USD 4.8 Billion | [98] |
| Compound Annual Growth Rate (CAGR) | ~7.5% - 12.5% (through 2033) | [97] [98] |
This growth is largely driven by the pharmaceutical and biotechnology industries, which account for approximately 60% of the market [96]. The stringent regulatory requirements for quality control and the increasing production of complex biologics and biosimilars make high-precision optical sensors indispensable [97] [96].
Table 2: Fermentation Monitoring Market by Technology & Application
| Segment | Analysis | Key Drivers |
|---|---|---|
| Sensing Technology | Optical sensors dominate for non-invasive biomass and metabolite tracking; Electrochemical sensors are robust for pH/DO2 [13]. | Non-invasiveness, real-time data, high sensitivity [4] [13]. |
| Key Applications | Pharmaceuticals: Largest segment (antibiotics, vaccines, biologics). Food & Beverage: Rapid growth (kombucha, yogurt, craft beer). Biofuels: Optimization for yield and viability [98] [96] [99]. | Regulatory compliance, product consistency, demand for sustainable products [97] [98]. |
| Deployment Mode | Shift towards In-line sensors for real-time control; Off-line/At-line for periodic validation [98] [99]. | Demand for continuous, real-time data for automated process control [99]. |
Technological Trends and Innovations
The field of optical fermentation monitoring is being transformed by several key technological advancements that enhance data granularity and process control.
- Integration of IoT and Advanced Analytics: The adoption of Internet of Things (IoT) architectures allows for real-time data capture from multiple sensor nodes, transmitting key parameters to centralized dashboards for instant analysis and visualization [13]. This facilitates rapid process adjustments and enhances operational resilience.
- Artificial Intelligence and Machine Learning: AI and ML algorithms are being leveraged to translate raw sensor data into actionable insights. These systems enable predictive analytics, forecasting fermentation trajectories, and flagging potential deviations before they impact the batch. This supports optimized feeding schedules, automated dosing, and proactive maintenance [99] [13].
- Miniaturization and Single-Use Technologies: Sensor miniaturization allows for more flexible deployment within bioreactors of all scales. Coupled with the trend towards single-use sensor modules, this minimizes cross-contamination risks, streamlines workflows, and reduces operational costs, especially in high-throughput biopharmaceutical applications [97] [99].
- Advancements in Optical Biosensor Designs: Research continues to enhance the sensitivity and specificity of optical biosensors. Key developments include:
- Surface Plasmon Resonance (SPR) and Localized SPR (LSPR): Used for label-free, real-time detection of pathogens and contaminants. LSPR, which incorporates metallic nanoparticles, offers enhanced sensitivity for trace-level detection [4].
- Fluorescence and Chemiluminescence Biosensors: Provide exceptional sensitivity for detecting low levels of toxins and specific metabolites [4].
- Fiber-Optic Biosensors: Utilize optical fibers to guide light, increasing sensitivity and enabling monitoring in complex, opaque fermentation matrices [4].
Leading Market Players and Competitive Landscape
The market comprises a mix of established multinational corporations and emerging specialized innovators.
Table 3: Key Players in the Fermentation Monitoring Sensor Market
| Company | Primary Focus & Key Characteristics |
|---|---|
| Sartorius AG | Extensive portfolio for biopharmaceutical workflows; scalable sensor arrays and validated software suites [96] [13]. |
| Thermo Fisher Scientific Inc. | Broad portfolio of analytical instruments and sensors for bioprocessing [99] [13]. |
| Mettler-Toledo International Inc. | Intelligent, self-diagnosing sensors that support lifecycle management and regulatory compliance [96] [13]. |
| Hamilton Company | Leader in electrochemical sensor innovation; modular pH and dissolved oxygen probes [96] [13]. |
| Danaher Corporation | Provides advanced monitoring solutions through its subsidiary, Pall Corporation [99] [13]. |
The competitive dynamic is characterized by technological innovation, with leading players focusing on integrating sensors with digital twins and control systems. There is a moderate level of merger and acquisition activity as larger companies acquire specialized sensor manufacturers to expand their portfolios and market reach [97] [96].
Application Note: Protocol for Real-Time Monitoring of Bacterial Fermentation Using an In-Line Optical Dissolved Oxygen Sensor
1. Application Overview This protocol describes the methodology for using an in-line optical dissolved oxygen (DO) sensor to monitor and control a bacterial fermentation process in a stirred-tank bioreactor. Real-time DO monitoring is essential for optimizing biomass yield and product formation, as oxygen is a critical substrate for aerobic microorganisms [97] [13].
2. Experimental Workflow The following diagram illustrates the core feedback control loop established by the sensor.
3. Materials and Reagents
Table 4: Research Reagent Solutions and Essential Materials
| Item | Function/Application |
|---|---|
| Sterilizable Optical DO Probe | In-line sensor with oxygen-sensitive fluorescent patch. Excited by LED, oxygen quenches fluorescence lifetime, which is correlated to DO concentration [13]. |
| Bioreactor System | Jacketed vessel with integrated ports for sensor installation and capability for agitation, aeration, and temperature control. |
| Calibration Solutions | Zero Solution: Saturated sodium sulfite (Na₂SO₃) solution. Span Solution: Sterile, air-saturated water. Used for two-point calibration pre-sterilization [13]. |
| Fermentation Media | Contains carbon source, nitrogen source, salts, vitamins, and other nutrients specific to the bacterial strain. |
| Bacterial Inoculum | Aseptically prepared culture of the target organism in exponential growth phase. |
4. Step-by-Step Protocol
- Step 1: Sensor Calibration: Perform a two-point calibration of the optical DO sensor prior to sterilization. Immerse the sensor in the zero solution until a stable reading is achieved, then set the 0% point. Rinse and place in the span solution, set the 100% point once stable. The 100% point corresponds to air saturation at the calibration temperature and pressure [13].
- Step 2: Bioreactor Setup and Sterilization: Install the calibrated sensor into the designated bioreactor port. Assemble the bioreactor, fill with the fermentation medium, and sterilize in-place (SIP) via autoclaving or steam-in-place cycles. Ensure the sensor and its cable connections are rated for the sterilization conditions.
- Step 3: Process Parameter Initialization: Post-sterilization, connect the sensor to the transmitter and control system. Initialize and verify all process parameters: set temperature, pressure, and agitation speed. The DO setpoint is typically defined as a percentage of air saturation (e.g., 20-40%).
- Step 4: Inoculation and Process Control: Aseptically transfer the prepared inoculum to the bioreactor. The control system will maintain the DO setpoint automatically. As biomass increases and consumes oxygen, the controller will increase agitation speed and/or the flow of oxygen-enriched air to maintain the setpoint.
- Step 5: Data Recording and Analysis: Record DO levels, agitation speed, and gas flow rates throughout the fermentation. Monitor the DO trend for deviations that may indicate contamination or metabolic shifts. Correlate the oxygen uptake rate (OUR) with other process data like pH and off-gas analysis.
5. Troubleshooting and Notes
- Drift in Readings: Can occur due to biofilm formation on the sensor patch. Ensure proper sterilization and consider sensor design that mitigates fouling.
- Signal Noise: Often caused by air bubbles adhering to the sensor tip. Optimize agitator placement and sparging to minimize bubble interference.
- Data Integrity: For GMP processes, ensure the sensor system is compliant with 21 CFR Part 11, with full data audit trails [99] [13].
Application Note: Protocol for Pathogen Detection in Fermented Food Products Using a Surface Plasmon Resonance (SPR) Biosensor
1. Application Overview This protocol details the use of a Surface Plasmon Resonance (SPR) biosensor for the rapid, label-free detection of specific pathogens, such as Salmonella spp., in liquid samples from fermented food products. This method significantly reduces detection time compared to traditional culture-based techniques [4] [7].
2. Experimental Workflow The workflow for SPR-based pathogen detection is outlined below.
3. Materials and Reagents
Table 5: Research Reagent Solutions for SPR Biosensing
| Item | Function/Application |
|---|---|
| SPR Biosensor System | Instrument with a gold-coated sensor chip, fluidics system, and optical detection unit. |
| Sensor Chip | Gold-coated glass slide, often pre-functionalized with carboxymethyl dextran for biomolecule immobilization. |
| Capture Antibodies | Monoclonal or polyclonal antibodies specific to the target pathogen (e.g., Salmonella). |
| Coupling Reagents | For covalent immobilization: e.g., N-ethyl-N'-(3-dimethylaminopropyl)carbodiimide (EDC) and N-hydroxysuccinimide (NHS). |
| Running Buffer | HEPES-buffered saline (HBS) or Phosphate-buffered saline (PBS) for sample dilution and chip conditioning. |
| Regeneration Solution | Glycine-HCl (pH 2.0-3.0) to dissociate the antigen-antibody complex, allowing for chip re-use without degrading the immobilized antibody. |
4. Step-by-Step Protocol
- Step 1: Sensor Surface Functionalization: Dock a new sensor chip into the SPR instrument. Prime the system with running buffer. For covalent immobilization, inject a mixture of EDC and NHS to activate the carboxyl groups on the dextran matrix. Follow by injecting the solution of purified capture antibody, which will form an amide bond with the activated surface. Finally, inject ethanolamine to deactivate any remaining ester groups [4].
- Step 2: Sample Preparation: The fermented food sample (e.g., liquid from yogurt or kefir) must be pre-processed. This typically involves centrifugation and filtration to remove large particulates and fat globules that could non-specifically bind to the sensor surface and interfere with the reading [4] [7].
- Step 3: Sample Injection and analyte binding: Dilute the prepared sample in running buffer. Inject the sample over the functionalized sensor surface at a constant flow rate. If the target pathogen is present, it will bind to the immobilized antibodies, causing an increase in the mass on the sensor surface. This mass change alters the local refractive index, which is detected as a shift in the resonance angle and recorded in real-time as a sensorgram [4].
- Step 4: Surface Regeneration: After the binding phase, inject the regeneration solution to break the antigen-antibody bonds and wash the pathogen from the surface. This returns the sensor response to baseline, making the chip ready for the next sample analysis [4].
- Step 5: Data Analysis: The sensorgram provides both qualitative (binding response shape) and quantitative (response unit magnitude) data. The maximum response unit (RU) value during sample injection can be correlated with pathogen concentration using a pre-established calibration curve.
5. Troubleshooting and Notes
- Non-Specific Binding: A major challenge in complex food matrices. Optimize the blocking step during surface preparation and include control flow cells to subtract background signals [4].
- Signal Drift: Can be caused by temperature fluctuations or improper buffer equilibration. Ensure the instrument and all buffers are thermally equilibrated before starting the experiment.
- Validation: For regulatory compliance, this method must be validated against standard reference methods for sensitivity, specificity, and limit of detection (LOD) [4].
Conclusion
Optical biosensors represent a transformative technology for fermentation monitoring, offering unparalleled capabilities for real-time, label-free, and product-specific analysis that is directly aligned with the needs of accelerated biopharmaceutical R&D. As demonstrated, technologies like RIfS and SPR provide direct pathways to monitor critical quality attributes, while the integration of AI-based soft sensors and IoT frameworks is evolving bioprocessing into adaptive, self-optimizing systems. The future of this field lies in the continued convergence of nanotechnology, advanced data analytics, and robust, miniaturized sensor designs. For biomedical and clinical research, these advancements promise not only to enhance yield and consistency in the production of therapeutics like monoclonal antibodies and antibiotics but also to pave the way for more agile and personalized medicine manufacturing platforms. Overcoming remaining challenges in standardization and matrix complexity will be key to unlocking the full potential of optical biosensors in safeguarding product quality and driving innovation.
References
- 1. Optical biosensors - PMC [https://pmc.ncbi.nlm.nih.gov/articles/PMC4986466/]
- 2. Optical Biosensors: A Revolution Towards Quantum ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC3205924/]
- 3. A Closer Look at Biosensors: A Practical Tutorial for ... [https://www.spectroscopyonline.com/view/a-closer-look-at-biosensors-a-practical-tutorial-for-spectroscopists]
- 4. Advancements in Optical Biosensor Technology for Food ... [https://www.mdpi.com/2673-4591/106/1/6]
- 5. Biosensors in food processing - PMC - PubMed Central [https://pmc.ncbi.nlm.nih.gov/articles/PMC3671056/]
- 6. Recent advances in biosensors: structure, principles ... [https://www.sciencedirect.com/science/article/abs/pii/S1046202325001665]
- 7. Biosensors in Microbial Ecology: Revolutionizing Food ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC12300841/]
- 8. Surface plasmon resonance biosensors and their medical ... [https://www.sciencedirect.com/science/article/pii/S0956566325001824]
- 9. The Development of Foodborne Pathogen Detection and ... [https://www.mdpi.com/2079-6374/15/12/774]
- 10. Fluorescent biosensors for the detection of foodborne ... [https://pubs.rsc.org/en/content/articlehtml/2025/ay/d5ay01025j]
- 11. Special Issue : Fiber Optic Biosensing Technology - Sensors [https://www.mdpi.com/journal/sensors/special_issues/3JPIG22BFA]
- 12. Integrating AI and advanced spectroscopic techniques for ... [https://www.sciencedirect.com/science/article/pii/S0924224424005260]
- 13. Fermentation Monitoring Sensors Market Size 2025-2032 [https://www.360iresearch.com/library/intelligence/fermentation-monitoring-sensors]
- 14. Optical sensor systems for bioprocess monitoring [https://www.sciencedirect.com/science/article/abs/pii/S0167779998012475]
- 15. A Fiber-Optic Carbon Dioxide Sensor for Fermentation ... [https://www.nature.com/articles/nbt0695-597]
- 16. Biosensors for fermentation control [https://www.sciencedirect.com/science/article/abs/pii/095816699390122D]
- 17. Biologics Market Size, Growth, Latest Trends and Forecast ... [https://www.pharmiweb.com/press-release/2025-11-13/biologics-market-size-growth-latest-trends-and-forecast-2025-2029]
- 18. Precision Fermentation simply explained [https://www.susupport.com/blogs/knowledge/precision-fermentation-simply-explained]
- 19. Achieving Scalable and Sustainable Precision ... [https://www.pharmasalmanac.com/articles/achieving-scalable-and-sustainable-precision-fermentation-using-process-analytical-technology]
- 20. Editorial to the Special Issue “Advances in Optical ... [https://www.mdpi.com/2079-6374/14/9/447]
- 21. Biologics Market Size to Exceed USD ... [https://www.biospace.com/press-releases/biologics-market-size-to-exceed-usd-1-144-20-billion-by-2034-amid-rising-demand-for-monoclonal-antibodies-vaccines-and-personalized-therapies]
- 22. Biologics Market Growth & Trends 2025 to 2035 [https://www.futuremarketinsights.com/reports/biologics-market]
- 23. Biosensors and their applications – A review - PMC [https://pmc.ncbi.nlm.nih.gov/articles/PMC4862100/]
- 24. Emerging applications of label - free optical biosensors [https://www.degruyterbrill.com/document/doi/10.1515/nanoph-2016-0158/html]
- 25. . Optical studies of glycopeptide antibiotic... biosensors Monitoring [https://pubmed.ncbi.nlm.nih.gov/11569825/]
- 26. Advances in optical biosensors as alternative diagnostics ... [https://www.sciencedirect.com/science/article/pii/S2666053925000839]
- 27. Emerging trends in AI-integrated optical biosensors for point ... [https://pubs.rsc.org/en/content/articlehtml/2025/cc/d5cc04899k]
- 28. - Label biosensing: going beyond the limits free optical [https://pubs.rsc.org/en/content/articlelanding/2023/cs/d3cs00155e]
- 29. - Label multiplexed detection of diabetic retinopathy biomarkers... free [https://researchprofiles.herts.ac.uk/en/publications/label-free-multiplexed-detection-of-diabetic-retinopathy-biomarke]
- 30. Optical Biosensor Market Report | Global Forecast From ... [https://dataintelo.com/report/optical-biosensor-market]
- 31. Optical Fiber Biosensor Market Size By Application 2025 [https://www.linkedin.com/pulse/optical-fiber-biosensor-market-size-application-jmihc/]
- 32. Reflectometric Interference Spectroscopy - an overview [https://www.sciencedirect.com/topics/biochemistry-genetics-and-molecular-biology/reflectometric-interference-spectroscopy]
- 33. Comparison of methods for quantitative biomolecular ... [https://link.springer.com/article/10.1007/s00216-021-03623-x]
- 34. Comparison of methods for quantitative biomolecular ... [https://pubmed.ncbi.nlm.nih.gov/34505164/]
- 35. Review Optical fiber SERS sensors: Unveiling advances ... [https://www.sciencedirect.com/science/article/abs/pii/S0010854524002078]
- 36. IoT-enabled biosensors for real-time monitoring and early ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC11811615/]
- 37. Ion- and substrate-selective optode membranes and ... [https://www.sciencedirect.com/science/article/pii/S0925400597801736]
- 38. Biosensor for Bacterial Detection Through Color Change in ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC12384555/]
- 39. Optical Methods for Label-Free Detection of Bacteria - PMC [https://pmc.ncbi.nlm.nih.gov/articles/PMC9775963/]
- 40. Principle and Protocol of Surface Plasmon Resonance (SPR) [https://www.creativebiomart.net/resource/principle-protocol-principle-and-protocol-of-surface-plasmon-resonance-spr-361.htm]
- 41. Cellular analysis and detection using surface plasmon ... [https://www.sciencedirect.com/science/article/abs/pii/S0165993618300256]
- 42. Algorithm assisted comprehensive optimization of SPR ... [https://www.sciencedirect.com/science/article/abs/pii/S0956566325007225]
- 43. Surface Plasmon Resonance Assay for Label-Free and ... [https://www.mdpi.com/2079-6374/11/6/180]
- 44. Development and Validation of an Artificial Neural-Network ... [https://www.mdpi.com/2227-9717/11/1/297]
- 45. Fermentation Process Monitoring [https://microbiosci.creative-biogene.com/fermentation-process-monitoring.html]
- 46. Real-time and on-line monitoring of ethanol fermentation ... [https://bioresourcesbioprocessing.springeropen.com/articles/10.1186/s40643-021-00391-5]
- 47. Based on vis-NIR combined with ANN for on-line detection ... [https://www.sciencedirect.com/science/article/abs/pii/S2212429224007764]
- 48. Application of Artificial Neural Network for predicting ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC10158627/]
- 49. Electrochemical biosensors for vancomycin monitoring in ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC12570155/]
- 50. Electrochemical biosensors for vancomycin monitoring in ... [https://pubs.rsc.org/en/content/articlelanding/2025/ra/d5ra07262j]
- 51. Comparison of methods for quantitative biomolecular ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC8748344/]
- 52. Real-Time and Label-Free Monitoring of Monoclonal ... [https://www.sciencedirect.com/science/article/abs/pii/S0956566325008553]
- 53. Rapid Detection and Identification of Vancomycin-Sensitive ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC10795129/]
- 54. Biosensors with vancomycin and polymetallic metal ... [https://www.sciencedirect.com/science/article/abs/pii/S0304389424031613]
- 55. Integrated hollow microneedle-optofluidic biosensor for therapeutic drug monitoring in sub-nanoliter volumes | Scientific Reports [https://www.nature.com/articles/srep29075]
- 56. A fluorescence biosensor for therapeutic drug monitoring of vancomycin using in vivo microdialysis - PubMed [https://pubmed.ncbi.nlm.nih.gov/33608077/]
- 57. Microbial Process Control in Traditional Fermented Foods [https://www.mdpi.com/2311-5637/11/6/323]
- 58. Precision Fermentation Sensors Market Size & Share 2025 ... [https://www.360iresearch.com/library/intelligence/precision-fermentation-sensors]
- 59. Matrix-insensitive protein assays push the limits of ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC4165514/]
- 60. Recent advances in optical biosensing approaches for ... [https://www.sciencedirect.com/science/article/pii/S2590137022001625]
- 61. Biochemistry strategies for label-free optical sensor ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC9242939/]
- 62. Surface Display Technology for Biosensor Applications - PMC [https://pmc.ncbi.nlm.nih.gov/articles/PMC7294428/]
- 63. Recent progress in biosensor regeneration techniques [https://pubs.rsc.org/en/content/articlehtml/2024/nr/d3nr05456j]
- 64. Immobilization strategies to develop enzymatic biosensors [https://www.sciencedirect.com/science/article/abs/pii/S073497501100156X]
- 65. Biosensors in Fermentation Applications [https://www.intechopen.com/chapters/52219]
- 66. Regenerable Biosensors for Small-Molecule Kinetic ... [https://www.sciencedirect.com/science/article/pii/S2472555222067077]
- 67. Frontiers | Automated Electrochemical Glucose Biosensor Platform as... [https://www.frontiersin.org/journals/bioengineering-and-biotechnology/articles/10.3389/fbioe.2020.00436/full]
- 68. A Review of Sensors for the Monitoring, Modeling, and ... [https://www.mdpi.com/2311-5637/11/6/329]
- 69. Emerging applications of biorecognition elements-based ... [https://link.springer.com/article/10.1007/s44397-025-00003-3]
- 70. The Application of State-of-the-Art Analytic Tools ... [https://www.mdpi.com/2311-5637/3/4/50]
- 71. No re-calibration required? Stability of a ... [https://www.sciencedirect.com/science/article/pii/S0956566321004292]
- 72. Automated Electrochemical Glucose Biosensor Platform as ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC7253623/]
- 73. (PDF) Pre- Calibrated for Single-Use Applications Biosensors [https://www.academia.edu/17604932/Pre_Calibrated_Biosensors_for_Single_Use_Applications]
- 74. Advancements and Perspectives in Optical Biosensors - PMC [https://pmc.ncbi.nlm.nih.gov/articles/PMC11170745/]
- 75. Optically Active Nanomaterials and Its Biosensing ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC9855722/]
- 76. AI-driven real-time monitoring and predictive control ... [https://www.sciencedirect.com/science/article/abs/pii/S0956713525007261]
- 77. Precision Fermentation: Fermentation Monitoring - Real-time ... [https://www.precisionfermentation.com/]
- 78. Voice Guidance System for Color Recognition Based on IoT [https://www.techscience.com/csse/v45n1/49331/html]
- 79. Fermentation Monitoring Software and Solutions [http://plaato.io/products/smart-fermentation?srsltid=AfmBOoo0QsAo5OYS-cZJDoxmZ73giexBPg_t3a65841RdMud10cA7h5P]
- 80. Optical biosensors: a decade in review [https://www.sciencedirect.com/science/article/pii/S1110016822008249]
- 81. Mathematical modeling of a MoSe₂-based SPR biosensor for ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC11907102/]
- 82. Biosensors, Volume 15, Issue 9 (September 2025) [https://www.mdpi.com/2079-6374/15/9]
- 83. Optical Biosensors and Applications - Special Issue [https://www.mdpi.com/journal/sensors/special_issues/Optical_Biosensor_Application]
- 84. Biosensors: Electrochemical Devices—General Concepts ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC9855974/]
- 85. Electrochemical vs. Optical Biosensors for Point-of-Care ... [https://www.mdpi.com/2227-9040/11/10/546]
- 86. Comparative analysis of electrochemical and optical ... [https://www.sciencedirect.com/science/article/pii/S095656632400099X]
- 87. Pedestal High-Contrast Gratings for Biosensing [https://www.mdpi.com/2079-4991/12/10/1748]
- 88. EBS 2025 [https://www.ebs2025.com/]
- 89. A Comparative Study of Optical Sensing Methods for ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC12251617/]
- 90. LED PEDD Discharge Photometry: Effects of Software ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC8879031/]
- 91. Advances in Optical Based Turbidity Sensing Using LED ... [https://www.mdpi.com/1424-8220/22/1/254]
- 92. Fermentation Process Validation Methods [https://editverse.com/fermentation-process-validation-methods/]
- 93. Biosensor Data Management → Term [https://pollution.sustainability-directory.com/term/biosensor-data-management/]
- 94. Recent advances in nanomaterial-based optical ... [https://www.sciencedirect.com/science/article/pii/S2095177925001662]
- 95. Development and Validation of a Custom-Built System for ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC12345559/]
- 96. Fermentation Monitoring Sensors 2025 Trends and ... [https://www.archivemarketresearch.com/reports/fermentation-monitoring-sensors-821565]
- 97. Fermentation Monitoring Sensors Strategic Insights [https://www.marketreportanalytics.com/reports/fermentation-monitoring-sensors-377249]
- 98. Fermentation Monitors Market Report | Global Forecast ... [https://dataintelo.com/report/fermentation-monitors-market]
- 99. Fermentation Monitoring Sensors Market by Parameter ... [https://www.researchandmarkets.com/reports/6132733/fermentation-monitoring-sensors-market-parameter?srsltid=AfmBOor3BVB8vH_3HI7SyT1T9I4as5hc_Th6K6LFGd_Ex7Leopsv6-G0]