PEG vs. Zwitterionic Coatings: A Comparative Analysis of Antifouling Performance for Biomedical Applications
This article provides a comprehensive comparison of polyethylene glycol (PEG) and zwitterionic polymer coatings, the two leading synthetic strategies for preventing biofouling on medical devices and in drug delivery systems.
PEG vs. Zwitterionic Coatings: A Comparative Analysis of Antifouling Performance for Biomedical Applications
Abstract
This article provides a comprehensive comparison of polyethylene glycol (PEG) and zwitterionic polymer coatings, the two leading synthetic strategies for preventing biofouling on medical devices and in drug delivery systems. Tailored for researchers and drug development professionals, it explores the fundamental mechanisms of protein resistance, evaluates current fabrication methodologies, and addresses key challenges such as PEG's oxidative degradation and immunogenicity versus the mechanical limitations of zwitterionic hydrogels. By synthesizing performance data and validation criteria, this review offers evidence-based guidance for selecting and optimizing antifouling coatings for specific biomedical applications, from implantable devices to nanomedicine.
Fundamental Antifouling Mechanisms: How PEG and Zwitterionic Coatings Resist Biofouling
For decades, polyethylene glycol (PEG) has served as the established "gold standard" for creating protein-resistant surfaces in biomedical applications. The widespread use of PEGylation—the covalent attachment of PEG to surfaces or molecules—stems from its exceptional ability to reduce nonspecific protein adsorption, thereby improving the biocompatibility and performance of medical devices, drug delivery systems, and biosensors. [1] [2] The antifouling efficacy of PEG primarily arises from two interconnected mechanisms: the formation of a steric repulsion barrier and the creation of a stable hydration layer. While PEG continues to be widely utilized, its limitations have spurred research into zwitterionic polymers as promising alternatives. This guide provides a detailed comparison of these materials, offering experimental methodologies and data to support objective performance evaluation for researchers and drug development professionals.
Fundamental Antifouling Mechanisms
PEG's Dual Defense System
PEG chains exert their protein-resistant effects through a combination of physical and chemical mechanisms:
Steric Repulsion: When tethered to a surface, flexible PEG chains extend into the aqueous environment, forming a dynamic, brush-like barrier. This creates a physical exclusion zone that prevents approaching proteins and biomolecules from reaching the underlying substrate. [3] [4] The random coil conformation of PEG chains generates an energetic penalty for compression when proteins approach, effectively pushing them away from the surface.
Hydration Layer Formation: The ether oxygen atoms in PEG's repeating ethylene oxide units (-CH₂CH₂O-) form hydrogen bonds with water molecules, creating a highly hydrated surface layer. [3] [4] This bound water layer presents an energetic barrier to protein adsorption, as proteins must displace the tightly bound water molecules to adhere to the surface—a thermodynamically unfavorable process. [4]
Zwitterionic Polymer Mechanisms
Zwitterionic polymers, containing both positive and negative charges within their repeating units, achieve superior hydration through ionic solvation. [1] [5] Unlike PEG, which binds approximately one water molecule per monomer via hydrogen bonding, zwitterionic polymers can bind 7-8 water molecules per repeating unit through stronger electrostatic interactions, forming a denser and more stable hydration layer. [5]
Comparative Performance Data
Table 1: Quantitative Comparison of PEG and Zwitterionic Polymer Antifouling Performance
| Performance Metric | PEG Systems | Zwitterionic Systems | Experimental Conditions | Reference |
|---|---|---|---|---|
| Protein Adsorption (BSA) | Moderate reduction | >90% reduction; superior to PEG at >1.5nm thickness | SPR measurement, coating thickness ~1-3.3nm | [6] |
| Interfacial Gelation Strength | 5x weaker than non-PEGylated protein | Not directly comparable (acts on proteins) | rhIL-1ra in siliconized syringes, mechanical perturbation | [7] |
| Fibrinogen Adsorption | Moderate reduction | Ultralow fouling at ≈3.6nm thickness | SPR measurement | [6] |
| Biomarker Capture Efficiency | Baseline | >2-fold increase vs. PEG | Wearable microprojection arrays in dengue-infected mice | [2] |
| Immunogenicity | Induces anti-PEG antibodies; accelerated blood clearance | Minimal reactivity against PEG antibodies | In vivo study, ELISA for anti-PEG antibody binding | [8] |
| Hydration Binding Energy | Higher | Lower than PEG | Molecular simulation and thermodynamic measurement | [5] |
Table 2: Mechanism and Property Comparison
| Characteristic | PEG | Zwitterionic Polymers |
|---|---|---|
| Primary Mechanism | Steric repulsion + hydrogen bonding | Ionic solvation + electrostatic hydration |
| Water Molecules Bound per Unit | ~1-3 [4] | 7-8 [5] |
| Hydration Type | Structural water network [4] | Dense hydration layer |
| Chemical Stability | Prone to oxidative degradation [5] | High chemical stability |
| Long-Term Stability | Limited in biological environments [9] | Excellent long-term stability |
| Immunogenicity | Evokes anti-PEG antibodies [8] | Minimal immune recognition [8] |
Experimental Protocols for Antifouling Assessment
Protein Aggregation Study in Siliconized Syringes
Objective: Quantify protein stabilization against interfacial aggregation by PEGylation. [7]
Materials: Recombinant human interleukin-1 receptor antagonist (rhIL-1ra), PEGylated rhIL-1ra (20 kDa PEG), siliconized glass syringes (BD Hypak SCF 27G1/2), silicone oil (Dow Corning 360), phosphate buffer, BCA assay kit, SE-HPLC equipment.
Method:
- Dialyze protein stocks against 10 mM phosphate buffer pH 6.5 overnight at 4°C.
- Filter solutions using 0.1 μm inorganic syringe filters.
- Dilute proteins to equimolar concentrations (30 μM) using phosphate buffer.
- Load 1.35 mL protein solution into syringes containing two glass beads (avoiding air bubbles).
- Divide syringes into two sets: rotate one set end-over-end at 3.5 rpm; keep the other set horizontal for quiescent incubation.
- Sample at predetermined time points using thin-tip pipettes to avoid disturbing adsorbed protein.
- Centrifuge samples at 20,000×g for 1 hour to separate silicone oil and insoluble aggregates.
- Analyze supernatant for soluble monomer content using SE-HPLC and BCA assay.
Key Measurements: Soluble monomer loss over time, intermolecular β-sheet content via FTIR, interfacial gel strength measurement.
Surface Plasmon Resonance (SPR) Coating Performance
Objective: Compare protein adsorption resistance of PEG and zwitterionic coatings with controlled thickness. [6]
Materials: SPR instrument sensor chips, polydopamine (PDA), carboxylic PEG, zwitterionic polymer (PMEN), bovine serum albumin (BSA), bovine plasma fibrinogen (Fg), phosphate buffered saline.
Method:
- Pre-coat SPR sensor chips with PDA intermediate layer via immersion in dopamine solution.
- Immobilize PEG and zwitterionic polymers via amidation coupling on PDA-coated chips.
- Control coating thickness by varying polymer concentration and reaction time (target: 1.0-3.6 nm).
- Prime SPR system with PBS buffer to establish baseline.
- Introduce protein solutions (1 mg/mL BSA or Fg in PBS) at constant flow rate.
- Monitor resonance unit changes in real-time during protein injection.
- Continue buffer flow to monitor dissociation phase.
- Regenerate surface with mild detergent or low pH buffer between experiments.
Key Measurements: Coating thickness by ellipsometry, protein adsorption amount from RU shift, initial adsorption rate, structural stability assessment.
In Vivo Performance in Wearable Devices
Objective: Evaluate antifouling and biomarker capture efficiency in complex biological environments. [2]
Materials: Polycarbonate microprojection arrays, amine-modified substrates, pSBMA polymer, linear PEG polymer (comparable molecular weight), dengue-infected mouse model, immunoglobulin G (IgG), bovine serum albumin, dengue NS1 protein.
Method:
- Functionalize polycarbonate arrays with amine groups using plasma treatment.
- Employ "grafting to" approach to attach pSBMA and PEG polymers.
- Characterize coating density and hydrodynamic radius.
- Incubate devices in single protein solutions, diluted plasma, and apply to mouse flank skin.
- Immobilize IgG or BSA on coated surfaces.
- Incubate in buffer or plasma solutions containing dengue NS1 protein.
- Apply wearable arrays to skin of dengue-infected mice.
- Quantify captured NS1 using fluorescent or enzymatic detection.
Key Measurements: Nonspecific adsorption in complex media, specific biomarker capture density, signal-to-noise ratio in disease detection.
The Scientist's Toolkit: Essential Research Reagents
Table 3: Key Reagents for Antifouling Coating Research
| Reagent/Chemical | Function/Application | Research Context |
|---|---|---|
| PLL-g-PEG | Comb-like copolymer for surface grafting; PLL backbone adsorbs to negative surfaces, PEG provides brush layer | Model protein-resistant surface for fundamental SFA studies [4] |
| Siliconized Syringes | Real-world interface for protein aggregation studies | Testing therapeutic protein stability under pharmaceutically relevant conditions [7] |
| Polydopamine (PDA) | Universal adhesive for substrate-independent coating | Enables reproducible PEG and zwitterionic polymer immobilization on various substrates [6] |
| SBMA Monomer | Primary component for polysulfobetaine zwitterionic polymers | Forming ultra-low fouling surfaces via surface-initiated polymerization [5] |
| Laponite XLG Nanosheets | Physical crosslinker for nanocomposite hydrogels | Enhancing mechanical properties of zwitterionic hydrogels [5] |
| Carboxybetaine Methacrylate | Monomer for PCB zwitterionic polymers | Creating blood-compatible coatings with high hydration capacity [9] |
While PEG remains the historical "gold standard" for antifouling applications, extensive experimental evidence demonstrates that zwitterionic polymers frequently surpass PEG in key performance metrics, including protein resistance, immunological inertness, and long-term stability. The superior performance of zwitterionic materials stems from their enhanced hydration capacity through ionic solvation rather than hydrogen bonding alone.
For researchers selecting coating strategies, the optimal choice depends on application-specific requirements:
- PEG may suffice for short-term applications where established protocols and regulatory pathways are advantageous.
- Zwitterionic polymers are preferable for long-term implants, blood-contacting devices, and applications where minimal immunogenicity is critical.
Future directions include developing hybrid systems that leverage the advantages of both materials and addressing the mechanical challenges of highly hydrated zwitterionic hydrogels through advanced nanocomposite strategies.
In the pursuit of advanced antifouling materials, the competition between traditional polyethylene glycol (PEG) coatings and emerging zwitterionic polymers represents a critical frontier in surface science. This review provides a comprehensive comparison of their performance, focusing on the unique biomimetic hydration mechanism that enables zwitterionic polymers to outperform PEG in many biomedical applications. While PEG relies on hydrogen bonding to form a hydration layer, zwitterionic polymers create a denser, more robust electrostatic hydration layer through their oppositely charged groups. We synthesize experimental data demonstrating superior protein resistance, enhanced stability, and improved diagnostic sensitivity offered by zwitterionic coatings, providing researchers with evidence-based guidance for material selection in drug delivery, biosensing, and medical device development.
Non-specific protein adsorption to biomedical surfaces triggers undesirable consequences including foreign body reactions, thrombosis, and biofilm formation that compromise device functionality and patient safety [10]. This fouling phenomenon represents a fundamental challenge for implantable medical devices, drug delivery vehicles, and diagnostic biosensors operating in biological environments. For decades, polyethylene glycol (PEG) has dominated antifouling strategies due to its hydrophilicity and biocompatibility, forming a protective hydration layer primarily through hydrogen bonding with water molecules [10]. However, PEG exhibits susceptibility to oxidative degradation and immunogenicity, limiting its long-term effectiveness in vivo [10] [11].
Zwitterionic polymers have emerged as superior alternatives by mimicking biological membrane structures through their balanced positively and negatively charged groups [10] [11]. These materials create an exceptionally dense electrostatic hydration layer via ionic solvation, demonstrating enhanced stability and antifouling performance compared to PEG-based systems [10]. This review systematically compares these competing technologies through experimental data and mechanistic insights, providing evidence-based guidance for researchers and product developers in the biomedical field.
Molecular Mechanisms: Contrasting Hydration Layer Formation
The fundamental distinction between PEG and zwitterionic polymers lies in their molecular interactions with water, which directly determines their antifouling efficacy and stability.
PEG Hydration Through Hydrogen Bonding
PEG forms hydration layers through hydrogen bonding interactions with water molecules. This mechanism creates a protective barrier that moderately resists protein adsorption. However, the hydrogen bonds in PEG-water systems are relatively weak and dynamically breaking and reforming, creating transient defects that can permit protein penetration. Additionally, PEG is susceptible to oxidative degradation in biological environments, leading to gradual loss of antifouling capability over time [10].
Zwitterionic Superhydration Through Electrostatic Interactions
Zwitterionic polymers contain equimolar cationic and anionic groups within their repeating units, creating a strongly electrostatically-driven hydration layer [12] [10]. The positively and negatively charged moieties interact strongly with water molecules through ionic solvation, forming a denser and more tightly bound hydration layer than PEG. This superhydrophilicity results from vigorous binding of water molecules via Coulombic forces, creating a physical and energy barrier that effectively repels proteins and other fouling agents [10]. The table below contrasts the fundamental hydration mechanisms:
Table: Comparison of Hydration Mechanisms Between PEG and Zwitterionic Polymers
| Feature | PEG | Zwitterionic Polymers |
|---|---|---|
| Primary Hydration Mechanism | Hydrogen bonding | Ionic solvation/Electrostatic interactions |
| Water-Binding Energy | Moderate (~20-40 kJ/mol) | Strong (~50-100 kJ/mol) |
| Hydration Layer Density | Moderate | High |
| Molecular Stability | Subject to oxidation | High chemical stability |
| Immunogenicity | Can induce antibodies | Low immunogenicity |
Experimental Performance Comparison
Direct comparative studies provide convincing evidence for the superior performance of zwitterionic coatings in complex biological environments.
Protein Resistance and Diagnostic Sensitivity
A critical study comparing poly(sulfobetaine-methacrylate) (pSBMA) with linear PEG of equivalent molecular weight and hydrodynamic radius demonstrated significant advantages for zwitterionic materials [13]. When functionalized with capture antibodies and deployed in wearable microprojection arrays for biomarker detection, the pSBMA-coated devices captured over twice the amount of target protein (dengue NS1) compared to PEG-coated devices in both plasma solutions and in vivo mouse models [13]. This enhanced performance is attributed to the higher density of immobilized proteins possible on zwitterionic surfaces while maintaining low non-specific adsorption.
Table: Experimental Performance Comparison of PEG vs. Zwitterionic Coatings
| Parameter | PEG Coatings | Zwitterionic Coatings | Experimental Context |
|---|---|---|---|
| Non-specific Adsorption | Low | Comparably low | Single protein solutions, diluted plasma [13] |
| Immobilized IgG Density | Baseline | Significantly higher | Surface functionalization [13] |
| Target Biomarker Capture | Baseline | >2-fold increase | Dengue NS1 detection in vivo [13] |
| Signal-to-Noise Ratio | Baseline | Significantly higher | Wearable biosensors [13] |
| Friction Coefficient | Not reported | 10⁻⁴ to 10⁻³ | Hydration lubrication [14] |
| Immunogenicity | Can produce antibodies | Low | Long-term circulation [10] [11] |
Tribological Performance and Stability
The exceptional lubricating properties of zwitterionic polymers stem from their robust hydration layers, with friction coefficients as low as 10⁻⁴ under appropriate confinement conditions [14]. This superlubrication state is critical for medical devices requiring insertion or movement through biological tissues. The structure and thickness of the hydration layer directly modulate energy dissipation pathways during friction, with zwitterionic polymers maintaining their lubricating properties even under substantial normal pressures up to 6-7 MPa [14].
Experimental Techniques for Investigating Hydration Layers
Advanced experimental methodologies provide insights into the structure-property relationships of zwitterionic hydration layers.
Key Investigation Techniques
- Surface Force Apparatus (SFA): Measures normal and shear forces between surfaces with sub-nanometer distance resolution, directly quantifying hydration layer thickness and lubricating properties [14].
- High-Resolution Atomic Force Microscopy (HR-AFM): Provides molecular-scale imaging of hydration layer structure and organization under varying ionic conditions [14].
- Molecular Dynamics (MD) Simulations: Offers atomistic insight into polymer-water interactions, chain mobility, and protein adhesion mechanisms at the molecular level [15].
Experimental Workflow for Hydration Layer Characterization
The following diagram illustrates a typical integrated approach to characterize zwitterionic polymer hydration layers:
The Scientist's Toolkit: Essential Research Reagents and Materials
Table: Essential Research Materials for Zwitterionic Polymer Investigations
| Material/Technique | Function/Application | Key Characteristics |
|---|---|---|
| Poly(sulfobetaine methacrylate) (pSBMA) | Antifouling coatings, drug delivery | Upper critical solution temperature (UCST) behavior, superior protein resistance [13] [15] |
| Polycarboxybetaine (PCB) | Biomedical devices, blood-contact applications | Excellent hemocompatibility, reduced thrombosis risk [10] |
| Phosphorylcholine-based Polymers (PMPC) | Biomimetic coatings, implantable devices | Mimics cell membrane structure, high biocompatibility [10] [15] |
| Surface Force Apparatus (SFA) | Hydration layer thickness and lubrication measurement | Sub-nanometer resolution, simultaneous normal and shear force measurement [14] |
| Frequency-Modulation AFM | Hydrated ion structure imaging | Atomic-scale resolution in liquid environments [14] |
| Molecular Dynamics Software | Atomistic simulation of polymer-water interactions | Predicts hydration layer structure and dynamics [15] |
Structural Varieties and Functional Adaptations
Zwitterionic polymers encompass several structural classes with distinct properties suited for different biomedical applications:
Major Zwitterionic Polymer Classes
- Polysulfobetaine (PSB): Exhibits Upper Critical Solution Temperature (UCST) behavior, transitioning from collapsed to extended chain conformation with increasing temperature, valuable for triggered drug delivery [10] [15].
- Polycarboxybetaine (PCB): Demonstrates exceptional hemocompatibility and resistance to thrombus formation, ideal for blood-contacting devices [10].
- Phosphorylcholine-based Polymers (PMPC): Closely mimics the outer surface of cell membranes, achieving superior biocompatibility for implantable devices [10].
Temperature Responsiveness and Applications
The UCST behavior of certain zwitterionic polymers enables innovative applications in drug delivery and smart coatings. Below the UCST, electrostatic interactions dominate, causing chain collapse, while above the UCST, thermal energy overcomes electrostatic associations, leading to chain expansion and increased solubility [15]. This property can be exploited for temperature-controlled drug release or tunable cell adhesion surfaces.
Zwitterionic polymers establish a new paradigm in antifouling technology through their biomimetic electrostatic hydration mechanism, outperforming traditional PEG coatings in key biomedical applications. The evidence from direct comparative studies reveals significant advantages in diagnostic sensitivity, protein immobilization capacity, and lubrication performance. While PEG remains a valuable antifouling material, zwitterionic polymers offer superior properties for demanding applications including implantable medical devices, targeted drug delivery systems, and high-sensitivity biosensors. Future research directions should focus on optimizing zwitterionic polymer architectures for specific biological environments and scaling up manufacturing processes to facilitate clinical translation.
In the ongoing research to develop superior antifouling coatings for biomedical applications, the comparison between traditional poly(ethylene glycol) (PEG) and emerging zwitterionic polymers remains a central focus. While PEG has long been the "golden standard" for preventing biofouling, its limitations, including susceptibility to oxidative degradation and the potential to elicit anti-PEG antibodies, have driven the investigation of high-performance alternatives. [16] Zwitterionic polymers, characterized by their unique molecular structures containing both positive and negative charges within the same monomer unit, have demonstrated exceptional hydrophilicity, biocompatibility, and robust antifouling performance. [17] [16] Their mechanism of action relies on the formation of a strong, stable hydration layer via electrostatic interactions, which creates a physical and thermodynamic barrier against the adsorption of proteins, platelets, and bacteria. [18] [17] This guide provides a structured comparison of the three primary classes of zwitterionic polymers—Phosphorylcholine (PC), Sulfobetaine (SB), and Carboxybetaine (CB)—to inform researchers and drug development professionals in their material selection process.
Comparative Analysis of Zwitterionic Polymer Classes
The following sections detail the key characteristics, performance data, and applications of PC, SB, and CB polymers. The structural differences in their charged groups significantly influence their hydration, stability, and functionality.
Structural Characteristics and Properties
- Phosphorylcholine (PC) Polymers: PC polymers mimic the phospholipid head groups found in cell membranes. The fundamental structure is a positively charged quaternary ammonium connected to a negatively charged phosphate group. [17] This biomimicry grants them excellent hemocompatibility, making them particularly suitable for blood-contacting devices like vascular grafts and stents. [17] A common monomer is MPC (2-methacryloyloxyethyl phosphorylcholine). [17]
- Sulfobetaine (SB) Polymers: SB polymers feature a positively charged quaternary ammonium group connected to a negatively charged sulfonate group. [17] They are known for being very hydrophilic, highly resistant to protein and bacteria adhesion, and able to tolerate high salt levels, maintaining their zwitterionic properties across a wide pH range. [18] [17] Common monomers include sulfobetaine methacrylate (SBMA). [18] [17]
- Carboxybetaine (CB) Polymers: CB polymers consist of a positively charged quaternary ammonium group with a negatively charged carboxylate. [17] [16] A key advantage of CB is its non-fouling nature combined with a reactive carboxylate group. This allows for the further attachment of bioactive molecules, such as peptides or drugs, without compromising its antifouling properties. [17] Monomers include carboxybetaine methacrylate (CBMA) and carboxybetaine acrylamide (CBAA). [17] Some CB variants, like CBMA-OH, can be pH-responsive, reversibly switching between an open carboxylate form and a closed lactone ring. [16]
Table 1: Fundamental Characteristics of Zwitterionic Polymer Classes
| Polymer Class | Positive Group | Negative Group | Key Traits | Common Monomers |
|---|---|---|---|---|
| Phosphorylcholine (PC) | Quaternary Ammonium | Phosphate | Excellent hemocompatibility, biomimetic | MPC (2-methacryloyloxyethyl phosphorylcholine) [17] |
| Sulfobetaine (SB) | Quaternary Ammonium | Sulfonate | High hydrophilicity, salt tolerance, wide pH stability | SBMA (sulfobetaine methacrylate) [18] [17] |
| Carboxybetaine (CB) | Quaternary Ammonium | Carboxylate | Reactive carboxylate for biofunctionalization, pH-responsive variants | CBMA (carboxybetaine methacrylate), CBAA (carboxybetaine acrylamide) [17] [16] |
Antifouling Performance and Experimental Data
Quantitative studies highlight the superior performance of zwitterionic coatings compared to PEG, with variations among PC, SB, and CB types.
- Performance vs. PEG: A comparative study found that a very thin (~1 nm) PMEN (a PC-based polymer) coating showed much stronger resistance to bovine serum albumin (BSA) adsorption than a PEG coating of similar thickness. While PEG's performance could exceed that of PMEN at thicker coatings (1.5-3.3 nm) due to a stronger steric repelling effect, both optimized thick coatings (~3.6 nm) achieved ultralow fouling by BSA and bovine plasma fibrinogen. [6]
- Molecular Dynamics Insights: Computational simulations provide a mechanistic understanding of zwitterionic superiority. Studies indicate that the antifouling performance ranking is often PCBMA > PMPC > PSBMA. [19] This is attributed to differences in their hydration structure and water residence times. For instance, random zwitterionic amphiphilic copolymer (r-ZAC) membranes containing SBMA sustain tightly bound, long-lived hydration layers that impose substantial free-energy barriers to foulant approach (e.g., ≈ 90 kcal/mol for alginate), whereas conventional polyamide membranes exhibit negligible barriers. [19]
- Dual-Functionality with Bactericidal Agents: Zwitterionic polymers are often combined with antimicrobial compounds to create coatings that both resist fouling and kill bacteria. For example, a study grafted a copolymer containing sulfobetaine (SBMA) and quaternary ammonium compound (MAPTAC) onto polyurethane catheters. The zwitterionic groups created a hydrated layer that reduced protein, platelet, and whole blood adsorption, while the quaternary ammonium cations provided strong antibacterial properties. [18]
Table 2: Experimental Antifouling Performance Data
| Coating Material | Test Method | Key Performance Metric | Result | Reference |
|---|---|---|---|---|
| PMEN (PC-based) | Surface Plasma Resonance (SPR) | BSA Adsorption (at ~1 nm thickness) | Much stronger resistance than PEG | [6] |
| PEG vs. PMEN | Surface Plasma Resonance (SPR) | BSA & Fibrinogen Adsorption (at ~3.6 nm thickness) | Both showed ultralow fouling | [6] |
| PCSA1 (SB & QAC) | Protein adsorption, platelet adhesion | Reduction in protein adsorption & platelet adhesion | Significant improvement vs. unmodified PU | [18] |
| r-ZAC (SBMA-based) Membrane | Steered Molecular Dynamics (SMD) | Free-energy barrier to alginate | ≈ 90 kcal/mol | [19] |
Experimental Protocols for Coating Fabrication and Evaluation
Common Coating Application Methodologies
Several robust strategies exist for grafting zwitterionic polymers onto medical devices, with the choice of method impacting coating stability and performance.
- Polydopamine-Assisted Coating: This versatile, substrate-independent method involves first coating the substrate with a polydopamine (PDA) intermediate layer, which acts as a universal adhesive. The zwitterionic polymer is then immobilized onto this PDA layer via amidation coupling. This method allows for quantitative fabrication and performance optimization using techniques like Surface Plasma Resonance (SPR). [6]
- Surface-Initiated Atom Transfer Radical Polymerization (SI-ATRP): This controlled radical polymerization technique grows polymer chains directly from an activated surface, creating a dense "polymer brush" structure. This method is valuable for creating high-performance, antifouling surfaces on various materials. [17]
- Covalent Grafting via Plasma Treatment: This two-step method is highly effective for polymer-based devices. The substrate (e.g., a polyurethane catheter) undergoes plasma treatment to generate surface functionalities. A polymer (like polyethyleneimine, PEI) is then grafted, providing active sites for the subsequent covalent attachment of the synthesized zwitterionic copolymer via amide coupling reactions. [18]
- Photopolymerization and Layer-by-Layer (LbL) Assembly: Photopolymerization is a promising technique for fabricating thin, stable zwitterionic films on various substrates. [17] LbL assembly involves the sequential deposition of oppositely charged polyelectrolytes to build up multilayer coatings, allowing for precise control over thickness and composition. [17]
Key Performance Evaluation Experiments
Rigorous in-vitro testing is crucial for evaluating the antifouling and biological performance of modified surfaces.
- Protein Adsorption Assay: This is a fundamental test for antifouling properties. The amount of protein (e.g., Bovine Serum Albumin - BSA, bovine plasma fibrinogen - Fg) adsorbed onto the coated surface is quantified using techniques like Surface Plasma Resonance (SPR) or fluorescence microscopy. Low protein adsorption is a key indicator of a successful antifouling coating. [6]
- Platelet Adhesion and Whole Blood Tests: For blood-contacting devices, coatings are incubated with platelet-rich plasma or whole blood. The surfaces are then examined, typically via scanning electron microscopy (SEM), to visualize and count the number of adhered platelets. A significant reduction in adhesion indicates improved hemocompatibility and reduced thrombosis risk. [18]
- Bacterial Adhesion and Bactericidal Assays: The coating's ability to resist bacterial colonization is tested by exposing it to bacterial cultures (e.g., E. coli, S. aureus). Bacterial adhesion is visualized and quantified. If the coating incorporates bactericidal agents like quaternary ammonium compounds, additional tests for zones of inhibition or direct contact-killing efficacy are performed to confirm antibacterial activity. [18]
- Hydrophilicity and Hydration Measurement: Water Contact Angle (WCA) measurements are a simple yet effective method to determine surface wettability. A lower WCA indicates higher hydrophilicity, which is correlated with the formation of a strong hydration layer. [18] Advanced techniques like molecular dynamics simulations can provide deeper insights into hydration layer structure and water dynamics. [19]
The Scientist's Toolkit: Essential Research Reagents and Materials
Table 3: Key Reagents and Materials for Zwitterionic Coating Research
| Item | Function/Description | Example Uses |
|---|---|---|
| SBMA Monomer | Sulfobetaine methacrylate; common monomer for synthesizing SB polymers. [18] | Free radical polymerization to create SB-based copolymers. [18] |
| MPC Monomer | 2-methacryloyloxyethyl phosphorylcholine; common monomer for PC polymers. [17] | Fabrication of biomimetic, hemocompatible coatings. [17] |
| CBMA Monomer | Carboxybetaine methacrylate; common monomer for CB polymers. [16] | Synthesis of functionalizable, pH-responsive zwitterionic materials. [16] |
| Polydopamine (PDA) | A universal adhesive polymer that forms coatings on virtually any substrate. [6] | Used as an intermediate layer for immobilizing zwitterionic polymers. [6] |
| ATRP Initiator | A chemical species that initiates Surface-Initiated Atom Transfer Radical Polymerization. | Growing dense, well-defined zwitterionic polymer brushes from surfaces. [17] |
The comparative data clearly positions zwitterionic polymers as powerful alternatives to PEG, particularly in demanding biological environments. Among the zwitterionic classes, Carboxybetaine (CB) polymers often demonstrate top-tier antifouling performance combined with unique functionality for biomolecule conjugation. Sulfobetaine (SB) polymers offer robust, salt-tolerant antifouling across a wide pH range, while Phosphorylcholine (PC) polymers excel in hemocompatibility for blood-contact applications. The selection of a specific zwitterionic chemistry depends on the target application's requirements, whether the priority is ultimate fouling resistance, surface functionalization, biomimicry, or integration of bactericidal properties. Future research will likely focus on hybrid coatings, stimuli-responsive systems, and scaling up manufacturing processes for widespread clinical translation.
In the development of advanced biomedical coatings, the interaction between material surfaces and water molecules dictates antifouling performance. Two primary mechanisms—hydrogen bonding and ionic solvation—enable polymers to form protective hydration layers that resist protein adsorption and cell attachment. This guide provides a comparative analysis of these mechanisms, drawing on experimental and simulation data to outline their distinct efficiencies, thermodynamic properties, and applicability in biomedical implants. Within the context of antifouling coatings, this comparison is critical for selecting between conventional polyethylene glycol (PEG)-based materials, which rely heavily on hydrogen bonding, and emerging zwitterionic polymers, which exploit intense ionic solvation.
Theoretical Background and Fundamental Interactions
Hydrogen Bonding in Aqueous Environments
Hydrogen bonding (H-bonding) is a fundamental interaction where a hydrogen atom covalently bound to an electronegative atom (e.g., O, N) experiences an attractive force with another electronegative atom [20]. In water, this leads to a dynamic, tetrahedral network that confers unique properties such as high cohesion and density anomalies. H-bonding can also occur between water and solutes; for instance, ions can act as structure-makers or structure-breakers of the native water network. A study on NaCl and CsI solutions demonstrated that ions can suppress or enhance water diffusion by altering the hydrogen-bonded structure, a phenomenon described as "structure making and breaking" [21]. The strength and geometry of H-bonds can be probed using quantum chemical (QC) methods, such as density functional theory (DFT) or Møller–Plesset perturbation theory (MP2), which provide insights into interaction energies and electronic structures [20].
Ionic Solvation and the Hydration Layer
Ionic solvation involves the interaction of charged species with the dipole moments of water molecules. This process is dominated by long-range Coulomb forces and results in the formation of a structured hydration shell around ions [22]. The efficiency of this process is influenced by the ion's charge density. A unique category within ionic liquids is the doubly ionic H-bond, where the hydrogen bond forms between two ions, a cation and an anion [20]. This interaction is stronger and possesses distinct characteristics compared to traditional H-bonds. Zwitterionic materials, which contain covalently linked cationic and anionic groups, exemplify this mechanism in antifouling coatings. Their strongly hydrated surfaces, achieved through ionic solvation, create a physical and energetic barrier to biomolecular adsorption [23].
Table 1: Fundamental Characteristics of Hydration Mechanisms
| Feature | Hydrogen Bonding | Ionic Solvation |
|---|---|---|
| Primary Interaction | Dipole-Dipole, Electrostatic [20] | Ion-Dipole, Coulombic [22] |
| Strength Range | Weaker (typically 1–30 kJ/mol) [20] | Stronger (can exceed 50 kJ/mol) [20] |
| Spatial Range | Short-Range (directionally specific) [20] | Long-Range (non-directional) [22] |
| Key Example | Water network around PEG [23] | Hydration shell of zwitterionic polymers [23] |
Comparative Experimental Data and Performance Metrics
The following tables synthesize experimental and simulation data to quantify the performance of both hydration mechanisms, particularly in the context of their antifouling efficacy and material properties.
Table 2: Experimentally Observed Hydration and Antifouling Performance
| Parameter | Hydrogen Bonding (PEG-like) | Ionic Solvation (Zwitterionic) | Experimental/Simulation Method |
|---|---|---|---|
| Protein Adsorption | Moderate reduction [23] | "Superlow" or undetectable (<5 ng/cm²) [23] | Surface Plasmon Resonance (SPR) [23] |
| Water Diffusion Anomaly | Structure-breaking (CsI) or making (NaCl) effects observed [21] | Not directly applicable (bulk property) | Machine Learning MD Simulations [21] |
| Hydration Energy | Lower (primarily H-bond energy) | Higher (strong ionic solvation) [23] | Quantum Chemical (QC) Calculations [20] |
| Mechanical Strength | Variable, can be good | Generally weak in pure form; requires reinforcement [23] | Tensile/Compression Testing [23] |
Table 3: Thermodynamic and Structural Properties from Simulation Studies
| Property | Hydrogen Bonding Hydration | Ionic Solvation Hydration |
|---|---|---|
| Primary Theory/Method | Thermodynamic Perturbation Theory (TPT), Integral Equation Theory (IET) [24] | QM/MM Molecular Dynamics [22] |
| Hydration Shell Definition | Less distinct, network perturbation [24] | Highly distinct, localized shell [22] |
| Impact on Water Structure | Can be disruptive (structure-breaking) or supportive (structure-making) [21] | Forms well-defined, stable primary hydration shell [22] |
| Entropic Contribution | Significant, can be unfavorable | Can be favorable due to strong enthalpic drive |
Molecular Mechanisms and Signaling Pathways in Antifouling
The superior antifouling performance of zwitterionic coatings stems from the molecular-level efficiency of ionic solvation. The balanced charged groups on the polymer chain create a strong electric field that immobilizes water molecules via ionic solvation, forming a dense and stable hydration layer [23]. This layer acts as a physical and energetic barrier. When a protein approaches this surface, it must displace these tightly bound water molecules, which is energetically unfavorable due to the high energy cost of dehydrating the ionic groups. In contrast, surfaces that hydrate primarily via hydrogen bonding (like PEG) form a less rigid hydration layer. The water molecules, while still bound, have higher mobility and can be more easily displaced by approaching proteins, leading to a higher probability of initial adsorption and subsequent biofouling [23].
Diagram 1: Molecular Antifouling Pathways: A comparison of the fouling resistance mechanisms driven by hydrogen bonding versus ionic solvation.
Detailed Experimental Protocols
Protocol 1: Machine Learning MD for Hydration Structure Analysis
This protocol is adapted from studies investigating ion-induced water dynamics [21].
- Objective: To quantify the "structure-making" or "breaking" effects of ions on water's hydrogen bond network and its diffusion.
- Methodology:
- Potential Development: Train a deep machine learning potential at the MP2 level of theory to achieve accurate quantum chemical force calculations at a feasible computational cost for molecular dynamics (MD) simulations [21].
- System Preparation: Construct simulation boxes containing water molecules (e.g., SPC/E or TIP4P models) and ions (e.g., Na⁺/Cl⁻ or Cs⁺/I⁻) at specified concentrations to mimic experimental conditions [21].
- Simulation Execution: Perform MD simulations under controlled temperature and pressure (NPT ensemble) for sufficient time (e.g., nanoseconds) to ensure equilibrium and proper sampling.
- Trajectory Analysis:
- Calculate the mean-squared displacement (MSD) of water molecules to determine diffusion coefficients.
- Analyze the radial distribution function (RDF), g(r), between ions and water oxygen/hydrogen atoms to define the hydration shell structure.
- Use geometric or energetic criteria to characterize the hydrogen bond network of water, noting changes in the number of H-bonds or their lifetime due to the presence of ions [21].
Protocol 2: QM/MM MD for Anionic Hydration Shell Study
This protocol is used for high-accuracy study of ion-specific hydration, particularly for anions [22].
- Objective: To determine the solvation structure, ligand exchange dynamics, and hydrogen-bonding characteristics of hydrated halide ions (F⁻, Cl⁻, Br⁻).
- Methodology:
- System Setup: Place a single ion in the center of a simulation box filled with a flexible water model (e.g., SPC-mTR2). Define the QM region as the ion and its first-shell water molecules. The rest of the water molecules are treated with MM potentials [22].
- Parameterization: Derive and validate Lennard-Jones parameters for the interaction between the QM ion and MM water molecules to ensure seamless QM/MM coupling [22].
- Simulation Execution: Run RIMP2/avTZ-based QM/MM MD simulations. After equilibration (e.g., 10 ps), collect data over a production run (e.g., 25 ps). Use a canonical (NVT) ensemble.
- Data Analysis:
- Plot ion-oxygen RDFs to identify the coordination number and shell boundaries.
- Analyze the angular distribution of the O-H vector of water molecules around the anion to characterize the geometry and strength of ion-water H-bonds.
- Calculate the velocity autocorrelation function to access vibrational spectra and dynamics [22].
Diagram 2: Computational Workflows: A side-by-side comparison of the two primary simulation methodologies used for investigating hydration mechanisms.
The Scientist's Toolkit: Key Reagents and Materials
Table 4: Essential Research Reagents and Computational Tools
| Item/Solution | Function/Description | Example Use Case |
|---|---|---|
| Zwitterionic Monomers | Building blocks for polymers with balanced charge; enable strong ionic solvation [23]. | Synthesizing pSBMA, pCBMA for antifouling coatings [23]. |
| Flexible Water Models (SPC-mTR2) | Molecular mechanical potential with internal degrees of freedom for water [22]. | QM/MM MD simulations of anion hydration where solvent geometry is key [22]. |
| Machine Learning Potentials | Force field trained on QC data for accurate and efficient MD simulations [21]. | Simulating ion-water dynamics at near-MP2 accuracy for nanoseconds [21]. |
| Debye-Hückel Theory | Theoretical model describing electrolyte behavior and electrostatic screening [25]. | Predicting zeta potential changes in charged surfaces with ionic strength [25]. |
| RIMP2/avTZ Method | High-level quantum chemical method for accurate electron correlation description [22]. | Benchmarking and core QM region treatment in QM/MM simulations [22]. |
Fabrication Techniques and Real-World Biomedical Applications
Surface immobilization of antifouling polymers is a critical step in the development of biomedical devices, drug delivery systems, and diagnostic platforms. The two predominant strategies—'grafting-to' and 'grafting-from'—offer distinct advantages and limitations that significantly impact coating performance. Within this context, polydopamine (PDA) has emerged as a versatile adhesion layer that enables substrate-independent modification, particularly valuable for applying uniform coatings on complex medical device geometries. This guide provides an objective comparison of these immobilization strategies, with a specific focus on their application in grafting poly(ethylene glycol) (PEG) and zwitterionic polymers for antifouling applications. The performance data, experimental protocols, and practical toolkit presented herein will assist researchers in selecting optimal surface engineering approaches for their specific biomedical applications.
Strategic Comparison: Grafting-To vs. Grafting-From
The choice between 'grafting-to' and 'grafting-from' methods involves fundamental trade-offs between structural control, grafting density, and experimental complexity, each directly influencing the resultant antifouling performance.
The 'grafting-to' approach involves conjugating pre-synthesized polymer chains to a substrate surface. This method benefits from well-defined polymer architecture and molecular weight, as polymers are characterized before grafting [26]. For instance, poly(sodium styrene sulfonate) (polyNaSS) with precise molecular weights (5, 10, and 35 kDa) has been successfully grafted to titanium surfaces using this technique [26]. However, this method typically achieves lower grafting densities due to steric hindrance as already-attached chains create a kinetic barrier for subsequent chains reaching the surface [27].
The 'grafting-from' approach involves immobilizing initiators on the substrate surface followed by in-situ polymerization. This technique typically yields higher grafting densities because small monomer molecules can more easily diffuse to the initiation sites than large pre-formed polymers [28] [26]. Studies modifying cross-linked polyethylene (CLPE) with zwitterionic polymers via surface-initiated polymerization have demonstrated excellent antifouling properties [28]. The drawback includes less control over polymer chain length and potential need for catalyst removal [28].
A hybrid approach, the Polymer Assembly-Assisted Grafting-To (PAAG) method, has recently been developed to balance these trade-offs. This technique utilizes pre-formed block copolymer assemblies that are then immobilized onto substrates, combining the synthetic control of 'grafting-to' with enhanced grafting density [27].
Table 1: Fundamental Characteristics of Grafting Methods
| Feature | Grafting-To Method | Grafting-From Method | Polymer Assembly-Assisted Grafting-To (PAAG) |
|---|---|---|---|
| Grafting Density | Lower due to steric hindrance [27] | Higher due to better monomer accessibility [28] | Improved density via pre-assembled structures [27] |
| Polymer Control | Excellent control over architecture and MW [26] | Less control over chain length distribution [28] | Controlled block copolymer architecture [27] |
| Experimental Complexity | Simpler; no in-situ polymerization [27] | More complex; requires surface initiation [28] | Moderate; combines synthesis and assembly [27] |
| Substrate Versatility | High, especially with PDA adhesion layers [29] | Moderate to high [28] | Demonstrated on gold surfaces [27] |
| Catalyst Removal | Not required | Often required (e.g., metal catalysts) [28] | Not required |
Polydopamine as a Universal Adhesion Platform
Polydopamine (PDA) serves as a remarkably versatile adhesion layer that effectively bridges inert substrates and functional polymer coatings. Inspired by mussel adhesive proteins, PDA forms conformal coatings on virtually any material surface—including metals, metal oxides, ceramics, and polymers—through a simple dip-coating process in an aqueous alkaline dopamine solution [30] [29]. This substrate-independent functionality makes it particularly valuable for modifying complex medical device geometries where uniform coverage is essential.
The surface chemistry of PDA provides secondary reactivity through catechol/quinone functional groups that enable covalent immobilization of polymers containing nucleophilic groups (e.g., amines, thiols) [30]. For antifouling applications, this universal adhesion capability has been successfully demonstrated for both PEG and zwitterionic polymers. Researchers have quantified that PDA-assisted immobilization creates stable coatings that maintain excellent antifouling properties across various substrates [29] [6].
Antifouling Performance: PEG vs. Zwitterionic Polymers
The antifouling performance of both PEG and zwitterionic polymers is highly dependent on grafting parameters, with significant implications for biomedical applications where protein resistance is critical.
Performance Comparison by Coating Thickness
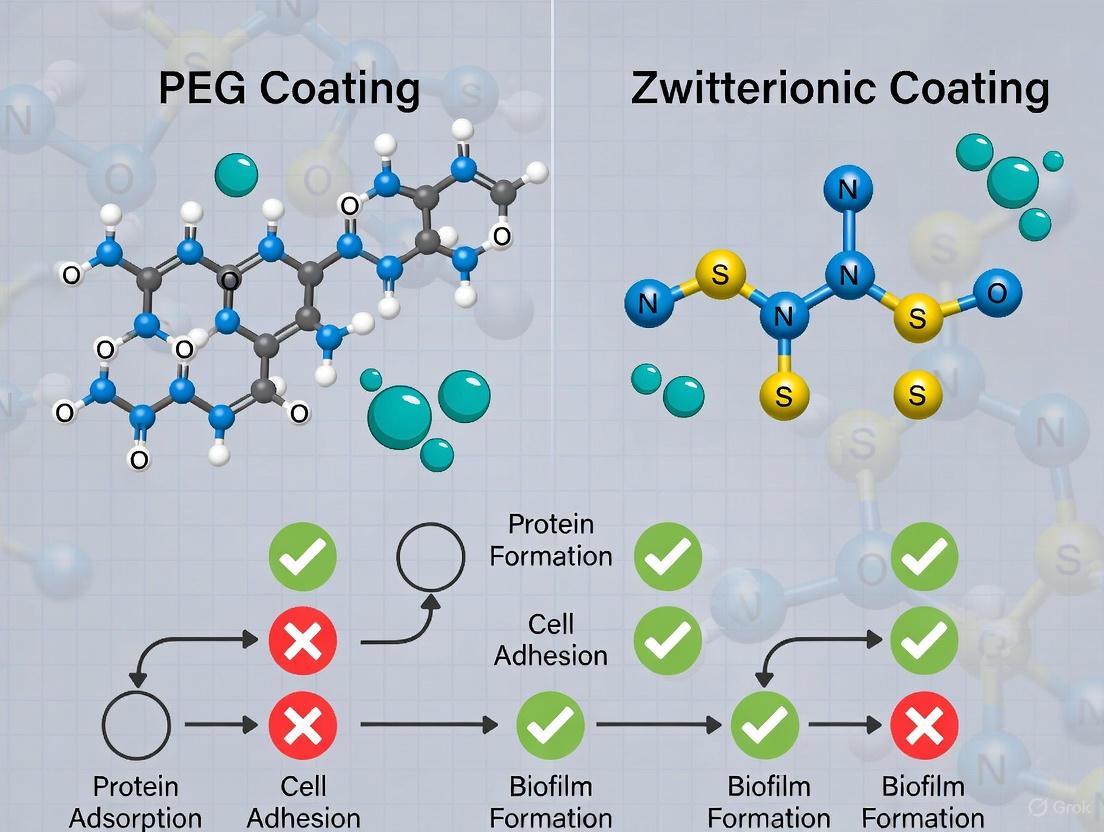
Direct comparative studies reveal that the superior antifouling polymer depends strongly on coating thickness. PMEN (phosphorylcholine zwitterion polymer) coatings approximately 1 nm thick demonstrated much stronger resistance to bovine serum albumin (BSA) adsorption than equivalent PEG coatings [29] [6]. However, as thickness increased to 1.5-3.3 nm, PEG coatings exceeded PMEN's protein resistance due to stronger steric repulsion effects. At approximately 3.6 nm thickness, both polymer types exhibited ultralow fouling against both BSA and bovine plasma fibrinogen (Fg) [29] [6].
Table 2: Antifouling Performance of PEG vs. Zwitterionic Coatings
| Parameter | PEG Coatings | Zwitterionic Coatings | Experimental Conditions |
|---|---|---|---|
| Optimal Thickness Range | 1.5-3.6 nm [29] | ~1 nm for thin coatings; 3.6 nm for thick coatings [29] | Surface plasmon resonance (SPR) measurement |
| BSA Adsorption (thin coatings ~1 nm) | Higher adsorption | Much stronger resistance [29] | 1 mg/mL BSA in PBS |
| BSA Adsorption (thick coatings ~3.6 nm) | Ultralow fouling | Ultralow fouling [29] | 1 mg/mL BSA in PBS |
| Fibrinogen Adsorption | Ultralow at ~3.6 nm | Ultralow at ~3.6 nm [29] | 1 mg/mL Fg in PBS |
| Effect of End Group (PEG) | -OH superior to -COOH (10-fold difference) [29] | Not applicable | SPR measurement |
| Platelet Adhesion | Excellent resistance | Excellent resistance [29] | In vitro platelet-rich plasma |
| Bacterial Adhesion | Excellent resistance | Excellent resistance [29] | S. aureus and other strains |
Impact of Grafting Density and Chain Length
For both PEG and zwitterionic polymers, grafting density often proves more critical than chain length for achieving optimal antifouling performance. High-density PEG brushes prepared at cloud point (CP) grafting conditions demonstrated undetectable protein adsorption, while non-CP grafted coatings showed significant protein adherence [31]. Similarly, zwitterionic polymer brushes require sufficient graft density to form an effective hydration barrier through electrostatic interactions [32].
Recent developments in zwitterionic grafting include innovative linker-free approaches such as plasma immersion ion implantation (PIII), which creates surface-embedded radicals for zwitterion anchoring without chemical initiators. This method has demonstrated a 9-fold reduction in fibrinogen adsorption and nearly 75% reduction in thrombosis on commercial polyurethane substrates [33].
Experimental Protocols and Methodologies
Polydopamine-Assisted Immobilization (Grafting-To)
The following protocol enables substrate-independent coating of both PEG and zwitterionic polymers:
- Substrate Preparation: Clean substrates (e.g., titanium, glass, polymers) thoroughly with acetone, ethanol, and water via sonication [26].
- PDA Deposition: Immerse substrates in 2 mg/mL dopamine solution in 10 mM Tris-HCl buffer (pH 8.5) for 4-24 hours under gentle agitation. The solution typically turns dark brown/black as PDA forms [29].
- Polymer Immobilization: For PEG coatings, incubate PDA-coated substrates in PEG-COOH solution (1-5 mg/mL in buffer) with coupling agents such as EDC/NHS for 12-24 hours [29]. For zwitterionic polymers, use active ester-functionalized zwitterions (e.g., PMEN copolymer) under similar conditions [29].
- Washing and Characterization: Rinse thoroughly with deionized water and characterize using surface plasmon resonance (SPR), X-ray photoelectron spectroscopy (XPS), or water contact angle measurement [29] [6].
Surface-Initiated Polymerization (Grafting-From)
This protocol describes zwitterionic polymer grafting on CLPE surfaces:
- Surface Activation: For thermal grafting method, immerse CLPE substrates in monomer solution (1 M zwitterionic monomer MPC or MEDSAH) with benzoyl peroxide initiator (5 mM) [28].
- Thermal Grafting: Heat at 70°C for 6-12 hours to initiate radical polymerization directly from the substrate surface [28].
- Alternative UV Grafting: For comparison, UV grafting can be performed using benzophenone initiator (1 mM) and UV irradiation (302 nm, 5-10 mW/cm²) for 5-30 minutes [28].
- Post-Processing and Validation: Remove substrates, rinse extensively with water/ethanol, and characterize graft layer thickness using ellipsometry or confocal microscopy. Validate uniformity on curved surfaces [28].
The Scientist's Toolkit: Essential Research Reagents
Table 3: Essential Reagents for Coating Immobilization Research
| Reagent/Chemical | Function/Application | Key Characteristics |
|---|---|---|
| Dopamine hydrochloride | PDA adhesion layer formation [30] [29] | Universal adhesive, substrate-independent, aqueous processing |
| MPC monomer (2-methacryloyloxyethyl phosphorylcholine) | Zwitterionic polymer grafting [28] [32] | Phosphorylcholine headgroup, biomimetic, excellent hydrophilicity |
| SBMA monomer (sulfobetaine methacrylate) | Zwitterionic polymer grafting [33] [32] | Sulfobetaine moiety, strong hydration, antifouling properties |
| PEG-COOH (carboxyl-terminated) | PEG grafting via PDA chemistry [29] [6] | Active terminal group for conjugation, various molecular weights |
| Benzophenone | Photoinitiator for UV grafting [28] | UV-activated radical generation for surface initiation |
| EDC/NHS (1-Ethyl-3-(3-dimethylaminopropyl)carbodiimide/N-hydroxysuccinimide) | Carboxyl-amine coupling chemistry [29] [34] | Zero-length crosslinkers for covalent immobilization |
| Azure A or Toluidine Blue O | Quantitative analysis of sulfonate groups [26] | Metachromatic dye for colorimetric quantification |
The selection between 'grafting-to' and 'grafting-from' immobilization strategies involves careful consideration of application-specific requirements. 'Grafting-from' methods generally provide higher grafting densities beneficial for demanding antifouling applications, while 'grafting-to' approaches offer superior control over polymer architecture. Polydopamine adhesion layers significantly enhance substrate versatility for both strategies. Performance data indicates that both PEG and zwitterionic polymers can achieve ultralow fouling with proper optimization of thickness, graft density, and chemical functionality. Zwitterionic coatings demonstrate particular advantage in thin film applications, while both systems perform comparably at optimal thicknesses. These findings provide a framework for researchers developing advanced antifouling coatings for biomedical devices, drug delivery systems, and diagnostic platforms.
The performance of biomedical devices, biosensors, and drug delivery systems is profoundly influenced by their surface interactions with biological environments. Nonspecific adsorption of proteins, cells, and other biomolecules—a phenomenon known as biofouling—can obstruct functionality and reduce efficacy. Polymer brush coatings have emerged as a powerful strategy to tune interfacial properties and mitigate fouling, with their performance critically dependent on the precision of their synthesis. This guide objectively compares three advanced polymerization techniques—Surface-Initiated Atom Transfer Radical Polymerization (SI-ATRP), Reversible Addition-Fragmentation Chain Transfer (RAFT), and Photopolymerization—for fabricating dense, well-defined brush architectures. Within the broader context of comparing the antifouling performance of poly(ethylene glycol) (PEG) and zwitterionic coatings, this review provides researchers with a structured comparison of synthetic methodologies, including quantitative performance data and detailed experimental protocols to inform material selection and process design.
Table 1: Core Characteristics of Advanced Polymerization Techniques
| Polymerization Method | Key Mechanism | Typical Catalysts | Tolerance to Oxygen | Spatiotemporal Control | Complex Patterning Capability |
|---|---|---|---|---|---|
| SI-ATRP | Reversible halogen transfer | Copper complexes | No (requires deoxygenation) | Limited (without external stimulus) | Moderate |
| RAFT | Reversible chain transfer | Thiocarbonylthio compounds (metal-free) | No (requires deoxygenation) | Moderate (via photoiniferter) | Good |
| SI-PET-RAFT | Photoinduced electron/energy transfer | Xanthene dyes (e.g., Eosin Y) | Yes (oxygen-tolerant) | Excellent | Excellent |
Polymer Brush Synthesis Techniques
Surface-Initiated Atom Transfer Radical Polymerization (SI-ATRP)
SI-ATRP is a workhorse technique for grafting polymer brushes from surfaces. Its mechanism relies on a reversible redox reaction catalyzed by a transition metal complex (typically copper) between a dormant alkyl halide initiator and an active radical species. The equilibrium between active and dormant species ensures controlled chain growth, enabling the synthesis of brushes with predetermined thickness, low dispersity, and complex architectures like block copolymers. A significant advancement is the development of electrochemically mediated ATRP (seATRP), which allows for precise control over the activator/deactivator ratio by applying an electrical potential, facilitating the use of ultra-low catalyst concentrations (as low as 6 ppm of Cu) [35]. This reduction in metal content is crucial for biomedical applications where copper contamination is a concern. However, traditional SI-ATRP requires rigorous oxygen removal and often uses metal catalysts, which can complicate manufacturing and raise biocompatibility questions [36] [37].
Surface-Initiated Photoinduced Electron/Energy Transfer RAFT (SI-PET-RAFT)
SI-PET-RAFT polymerization represents a significant evolution of the RAFT technique, combining the molecular weight control of conventional RAFT with the mild conditions of photoredox catalysis. This method uses visible light-absorbing photocatalysts (e.g., Eosin Y, Erythrosin B) to catalyze the RAFT process via an electron or energy transfer mechanism [36] [38]. A key advantage is its robust tolerance to oxygen. The reaction proceeds in an aqueous environment under atmospheric conditions without prior degassing, as the photocatalyst and a sacrificing electron donor (e.g., triethanolamine, TEOA) work together to consume ambient oxygen [36]. This "open-air" operation dramatically simplifies experimental setup and is highly amenable to scaling up. Furthermore, the light-triggered nature of SI-PET-RAFT provides unparalleled spatiotemporal control, allowing for the creation of complex three-dimensional brush patterns simply by modulating light exposure through photomasks or varying light intensity [36]. Recent innovations, such as conducting SI-PET-RAFT under continuous flow conditions, have further improved control, enabling prolonged linear growth of brushes up to 250 nm—a five-fold increase compared to static (no-flow) conditions which often plateau below 50 nm [39].
Comparative Performance Data
The following tables synthesize quantitative data from key studies to facilitate direct comparison of the antifouling performance and structural outcomes achievable with different polymers and polymerization methods.
Table 2: Antifouling Performance of PEG vs. Zwitterionic Polymer Brushes
| Polymer Coating | Coating Thickness | Protein Challenge | Resulting Adsorption | Key Finding | Source |
|---|---|---|---|---|---|
| PMEN (Zwitterionic) | ~1.0 nm | Bovine Serum Albumin (BSA) | Very low adsorption | Superior performance at ultrathin thicknesses. | [6] |
| PEG (COOH terminus) | ~1.5 - 3.3 nm | BSA | Low adsorption | Performance exceeds PMEN due to stronger steric repulsion at this thickness. | [6] |
| PEG (OH terminus) | ~3.6 nm | BSA & Fibrinogen (Fg) | Ultralow fouling | End-group chemistry critically affects performance; -OH superior to -COOH. | [6] |
| PMEN (Zwitterionic) | ~3.6 nm | BSA & Fibrinogen (Fg) | Ultralow fouling | At optimal thickness, performance is comparable to best PEG coatings. | [6] |
Table 3: Structural Control in Polymer Brush Synthesis via SI-PET-RAFT
| Monomer | Polymerization Conditions | Maximum Brush Thickness Achieved | Key Enabling Factor | Source |
|---|---|---|---|---|
| Poly(MeOEGMA) | No-flow (static) | Plateaus at < 50 nm | Standard batch reaction. | [39] |
| Poly(MeOEGMA) | Continuous flow | Up to 250 nm | Continuous refreshment of monomers and catalysts. | [39] |
| HPMA, CBMA, MeOEGMA | Aqueous SI-PET-RAFT | Not Specified | Oxygen-tolerant polymerization in water. | [36] |
Experimental Protocols
This protocol describes the synthesis of poly(MeOEGMA) brushes with enhanced control and thickness under continuous flow conditions.
Workflow Overview:
Step-by-Step Procedure:
Substrate Preparation and Functionalization:
- Begin with clean silicon wafers (~1 × 1 cm). Rinse sequentially with acetone, absolute ethanol, and Milli-Q water, then dry under a gentle stream of argon.
- Surface Oxidation: Expose the wafers to oxygen plasma for 5 minutes (e.g., 100 W, 5 mbar O₂) to generate surface hydroxyl groups.
- APTES Silanization: Immediately immerse the activated surfaces in a fresh solution of (3-aminopropyl)triethoxysilane (APTES) (1 mg·mL⁻¹ in ethanol) for 16 hours at room temperature. This forms an amine-terminated monolayer.
- RAFT Agent Immobilization: Submerge the aminated substrates in a solution of RAFT-NHS ester (e.g., 4-cyano-4-(phenylcarbonothioylthio)pentanoic acid N-succinimidyl ester, 20 mg in 1 mL dry THF) with a base like triethylamine (TEA, 7 mg) as a proton scavenger. React for 16 hours at room temperature. Rinse thoroughly with THF, acetone, ethanol, and water, then dry under argon. Store under an inert atmosphere until use.
Polymerization Solution Preparation:
- Prepare a photocatalyst stock solution by dissolving Eosin Y (25 mg, 39 μmol) and triethanolamine (TEOA, 160 mg, 1.6 mmol) in 10 mL of Milli-Q water.
- In a separate vial, dissolve the monomer (e.g., MeOEGMA, 94 mg, 0.3 mmol) in 1 mL of Milli-Q water.
- Add 10 μL of the photocatalyst stock solution to the monomer solution and mix thoroughly (e.g., by vortexing).
SI-PET-RAFT Polymerization in Flow:
- Assemble a flow chamber and place the RAFT-functionalized substrate inside.
- Load the polymerization solution into a syringe pump and connect it to the flow chamber.
- Pump the solution over the surface at a controlled flow rate (e.g., 30 μL min⁻¹). The solution height over the surface should be consistent (e.g., 2 mm).
- Initiate polymerization by irradiating the chamber with visible light (e.g., a blue LED light source at 410 nm). The light source should be positioned 3-4 cm away to prevent heating.
- To stop the reaction, turn off the light and stop the flow. Remove the samples and rinse copiously with Milli-Q water and ethanol, then dry under an argon stream.
This protocol uses a polydopamine (PDA) intermediate layer for substrate-independent coating application, allowing for direct performance comparison.
Workflow Overview:
Step-by-Step Procedure:
Polydopamine (PDA) Priming:
- Clean the substrate (e.g., SPR sensor chip, other inert materials) thoroughly.
- Immerse the substrate in a freshly prepared alkaline solution of dopamine hydrochloride (typically 2 mg mL⁻¹ in 10 mM Tris-HCl buffer, pH 8.5) for several hours. This results in the spontaneous deposition of a thin, universal PDA adhesive layer.
Polymer Coating Immobilization:
- For Zwitterionic Coating (PMEN): Immerse the PDA-coated substrate in an aqueous solution of a random copolymer bearing phosphorylcholine zwitterion and active ester side chains (PMEN). The active esters will react with the amine and catechol groups of the PDA layer via amidation coupling.
- For PEG Coating: Similarly, immobilize carboxylic-terminated PEG (or other functional variants) onto the PDA layer via the same coupling chemistry.
- Key Consideration: The coating thickness can be precisely monitored and optimized in real-time using Surface Plasmon Resonance (SPR). This quantitative feedback is crucial for achieving reproducible, high-performance coatings.
Performance Validation:
- Challenge the optimized coatings with single-protein solutions (e.g., BSA, fibrinogen) or complex biological media (e.g., diluted bovine serum, platelets, bacteria).
- Quantify the antifouling performance using SPR or fluorescence microscopy to measure the amount of non-specifically adsorbed material.
The Scientist's Toolkit: Essential Research Reagents
Table 4: Key Reagent Solutions for Polymer Brush Synthesis
| Reagent Category | Specific Example | Function/Purpose | Key Consideration |
|---|---|---|---|
| Photocatalyst | Eosin Y (EY) | Absorbs visible light to initiate PET-RAFT polymerization; enables oxygen tolerance. | Affordable and organic; part of a reductive quenching cycle with TEOA. [36] |
| Sacrificial Electron Donor | Triethanolamine (TEOA) | Consumes dissolved oxygen, allowing polymerization to proceed in open air. | Critical for achieving oxygen-tolerant polymerization in aqueous systems. [36] |
| Silane Coupling Agent | (3-aminopropyl)triethoxysilane (APTES) | Provides surface-anchored amine groups for subsequent immobilization of initiators. | Forms a monolayer on hydroxyl-rich surfaces (e.g., plasma-treated Si wafers). [36] [35] |
| RAFT Agent | RAFT-NHS Ester (e.g., 4-cyano-4-(phenylcarbonothioylthio)pentanoic acid N-succinimidyl ester) | Mediates the controlled radical polymerization; NHS ester group reacts with surface amines. | The immobilized agent defines the grafting-from sites for brush growth. [36] [39] |
| Universal Adhesive | Polydopamine (PDA) | Forms a strong, versatile coating on virtually any substrate, providing a reactive platform. | Allows application of antifouling polymers to inert materials (e.g., implants). [6] |
| Antifouling Monomers | Oligo(ethylene glycol) methacrylate (MeOEGMA) | Forms hydrated brushes that resist protein adsorption via steric repulsion and hydration. | Performance is highly dependent on brush thickness and end-group. [6] [36] |
| Antifouling Monomers | Carboxybetaine methacrylamide (CBMA) | Forms zwitterionic brushes that resist fouling through the formation of a tightly bound water layer. | Excellent performance, especially at very low coating thicknesses. [6] [36] |
The selection of a polymerization technique for fabricating antifouling polymer brushes involves critical trade-offs between control, complexity, and application requirements. While SI-ATRP remains a robust method for achieving well-defined brushes, its reliance on metal catalysts and oxygen-free environments presents limitations for biomedical scaling. SI-PET-RAFT emerges as a highly promising alternative, offering metal-free conditions, inherent oxygen tolerance, and superior spatiotemporal control for complex patterning. The experimental data underscores that beyond the synthesis method, the final antifouling performance is dictated by a triad of factors: chemical composition (PEG vs. zwitterion), brush architecture (thickness, density), and surface chemistry (end-group functionality). Zwitterionic polymers can outperform PEG at ultrathin dimensions, but PEG can achieve ultralow fouling with optimized, thicker brushes. The provided protocols and reagent toolkit offer a foundation for researchers to systematically explore these parameters, advancing the development of advanced non-fouling surfaces for biomedical and biotechnological applications.
Blood-contacting medical devices, including stents, catheters, and oxygenators, are foundational tools in modern clinical practice for managing cardiovascular disease, respiratory failure, and critical care needs. However, when these devices contact blood, the initial nonspecific adsorption of proteins triggers a cascade of detrimental biological responses, including thrombus formation, inflammatory reactions, and bacterial infections [40]. These complications significantly impact patient safety by increasing risks of device failure, pulmonary embolism, and other life-threatening conditions, while also elevating healthcare costs due to extended hospitalization and additional interventions [41]. Consequently, the development of advanced antifouling surface coatings represents a critical research frontier in biomedical engineering, aiming to create bioinert interfaces that mitigate undesirable biological responses.
Within this field, a key scientific debate centers on comparing the performance and applicability of established polyethylene glycol (PEG) coatings against emerging zwitterionic polymers. For years, PEG has been considered the "gold standard" for nonfouling applications due to its hydrophilicity and effectiveness in resisting protein adsorption [42]. However, inherent limitations of PEG, including its susceptibility to oxidative degradation and potential to elicit antibody responses in vivo, have motivated the exploration of superior alternatives [5]. Zwitterionic polymers, featuring balanced positive and negative charges within their molecular structure, have emerged as promising next-generation candidates. This review systematically compares the antifouling performance of PEG and zwitterionic polymer coatings across various blood-contacting devices, providing researchers and product developers with objective, data-driven insights to inform material selection and innovation.
Antifouling Mechanisms: Fundamental Principles
The antifouling performance of both PEG and zwitterionic polymers fundamentally derives from their ability to form hydration layers at the material-water interface. However, the molecular mechanisms through which they achieve this hydration differ significantly, leading to important practical implications for their performance and stability.
PEG-based coatings function primarily as hydrogen bond acceptors (but not donors), organizing surrounding water molecules through hydrogen bonding to form a hydration barrier that provides steric repulsion against approaching proteins and biomolecules [42]. While effective initially, this hydration layer is relatively less robust, and PEG chains are known to undergo oxidative degradation in biological environments, potentially compromising long-term performance [5].
In contrast, zwitterionic polymers, including poly(carboxybetaine) (PCB), poly(sulfobetaine) (PSB), and poly(2-methacryloyloxyethyl phosphorylcholine) (PMPC), create ionic solvation layers. Their molecular structure contains balanced cationic and anionic groups that electrostatically interact with water molecules, resulting in a more tightly bound and denser hydration layer than PEG [5]. This stronger hydration forms a formidable energy barrier that prevents the adsorption of proteins, bacteria, and other fouling agents. Furthermore, the balanced charge distribution ensures electrical neutrality across a wide pH range (approximately 4-10), effectively eliminating electrostatic interactions with charged biomolecules [43]. Research indicates that while a PEG unit binds approximately one water molecule via hydrogen bonding, each zwitterionic repeating unit can bind at least 7-8 water molecules through ionic solvation, resulting in significantly stronger hydration and superior fouling resistance [5].
The following diagram illustrates these fundamental antifouling mechanisms and their consequences for biological interactions:
Performance Comparison: Quantitative Data Analysis
Extensive research has quantified the antifouling performance of PEG and zwitterionic coatings across multiple biological challenges. The following tables summarize key experimental findings from recent studies, providing direct comparison of their effectiveness against protein adsorption, bacterial adhesion, and thrombus formation.
Table 1: Protein and Bacterial Fouling Resistance Performance
| Coating Type | Specific Formulation | Test Organism/Protein | Reduction Efficiency | Reference |
|---|---|---|---|---|
| PEG | PEG brush on silicon wafers | E. coli, S. aureus, P. aeruginosa | 99% suppression after 1 day; reduced performance over 7 days | [42] |
| Zwitterionic | PMPC-based coating | E. coli, S. aureus, P. mirabilis | 72.98%, 75.11%, and 88.23% antibacterial rates after 7 days | [44] |
| Zwitterionic | Poly(MPC-co-HEMA-co-BP) | Bovine Serum Albumin (BSA) | Significant reduction in protein adsorption demonstrated | [44] |
| Zwitterionic | Grafted phosphorylcholine polymer | Broad-spectrum proteins | Near-zero protein fouling across pH 4-10 | [43] |
Table 2: Hemocompatibility and Thrombogenic Performance
| Coating Type | Application | Key Hemocompatibility Metrics | Performance Outcome | |
|---|---|---|---|---|
| PEG (with PTX drug) | WE43 Mg alloy cardiovascular stent | Hemolysis rate: 0.6% (clinical requirement <5%) | Prevents non-specific protein adsorption; enhances hemocompatibility | [45] |
| Zwitterionic | Blood-contacting catheters | Platelet adhesion, thrombus formation | Superior anticoagulation; maintains patency under blood flow | [43] |
| Phosphorylcholine-based | ECMO circuits | Platelet consumption, fibrinogen adsorption | Reduces postoperative bleeding and transfusion requirements | [41] |
| PEG | Glass and SPR chips | Protein adsorption, platelet activation | ~80-90% reduction in bacterial adhesion | [42] |
The data demonstrates that while PEG coatings provide excellent initial antifouling performance, their effectiveness can diminish over time. In contrast, zwitterionic coatings maintain strong antifouling and antimicrobial activity even after prolonged exposure (7 days), with the added advantage of stability across a broad pH range. In hemocompatibility applications, both coating types can meet clinical requirements for hemolysis rates, but zwitterionic coatings exhibit superior anticoagulation properties, making them particularly valuable for devices with prolonged blood contact, such as catheters and ECMO circuits.
Application-Specific Performance in Medical Devices
Vascular Stents
Vascular stents require coatings that prevent restenosis (re-narrowing of blood vessels) and thrombosis (blood clot formation), while also supporting endothelialization (healing of the vessel lining). Drug-eluting stents (DES) represent a major application, where coatings serve as drug reservoirs in addition to providing antifouling properties.
For magnesium alloy biodegradable stents, research on WE43 Mg alloy with PTX-PEG coatings demonstrated excellent corrosion resistance and a stable drug release profile, with a hemolysis rate of 0.6% - well within clinical requirements (<5%) [45]. The PEG component effectively prevented non-specific protein adsorption and nanoparticle aggregation, enhancing surface hemocompatibility. In gastrointestinal cancer stents, polyurethane-silicone (PUS) elastomers with 5-fluorouracil (5FU) demonstrated controlled drug release profiles varying by stent structure, with one design plateauing after ~12 days while another provided gradual release over 150 days [46].
Zwitterionic coatings applied to stents leverage their superior anticoagulant properties by mimicking the electroneutral characteristics of the endothelial glycocalyx, thereby minimizing electrostatic interactions with plasma proteins and consequently reducing platelet consumption and fibrinogen adsorption [41]. This mechanism is particularly valuable for preventing stent thrombosis.
Blood-Contacting Catheters
Catheters face unique challenges due to their extended contact with blood and complex geometries, particularly with long, narrow lumens that are difficult to coat uniformly. Thrombus formation within catheters can lead to serious complications including pulmonary embolism and vascular access failure.
Recent advances in zwitterionic coatings have addressed these challenges through innovative application methods. A universal zwitterion coating approach utilizing a wet-adhesive initiator-bearing polymer rapidly assembles on catheter surfaces in aqueous environments, facilitating grafting of superhydrophilic zwitterionic polymers onto complex geometries [43]. This technique demonstrates broad substrate adaptability, successfully coating ten different substrate materials with near-zero protein fouling across a wide pH range, and superior resistance to blood cells and bacteria while maintaining stability under simulated bloodstream conditions.
Compared to conventional heparin coatings that dominate the catheter market but carry risks of heparin-induced thrombocytopenia and bleeding complications, zwitterionic alternatives provide excellent anticoagulation without releasing bioactive molecules into the bloodstream [43]. This safety profile, combined with durable antifouling performance, positions zwitterionic polymers as promising alternatives for next-generation catheter coatings.
Extracorporeal Membrane Oxygenation (ECMO) Circuits
ECMO circuits present particularly challenging environments for antifouling coatings due to prolonged blood contact and the high shear stresses imposed by centrifugal pumps. Circuit components—including cannulas, oxygenators, and pumps—require exceptional hemocompatibility to prevent thrombosis while minimizing bleeding risks from systemic anticoagulation.
Phosphorylcholine (PC)-based zwitterionic coatings have demonstrated significant promise in ECMO applications by mimicking the outer leaflet of cell membranes, which naturally exhibits thromboresistant properties [41]. Several clinical studies have associated PC-coated circuits with reductions in postoperative bleeding, transfusion requirements, total heparin dosage, and severe complications.
Comparative studies between zwitterionic and PEG-based coatings in ECMO environments have shown that zwitterionic polymers typically exhibit superior hydration capacity and fouling resistance under dynamic flow conditions. For example, poly(sulfobetaine methacrylate) (PSBMA) modifications on poly-4-methyl-1-pentene (PMP) oxygenator membranes reduced protein adsorption by 70.58% while significantly enhancing blood compatibility [47]. This improved performance is attributed to the stronger hydration layer formed through ionic solvation in zwitterionic polymers compared to hydrogen bonding in PEG.
Experimental Protocols and Methodologies
Coating Fabrication Techniques
Zwitterionic Coating via Tannic Acid Adhesion
A robust method for applying zwitterionic coatings to various substrates utilizes tannic acid (TA)-Fe³⁺ complexation followed by zwitterionic polymer grafting [47]. The experimental workflow comprises:
- Surface Preparation: Polyethylene terephthalate (PET) substrates are pre-soaked in ethanol to clean and activate the surface.
- TA-Fe³⁺ Deposition:
- One-pot method: TA aqueous solution (4 mg/mL) is mixed with FeCl₃·6H₂O solution (1 mg/mL). The substrate is immersed for 5 minutes.
- Two-pot method: Sequential immersion in TA solution (4 mg/mL, 5 minutes) followed by Fe³⁺ solution (1 mg/mL, 5 minutes).
- Zwitterionic Copolymer Synthesis: Poly(ethylenimine)-g-sulfobetaine methacrylate (PEI-g-SBMA) is synthesized via Michael addition reaction by grafting SBMA onto branched PEI (MW=10,000).
- Surface Grafting: TA-Fe³⁺ modified substrates are immersed in PEI-g-SBMA solution (20 mg/mL in PBS buffer, pH 8.5) for 12 hours to allow Schiff-base reaction.
- Post-processing: Samples are rinsed with deionized water and dried under nitrogen.
This method creates coatings with significantly improved hydrophilic and lubricating properties, demonstrating excellent resistance to bovine serum albumin (BSA) adsorption in antifouling tests [47].
Photocurable Zwitterionic Coatings
For urinary catheter applications, a photocurable zwitterionic coating with enhanced antifouling and lubrication properties has been developed [44]:
- Polymer Synthesis: Copolymer poly(MPC-co-HEMA-co-BP) (PMHB) is synthesized through free radical copolymerization of 2-methacryloyloxyethyl phosphatidylcholine (MPC), hydroxyethyl methacrylate (HEMA), and 3-(benzoyl)phenyl methacrylate (BP).
- Solution Preparation: The PMHB copolymer is dissolved in solvent to create a pre-gel solution.
- Application: The solution is spin-coated onto polyurethane substrates.
- UV Curing: Coatings are crosslinked under UV irradiation, with benzophenone groups enabling covalent bonding to the substrate.
This approach creates hydrophilic coatings with hygroscopic properties that facilitate hydrated layer formation, resulting in friction reduction and antifouling performance demonstrated by 72.98-88.23% antibacterial rates against common pathogens after 7 days [44].
The following diagram illustrates the sequence of these coating fabrication processes:
Characterization and Testing Methods
Standardized experimental protocols are essential for objectively comparing antifouling performance:
Surface Analysis:
- Water Contact Angle (WCA): Measures hydrophilicity/hydrophobicity using goniometers.
- X-ray Photoelectron Spectroscopy (XPS): Determines elemental surface composition and chemical states.
- Fourier Transform Infrared (FTIR) Spectroscopy: Identifies chemical functional groups on coated surfaces.
- Scanning Electron Microscopy (SEM): Visualizes surface morphology and coating uniformity.
Antifouling Performance:
- Protein Adsorption Tests: Quantify nonspecific adsorption of bovine serum albumin (BSA) or fibrinogen using spectrophotometric methods (e.g., BCA Protein Assay Kit) or fluorescence labeling [47].
- Bacterial Adhesion Assays: Evaluate colonization resistance against clinically relevant strains (E. coli, S. aureus, P. aeruginosa) through plate counting and fluorescence microscopy after incubation.
- Antibacterial Efficacy: Determine bacterial viability on coated surfaces using live/dead staining or metabolic activity assays (e.g., MTT).
Hemocompatibility Assessment:
- Hemolysis Rate: Measure red blood cell rupture after contact with coatings; clinical requirement <5% [45].
- Platelet Adhesion: Quantify platelet attachment and activation using SEM or lactate dehydrogenase (LDH) assay.
- Thrombogenicity: Evaluate clot formation under dynamic flow conditions using simulated bloodstream models [43].
Mechanical and Stability Testing:
- Abrasion Resistance: Assess coating durability under mechanical stress.
- Hydration Stability: Evaluate long-term performance in physiological solutions.
- Sterilization Resistance: Validate coating integrity after gamma irradiation or ethylene oxide sterilization [46].
The Scientist's Toolkit: Essential Research Reagents and Materials
Table 3: Key Research Reagents for Antifouling Coating Development
| Reagent/Material | Function/Application | Research Context |
|---|---|---|
| Sulfobetaine methacrylate (SBMA) | Zwitterionic monomer for copolymer synthesis | Building block for PSBMA-based antifouling polymers [47] |
| 2-Methacryloyloxyethyl phosphorylcholine (MPC) | Zwitterionic monomer mimicking cell membranes | Primary component for PMPC-based coatings [44] |
| Tannic Acid (TA) - Fe³⁺ Complex | Adhesive layer for surface priming | Enables zwitterionic polymer grafting on diverse substrates [47] |
| Polyethyleneimine (PEI) | Cationic polymer for grafting zwitterionic groups | Serves as backbone for PEI-g-SBMA graft copolymers [47] |
| Poly(ethylene-co-vinyl acetate) (PEVA) | Drug diffusion-regulating membrane | Controls drug release rate in drug-eluting stent coatings [46] |
| Polyurethane-silicone (PUS) elastomer | Drug-encapsulating polymer reservoir | Basecoat for drug-eluting gastrointestinal stents [46] |
| Benzophenone derivatives | Photosensitive crosslinking agents | Enable UV-curing of coatings on various substrates [44] |
Based on comprehensive analysis of current research, zwitterionic polymer coatings demonstrate superior overall performance compared to PEG-based coatings for most blood-contacting applications. The key advantages of zwitterionic materials include:
- Stronger hydration layers through ionic solvation versus hydrogen bonding
- Enhanced long-term stability against oxidative degradation
- Broader pH stability maintaining effectiveness across physiological ranges
- Superior anticoagulation properties approaching near-zero fouling performance
However, PEG-based coatings remain valuable for specific applications, particularly drug-eluting systems where their well-characterized drug release kinetics and compatibility with various therapeutic agents provide practical advantages. PEG coatings also typically present fewer mechanical challenges than highly hydrophilic zwitterionic hydrogels, which can be brittle and require reinforcement strategies [5].
Future research directions should address critical remaining challenges, including:
- Mechanical reinforcement of zwitterionic hydrogels for load-bearing applications
- Scalable manufacturing processes for complex device geometries
- Multifunctional coating systems combining antifouling with bioactive capabilities
- Long-term in vivo validation of coating durability and safety
As the field advances, the optimal selection between PEG and zwitterionic coatings will continue to depend on specific device requirements, balancing antifouling performance, mechanical properties, manufacturing considerations, and clinical application needs.
Enabling Long-Circulating Nanomedicines and Drug Delivery Systems
The efficacy of intravenously administered nanomedicines is fundamentally constrained by the body's biological recognition and clearance mechanisms. Upon introduction into the bloodstream, nanoparticles are rapidly coated by plasma proteins, a process known as opsonization, which marks them for removal by the mononuclear phagocyte system (MPS). This process significantly shortens their circulation half-life, limiting their ability to reach and accumulate at the target site, such as a tumor. The application of antifouling coatings creates a "stealth" barrier that minimizes nonspecific protein adsorption, thereby prolonging circulation time and enhancing therapeutic efficacy [32] [48].
For over two decades, poly(ethylene glycol) (PEG) has been the gold standard for creating stealth nanocarriers through a process called PEGylation. However, the emergence of PEG-specific antibodies and accelerated blood clearance (ABC) phenomenon with repeated dosing has spurred the search for alternatives [5]. Zwitterionic polymers, inspired by the outer membrane of mammalian cells, have recently emerged as a powerful alternative. These polymers, bearing pairs of oppositely charged groups, confer superior antifouling properties through the formation of a robust hydration layer via ionic solvation [32] [5]. This guide provides an objective, data-driven comparison of PEG and zwitterionic polymer coatings to inform the design of next-generation long-circulating nanomedicines.
Material Design and Antifouling Mechanisms
Polyethylene Glycol (PEG) Coatings
PEG is a synthetic, hydrophilic, and uncharged polymer that is covalently grafted onto nanoparticle surfaces. Its antifouling action is primarily attributed to the formation of a steric hydration barrier. The ethylene oxide repeating units bind water molecules predominantly through hydrogen bonding, creating a physical and energetic barrier that prevents approaching proteins from adsorbing to the surface. The dynamic flexibility of PEG chains further contributes to repelling foulants [32] [48].
A key advancement in PEGylation is the formation of a dense "PEG corona" on the nanoparticle surface. This corona reduces opsonization and subsequent immune recognition, significantly extending systemic circulation time. This is particularly crucial in cancer therapy, as it increases the likelihood of nanoparticles reaching the tumor site via the Enhanced Permeability and Retention (EPR) effect [48].
Zwitterionic Polymer Coatings
Zwitterionic polymers, such as poly(carboxybetaine) (PCB), poly(sulfobetaine) (PSB), and poly(2-methacryloyloxyethyl phosphorylcholine) (PMPC), contain both cationic and anionic groups within their repeating units [5]. Unlike PEG, which binds water via hydrogen bonding, zwitterions bind water molecules through stronger, more robust ionic solvation. Each zwitterionic unit can bind at least 7-8 water molecules, forming a denser and more tightly bound hydration layer than PEG. This layer presents a formidable steric and energetic barrier to protein adsorption, effectively preventing the initial step in the biofouling cascade [32] [5].
Table 1: Key Characteristics of PEG and Zwitterionic Coatings
| Feature | PEG (and derivatives) | Zwitterionic Polymers |
|---|---|---|
| Chemical Moieties | Ethylene oxide repeats [32] | Sulfobetaine (SBMA), Carboxybetaine (CBMA), Phosphorylcholine (MPC) [6] [5] |
| Hydration Mechanism | Hydrogen bonding [5] | Ionic solvation [5] |
| Primary Antifouling Mechanism | Steric repulsion & dynamic flexibility [32] [48] | Dense, tightly bound hydration layer [32] [5] |
| Key Material Property | High chain mobility and flexibility [32] | Charge neutrality and superhydrophilicity [5] |
Diagram 1: Antifouling hydration layer mechanisms for PEG and zwitterionic coatings.
Quantitative Performance Comparison
Direct, quantitative comparisons are essential for evaluating coating performance. Surface Plasmon Resonance (SPR) is a key tool for real-time, label-free analysis of protein adsorption onto coated surfaces.
Protein Adsorption and Coating Thickness
A comparative study using SPR provided a detailed analysis of PEG and zwitterionic (PMEN) coatings of controlled thickness. The findings highlight a nuanced, thickness-dependent performance.
Table 2: Protein Adsorption Performance vs. Coating Thickness [6]
| Coating Type | Coating Thickness | Performance against BSA | Key Finding |
|---|---|---|---|
| Zwitterionic (PMEN) | ~1.0 nm | Much stronger resistance | Superior at ultra-thin dimensions. |
| PEG-OH | ~1.0 nm | Weaker resistance | Less effective at minimal thickness. |
| PEG-OH | 1.5 - 3.3 nm | Resistance exceeds PMEN | Performance improves with thicker, steric-repelling layers. |
| Both PEG & PMEN | ~3.6 nm | Ultralow fouling (BSA & Fg) | Both achieve excellent performance at sufficient thickness. |
A critical finding was the significant impact of PEG's terminal chemistry. Changing the end group from -OH to -COOH resulted in a 10-fold increase in protein adsorption, underscoring that surface chemistry details are as critical as the polymer choice itself [6].
Long-Term Stability and In Vivo Performance
While both coatings can achieve excellent initial antifouling, their long-term stability differs. PEG is susceptible to oxidative degradation in vivo, which can compromise its performance over time [5]. In contrast, zwitterionic coatings demonstrate superior stability. For instance, a zwitterionic SBMA-poly dopamine (PDA) coating on a biosensor exhibited less than 8.5% signal drift over 24 hours in serum, compared to over 27% for an uncoated control. Furthermore, this zwitterionic coating displayed greater resistance to hydrolytic degradation and signal loss over time compared to a PEG-based control [49].
Regarding immune response, the generation of PEG-specific antibodies is a documented concern that can lead to accelerated blood clearance of PEGylated nanocarriers upon repeated administration. Zwitterionic materials, by mimicking the cell membrane, show low immunogenicity and do not currently face this issue, making them promising for chronic therapies requiring multiple doses [5].
Table 3: Comparative In Vivo & Stability Profile
| Parameter | PEG Coatings | Zwitterionic Coatings |
|---|---|---|
| Circulation Half-life | Extended (classic stealth) [48] | Comparable or superior in model systems [50] |
| Immune Response | Can induce PEG-specific antibodies [5] | Low immunogenicity reported [5] |
| Chemical Stability | Susceptible to oxidative degradation [5] | High hydrolytic and metabolic stability [49] |
| Mechanical Stability | Good on nanoparticles | Can be brittle in hydrogels; requires reinforcement [5] |
Experimental Protocols for Performance Evaluation
Quantitative Coating Fabrication and SPR Analysis
This protocol, adapted from Xing et al., describes a substrate-independent method for fabricating and quantitatively comparing coatings [6].
Methodology:
- Substrate Preparation: Sensor chips (e.g., gold for SPR) are pre-coated with a polydopamine (PDA) intermediate layer via immersion in a dopamine solution (e.g., 2 mg/mL in Tris-HCl buffer, pH 8.5) for several hours. PDA acts as a universal adhesive.
- Polymer Immobilization: The PDA-coated chips are incubated in aqueous solutions of the functional polymers.
- Zwitterionic Coating: Use a random copolymer (e.g., PMEN) bearing phosphorylcholine and active ester side chains. The active esters react with PDA amines via amidation coupling.
- PEG Coating: Use carboxylic acid-terminated PEG (e.g., HOOC-PEG-COOH) which also couples to PDA via amidation.
- Performance Optimization: The SPR instrument is used to monitor the coating deposition in real-time, allowing precise control over the final coating thickness by varying polymer concentration and immobilization time.
- Antifouling Assay: The coated sensor is exposed to challenging media like undiluted blood serum or a solution of a model protein (e.g., Bovine Serum Albumin, BSA, at 1 mg/mL). The SPR system quantifies the mass of adsorbed protein in real-time, providing a direct measure of antifouling efficacy.
Evaluating Colloidal Stability of Coated Nanoparticles
This protocol, based on PLGA nanoparticle studies, assesses the stability of coated nanocarriers in physiologically relevant conditions [51] [48].
Methodology:
- Nanoparticle Synthesis and Coating:
- PEGylated NPs: Synthesize PLGA nanoparticles using a single or double emulsion-solvent evaporation method. PEGylation is achieved by using a PEG-PLGA diblock copolymer or by post-synthesis grafting of PEG-amine to surface carboxyls via EDC/NHS chemistry [48].
- Zwitterionic NPs: Coat pre-formed nanoparticles by grafting zwitterionic polymers (e.g., PSB) onto the surface using similar carbodiimide chemistry or by incorporating zwitterionic monomers during formulation.
- Characterization: Determine the hydrodynamic diameter, polydispersity index (PDI), and zeta potential of the nanoparticles using Dynamic Light Scattering (DLS) in deionized water.
- Stability Test: Incubate the nanoparticles in a biologically relevant buffer (e.g., phosphate-buffered saline, PBS, or cell culture medium with 10% serum) at 37°C under gentle agitation.
- Analysis: Monitor the particle size and PDI over time (e.g., 0, 1, 4, 8, 24, 48 hours) using DLS. A stable coating will prevent aggregation, manifesting as minimal change in hydrodynamic diameter and PDI. A shift in zeta potential towards neutrality also indicates a successful stealth coating.
Diagram 2: Experimental workflow for coating performance evaluation.
The Scientist's Toolkit: Essential Research Reagents
Table 4: Key Reagents for Antifouling Coating Research
| Reagent / Material | Function in Research | Application Notes |
|---|---|---|
| Polydopamine (PDA) | Universal adhesive primer for coating diverse substrates (metals, polymers, ceramics) [6]. | Enables substrate-independent comparison of polymer coatings. |
| Carboxyl-terminated PEG | Model PEG coating polymer; couples to amine-rich surfaces via EDC/NHS chemistry [6] [48]. | Terminal group (-OH vs -COOH) drastically impacts performance [6]. |
| Poly(sulfobetaine methacrylate) (PSBMA) | Common zwitterionic polymer; provides excellent antifouling via ionic hydration [49] [5]. | Often grafted from surfaces via ATRP or coupled via PDA layer. |
| Poly(MPC) (PMPC) | Phosphorylcholine-based zwitterionic polymer; mimics cell membrane structure [6] [32]. | Known for its high biocompatibility and hemocompatibility. |
| EDC / NHS Crosslinkers | Activate carboxyl groups for covalent conjugation to amine groups on surfaces or nanoparticles [48]. | Essential for grafting polymers onto pre-formed nanoparticles. |
| PLGA-PEG Diblock Copolymer | For creating PEGylated nanoparticles with a dense PEG corona [48]. | Standard material for formulating long-circulating nanocarriers. |
| Bovine Serum Albumin (BSA) | Model protein for in vitro antifouling adsorption assays [6]. | Used in SPR and fluorescence-based quantification methods. |
The choice between PEG and zwitterionic coatings is not a simple binary selection but a strategic decision based on the specific application requirements. PEG remains a well-established, versatile, and effective option, particularly when a thick, steric brush layer can be formed. However, researchers must contend with its potential immunogenicity and oxidative degradation.
Zwitterionic coatings offer a compelling alternative, especially for applications demanding the highest level of fouling resistance, superior stability, and low immunogenicity. Their performance at ultra-thin dimensions and in complex biological environments makes them ideal for sensitive applications like biosensors and implantable devices [49]. The main challenge for zwitterionic materials lies in improving the mechanical properties of bulk hydrogels for load-bearing applications, an area of active research involving nanocomposites and double-network hydrogels [5].
Future research will likely focus on advanced material designs, including the development of hybrid coatings that leverage the strengths of both polymer families. Furthermore, high-throughput screening methods are being employed to rapidly optimize synthesis parameters and uncover new structure-property relationships, accelerating the discovery of next-generation antifouling materials [52]. As the field progresses, zwitterionic polymers are poised to play an increasingly central role in revolutionizing targeted drug delivery and diagnostic technologies.
Performance in Biosensors, Ophthalmic Implants, and Tissue Engineering Scaffolds
The performance of biomedical devices and constructs is profoundly influenced by their interactions with the complex biological environment. A critical challenge is biofouling—the nonspecific adsorption of proteins, cells, and other biomolecules—which can compromise device functionality, trigger immune responses, and lead to implant failure [53]. For decades, polyethylene glycol (PEG) has been the gold standard antifouling polymer [53]. However, a new class of materials, zwitterionic polymers, has emerged as a powerful alternative, often demonstrating superior performance [5] [54]. This guide provides an objective, data-driven comparison of PEG and zwitterionic coatings across three key biomedical applications: biosensors, ophthalmic implants, and tissue engineering scaffolds, framing the analysis within the broader thesis of antifouling performance.
Material Properties and Antifouling Mechanisms
The fundamental differences in the structure and hydration mechanisms of PEG and zwitterionic polymers underpin their performance variations.
Polyethylene Glycol (PEG)
PEG is a non-ionic, hydrophilic polymer that resists fouling primarily through hydrogen bonding with water molecules, forming a hydration layer that acts as a steric barrier [53]. Each ethylene glycol (EG) unit can bind approximately one water molecule [53]. While effective, this hydration is weaker than that of zwitterionic materials, and PEG is susceptible to oxidative degradation in biological environments, which can diminish its antifouling properties over time [5] [53]. Furthermore, the emergence of PEG-specific antibodies in some patients can lead to accelerated blood clearance and immune reactions [5].
Zwitterionic Polymers
Zwitterionic polymers, such as poly(carboxybetaine) (PCB), poly(sulfobetaine) (PSB), and poly(2-methacryloyloxyethyl phosphorylcholine) (PMPC), contain both cationic and anionic groups within a single monomeric unit, resulting in overall charge neutrality [53] [54]. They bind water molecules via stronger ionic solvation, creating a more robust and dense hydration layer. Each sulfobetaine unit, for example, can bind 7–8 water molecules [53]. This intense hydration forms a physical and energetic barrier that is highly effective at repelling biomolecules [54]. Zwitterionic materials also exhibit an "anti-polyelectrolyte effect", where their chains expand in saline solutions, enhancing their stability and antifouling performance in physiological conditions [5].
Table 1: Fundamental Properties of PEG and Zwitterionic Polymers
| Property | Polyethylene Glycol (PEG) | Zwitterionic Polymers (e.g., PSB, PCB) |
|---|---|---|
| Chemical Nature | Non-ionic | Equimolar positive & negative charges; net neutral |
| Primary Hydration Mechanism | Hydrogen Bonding | Ionic Solvation |
| Approx. Water Molecules Bound per Unit | ~1 (per EG unit) [53] | 7–8 (per SB unit) [53] |
| Hydration Binding Energy | Lower | Higher [53] |
| Susceptibility to Oxidation | Yes, vulnerable [5] | Highly chemically stable [54] |
| Immunogenicity | Can elicit PEG-specific antibodies [5] | Minimal immunogenicity reported [53] |
The following diagram illustrates the distinct hydration mechanisms and subsequent antifouling performance of PEG and zwitterionic polymers.
Performance Comparison in Biomedical Applications
Direct comparative studies and application-specific outcomes reveal clear performance differences.
Biosensors
Antifouling coatings are critical for biosensors to function accurately in complex biological fluids like blood, serum, or interstitial fluid by minimizing nonspecific signal noise [55].
A seminal study directly compared linear PEG and poly(sulfobetaine methacrylate) (pSBMA) coatings of equivalent molecular weight and hydrodynamic radius on wearable microprojection array biosensors [2] [13]. The goal was the selective capture of the dengue virus non-structural protein 1 (NS1) biomarker.
Table 2: Performance in Biosensor Antigen Capture
| Experimental Metric | PEG Coating | pSBMA (Zwitterionic) Coating |
|---|---|---|
| Non-specific Protein Adsorption (in single protein sol., diluted plasma, skin) | Comparable to pSBMA [2] | Comparable to PEG [2] |
| Density of Immobilized Capture Antibody (IgG) | Significantly Lower [2] | Significantly Higher [2] |
| Capture of Target Antigen (Dengue NS1) in buffer/plasma | Baseline | Significantly more NS1 captured [2] |
| In Vivo Signal-to-Noise Ratio (in dengue-infected mice) | Baseline | >2-fold increase compared to PEG [2] [13] |
Key Experimental Protocol (Summarized) [2]:
- Surface Functionalization: Polycarbonate microprojection arrays were amine-modified. pSBMA or linear PEG polymers were then attached using a "grafting to" approach.
- Assay Conditions: Coated devices were incubated in solutions containing dengue NS1 or applied to the skin of dengue-infected mice.
- Measurement: The captured NS1 was quantified, and the signal-to-noise ratio was calculated to compare the specific detection efficiency against non-specific background.
Conclusion for Biosensors: While both coatings resist non-specific adsorption, pSBMA supports a higher density of functional capture ligands and enables more efficient target binding, resulting in a significantly higher diagnostic signal (>2-fold) in a realistic, complex biological environment [2] [13].
Ophthalmic Implants
In ophthalmic applications such as contact lenses, intraocular lenses, and drug-delivery implants, antifouling surfaces are essential to prevent the adsorption of proteins, lipids, and bacteria, which can cause inflammation, infection, and device failure [56] [57].
While direct head-to-head comparisons in clinical ophthalmic implants are less common, the superior hydration and stability of zwitterionic materials like PMPC (inspired by the cell membrane lipid phosphatidylcholine) confer significant advantages. PMPC-based hydrogels exhibit exceptionally low friction and stable antifouling properties even under high ionic strength, making them ideal for comfortable contact lenses and implants that move within the eye [54].
Furthermore, zwitterionic carboxybetaine (CB) hydrogels are particularly useful for functionalized ophthalmic devices. The carboxyl groups on CB allow for the straightforward covalent conjugation of therapeutic agents, enabling the development of drug-eluting contact lenses with sustained release profiles [54]. This contrasts with PEG, which lacks readily functionalizable groups for stable bioconjugation without prior modification.
Conclusion for Ophthalmic Implants: Zwitterionic polymers, particularly PMPC and PCB, offer superior lubricity, stability, and a platform for integrated drug delivery, positioning them as next-generation materials for smart ophthalmic implants that go beyond simple inertness to active therapy [56] [54].
Tissue Engineering Scaffolds
For tissue engineering, the scaffold material must not only resist fouling to prevent fibrotic encapsulation but also often support specific cellular adhesion and tissue integration [58] [59].
A significant challenge for pure zwitterionic hydrogels has been their poor mechanical properties—they are often brittle and weak due to their superhydrophilicity and high water content, limiting their use in load-bearing applications [5]. However, advanced reinforcement strategies have been developed to create high-strength zwitterionic hydrogels.
Table 3: Performance in Tissue Engineering Scaffolds
| Aspect | PEG-based Hydrogels | Zwitterionic Hydrogels |
|---|---|---|
| Inherent Mechanical Strength | Moderate, tunable via crosslinking | Generally low and brittle without modification [5] |
| Bioinertness | High, can inhibit desired cell adhesion | High, but can be functionally modified (e.g., via CB's COOH groups) [54] |
| Key Reinforcement Strategies | Double networks, nanocomposites | Nanocomposites (e.g., Laponite clay, CNC) [5], Double networks, Macromolecular microspheres [5] |
| Mechanical Performance Post-Reinforcement | Good | Excellent: Can achieve >1750% stretchability and breaking strength of ~0.27 MPa [5] |
| Additional Functionality | Limited | Ionic conductivity, Self-healing properties (via dipole-dipole interactions) [54] |
Key Experimental Protocol (Summarized) for Zwitterionic Nanocomposite Hydrogel [5]:
- Preparation: Zwitterionic monomer (e.g., SBMA) and acrylamide (AM) are polymerized in the presence of reinforcing agents like exfoliated Laponite XLG nanosheets or Cellulose Nanocrystals (CNCs).
- Mechanism: The nanosheets act as physical crosslinkers via ionic interactions with the zwitterions, forming a dense, energy-dissipating network.
- Outcome: This strategy produces hydrogels with high toughness, stretchability (up to 1750%), and self-healing capabilities, making them suitable for dynamic physiological environments.
Conclusion for Tissue Engineering: While PEG hydrogels are mechanically competent, advanced zwitterionic composites can achieve superior mechanical toughness and additional functionalities like self-healing and conductivity, making them promising for load-bearing and dynamic tissue constructs [5] [59].
The Scientist's Toolkit: Key Research Reagents
This table details essential materials and their functions for researching PEG and zwitterionic coatings.
Table 4: Essential Research Reagents and Materials
| Reagent/Material | Function in Research | Key Considerations |
|---|---|---|
| Laponite XLG Nanosheets | Nanocomposite physical crosslinker for reinforcing zwitterionic hydrogels [5]. | Enhances mechanical strength, toughness, and can introduce self-healing properties. |
| Carboxybetaine (CB) Methacrylate | Zwitterionic monomer for creating hydrogels with functionalizable carboxyl groups [54]. | Enables covalent conjugation of peptides, proteins, and drugs for bioactive scaffolds. |
| Sulfobetaine (SB) Methacrylate | A readily accessible zwitterionic monomer for creating antifouling hydrogels and coatings [5]. | Imparts strong antifouling and ionic conductivity; exhibits thermo-responsiveness. |
| Poly(ethylene glycol) Diacrylate (PEGDA) | A crosslinkable PEG derivative for forming hydrogel networks. | Molecular weight determines mesh size and mechanical properties; susceptible to degradation. |
| Dengue NS1 Antigen & Anti-NS1 IgG | Model target and capture antibody for benchmarking biosensor performance [2]. | Used in validation studies to quantify specific capture efficiency vs. non-specific adsorption. |
| Amine-Modified Substrates (e.g., polycarbonate, gold chips) | Foundation for polymer grafting via "grafting to" or "grafting from" strategies [2]. | Surface density of amine groups controls the final density of the grafted polymer brush. |
Integrated Experimental Workflow
The following diagram outlines a generalized experimental workflow for preparing and evaluating antifouling coatings, integrating key steps from the protocols discussed.
The comparative analysis of PEG and zwitterionic polymers across biosensors, ophthalmic implants, and tissue engineering reveals a nuanced but clear trend. While PEG remains a viable and well-understood antifouling material, zwitterionic polymers frequently demonstrate superior performance in demanding biological settings.
- In Biosensors, zwitterionic pSBMA coatings provide a higher signal-to-noise ratio and more efficient target capture than PEG in complex, real-world samples [2] [13].
- In Ophthalmic Implants, zwitterionic materials like PMPC and PCB offer exceptional lubricity, stability, and a platform for integrated drug delivery [56] [54].
- In Tissue Engineering, although initially mechanically inferior, zwitterionic hydrogels can be engineered into tough, functional composites with properties surpassing many PEG-based systems [5].
The choice between PEG and zwitterionic polymers ultimately depends on the specific application requirements. However, for applications demanding the highest fouling resistance in complex media, long-term stability, and additional functionalities like conductivity or bio-conjugation, zwitterionic materials represent the leading edge of antifouling biomaterial science.
Addressing Limitations: Immunogenicity, Stability, and Mechanical Performance
Polyethylene glycol (PEG) has long been considered a gold standard polymer in biomedical science for improving the pharmacokinetic properties and stability of therapeutics. Through a process known as PEGylation, this flexible, hydrophilic polymer is covalently attached to proteins, peptides, and nanocarriers to confer 'stealth' properties, prolonging circulation time by reducing opsonization and recognition by the mononuclear phagocyte system [60] [61]. Its extensive application history, from early FDA-approved drugs like Adagen to the recent PEGylated lipid nanoparticles in mRNA COVID-19 vaccines, underscores its foundational role in drug delivery and bioconjugation [60] [62].
However, the very properties that made PEG ubiquitous are now shadowed by significant clinical challenges. Emerging research has uncovered that PEG is not immunologically inert as once presumed [60]. The rise of anti-PEG antibodies (APAs) in treated individuals—and surprisingly, even in treatment-naïve populations—poses a substantial threat to the efficacy and safety of PEGylated therapeutics [63] [64]. This immunogenic response can lead to accelerated blood clearance (ABC) of PEGylated drugs, reduced therapeutic efficacy, and potentially severe hypersensitivity reactions [64] [62]. Within this context, zwitterionic polymers have emerged as a promising alternative, demonstrating superior antifouling performance and potentially reduced immunogenicity [2] [65]. This guide provides a objective comparison of PEG and zwitterionic polymer performance, supported by experimental data relevant to antifouling coating research.
PEG's Mechanism and Limitations
The Conventional Working Mechanism of PEG
PEG functions primarily by forming a highly hydrated steric barrier around the conjugated therapeutic agent or surface. The chemical structure of PEG, HO-[CH2-CH2-O]n-H, confers significant chain flexibility and hydrophilicity, with each subunit binding 2-3 water molecules [60]. This hydration sheath creates a physical barrier that sterically hinders the approach and binding of biomacromolecules, such as opsonizing proteins, to the underlying surface [60]. Consequently, PEGylation slows clearance by the mononuclear phagocyte system, leading to longer circulation times for drugs and improved accumulation at target sites [60] [61]. For nanoparticles, PEG coatings also enhance colloidal stability, as demonstrated by improved zeta potential and reduced aggregation in PEG-coated magnetite nanoparticles [66].
The Critical Limitations of PEG
Immunogenicity and Anti-PEG Antibodies
A primary limitation of PEG is its unexpected immunogenicity. Contrary to initial beliefs of being immunologically inert, PEG can trigger the production of specific anti-PEG antibodies (APAs) [64]. The mechanisms are complex:
- Thymus-Dependent (TD) Response: PEGylated proteins and large nanoparticles can be processed by B cells, which present PEG-related antigens to helper T cells. This collaboration triggers a robust immune response, including class switching to produce high-affinity anti-PEG IgG antibodies [64].
- Thymus-Independent (TI) Response: The repetitive, linear structure of the PEG backbone itself can act as a TI-2 antigen, directly cross-linking B cell receptors and leading to the production of anti-PEG IgM with minimal T cell involvement [64].
The consequences of APA development are clinically significant. APAs can bind to PEGylated therapeutics, leading to their accelerated blood clearance (ABC) via complement activation and uptake by phagocytic cells in the liver and spleen. This phenomenon is particularly pronounced after repeated administration, drastically reducing the drug's half-life and efficacy upon subsequent doses [64] [62]. Furthermore, the formation of immune complexes can trigger hypersensitivity reactions, including potentially life-threatening anaphylaxis [64].
Alarmingly, pre-existing APAs are found in a substantial portion of the population, attributed to exposure to PEG in consumer products like cosmetics and processed foods [63] [64]. One study reported a pre-existing APA detection rate as high as 72% in healthy individuals [64], posing a challenge for first-time administration of PEGylated drugs.
Oxidative Degradation
While the search results provided do not detail PEG's oxidative degradation, it is a well-documented challenge in the field. PEG chains are susceptible to oxidative damage, especially in the presence of oxygen or reactive oxygen species, which can lead to chain scission and loss of polymeric properties. This degradation compromises the steric hydration barrier over time, diminishing the long-term stability and performance of PEGylated coatings and therapeutics.
Performance Comparison: PEG vs. Zwitterionic Polymers
Antifouling Performance in Complex Biological Environments
Direct comparative studies reveal distinct performance differences between PEG and zwitterionic polymers, particularly in demanding applications.
Table 1: Comparative Antifouling Performance of PEG and Zwitterionic Polymers
| Performance Metric | PEG Coating | Zwitterionic Polymer (pSBMA) Coating | Experimental Context |
|---|---|---|---|
| Protein Adsorption (Non-specific) | Effective reduction [2] | Effective reduction, comparable to PEG [2] | Single protein solutions & diluted plasma [2] |
| Immobilized Protein Density | Baseline | Significantly higher [2] | Immobilization of IgG and BSA on coated surfaces [2] |
| Biomarker Capture Efficiency | Baseline | >2-fold increase in signal [2] | Capture of dengue NS1 biomarker from buffer, plasma, and in vivo [2] |
| Signal-to-Noise Ratio | Baseline | Significantly higher [2] | Detection of dengue NS1 in a preclinical model [2] |
| Colloidal Stability Enhancement | Effective (e.g., improves SAR) [66] | Information Not Covered in Search Results | PEG-coated magnetite nanoparticles [66] |
As shown in Table 1, while both polymers effectively resist non-specific protein adsorption, a key finding is that zwitterionic pSBMA surfaces allow for a significantly higher density of immobilized functional proteins (e.g., capture antibodies) while maintaining low non-fouling backgrounds [2]. This translates directly to enhanced performance in biosensing, as evidenced by the superior capture of the dengue NS1 biomarker, where pSBMA-coated devices yielded a more than two-fold signal increase compared to PEG controls [2].
Molecular Mechanisms and Interfacial Behavior
The superior performance of zwitterionic polymers is rooted in their distinct molecular-level interactions with water and solutes.
Table 2: Underlying Mechanisms and Stability Profile
| Aspect | PEG | Zwitterionic Polymers |
|---|---|---|
| Primary Mechanism | Steric hindrance & formation of a hydration sheath [60] | Formation of a densely bound hydration layer via electrostatic interactions [65] |
| Water Interaction | Hydrogen bonding with ether oxygen [60] | Strong ionic solvation; water molecules are tightly bound to both positive and negative charges [65] |
| Effect on By-product Adsorption | Information Not Covered in Search Results | Reduces irreversible adsorption of hydrophobic by-products [65] |
| Immunogenicity Potential | Can induce anti-PEG antibodies, leading to ABC [64] [62] | Considered to have low immunogenicity; seen as a promising alternative [62] |
Zwitterionic polymers possess equally balanced positive and negative charge groups within their repeating units, resulting in overall charge neutrality [65]. This structure enables a uniquely strong interaction with water molecules. The charged groups create a robust and dense hydration layer through ionic solvation, which forms a more formidable energy barrier against the approach and adsorption of proteins and other fouling organisms compared to the hydrogen-bonded water network associated with PEG [65]. Molecular dynamics simulations indicate that zwitterionic groups significantly reduce the interaction energy between a resin matrix and degraded products, facilitating the stripping of foulants and maintaining a self-cleaning surface [65].
Experimental Protocols for Performance Evaluation
To objectively compare the antifouling performance of PEG and zwitterionic coatings, researchers employ a suite of standardized experiments. Below are detailed protocols for key assays cited in this guide.
In Vitro Antifouling and Biosensor Performance Assay
This protocol is adapted from the study that directly compared PEG and pSBMA-coated microprojection arrays [2].
- Objective: To evaluate the non-specific protein adsorption and specific biomarker capture efficiency of coated surfaces in complex biological fluids.
- Materials:
- Coated Substrates: Polycarbonate surfaces functionalized with linear PEG (e.g., MW ~2k Da) or poly(sulfobetaine methacrylate) (pSBMA) of comparable hydrodynamic radius [2].
- Proteins: Single protein solutions (e.g., IgG, BSA), diluted plasma, target antigen (e.g., dengue NS1 protein).
- Detection Reagents: Fluorescently-labeled or enzyme-linked detection antibodies.
- Methodology:
- Protein Immobilization: Immobilize specific capture proteins (e.g., anti-NS1 IgG) onto the PEG and pSBMA-coated surfaces.
- Non-specific Adsorption Test: Incubate coated surfaces in single protein solutions or diluted plasma. Quantify non-specifically adsorbed protein using spectroscopic or fluorescent methods.
- Specific Capture Test: Incubate the functionalized devices in buffer or plasma spiked with a known concentration of the target biomarker (e.g., NS1).
- Signal Detection: Apply a labeled detection antibody and measure the signal intensity (e.g., fluorescence, colorimetry).
- In Vivo Validation (Preclinical): Apply the wearable coated devices to the skin of infected and naive animal models. After a set period, retrieve the devices and quantify the captured biomarker.
- Key Measurements: Density of immobilized capture antibody, amount of non-specifically adsorbed protein, signal intensity from captured target, and signal-to-noise ratio.
Molecular Dynamics (MD) Simulation of Coating Hydration and Interaction
This protocol is based on simulations used to reveal the mechanism of zwitterionic coatings [65].
- Objective: To model and visualize the atomic-level interactions at the coating-water interface, including hydration layer structure and binding energies.
- Software: Simulation packages like GROMACS or LAMMPS [65].
- Methodology:
- Model Construction: Build atomistic models of the polymer resin with varying degrees of hydrolysis to introduce zwitterionic groups. Create a simulation box containing the resin and water molecules.
- Simulation Run: Perform steered molecular dynamics (SMD) simulations to analyze the swelling behavior of the resin and the adsorption/stripping mechanism of degraded products.
- Data Analysis:
- Calculate the interaction energy between the resin matrix and degraded products.
- Quantify the number and lifetime of hydrogen bonds.
- Compute the potential of mean force (PMF) to determine the energy barrier for product desorption.
- Key Outputs: Interaction energies, hydrogen bond dynamics, PMF energy barriers, and visual representations of the hydration layer and molecular interactions [65].
Visualization of Key Mechanisms
Antibody Response to PEGylated Therapeutics
The following diagram illustrates the two primary pathways by which PEGylated therapeutics trigger the production of anti-PEG antibodies.
Diagram Title: Pathways for Anti-PEG Antibody Production
Molecular Antifouling Mechanism Comparison
This diagram contrasts the distinct molecular-level mechanisms by which PEG and zwitterionic polymers achieve their antifouling effects.
Diagram Title: Molecular Antifouling Mechanisms Compared
The Scientist's Toolkit: Essential Research Reagents and Materials
Table 3: Key Materials for Antifouling Coating Research
| Item | Function in Research | Application Example |
|---|---|---|
| Linear mPEG Derivatives (e.g., mPEG-NHS, mPEG-MAL) | Covalently attaches to amine or thiol groups on surfaces or biomolecules for "grafting to" PEGylation [2]. | Creating PEGylated control surfaces for comparative antifouling studies [2]. |
| Zwitterionic Monomers (e.g., Sulfobetaine methacrylate - SBMA) | Polymerized to form antifouling brushes or hydrogels via "grafting from" or "grafting to" approaches [2] [65]. | Fabricating pSBMA coatings for evaluating hydration and non-specific adsorption [2]. |
| PEGylated Liposomes (e.g., Doxil) | Model nanocarrier system for studying in vivo pharmacokinetics and the Accelerated Blood Clearance (ABC) phenomenon [64]. | Investigating the impact of anti-PEG antibodies on circulation half-life [62]. |
| Anti-PEG ELISA Kits | Detects and quantifies the concentration of anti-PEG IgM and IgG antibodies in serum or plasma [64]. | Assessing immunogenicity of PEGylated formulations in animal models or screening for pre-existing APAs [64]. |
| Model Proteins (e.g., IgG, BSA, Fibrinogen) | Challenging coated surfaces to quantify the level of non-specific protein adsorption in in vitro assays [2]. | Standard fouling tests to benchmark the performance of new antifouling coatings [2]. |
PEG has undeniably revolutionized drug delivery, but its limitations, particularly its propensity to induce immunogenicity, are now significant concerns for the scientific community. The rise of anti-PEG antibodies presents a tangible risk of accelerated clearance and reduced efficacy for a wide range of therapeutics.
As the comparative data shows, zwitterionic polymers like pSBMA represent a powerful and often superior alternative, especially in applications requiring exquisite sensitivity and low fouling in complex biological environments. Their mechanism of action, rooted in the formation of a dense, energetically stable hydration layer, enables not only excellent resistance to non-specific adsorption but also, critically, allows for higher functional density of capture agents—a key advantage for diagnostic and biosensing applications [2] [65].
While PEG will likely remain a useful tool in the biomedical arsenal, the future of advanced antifouling coatings and stealth therapeutics lies in the rational design and adoption of next-generation materials, with zwitterionic polymers leading the way. Overcoming the limitations of PEG is not merely an academic exercise but a necessary step to ensure the safety and efficacy of future biotechnologies.
Strategies for Enhancing the Mechanical Strength of Zwitterionic Hydrogels and Coatings
Zwitterionic polymers, characterized by their balanced cationic and anionic groups, have emerged as premier antifouling materials for biomedical implants due to their strong hydration capabilities through ionic solvation [23] [5]. These polymers exhibit unprecedented biological interface performance, forming tightly bound hydration layers that demonstrate superior resistance to nonspecific protein adsorption, biofilm formation, thrombosis, and foreign body reactions—often surpassing conventional polyethylene glycol (PEG)-based materials [23] [2]. However, the same superhydrophilicity that confers remarkable antifouling properties also severely limits their mechanical performance. The strong interaction between zwitterionic polymer chains and water molecules impedes chain entanglement and weakens intermolecular physical interactions, typically resulting in weak, brittle materials with Young's moduli often under 50 kPa, breaking stress below 100 kPa, and elongation at break rarely exceeding 200% [5]. This mechanical inadequacy poses significant challenges for sustained physiological functionality in load-bearing applications, prompting extensive research into reinforcement strategies that balance mechanical robustness with biofunctionality for next-generation zwitterionic biomedical implants [23] [67].
Comparative Antifouling Performance: PEG vs. Zwitterionic Polymers
While both PEG and zwitterionic polymers serve as effective antifouling materials, their underlying mechanisms and performance characteristics differ significantly. PEG resists fouling through a hydration layer maintained by hydrogen bonding, whereas zwitterionic polymers bind water through stronger ionic solvation, creating a more robust hydration barrier [5]. This fundamental difference translates to distinct performance advantages in biomedical applications.
Table 1: Comparative Antifouling Performance of PEG and Zwitterionic Polymer Coatings
| Performance Characteristic | PEG Coatings | Zwitterionic Coatings | Experimental Context |
|---|---|---|---|
| Protein Adsorption (Thin Coatings ~1 nm) | Higher BSA adsorption [6] | "Much stronger resistance to BSA adsorption" [6] | Surface plasma resonance (SPR) measurement [6] |
| Protein Adsorption (Thick Coatings ~3.6 nm) | "Ultralow fouling" by BSA and fibrinogen [6] | "Ultralow fouling" by BSA and fibrinogen [6] | Surface plasma resonance (SPR) measurement [6] |
| End Group Sensitivity | "Protein adsorption amount could increase by 10-fold" when end group changes from -OH to -COOH [6] | Performance more stable against chemical modifications | Surface plasma resonance (SPR) measurement [6] |
| Specific Biomarker Capture | Lower signal for dengue NS1 capture [2] | "Significantly higher capture efficiency (>2-fold increase in signal)" [2] | Wearable microprojection arrays in dengue-infected mice [2] |
| Macrophage Adhesion | Not specified in results | "Reduced by 30-fold" relative to uncoated surfaces [68] | Cell culture on grafted zwitterionic films [68] |
| Fibroblast Adhesion | Not specified in results | "Reduced nearly 10-fold" relative to uncoated surfaces [68] | Cell culture on grafted zwitterionic films [68] |
| Fibrinogen Adsorption | Not specified in results | "Nearly a 20-fold reduction" across wide cross-link density range [68] | Fluorescently labeled fibrinogen on photografted films [68] |
Comparative studies reveal that very thin zwitterionic coatings (approximately 1 nm) exhibit significantly stronger resistance to bovine serum albumin (BSA) adsorption than equivalent PEG coatings [6]. However, this advantage diminishes with increasing thickness, as PEG coatings leverage stronger steric repulsion at greater dimensions (1.5-3.3 nm), eventually matching zwitterionic performance at approximately 3.6 nm where both demonstrate ultralow fouling against BSA and bovine plasma fibrinogen [6]. Notably, zwitterionic coatings demonstrate significantly higher capture efficiency for specific disease biomarkers like dengue non-structural protein 1 (NS1), showing more than a two-fold increase in signal compared to PEG-coated devices in wearable microprojection arrays applied to dengue-infected mice [2]. This enhanced performance in complex biological environments, combined with greater stability against chemical modification, positions zwitterionic polymers as superior candidates for advanced biomedical applications requiring specific molecular recognition alongside general antifouling properties.
Mechanical Reinforcement Strategies for Zwitterionic Materials
Nanocomposite Reinforcement
Incorporating nanoscale fillers represents one of the most straightforward approaches for enhancing zwitterionic hydrogel mechanical properties. This strategy leverages ionic interactions between zwitterions and nanoparticles to create dense physical crosslinking networks that significantly improve strength and toughness while maintaining antifouling performance.
Table 2: Nanocomposite Reinforcement Strategies for Zwitterionic Hydrogels
| Nanomaterial Type | Mechanical Performance | Key Interactions | Additional functionalities |
|---|---|---|---|
| Laponite XLG Nanosheets [5] | "Stretch up to 1750%" with "breaking strength of 0.27 MPa" [5] | "Ionic interactions between zwitterions and clay platelets" with "inter-chain bonds among zwitterionic groups" [5] | "Flexible adhesion, robust self-healing capability, and high strain sensitivity" [5] |
| Cellulose Nanocrystals (CNCs) [5] | Improved compressive and tensile properties (specific values not provided in results) | Hydrogen bonding and physical entanglement within polymer network | Enhanced biocompatibility and renewability from natural source |
| Exfoliated Clay Nanosheets [23] | "Tensile strength improved to 71.3 kPa from 19.0 kPa" for sulfobetaine polyacrylamide composites [23] | Physical cross-linking through ionic and van der Waals interactions | Maintains high transparency and swelling capacity |
The mechanical properties of nanocomposite zwitterionic hydrogels can be precisely tuned by varying the concentration of both zwitterionic monomers and nanofillers. Increasing the content of sulfobetaine methacrylate (SBMA) and Laponite XLG clay facilitates the formation of a denser network, enhancing energy dissipation and thereby toughening the hydrogel [5]. This tunability enables customization for specific applications, from soft tissue implants requiring low modulus to load-bearing applications demanding higher strength. The resulting materials often exhibit additional functionalities including flexible adhesion, robust self-healing capability, and high strain sensitivity, making them suitable for advanced applications like wearable sensors and electronic skin systems [5].
Microgel Reinforcement Architecture
A particularly innovative approach to mechanical reinforcement involves creating zwitterionic microgel-reinforced pure zwitterionic hydrogels, which maintain excellent antifouling properties by avoiding the introduction of non-zwitterionic components. In this architecture, poly(carboxybetaine) microgels (pCBM) are first prepared via inverse miniemulsion polymerization, then combined with a poly(sulfobetaine) (pSB) continuous phase to form a composite hydrogel (pCBM/pSB) [69].
Figure 1: Microgel-Reinforced Zwitterionic Hydrogel Fabrication Workflow
This hierarchical structure provides multiple mechanical enhancement mechanisms. The pCBM microgels act as multifunctional crosslinking points, with the continuous pSB network transmitting stress throughout the structure [69]. Under deformation, energy dissipates through the destruction and reconstruction of electrostatic interactions between pCB and pSB chains, avoiding stress concentration that typically leads to failure in conventional zwitterionic hydrogels [69]. The cross-linking degree of pCBM significantly influences mechanical performance, with optimal compression strength (1.68 ± 0.15 MPa) achieved at 4 mol% methylenebisacrylamide (MBA) cross-linker concentration [69]. This represents a substantial improvement over pure pSB hydrogels (0.23 ± 0.05 MPa), demonstrating the effectiveness of microgel reinforcement for creating robust zwitterionic materials capable of withstanding physiological stresses.
Cross-Linking Density Optimization
Strategic control of cross-linking density represents a fundamental approach for balancing the mechanical and antifouling properties of zwitterionic materials. Cross-link density directly modulates key mechanical properties including swelling behavior, stress-strain response, and lubricity, while theoretically potentially compromising antifouling performance by reducing chain mobility and hydration capacity [68].
Table 3: Effect of Cross-Link Density on Zwitterionic Hydrogel Properties
| Property | Low Cross-Link Density | High Cross-Link Density | Optimal Range |
|---|---|---|---|
| Protein Adsorption [68] | Increased fibrinogen adsorption at very low densities | Increased fibrinogen adsorption at very high densities | Broad range (5-50% cross-linker) maintains low fouling |
| Cell Adhesion [68] | Low macrophage and fibroblast adhesion | "Increased degrees of cell adhesion" above 50% cross-linker | Below 50% cross-linker concentration |
| Swelling Ratio [68] | Higher swelling | "Reduced swelling" with increased cross-link density | Application-dependent |
| Compressive Modulus [68] | Lower modulus | "Greater compressive modulus" with increased cross-link density | Application-dependent |
| Lubricity [68] | Maintains low friction | Maintains low friction across broad density range | Broad effective range |
Research demonstrates that zwitterionic thin films maintain nearly a 20-fold reduction in fibrinogen adsorption compared to uncoated polydimethylsiloxane (PDMS) across a wide range of cross-link densities [68]. Similarly, macrophage and fibroblast adhesion remain significantly reduced (30-fold and 10-fold, respectively) across broad cross-linking ranges [68]. However, this antifouling performance can be compromised at extreme cross-linking levels, with increased protein adsorption observed at both very high and very low cross-link densities, and increased cell adhesion noted when cross-linker concentration exceeds 50% [68]. This creates a well-defined optimization window where mechanical properties can be significantly enhanced without substantially compromising the antifouling capabilities that make zwitterionic materials valuable for biomedical applications.
Experimental Protocols for Mechanical Enhancement
Microgel-Reinforced Hydrogel Fabrication Protocol
The fabrication of microgel-reinforced zwitterionic hydrogels follows a multi-step process that ensures proper integration of the microgel reinforcement phase with the continuous hydrogel matrix [69]:
Poly(carboxybetaine) Microgel (pCBM) Synthesis: Prepare pCBM via inverse miniemulsion polymerization with carboxybetaine methacrylate (CBMA) monomer and methylenebisacrylamide (MBA) cross-linker (optimal concentration: 4 mol% relative to CBMA). Purify the resulting microgels to remove unreacted components.
Pre-gel Solution Preparation: Dry the synthesized pCBM microgels (achieving diameter of 641 ± 65 nm), then disperse and swell them in SBMA monomer solution to obtain a paste-like consistency. The high specific surface area and superhydrophilic nature of pCBM enable rapid absorption of the SBMA monomer solution.
Hydrogel Polymerization: Initiate polymerization of the SBMA monomer within the pCBM-containing pre-gel solution. During this process, the forming pSB chains pass through the pCBM microgels, creating a uniform network structure with a two-phase architecture.
Characterization and Validation: Assess mechanical properties through compression testing (optimal pCBM20/pSB hydrogel demonstrates compressive strength of 1.03 ± 0.11 MPa) and evaluate antifouling performance through protein adsorption and cell adhesion assays.
This protocol produces hydrogels with the continuous pSB phase restricting deformation through "locking" the entire structure via strong inter- and intramolecular electrostatic interactions, while the dispersed pCBM microgel phase acts as both cross-linking sites and energy dissipation centers [69].
Cross-Link Density Optimization Protocol
For zwitterionic thin films grafted onto biomaterial substrates such as polydimethylsiloxane (PDMS), the following protocol enables systematic optimization of cross-linking density [68]:
Substrate Functionalization: Soak PDMS substrates in a 50 g/L solution of benzophenone in acetone for 1 hour to enable subsequent surface photografting. Remove substrates and evaporate residual solution using a nitrogen gas stream, then place under vacuum for 1 hour to ensure complete acetone removal.
Monomer Solution Preparation: Prepare solutions with total monomer concentration maintained at 35 wt% in deionized water. Vary the proportion of poly(ethylene glycol) dimethacrylate (PEGDMA) cross-linker to sulfobetaine methacrylate (SBMA) or carboxybetaine methacrylate (CBMA) from 0 to 100% of total monomer. Add 0.05 wt% Irgacure 2959 photoinitiator to each solution.
Photografting Process: Pipette 20 μL of monomer solution onto functionalized PDMS substrates and disperse via capillary action using a coverslip. Expose to 30 mW/cm² of 365 nm UV light for polymerization and grafting.
Performance Evaluation:
- Protein Adsorption: Apply fluorescently labeled fibrinogen solution (1.0 mg/mL) to films for 1 hour, rinse, and quantify fluorescence via epifluorescence microscopy.
- Cell Adhesion: Culture macrophages and fibroblasts on coated surfaces and quantify adhesion counts.
- Mechanical Testing: Evaluate swelling ratio, compressive modulus, and lubricity across the cross-linking density spectrum.
This systematic approach identifies the optimal cross-linking window (typically below 50% cross-linker concentration) that maintains excellent antifouling performance while achieving significant mechanical enhancement [68].
The Scientist's Toolkit: Essential Research Reagents
Table 4: Essential Reagents for Zwitterionic Hydrogel Research
| Reagent/Category | Specific Examples | Function in Research | Key Considerations |
|---|---|---|---|
| Zwitterionic Monomers [68] [5] | Sulfobetaine methacrylate (SBMA), Carboxybetaine methacrylate (CBMA) | Primary building blocks for hydrogel network | Choice affects antifouling performance and mechanical properties |
| Cross-linkers [68] [69] | Poly(ethylene glycol) dimethacrylate (PEGDMA), Methylenebisacrylamide (MBA) | Control network density and mechanical properties | Concentration significantly impacts balance between strength and antifouling |
| Nanocomposite Fillers [5] | Laponite XLG nanosheets, Cellulose nanocrystals (CNCs) | Enhance mechanical properties through reinforcement | Provide additional functionality (self-healing, adhesion) |
| Photoinitiators [68] | Irgacure 2959 | Enable photopolymerization for patterning and grafting | Biocompatibility and solubility important for biomedical applications |
| Substrate Primers [68] | Benzophenone | Enable surface grafting on various substrates | Universal adhesion important for device coatings |
| Characterization Reagents [68] | Fluorescently labeled fibrinogen, Cell staining dyes | Evaluate antifouling performance and biocompatibility | Enable quantification of protein adsorption and cell adhesion |
The field of zwitterionic biomaterials continues to evolve with emerging strategies including mechanically trained anisotropic hydrogels that leverage asymmetric response of zwitterionic chains to external stimuli [70], porous hierarchically ordered hydrogels that mimic natural tissue structure through controlled microarchitecture [71], and multi-network systems that combine various reinforcement mechanisms for synergistic property enhancement [5]. While significant progress has been made in enhancing the mechanical properties of zwitterionic hydrogels and coatings, challenges remain in standardizing long-term in vivo evaluations, optimizing large-scale manufacturing processes, and further improving the balance between mechanical robustness, antifouling performance, and additional functionalities like drug elution or electrical conductivity [23] [72]. As research advances, zwitterionic materials with enhanced mechanical properties are poised to enable transformative applications in regenerative medicine, implantable devices, biosensing, and drug delivery systems that require the unique combination of superior antifouling performance and mechanical durability.
The prevention of biofouling—the unwanted adhesion of proteins, cells, and microorganisms to surfaces—is a critical challenge across biomedical and marine applications. Biofilm formation on medical implants, biosensors, and drug delivery carriers can lead to device failure, infections, and compromised functionality [42] [23]. Among the most extensively studied antifouling materials are poly(ethylene glycol) (PEG) and zwitterionic polymers, both creating a hydration layer that forms a physical and energetic barrier against fouling agents [42]. However, the performance of these coatings is not merely a function of their chemical composition; it is profoundly influenced by physical parameters including graft density, chain length, and overall thickness. Optimization of these parameters is essential for achieving ultralow fouling surfaces, as they directly impact the steric repulsion, hydration capacity, and mechanical stability of the polymer brush layer [6] [73]. This guide objectively compares the antifouling performance of PEG and zwitterionic coatings by examining experimental data that delineates how these critical parameters govern their efficacy.
Comparative Performance Data: PEG vs. Zwitterionic Coatings
The antifouling performance of PEG and zwitterionic coatings is highly dependent on their physical parameters. The following table summarizes key experimental findings from comparative studies, highlighting the role of thickness, chain length, and graft density.
Table 1: Comparative Antifouling Performance of PEG and Zwitterionic Coatings
| Coating Type | Optimal Thickness/Chain Length | Graft Density Control | Tested Fouling Agents | Key Performance Findings | Research Source |
|---|---|---|---|---|---|
| Phosphorylcholine Zwitterion (PMEN) | ~1 nm | Via polydopamine immobilization & ATRP time | Bovine Serum Albumin (BSA), blood plasma fibrinogen, platelets, bacteria | Superior BSA resistance at ~1 nm thickness compared to PEG; ultralow fouling with BSA/Fg at ~3.6 nm [6]. | Cheng-Mei Xing et al., Acta Biomaterialia [6] |
| PEG (PEG-OH) | 1.5 - 3.3 nm | Via polydopamine immobilization | Bovine Serum Albumin (BSA), blood plasma fibrinogen, platelets, bacteria | Performance exceeds PMEN at 1.5-3.3 nm due to stronger steric repulsion; ultralow fouling at ~3.6 nm. End-group (-OH vs. -COOH) critically affects performance [6]. | Cheng-Mei Xing et al., Acta Biomaterialia [6] |
| Sulfobetaine Methacrylate (pSBMA) | Polymer brush length tuned by ATRP time (1-24 h) | Aminolysis pre-treatment time (15 min - 3.5 h) on PET fabrics | Protein adhesion | Polymerization time is a major factor in preventing protein adhesion; longer times yield denser, more robust brushes [73]. | PMC, Polymers Journal [73] |
| PEG (Various) | N/A | Spin-coating, immersion, pulsed electrodeposition | E. coli, S. aureus, P. aeruginosa | Demonstrates up to 99% reduction in bacterial adhesion, often used as a control standard in fouling experiments [42]. | Multiple Authors [42] |
Detailed Experimental Protocols and Methodologies
Quantitative Coating Fabrication and Optimization via SPR
A foundational protocol for the quantitative fabrication and optimization of PEG and zwitterionic coatings leverages a Surface Plasma Resonance (SPR) instrument for in-situ monitoring [6].
- Substrate Preparation: SPR sensor chips (often gold-coated) are first pre-coated with a polydopamine (PDA) intermediate layer. This is achieved by immersing the chips in an aqueous dopamine solution (e.g., 2 mg/mL in Tris-HCl buffer, pH 8.5) for several hours. The PDA layer acts as a universal, adherent platform rich in functional groups for subsequent chemical grafting [6].
- Polymer Immobilization: The PDA-coated chips are then incubated in aqueous solutions of the active polymers. For zwitterionic coatings, a random copolymer bearing phosphorylcholine zwitterion and active ester side chains (PMEN) is used. For PEG coatings, carboxylic PEG derivatives are utilized. The active esters in PMEN and the carboxylic groups in PEG react with the amine groups of the PDA layer via amidation coupling, covalently anchoring the polymers [6].
- Performance Optimization: The SPR system is used to monitor the polymer grafting process in real-time, allowing for precise control over the coating thickness by varying solution concentration or incubation time. Following deposition, the same SPR setup is used to quantify the adsorption of model proteins like Bovine Serum Albumin (BSA) or fibrinogen onto the modified surfaces, directly correlating coating parameters with antifouling efficacy [6].
"Grafting From" Zwitterionic Brushes via ATRP
For high-density polymer brush coatings, surface-initiated Atom Transfer Radical Polymerization (ATRP) is a preferred "grafting from" technique, as demonstrated on polyester (PET) fabrics [73].
- Surface Pre-functionalization: Inert PET substrates undergo aminolysis to generate surface amine groups. This involves treating the PET with a 40% v/v solution of ethylenediamine in ethanol at 55°C for a duration between 15 minutes and 3.5 hours. Critically, this aminolysis time directly controls the density of the resulting surface amine groups, which in turn dictates the final graft density of the polymer brush [73].
- Initiator Immobilization: The amine-functionalized samples (PETEDA) are reacted with 2-bromoisobutyryl bromide (BIBB) in tetrahydrofuran (THF) with triethylamine (TEA) as a base. This step installs bromine-containing ATRP initiator groups (PETBr) onto the surface [73].
- Polymer Brush Growth: The initiator-functionalized substrates are placed in a Schlenk flask containing a degassed mixture of the zwitterionic monomer (e.g., sulfobetaine methacrylate - SBMA), a copper(I) bromide catalyst, and a bipyridyl ligand in a methanol-water solvent. The polymerization proceeds at room temperature for a set time, ranging from 1 to 24 hours. The polymerization time is a key factor controlling the chain length and thickness of the resulting zwitterionic polymer brush [73].
Antifouling Performance Assays
The efficacy of optimized coatings is validated through a suite of biological assays.
- Protein Adsorption Tests: Coated surfaces are exposed to protein solutions such as BSA or blood plasma fibrinogen (Fg). The amount of adsorbed protein is quantified using label-free techniques like SPR [6] or indirectly through methods like SDS-PAGE and LC-MS/MS [74].
- Bacterial Adhesion Assays: Coatings are challenged with common pathogenic bacteria, including E. coli (Gram-negative) and S. aureus (Gram-positive). After incubation for periods ranging from 2 hours to 7 days, adhered bacteria are counted via fluorescence microscopy or plate counting to determine the percentage reduction compared to a control surface [6] [42].
- Complex Biofluid Exposure: For biomedical applications, coatings are tested in complex media like undiluted blood plasma or serum to assess performance under clinically relevant conditions [75].
Visualizing Coating Fabrication and Structure-Performance Relationships
The following diagrams illustrate the core experimental workflows and the relationship between coating parameters and antifouling performance.
Polymer Brush Fabrication via ATRP
Coating Parameter Impact on Performance
The Scientist's Toolkit: Essential Research Reagents and Materials
The experimental protocols rely on a set of core materials and reagents, each with a specific function in creating and analyzing optimized antifouling coatings.
Table 2: Essential Reagents for Antifouling Coating Research
| Reagent/Material | Function in Research | Key Consideration |
|---|---|---|
| Sulfobetaine Methacrylate (SBMA) | A zwitterionic monomer for creating ultra-low fouling polymer brushes via ATRP [73]. | Forms brushes with strong hydration via ionic solvation; exhibits Upper Critical Solution Temperature (UCST) behavior [73]. |
| Poly(ethylene glycol) (PEG) | The benchmark polymer for antifouling comparisons; often functionalized with -OH or -COOH end groups [6] [42]. | Performance is highly dependent on end-group chemistry and chain length; susceptible to oxidative degradation [6] [23]. |
| Polydopamine (PDA) | A universal adhesive primer layer that facilitates the coating of diverse substrates [6]. | Provides a versatile platform for immobilizing polymers via its amine-reactive surface; coating time controls layer thickness and group density [6]. |
| ATRP Initiator (e.g., BIBB) | Installs bromine-containing groups on a pre-functionalized surface to initiate polymer brush growth [73]. | Essential for the "grafting from" approach; density is controlled by the preceding pre-functionalization step (e.g., aminolysis time) [73]. |
| Aminolysis Agent (e.g., Ethylenediamine) | Introduces amine functional groups onto inert polymer surfaces like PET to enable further chemistry [73]. | Reaction time is a critical parameter to tune the density of surface amines, which directly dictates the final graft density of the polymer brush [73]. |
The optimization of graft density, chain length, and thickness is paramount to unlocking the full potential of both PEG and zwitterionic antifouling coatings. While PEG relies more on chain length and thickness for steric repulsion, zwitterionic polymers demonstrate exceptional performance at very low thicknesses due to their intense ionic hydration [6]. The choice between these materials is therefore application-dependent. A significant challenge for zwitterionic polymers lies in their often-inadequate mechanical strength for long-term implants, driving research into strategies like crosslinking, nanocomposite reinforcement, and copolymer design to enhance durability without sacrificing antifouling properties [23]. Future progress will hinge on the development of standardized in-vivo testing protocols and the creation of multifunctional, smart coatings that integrate robust antifouling with enhanced mechanical performance for real-world biomedical and industrial applications.
Ensuring Long-Term Stability in Complex Biological Fluids and Under Flow Shear Stress
For researchers and scientists developing medical devices, drug delivery systems, or implantable sensors, achieving long-term stability of antifouling coatings represents a significant translational challenge. These surfaces must resist not only the initial adsorption of proteins, cells, and microorganisms but also maintain this antifouling performance under the dynamic conditions of physiological environments. Complex biological fluids introduce a complex mixture of proteins, lipids, and electrolytes, while flow shear stress continuously challenges the integrity of surface modifications. For decades, polyethylene glycol (PEG) has been the benchmark antifouling polymer. However, a new generation of zwitterionic materials is emerging, demonstrating superior stability and performance in demanding biological applications. This guide provides an objective, data-driven comparison of these competing technologies, focusing on their long-term stability for research and development professionals.
Antifouling Coating Performance: A Quantitative Comparison
The efficacy of an antifouling coating is measured by its ability to reduce the adhesion of biological material. The following table summarizes key performance metrics for PEG and zwitterionic coatings based on recent experimental studies.
Table 1: Quantitative Comparison of Antifouling Coating Performance
| Coating Type | Test Organism/Condition | Performance Metric | Reduction vs. Control | Key Findings | Citation |
|---|---|---|---|---|---|
| PEG (Polyethylene Glycol) | Platelet adhesion under physiological flow | Platelet Adhesion | 69% reduction | Effective, but outperformed by zwitterionic coatings. | [76] |
| Zwitterionic SBSi (Sulfobetaine Silane) | Platelet adhesion under physiological flow | Platelet Adhesion | 96% reduction | Superior resistance to platelet adhesion, indicating high blood compatibility. | [76] |
| Zwitterionic PEG-SBSi (Hybrid) | Platelet adhesion under physiological flow | Platelet Adhesion | 80% reduction | Performance between pure PEG and SBSi. | [76] |
| PEG | E. coli, S. aureus, P. aeruginosa | Bacteria Adhesion | ~99% suppression (1-day); ~99% reduction (7-day) | Excellent initial performance, though stability can be compromised over time in complex media. | [42] |
| Zwitterionic Polymers | Various bacterial species | Bacteria Adhesion | Up to 99% reduction overall | Consistently high performance; noted as the most promising for preventing bacterial adhesion. | [42] |
Beyond adhesion metrics, the long-term stability of the coating itself is paramount. The following table compares the characteristics of both classes of materials under different environmental stressors.
Table 2: Comparison of Long-Term Stability and Material Properties
| Parameter | PEG-Based Coatings | Zwitterionic Coatings |
|---|---|---|
| Primary Hydration Mechanism | Hydrogen bonding [77] | Ionic solvation (stronger electrostatic interaction) [77] |
| Hydration Capacity | ~1 water molecule per ethylene glycol (EG) unit [77] | ~7-8 water molecules per sulfobetaine (SB) unit [77] |
| Stability under Ambient Storage | Maintains hydrophilicity (contact angle <20° for 35 days) [76] | Maintains hydrophilicity (contact angle <20° for 35 days) [76] |
| Stability under Static Fluid | Good performance, but may degrade over time [76] [1] | Increased stability and hydrophilicity after 7 days [76] |
| Stability under Flow Shear Stress | Effective but can be susceptible to oxidation [77] | High stability; retains hydrophilicity (contact angle ≤40°) after 7 days under flow [76] |
| Mechanical Strength | Generally good | Inherently weak; requires reinforcement strategies (e.g., cross-linking, nanofillers) [23] |
| Immunogenicity | Can trigger anti-PEG antibodies, accelerating blood clearance [77] [1] | Minimal immunogenicity reported [77] |
Experimental Protocols for Evaluating Stability and Performance
To ensure the reliability and reproducibility of stability data, researchers employ standardized experimental protocols. Below are detailed methodologies for key tests cited in this guide.
Protocol: Stability and Antifouling under Flow Shear Stress
This protocol is adapted from studies evaluating coatings for blood-contacting devices [76].
- 1. Coating Application: PDMS microfluidic flow chambers are used as substrates. Coatings (PEG, SBSi, PEG-SBSi) are grafted via a sequential flow chemistry process at room temperature.
- 2. Dynamic Flow Conditioning: Coated chambers are subjected to physiologically relevant flow conditions using a peristaltic or syringe pump. A typical experiment involves circulating phosphate-buffered saline (PBS) or blood plasma for 7 days at various shear rates (e.g., 0-1000 s⁻¹).
- 3. Contact Angle Measurement: Surface hydrophilicity is measured before and after flow conditioning using a contact angle goniometer. A low contact angle (<40°) indicates maintained hydrophilicity and coating stability.
- 4. Platelet Adhesion Test: After flow conditioning, the chambers are perfused with platelet-rich plasma under physiological shear stress. The surfaces are then imaged (e.g., via fluorescence or scanning electron microscopy), and adhered platelets are counted. The percentage reduction in adhesion is calculated versus an uncoated PDMS control.
Protocol: Single Thread Adhesion Test (STAT)
This quantitative method is used to measure the adhesion strength of macrofoulers like mussels, providing high resolution between high-performance coatings [78].
- 1. Coating Preparation: Standard and high-performance coatings are applied to relevant substrates (e.g., commercial foul-release coatings).
- 2. Mussel Placement: Mussels are placed on the coated surfaces and allowed to attach, forming byssal threads and adhesive plaques.
- 3. Tensile Measurement: Individual byssal threads are carefully detached from the surface using a microtensile tester, which measures the force required for detachment.
- 4. Data Analysis: The tensile adhesion force for each thread is recorded. This method provides finer resolution than shear-based tests, allowing for meaningful comparisons between coatings that exhibit very low adhesion.
Protocol: Bacterial Adhesion Resistance
This is a common assay for evaluating antifouling performance against microbial biofilms [42].
- 1. Substrate Preparation: Coatings are applied to substrates like glass, silicon wafers, or medical-grade metals (e.g., titanium).
- 2. Bacterial Culture and Exposure: Model bacterial species (e.g., E. coli, S. aureus, P. aeruginosa) are grown in a suitable medium. Coated substrates are immersed in the bacterial suspension for a set period (30 minutes to 7 days) under static or gentle agitation conditions.
- 3. Sample Analysis: After incubation, substrates are gently rinsed to remove non-adhered bacteria. Adhered bacteria are quantified via:
- Viable Cell Count: Bacteria are dislodged by sonication or scraping, serially diluted, and plated on agar to count Colony Forming Units (CFU).
- Microscopy: Surfaces are stained (e.g., with DAPI or LIVE/DEAD stain) and imaged under fluorescence or confocal microscopy to count and visualize adhered cells.
- 4. Efficacy Calculation: The reduction in adhered bacteria is calculated as a percentage compared to an uncoated control.
Mechanisms of Action and Performance Workflow
The fundamental difference in performance and stability between PEG and zwitterionic coatings stems from their distinct mechanisms of interaction with water, which forms the basis of their antifouling properties.
The Scientist's Toolkit: Essential Research Reagents and Materials
Successfully developing and testing long-term stable antifouling coatings requires a specific set of reagents and instruments. The following table details key solutions and materials used in the featured experiments.
Table 3: Key Research Reagents and Materials for Antifouling Coating Development
| Item Name | Function/Application | Brief Explanation |
|---|---|---|
| PDMS (Polydimethylsiloxane) | Substrate for microfluidic devices | An elastomeric polymer used to create flow chambers for simulating physiological shear stress. [76] |
| Sulfobetaine Silane (SBSi) | Zwitterionic coating precursor | A molecule used to graft zwitterionic surfaces onto substrates, providing excellent antifouling and stability. [76] |
| Atom Transfer Radical Polymerization (ATRP) Initiator | Surface initiation for polymer brushes | A technique (often using bromine-terminated silanes or thiols) to create a tightly anchored layer for growing polymer brushes like PEG or zwitterions. [42] |
| Microfluidic Flow System | Applying dynamic shear stress | A setup (pumps, tubing, chambers) to subject coated surfaces to controlled, physiologically relevant flow conditions. [76] |
| Contact Angle Goniometer | Measuring surface wettability | An instrument that measures the contact angle of a water droplet on a surface, indicating its hydrophilicity and coating stability. [76] |
| Mussel Single Thread Adhesion Test (STAT) | Quantifying macrofouler adhesion | A specialized tensile test that measures the force required to detach an individual mussel byssal thread, offering high resolution for low-adhesion coatings. [78] |
The experimental data clearly indicates that while PEG remains a competent antifouling material, zwitterionic coatings offer a compelling advantage for applications requiring long-term stability in complex biological fluids and under flow shear stress. Their superior performance is rooted in a stronger, more robust hydration mechanism via ionic solvation. This translates to significantly reduced platelet adhesion and exceptional retention of hydrophilicity under dynamic flow.
However, a key challenge for zwitterionic materials is their inherent mechanical strength. Future research is focused on developing reinforced zwitterionic hydrogels, copolymer systems, and nanofiller composites to enhance durability without compromising antifouling performance [23]. Furthermore, standardized long-term in vivo evaluations are needed to fully bridge the gap between laboratory research and clinical translation. For researchers and drug development professionals, zwitterionic polymers represent a promising frontier in developing more reliable, longer-lasting medical devices, implants, and drug delivery systems.
In the fields of drug delivery and biomedical implants, creating surfaces that can effectively resist the nonspecific adsorption of proteins, cells, and other biological foulants is a fundamental challenge. For decades, poly(ethylene glycol) (PEG) has been the gold standard for antifouling coatings, prized for its ability to bind water via hydrogen bonding and create a steric barrier that minimizes unwanted interactions. [79] [10] However, growing concerns about PEG's immunogenicity and chemical instability have spurred the search for alternatives. [8] [79] [10] Zwitterionic polymers, which contain pairs of oppositely charged groups, have emerged as a powerful candidate. They bind water more tightly than PEG through ionic solvation, leading to exceptional antifouling performance. [23] [5] [10] Despite their superior bio-inertness, zwitterionic materials often suffer from inherently weak mechanical strength, limiting their use in load-bearing applications such as implants. [23] [5]
To overcome these limitations, researchers are developing advanced hybrid and copolymer approaches. These strategies aim to merge the unparalleled antifouling capacity of zwitterions with the proven performance of PEG or the robust mechanical properties of elastomers and other reinforcing agents. This guide provides a objective comparison of these emerging strategies, detailing their performance, experimental protocols, and applications for researchers and drug development professionals.
Comparative Performance of Polymer Modifiers
The following tables summarize key experimental data comparing the properties of PEG, zwitterionic polymers, and their hybrids.
Table 1: Comparison of Key Properties and Performance Metrics
| Polymer Type | Hydration Mechanism | Anti-PEG Antibody Reactivity | Key Mechanical Properties | Primary Limitations |
|---|---|---|---|---|
| PEG | Hydrogen bonding (~3 H₂O per EO unit) [79] | High (reference level) [8] | Dependent on molecular weight and density; "mushroom" vs. "brush" conformation [79] | Immunogenicity; oxidative degradation; induces anti-PEG antibodies [79] [10] |
| Zwitterionic Polymers (e.g., PMPC) | Ionic solvation (~7-8 H₂O per repeating unit) [5] | Minimal (significantly lower than PEG and PTEGMA) [8] | Generally weak and brittle in pure hydrogels (e.g., Young's modulus < 50 kPa) [5] | Poor mechanical strength; high swelling tendency [23] [5] |
| PEG-like Brush Polymer (PTEGMA) | Hydrogen bonding [8] | Moderate (less than PEG, but non-negligible) [8] | Can be tuned via polymerization; potentially superior to PEG [8] | Retains some anti-PEG antibody reactivity due to ethylene glycol units [8] |
Table 2: Experimental In Vivo and Functional Performance Data
| Polymer Modifier | Model System | Key Functional Outcome | Reference Experiment |
|---|---|---|---|
| PMPC206 | DNA aptamer (TXB0063) targeting IFN-γ [8] | No impairment of aptamer's neutralizing activity [8] | In vitro binding assay |
| PMPC206 | C57BL/6 mice [8] | Extended blood circulation time (comparable or longer than similar-sized PEG) [8] | Pharmacokinetic study |
| PMPC206 | LPS-induced endotoxic shock mouse model [8] | Significantly superior survival rate vs. unmodified aptamer [8] | In vivo therapeutic efficacy study |
| Fiber-Reinforced Zwitterionic Elastomer | Rat subcutaneous implantation [80] | Remarkable anti-inflammatory and anti-calcification effects [80] | 60-day in vivo biocompatibility test |
| Zwitterionic Nanocomposite Hydrogel | Material testing [5] | Breaking strength: 0.27 MPa; Elongation at break: 1750% [5] | Mechanical stress-strain measurement |
Detailed Experimental Protocols and Methodologies
Assessing Immunogenicity: Anti-PEG Antibody Reactivity Assay
A critical test for any PEG alternative is its ability to evade the immune system, specifically pre-existing anti-PEG antibodies. [8]
- Step 1: Polymer Coating. The polymers to be tested (e.g., PMPC, PTEGMA, PEG) are dissolved in phosphate-buffered saline (PBS) at a defined concentration (e.g., 20 µg mL⁻¹) and used to coat the wells of a microtiter plate.
- Step 2: Antibody Incubation. A solution containing horseradish peroxidase (HRP)-conjugated anti-PEG antibody (e.g., clone 1D9-6) is added to the polymer-coated wells.
- Step 3: Detection. The binding of the antibody is quantified using a colorimetric substrate like 3,3',5,5'-tetramethylbenzidine (TMB). The resulting signal intensity is directly proportional to the level of antibody binding to the polymer. [8]
This protocol clearly demonstrated that PMPC had significantly lower reactivity against anti-PEG antibodies than both PEG and the PEG-alternative PTEGMA. [8]
Enhancing Mechanical Strength: Fabrication of Fiber-Reinforced Zwitterionic Elastomers
For implant applications like heart valves, mechanical robustness is as important as bio-inertness. A representative protocol involves creating a composite material.
- Step 1: Electrospun Fiber Fabrication. A solution of a durable polymer (e.g., poly(ether-ester-urethane) urea) is electrospun to create a mat of orthogonally aligned microfibers. This mat acts as the mechanical scaffold.
- Step 2: Zwitterionic Elastomer Infiltration. The fibrous scaffold is immersed in a mixture containing a urethane-based diol, a zwitterionic diol monomer, and a disocyanate crosslinker.
- Step 3: In Situ Polymerization and Curing. The infiltrated scaffold is subjected to a thermal curing cycle (e.g., 80°C for 12 hours) to form a crosslinked zwitterionic poly(urethane-urea) elastomer within the fiber network. This process integrates the antifouling surface in situ with the mechanical reinforcement. [80]
- Step 4: Validation Testing. The resulting composite is subjected to accelerated fatigue testing (e.g., 100 million cycles) and long-term immersion in serum (e.g., 60 days) to validate its mechanical and chemical integrity. [80]
In Vivo Therapeutic Efficacy Model
To validate the performance of a zwitterionic-modified therapeutic, an animal model of disease is used.
- Model: LPS-induced inflammation/endotoxic shock in mice.
- Intervention: Treatment with a therapeutic aptamer (e.g., IFN-γ targeting TXB0063) conjugated to PMPC.
- Control: Treatment with the unmodified aptamer.
- Primary Outcome Measure: Survival rate of the animals over time. Studies have shown a statistically significant superior survival rate for the PMPC-aptamer conjugate group compared to the unmodified aptamer group. [8]
Schematic Workflows and Structural Relationships
Creating a Fiber-Reinforced Zwitterionic Composite
The following diagram illustrates the fabrication process for a mechanically robust, antifouling zwitterionic elastomer composite.
Diagram 1: Fabrication of a fiber-reinforced zwitterionic composite. This process integrates a strong microfiber scaffold with a zwitterionic elastomer matrix to achieve both mechanical robustness and antifouling properties. [80]
Structure-Property Relationships in Hybrid Hydrogels
The mechanical and antifouling properties of hybrid materials are dictated by their internal molecular interactions.
Diagram 2: Structure-property relationships in hybrid hydrogels. This shows how different reinforcement strategies contribute to improved mechanical performance at a molecular level. [23] [5]
The Scientist's Toolkit: Essential Research Reagents
Table 3: Key Research Reagents for Hybrid Zwitterionic Material Development
| Reagent / Material | Function / Role | Example Use Case |
|---|---|---|
| Zwitterionic Monomers (SBMA, CBMA, MPC) | The core building block providing antifouling properties via ionic solvation. [23] [10] | Synthesizing zwitterionic polymer brushes, hydrogels, or copolymer blocks for drug conjugation. [8] |
| Electrospun Polymer Microfibers (e.g., Poly(ether-ester-urethane) urea) | Provides a mechanically robust, anisotropic scaffold to reinforce soft zwitterionic matrices. [80] | Creating composite materials for load-bearing implants like heart valves. [80] |
| Nanofillers (Laponite XLG, Cellulose Nanocrystals - CNC) | Acts as a physical crosslinker and reinforcement agent to dissipate energy and enhance toughness. [5] | Forming tough zwitterionic nanocomposite hydrogels for wearable sensors or tissue engineering. [5] |
| Atom Transfer Radical Polymerization (ATRP) Initiator | A controlled radical polymerization technique to synthesize well-defined polymers with target molecular weights. [8] | Preparing PMPC or PTEGMA modifiers with a specific degree of polymerization and terminal amino groups for bioconjugation. [8] |
| Anti-PEG Antibodies (e.g., 1D9-6 clone) | A critical bioreagent for assessing the immunogenic potential and safety profile of PEG-alternative polymers. [8] | In vitro ELISA-style assays to quantify polymer reactivity and potential for immune recognition. [8] |
Hybrid and copolymer approaches represent a sophisticated materials engineering strategy that moves beyond the limitations of single-component systems. While PEG continues to be useful, its immunogenicity risk is a significant drawback. Pure zwitterionic materials, though highly bio-inert, often lack the mechanical integrity required for many clinical applications. The data shows that zwitterionic polymers like PMPC offer a compelling alternative to PEG, demonstrating minimal immunogenicity and excellent in vivo performance in therapeutic aptamer delivery. [8]
The future of antifouling coatings lies in smart combinations. Integrating zwitterions into fiber-reinforced elastomers or nanocomposite hydrogels successfully bridges the gap between superior antifouling and mechanical resilience, as evidenced by prototypes that withstand millions of fatigue cycles. [80] For researchers, the path forward involves continued innovation in material design—such as developing stimuli-responsive zwitterionic copolymers—and a stronger emphasis on standardized long-term in vivo evaluations to ensure these promising laboratory innovations can be reliably translated into clinical products. [23]
Performance Validation: Direct Comparison and Selection Criteria
The nonspecific adsorption of proteins and subsequent adhesion of bacteria and formation of biofilms on material surfaces represent a fundamental challenge across biomedical applications, from implantable medical devices and biosensors to drug delivery systems [81] [82] [9]. The initial layer of adsorbed protein dramatically alters the interface, potentially compromising device functionality, reducing sensor sensitivity, and increasing the risk of infection and thrombosis [9]. For decades, poly(ethylene glycol) (PEG) and its derivatives have served as the gold standard for creating antifouling surfaces, resisting biofouling through steric repulsion and the formation of a hydration layer [83] [84]. However, PEG's susceptibility to oxidative degradation and its tendency to elicit immune responses have prompted the search for more robust alternatives [83] [5] [84].
Zwitterionic polymers, bearing both positive and negative charges within the same molecular unit, have emerged as a leading next-generation platform. These materials, including poly(carboxybetaine) (PCB), poly(sulfobetaine) (PSB), and poly(2-methacryloyloxyethyl phosphorylcholine) (PMPC), resist fouling by forming a dense and tightly bound hydration layer via ionic solvation [9] [5]. This guide provides a quantitative, data-driven comparison of the antifouling performance of PEG and zwitterionic coatings, focusing on the critical metrics of protein adsorption (ng/cm²) and bacterial adhesion reduction (%).
Quantitative Performance Data
Protein Adsorption Resistance
Protein adsorption is the primary event leading to the failure of biomaterials. The following table summarizes quantitative data on the protein adsorption resistance of various coatings.
Table 1: Quantitative protein adsorption data for antifouling coatings.
| Coating Type | Specific Material | Protein / Biofluid Tested | Adsorption (ng/cm²) | Reference / Control |
|---|---|---|---|---|
| Zwitterionic Polymer Brush | pCBMA, pSBMA | Fibrinogen | < 0.3 ng/cm² [83] | Not Specified |
| Zwitterionic Peptide on PSi | EKEKEKEKEKGGC | Gastrointestinal Fluid | ~80% reduction vs. PEG [85] | PEG-coated PSi |
| Mixed Polymer Brush | PCBMA/PMTFPS | Not Specified | Excellent resistance after dry/wet treatment [81] | Specific structural conditions |
| PEG Coating | PEG (Various MW) | Not Specified | > 0.3 ng/cm² (Inferred) | Zwitterionic Coating |
Key Findings:
- Ultra-Low Fouling of Zwitterionic Brushes: Surfaces grafted with zwitterionic polymers like pCBMA and pSBMA demonstrate exceptional resistance to nonspecific protein adsorption, with fibrinogen adsorption reported to be less than 0.3 ng/cm² [83].
- Superiority of Zwitterionic Peptides: When applied to challenging high-surface-area substrates like porous silicon (PSi), a designed zwitterionic peptide (EKEKEKEKEKGGC) provided approximately 80% greater reduction in biofouling from complex gastrointestinal fluid compared to conventional PEG coatings [85].
- Synergistic Effects in Mixed Brushes: Combining hydrophilic zwitterionic polymers with hydrophobic components like fluorosiloxane (PMTFPS) can yield synergistic antifouling effects. Optimal performance is achieved with specific architectural conditions, such as when the zwitterionic PCBMA chains are shorter than the PMTFPS chains and at a moderately higher grafting density [81].
Bacterial Adhesion and Biofilm Formation Resistance
Preventing the initial adhesion of bacteria is crucial for inhibiting biofilm formation. The following table quantifies the performance of various coatings against bacterial adhesion.
Table 2: Quantitative bacterial adhesion reduction data for antifouling coatings.
| Coating Type | Specific Material | Bacterial Species | Adhesion Reduction (%) | Test Conditions |
|---|---|---|---|---|
| Zwitterionic Polymer Brush | pSBMA (grafted via ATRP) | S. epidermidis | 92% [83] | 3 hours, PBS buffer |
| Zwitterionic Polymer Brush | pSBMA (grafted via ATRP) | P. aeruginosa | 96% [83] | 3 hours, PBS buffer |
| PEG Coating | pOEGMA (grafted via ATRP) | S. epidermidis, P. aeruginosa | Significant reduction (exact % not specified) [83] | 3 hours, PBS buffer |
| Self-Assembled Monolayer (SAM) | OEG-SAM | S. epidermidis, P. aeruginosa | Significant reduction (short-term) [83] | 3 hours, PBS buffer |
Key Findings:
- High-Efficiency Resistance: Long-chain zwitterionic pSBMA brushes grafted via surface-initiated Atom Transfer Radical Polymerization (ATRP) reduce short-term adhesion of clinically relevant bacteria by over 90% compared to glass references [83].
- Long-Term Biofilm Prevention: Qualitative assessments confirm that pSBMA coatings dramatically reduce long-term (24-48 hour) biofilm formation of both S. epidermidis and P. aeruginosa [83].
- Coating Stability is Critical: While short-chain OEG self-assembled monolayers (SAMs) can resist initial bacterial adhesion, they often fail to prevent long-term biofilm formation due to a lack of stability on the substrate surface, unlike covalently grafted polymer brushes [83].
Experimental Protocols for Key Studies
Fabrication of Zwitterionic Polymer Brushes via ATRP
The "grafting from" method using ATRP is a widely used and effective technique for creating dense, long-chain zwitterionic polymer brushes on surfaces [83].
- Surface Preparation and Initiation Immobilization: Substrates (e.g., gold-coated chips for SPR, silicon wafers) are thoroughly cleaned. An ATRP initiator (e.g., 2-bromoisobutyryl bromide) is immobilized onto the surface, creating a monolayer of initiation sites.
- Polymerization Reaction: The initiator-functionalized substrate is placed in a deoxygenated reaction solution containing the zwitterionic monomer (e.g., sulfobetaine methacrylate - SBMA), a catalyst (e.g., CuBr), and a ligand (e.g., 2,2'-bipyridine - BPY).
- Reaction Conditions: The polymerization is typically allowed to proceed for several hours at a controlled temperature (e.g., room temperature or 30°C).
- Post-processing: After polymerization, the substrate is removed and rigorously rinsed with appropriate solvents (e.g., Milli-Q water, ethanol) to remove physisorbed monomers, catalyst, and untethered polymer chains, leaving a covalently attached brush layer [83].
Preparation of Mixed Polymer Brushes
Amphiphilic mixed brushes combine the fouling-resistance of hydrophilic components with the fouling-release properties of hydrophobic components [81].
- Sequential Grafting: A mixed brush of hydrophilic poly(carboxybetaine methacrylate) (PCBMA) and hydrophobic poly[methyl(3,3,3-trifluoropropyl)siloxane] (PMTFPS) can be fabricated sequentially.
- "Grafting From" for PCBMA: The PCBMA block is first grown directly from the silicon substrate using a "grafting from" approach, allowing control over grafting density and chain length.
- "Grafting To" for PMTFPS: The PMTFPS chains, pre-synthesized and end-functionalized with a trimethoxysilane group (PMTFPS-TMS), are then coupled to the surface in a "grafting to" step.
- Architectural Control: This sequential method enables precise control over the structure and composition of the mixed brush, which is critical for achieving synergistic antifouling effects [81].
Protein Adsorption Measurement via Surface Plasmon Resonance (SPR)
SPR is a label-free optical technique that sensitively measures biomolecular interactions on a metal surface in real-time.
- Baseline Establishment: The coated sensor chip is mounted in the SPR instrument. A running buffer (e.g., PBS) is flowed over the surface until a stable baseline is achieved.
- Sample Injection: A solution of the target protein (e.g., fibrinogen, human serum) is injected and flowed over the sensor surface.
- Real-Time Monitoring: The SPR instrument monitors the change in the refractive index at the sensor surface, which is directly proportional to the mass of adsorbed protein. This is recorded as a sensorgram.
- Data Analysis: The response difference between the active sensor surface and a reference flow cell is calculated. The shift in resonance units (RU) is converted to mass per unit area (ng/cm²), with 1000 RU corresponding to approximately 1 ng/mm² of adsorbed protein [83].
Bacterial Adhesion Assay under Laminar Flow
Quantifying bacterial adhesion under flow conditions mimics physiological and industrial environments more realistically than static assays.
- Bacterial Culture and Preparation: Model bacterial strains (e.g., S. epidermidis, P. aeruginosa) are cultured and suspended in an appropriate buffer like PBS.
- Flow Chamber Setup: The coated substrate is assembled into a laminar flow chamber. The bacterial suspension is pumped through the chamber at a defined, controlled shear rate.
- Adhesion Phase: The suspension is flowed for a set period (e.g., 3 hours for short-term adhesion studies).
- Rinsing and Enumeration: After the adhesion phase, sterile buffer is flowed through the chamber to remove non-adhered cells. The number of adhered bacteria on the surface is then quantified, typically using microscopy image analysis or by detaching and plating the cells for colony-forming unit (CFU) counts [83].
Visualization of Antifouling Mechanisms
The superior antifouling performance of zwitterionic coatings stems from their unique mechanism of surface hydration. The following diagram illustrates and contrasts the hydration layers of PEG and zwitterionic coatings.
Diagram 1: Molecular hydration mechanisms of PEG versus zwitterionic coatings.
The Scientist's Toolkit: Essential Research Reagents
Table 3: Key reagents and materials for fabricating and testing antifouling coatings.
| Reagent/Material | Function in Research | Example Use Case |
|---|---|---|
| Sulfobetaine Methacrylate (SBMA) | Zwitterionic monomer for creating antifouling polymer brushes. | Grafted via ATRP to form pSBMA brushes for protein and bacterial adhesion studies [83]. |
| Carboxybetaine Methacrylate (CBMA) | Zwitterionic monomer for creating antifouling polymer brushes. | Used in mixed brush systems with fluorosiloxane polymers to study synergistic effects [81]. |
| 2-Methacryloyloxyethyl Phosphorylcholine (MPC) | Zwitterionic monomer mimicking phospholipid headgroups. | Polymerized to form PMPC, used in hydrogels and as a stealth corona in nanocarriers [5] [84]. |
| ATRP Initiator (e.g., BiBB) | Immobilized on surfaces to initiate controlled radical polymerization. | Used to covalently "graft from" surfaces for creating dense polymer brush layers [81] [83]. |
| Poly(ethylene glycol) Methyl Ether Methacrylate (OEGMA) | Monomer for creating PEG-like polymer brushes as a experimental control. | Grafted via ATRP to form pOEGMA brushes for comparative performance evaluation [83]. |
| Laminar Flow Chamber | Device to study bacterial adhesion under controlled shear stress. | Used to quantify bacterial adhesion reduction (%) over defined time periods [83]. |
| SPR Sensor Chip (Gold) | Transducer for label-free, real-time measurement of biomolecular adsorption. | Functionalized with coatings to quantify protein adsorption in ng/cm² [83]. |
The evaluation of antifouling coatings for biomedical devices relies heavily on robust laboratory models that predict their performance in complex biological environments. Among the most promising materials are polyethylene glycol (PEG) and zwitterionic polymers, both celebrated for their ability to resist protein adsorption, bacterial adhesion, and thrombus formation. This guide objectively compares the antifouling performance of PEG versus zwitterionic coatings across standardized in vitro and ex vivo validation models, providing researchers with structured experimental data, detailed methodologies, and essential technical considerations for their comparative studies.
Performance Comparison of PEG and Zwitterionic Coatings
The following tables summarize key performance metrics for PEG and zwitterionic coatings derived from published experimental data.
Table 1: Quantitative Antifouling Performance of PEG vs. Zwitterionic Coatings
| Performance Parameter | PEG Coatings | Zwitterionic Coatings | Test Model | Reference |
|---|---|---|---|---|
| Protein Adsorption (BSA) | Varies with thickness; ~1.5-3.3 nm thickness can exceed PMEN performance [6] | Superior resistance at ~1 nm thickness; comparable at ~3.6 nm [6] | Surface Plasma Resonance (SPR) with BSA [6] | Cheng-Mei Xing et al., 2017 [6] |
| Effect of End Group | 10-fold increase in protein adsorption with -COOH vs. -OH [6] | Performance stable against end-group variations [6] | SPR sensor chip [6] | Cheng-Mei Xing et al., 2017 [6] |
| Long-term Sensor Signal Stability | >27% signal drift over 24 hours [49] | <8.5% signal drift over 24 hours [49] | Electrochemical aptamer-based sensor in serum [49] | Duan H. et al., 2025 [49] |
| Mechanical/Hydrolytic Stability | Prone to oxidative degradation [17] [5] | Superior resistance to hydrolytic degradation [49] | Wearable microneedle patch, mechanical testing [49] | Duan H. et al., 2025 [49] |
| Immunogenic Potential | Can elicit PEG-specific antibodies [5] | Low immunogenic potential reported [17] | In vivo models (literature review) [5] | Various Studies [17] [5] |
Table 2: Application-Based Efficacy in Complex Biological Models
| Application Context | PEG Coating Performance | Zwitterionic Coating Performance | Key Findings |
|---|---|---|---|
| Biosensors | Signal drift >27% in serum; hydrolytic degradation [49] | Signal drift <8.5% in serum; maintains >90% initial signal in tissue phantom [49] | Zwitterionic coatings (SBMA@PDA) offer superior stability for continuous monitoring. [49] |
| Blood-Contacting Devices | Reduces protein fouling [6] | Significantly reduces protein adsorption and platelet adhesion, lowering thrombosis risk [86] | Zwitterionic coatings can improve hemocompatibility without long-term anticoagulation therapy. [86] |
| Implantable Devices | Antifouling efficacy can degrade over time [5] | Supports tissue integration while resisting biofilm formation and inflammation [86] | Titanium implants with zwitterionic treatments show enhanced biocompatibility. [86] |
Experimental Models and Methodologies
In Vitro Protein Adsorption Assay (SPR Model)
This model quantitatively evaluates the non-specific adsorption of proteins like Bovine Serum Albumin (BSA) and fibrinogen onto coating surfaces [6].
Detailed Protocol:
- Coating Fabrication: Immobilize PEG or zwitterionic polymers (e.g., PMEN, a phosphorylcholine-based polymer) onto a SPR sensor chip. This is often achieved by first depositing a polydopamine (PDA) intermediate layer, which acts as a universal adhesive, followed by covalent coupling of the antifouling polymer via amidation [6].
- Thickness Measurement: Use the SPR instrument or an ellipsometer to ensure coatings are fabricated with comparable, controlled thickness (e.g., ~1 nm, ~3.6 nm), as performance is highly thickness-dependent [6].
- Protein Exposure: Flow a solution of the target protein (e.g., 1 mg/mL BSA in a buffer like PBS) over the coated sensor chip at a controlled rate and temperature (e.g., 25°C) [6].
- Quantitative Measurement: The SPR instrument monitors the change in the refractive index at the sensor surface in real-time, which is directly proportional to the mass of adsorbed protein. The response is measured in Resonance Units (RU) [6].
- Data Analysis: Compare the final resonance unit (RU) values or the mass of adsorbed protein (ng/cm²) for PEG and zwitterionic coatings after a stable signal is achieved. A lower value indicates superior antifouling performance [6].
Bacterial Culture and Biofilm Models
These models assess the coating's ability to resist bacterial adhesion and biofilm formation, which is critical for preventing device-associated infections [87].
Detailed Protocol:
- Strain Selection: Select representative Gram-positive (e.g., Staphylococcus aureus) and Gram-negative (e.g., Escherichia coli) strains [87] [88]. Culture them to mid-log phase in appropriate broth (e.g., TSB for S. aureus, LB for E. coli) [89].
- Sample Inoculation: Prepare a standardized bacterial suspension (e.g., adjusted to an optical density of 0.1 at 600 nm). Apply the suspension to coated and uncoated (control) substrates at a defined Multiplicity of Infection (MOI), e.g., 0.2 [88].
- Incubation: Incubate the samples under static or dynamic (with flow) conditions for a set period (e.g., 2-6 hours for initial adhesion; up to 24-48 hours for mature biofilm formation) at 37°C [87] [88].
- Analysis:
- Adhesion Quantification: After incubation, gently rinse samples to remove non-adhered bacteria. Detach adhered bacteria by sonication in a neutral buffer, then plate the serially diluted suspension on agar plates for colony-forming unit (CFU) counting.
- Biofilm Staining: For mature biofilms, use fluorescent dyes (e.g., SYTO 9 for live cells in a BacLight kit) and visualize via confocal laser scanning microscopy (CLSM) to analyze biofilm thickness and biovolume.
- Data Analysis: Compare CFU counts or biofilm biovolume between PEG-coated, zwitterionic-coated, and control surfaces. Superior coatings show significantly lower bacterial counts and biofilm formation [87].
Ex Vivo Whole Blood Circulation Model
This model evaluates the hemocompatibility of coatings, including their resistance to plasma protein adsorption and platelet adhesion, which is crucial for blood-contacting devices [88].
Detailed Protocol:
- Blood Collection: Draw fresh human whole blood from healthy volunteers into anticoagulant-containing vacutainers (e.g., Lithium Heparin) with informed consent and ethical approval [88].
- Sample Setup: Place coated substrates (e.g., sensor chips, catheter segments) in sterile vials or a flow chamber system.
- Blood Incubation: Add the whole blood directly to the samples (e.g., 1 mL blood per sample). Incubate on a rotator (to prevent sedimentation and simulate low flow) at 37°C for defined periods (e.g., 2-6 hours) [88].
- Post-Incubation Analysis:
- Centrifuge the blood-sample mixture and collect the plasma supernatant [88].
- Analyze Plasma: Use enzyme-linked immunosorbent assays (ELISAs) to quantify the release of key immune and coagulation markers, such as IL-6 (inflammation), C5a (complement activation), and TF3 (tissue factor, coagulation) [88].
- Analyze Surface: Examine the coated surface itself post-incubation using microscopy or protein assays to quantify platelet adhesion and fibrin clot formation.
- Data Analysis: Compare the levels of biomarkers in plasma and the degree of surface fouling. Superior antifouling coatings will result in significantly lower levels of pro-inflammatory and pro-coagulant markers and reduced cellular adhesion on the surface [88] [86].
The Scientist's Toolkit: Essential Research Reagents and Materials
Table 3: Key Reagents and Materials for Antifouling Validation Experiments
| Item | Function/Application | Examples & Notes |
|---|---|---|
| SPR Sensor Chip | Gold-coated glass slide for real-time, label-free biomolecular interaction analysis [6]. | Biacore chips or similar. The foundation for quantitative protein adsorption studies [6]. |
| Polydopamine (PDA) | A universal bio-adhesive primer layer that facilitates the covalent immobilization of polymers onto diverse substrates [6] [49]. | Prepared by dissolving dopamine hydrochloride in a mild Tris-HCl buffer (pH ~8.5) and exposing it to the substrate [6]. |
| Zwitterionic Monomers | Building blocks for synthesizing antifouling polymers [17]. | Sulfobetaine methacrylate (SBMA), Carboxybetaine methacrylate (CBMA), 2-methacryloyloxyethyl phosphorylcholine (MPC) [17]. |
| Model Proteins | Used for in vitro fouling challenges to simulate biological exposure [6]. | Bovine Serum Albumin (BSA), Bovine Plasma Fibrinogen (Fg). Use high-purity grades [6]. |
| Model Bacterial Strains | Used for bacterial adhesion and biofilm assays [88]. | Staphylococcus aureus (Gram-positive), Escherichia coli (Gram-negative). Use standard lab strains (e.g., ATCC) for reproducibility [88]. |
| ELISA Kits | Quantify specific biomarkers (cytokines, complement factors) in plasma from ex vivo blood models [88]. | Commercial Duoset kits for IL-6, C5a, TF3, etc. Essential for measuring immune response [88]. |
| SDT Lysis Buffer | Efficiently extracts proteins from bacterial cells for proteomic analysis in biofilm studies [89]. | Composition: 4% (w/v) SDS, 100 mM DTT, 100 mM Tris-HCl (pH 7.6). Often used with boiling/ultrasonication [89]. |
Discussion and Technical Considerations
Performance Under Physiologically Relevant Conditions
While PEG is a well-established antifouling polymer, zwitterionic materials demonstrate several advantages in complex test models. Their hydration via ionic solvation creates a more robust barrier than PEG's hydrogen bonding, which translates to superior performance in high-ionic-strength environments like blood and serum [17] [5]. This is evidenced by significantly lower sensor signal drift (<8.5% vs. >27% for PEG) in continuous serum exposure tests [49]. Furthermore, the charge-neutral character of zwitterions effectively eliminates non-specific electrostatic interactions with charged biomolecules, a common fouling mechanism that PEG coatings are less effective at mitigating [17].
Addressing Mechanical and Chemical Stability
A significant challenge for highly hydrophilic zwitterionic hydrogels is their inherent brittleness and weak mechanical properties, often characterized by low Young's modulus and tensile strength [5]. This can limit their use in load-bearing applications like certain implants. Advanced reinforcement strategies are being actively researched to overcome this limitation, including:
- Nanocomposite Integration: Incorporating laponite nanosheets or cellulose nanocrystals (CNCs) as physical cross-linkers to enhance strength and toughness [5].
- Interpenetrating Networks: Creating double-network or interpenetrating polymer networks with other robust polymers to improve mechanical integrity [5]. In contrast, a critical limitation of PEG is its susceptibility to oxidative degradation in vivo, leading to a loss of antifouling efficacy over time, whereas zwitterionic polymers are generally more chemically robust [5] [49].
The selection of an optimal antifouling coating for biomedical devices is critical for patient outcomes, with polyethylene glycol (PEG) and zwitterionic polymers representing the leading contenders. For years, the scientific community has debated their relative performance, with reported comparisons often being contradictory. This guide demonstrates that these apparent contradictions frequently stem from comparisons made at unequal coating thicknesses. By systematically analyzing head-to-head studies that implement strict thickness control, we reveal a critical performance crossover point: zwitterionic coatings dominate in the ultrathin regime (<1.5 nm), while PEG coatings can surpass them at greater thicknesses (1.5-3.3 nm) due to enhanced steric repulsion. Ultimately, both can achieve ultralow fouling at optimized thicknesses around 3.6 nm. The following data, protocols, and analyses provide researchers with a framework for the direct, equitable comparison of these advanced antifouling materials.
Performance Data at Controlled Thicknesses
The table below summarizes key findings from studies that directly compared PEG and zwitterionic polymer coatings while carefully controlling thickness.
Table 1: Direct Performance Comparison of PEG and Zwitterionic Coatings at Controlled Thicknesses
| Coating Type | Specific Polymer | Coating Thickness | Tested Foulant(s) | Key Quantitative Result | Performance Interpretation |
|---|---|---|---|---|---|
| Zwitterionic | PMEN (Phosphorylcholine) | ~1.0 nm | Bovine Serum Albumin (BSA) | "Much stronger resistance... than PEG" [6] | Superior performance at ultrathin scale |
| Polyethylene Glycol (PEG) | PEG-OH | ~1.0 nm | Bovine Serum Albumin (BSA) | Significantly higher adsorption than PMEN [6] | Inferior performance at ultrathin scale |
| Zwitterionic | PMEN (Phosphorylcholine) | 1.5 - 3.3 nm | Bovine Serum Albumin (BSA) | Resistance efficacy surpassed by PEG [6] | Performance overtaken by PEG at higher thickness |
| Polyethylene Glycol (PEG) | PEG-OH | 1.5 - 3.3 nm | Bovine Serum Albumin (BSA) | Resistance efficacy exceeded PMEN [6] | Superior performance due to stronger steric repulsion |
| Polyethylene Glycol (PEG) | PEG-OH | ~3.6 nm | BSA & Bovine Plasma Fibrinogen (Fg) | "Ultralow fouling" [6] | Performance equalized at optimal thickness |
| Zwitterionic | PMEN (Phosphorylcholine) | ~3.6 nm | BSA & Bovine Plasma Fibrinogen (Fg) | "Ultralow fouling" [6] | Performance equalized at optimal thickness |
| Zwitterionic | pSBMA | Equivalent Hydrodynamic Radius to PEG | Dengue NS1 biomarker in wearable devices | "Significantly higher capture efficiency (>2-fold increase)" [13] | Superior for specific biomarker capture in complex biological tissue |
Decoding the Thickness-Performance Relationship
The experimental data reveals a nuanced narrative that moves beyond a simple "which is better" dichotomy. The relationship between coating thickness and antifouling performance is governed by distinct mechanisms that dominate at different physical scales.
Figure 1: The thickness-dependent performance crossover between PEG and zwitterionic coatings.
The Ultrathin Regime (<1.5 nm): Zwitterionic polymers, such as PMEN (phosphorylcholine-based) and pSBMA (sulfobetaine methacrylate), demonstrate superior resistance to protein adsorption at thicknesses around 1 nm [6]. Their exceptional performance is attributed to a strong electrostatic hydration mechanism. The zwitterions contain both positive and negative charges within the same molecular unit, creating a powerful, tightly bound water layer that provides a formidable osmotic and energetic barrier against approaching proteins, even at a minimal physical presence [32].
The Crossover Point (1.5 - 3.3 nm): As coating thickness increases, the antifouling mechanism begins to shift. PEG coatings, when grown to a thickness between 1.5 and 3.3 nm, begin to surpass the performance of their zwitterionic counterparts. This is due to the increasing dominance of steric repulsion [6]. The long, flexible PEG chains, when given sufficient space, create a physical and energetic barrier that prevents foulants from reaching the substrate. The performance of a coating is not intrinsic to its chemistry alone but is a function of its deployed physical dimensions.
The Saturation Point (~3.6 nm): When both types of coatings are optimized and fabricated to a sufficient thickness—approximately 3.6 nm in the case of the SPR study—their performance converges, and both demonstrate "ultralow fouling" against challenging foulants like undiluted blood plasma [6]. At this point, both mechanisms (electrostatic hydration and steric repulsion) are fully mobilized, providing comprehensive fouling resistance.
Experimental Protocols for Equitable Comparison
To ensure reproducible and meaningful comparisons, researchers must adhere to rigorous protocols for coating fabrication and testing. The following workflow, adapted from a seminal SPR study, outlines a robust methodology for quantitative comparison [6].
Figure 2: Workflow for quantitative coating fabrication and testing.
Detailed Coating Fabrication via Polydopamine Intermediate Layer
The use of a polydopamine (PDA) intermediate layer is a widely adopted and substrate-independent strategy for ensuring uniform and stable adhesion of antifouling polymers [6]. The procedure is as follows:
- Substrate Cleaning: Standard clean with oxygen plasma or piranha solution.
- PDA Deposition: Immerse the substrate in a freshly prepared aqueous solution of dopamine hydrochloride (2 mg/mL in 10 mM Tris-HCl buffer, pH 8.5) for a defined period (e.g., 30-60 minutes) to form a thin, adherent PDA film.
- Polymer Grafting: Incubate the PDA-coated substrate in an aqueous solution of the desired polymer.
- For Zwitterionic PMEN: Use a solution of the random copolymer bearing phosphorylcholine zwitterion and active ester side chains. The active esters readily form amide bonds with the amine groups of the PDA layer.
- For PEG Coatings: Use a solution of carboxylic PEG. The terminal carboxyl groups can be activated for coupling to the PDA amines.
- Thickness Control: The final dry thickness of the polymer brush is precisely controlled by varying the polymer concentration, reaction time, and temperature during the grafting step. This is monitored in real-time using techniques like Surface Plasmon Resonance (SPR) or Ellipsometry.
Critical Consideration: PEG End-Group Chemistry
A critical finding from controlled studies is that the end-group chemistry of PEG significantly impacts its performance. Changing the PEG terminal group from -OH to -COOH can cause a 10-fold increase in protein adsorption [6]. This is attributed to the introduction of a charged moiety that can interact electrostatically with proteins. Therefore, PEG-OH is generally the preferred chemistry for antifouling applications, and this must be specified and controlled in any comparative study.
The Scientist's Toolkit: Essential Research Reagents
Table 2: Key Reagents and Materials for Antifouling Coating Research
| Reagent/Material | Function in Research | Specific Examples & Notes |
|---|---|---|
| Zwitterionic Monomers | Primary building block for zwitterionic polymer brushes and hydrogels. | Sulfobetaine methacrylate (SBMA) [52], Carboxybetaine methacrylate (CBMA), Phosphorylcholine-based monomers (e.g., MPC) [32]. |
| PEG Derivatives | Primary building block for PEG-based coatings. | PEG-diacrylate (crosslinker), PEG-acrylate, PEG-OH (optimal end-group), PEG-COOH (use with caution) [6] [90]. |
| Surface Initiators | Anchor polymer chains to substrate for "grafting from" techniques. | Dopamine-based initiators [42], Bromine-terminated silanes [42] [32], Thiols (for gold surfaces) [32]. |
| Universal Adhesive | Provides a substrate-independent platform for "grafting to" methods. | Polydopamine (PDA) [6]. Simplifies coating of diverse materials (metals, polymers, ceramics). |
| Crosslinkers | Tune the mesh size and mechanical stability of hydrogel coatings. | Poly(ethylene glycol) dimethacrylate (PEGDMA) of varying molecular weights [52]. |
| High-Throughput Screening Tools | Accelerate optimization of parameters like composition and crosslinking. | Droplet Microarray (DMA) systems [52]. Enable testing of hundreds of combinations with minimal reagent use. |
The quest to crown a definitive "winner" between PEG and zwitterionic antifouling coatings is a misdirection. The evidence from controlled studies confirms that the optimal choice is context-dependent. Researchers and developers must prioritize the operational constraints of their specific application. For ultrathin coatings on miniaturized sensors or devices where every nanometer counts, zwitterionic polymers offer unparalleled performance. For applications where a thicker hydrogel layer is permissible and maximum steric repulsion is desired, PEG is an excellent candidate. Furthermore, long-term chemical stability must be considered; while PEG is susceptible to autoxidation, particularly in the presence of transition metals, zwitterionic polymers often demonstrate superior stability [91]. Ultimately, this analysis underscores that a sophisticated understanding of the relationship between thickness, mechanism, and performance is paramount for designing next-generation antifouling biomedical devices.
Durability and Stability Assessment Under Physiological Conditions
This guide provides an objective comparison of the durability and stability of polyethylene glycol (PEG) and zwitterionic polymer coatings under physiological conditions, crucial for selecting materials for biomedical devices and drug delivery systems.
Performance Comparison in Physiological Environments
The long-term efficacy of an antifouling coating is determined by its chemical stability and ability to maintain a non-fouling surface when exposed to complex biological fluids. The table below summarizes key performance metrics from comparative studies.
Table 1: Comparative Stability and Antifouling Performance of PEG and Zwitterionic Coatings
| Performance Metric | Polyethylene Glycol (PEG) | Zwitterionic Polymers | Test Conditions | Citation |
|---|---|---|---|---|
| Long-term Signal Stability (in serum) | ~27% signal drift over 24 hours | <8.5% signal drift over 24 hours | Electrochemical biosensor for drug monitoring | [49] |
| Hydrolytic Degradation Resistance | Lower resistance to hydrolysis; chain depolymerization | Superior resistance to hydrolytic degradation | Exposure to aqueous, ion-rich biological solutions | [1] [49] |
| Structural Stability | Autoxidizes and degrades in presence of transition metal ions | High chemical stability; retains structure during storage/handling | Room temperature storage in biologically relevant solutions | [1] [91] |
| Macrophage Adhesion Reduction | Not specified | 30-fold reduction vs. uncoated surface | Photografted thin films on PDMS | [68] |
| Fibroblast Adhesion Reduction | Not specified | 10-fold reduction vs. uncoated surface | Photografted thin films on PDMS | [68] |
| Protein Adsorption (Fibrinogen) | Varies with cross-linking | ~20-fold reduction vs. uncoated PDMS | Fluorescently labeled fibrinogen adhesion test | [68] |
Experimental Protocols for Durability Assessment
To obtain the data presented, researchers employ rigorous experimental protocols that simulate physiological stresses.
Continuous Electrochemical Interrogation in Serum
This protocol assesses the stability of coatings on biosensors, crucial for therapeutic drug monitoring [49].
- Sensor Functionalization: Coat the electrochemical sensor (e.g., a gold nanoparticle/MXene electrode) with the material (PEG or zwitterionic SBMA via a polydopamine adhesive layer).
- Aptamer Immobilization: Functionalize the coated sensor with a specific aptamer (e.g., for vancomycin).
- Exposure to Serum: Immerse the functionalized sensor in undiluted blood serum or serum-spiked buffer at 37°C.
- Continuous Measurement: Use cyclic voltammetry and square wave voltammetry to interrogate the sensor continuously over 24+ hours.
- Data Analysis: Calculate the percentage of signal drift from the initial measurement. Lower drift indicates superior coating stability and fouling resistance.
Tribological Long-Term Durability Testing
This method uses friction and wear cycles to simulate mechanical aging and evaluate long-term antifouling performance [91].
- Coating Application: Apply the coating (e.g., PSBM terpolymer) to a substrate (e.g., PMMA) via drop-casting.
- Mechanical Aging: Subject the coated substrate to controlled abrasion or wear cycles to induce surface stress.
- Biofouling Challenge: Expose both aged and pristine coated surfaces to fouling organisms (e.g., Chlorella sp., Nannochloropsis sp.) for a set period (e.g., 7 days).
- Performance Evaluation: Quantify the biomass adhered to the surface after exposure.
- Cycle Repetition: Repeat the stress and challenge cycles multiple times. A coating that maintains low biomass adhesion over many cycles demonstrates high durability.
Cross-Link Density Optimization
This protocol investigates the trade-off between mechanical stability and antifouling performance in hydrogel coatings [68].
- Varying Cross-linker: Prepare a series of zwitterionic (e.g., SBMA) monomer solutions with a fixed total monomer concentration but varying ratios of cross-linker (e.g., PEGDMA) from 0% to 100%.
- Photografting: Graft the monomer solutions onto a substrate (e.g., benzophenone-soaked PDMS) using UV light.
- Mechanical Testing: Measure the equilibrium swelling ratio and compressive modulus of the resulting thin films. Higher cross-link density reduces swelling and increases modulus.
- Biological Testing: Quantify protein adsorption (e.g., fluorescent fibrinogen) and cell adhesion (macrophages, fibroblasts) on the films.
- Identify Optimal Range: Determine the cross-link density range that provides the best balance of mechanical strength and antifouling efficacy.
Coating Stability and Performance Pathways
The durability and antifouling performance of a coating are direct outcomes of its chemical structure and its interaction with the physiological environment. The following diagram illustrates the causal pathways from material composition to in-situ performance.
The Scientist's Toolkit: Key Research Reagents
The following table lists essential materials and their functions for preparing and testing durable antifouling coatings.
Table 2: Essential Reagents for Antifouling Coating Research
| Reagent/Material | Function in Research | Key Characteristics |
|---|---|---|
| SBMA (Sulfobetaine Methacrylate) | Zwitterionic monomer for creating ultra-low fouling polymer brushes and hydrogels. | Net neutral charge, super-hydrophilicity, forms a dense hydration layer. [1] [68] |
| PEGDMA (Poly(Ethylene Glycol) Dimethacrylate) | Cross-linker for tuning the mechanical properties and swelling ratio of hydrogel networks. | Bi-functional monomer; concentration controls mesh density and durability. [68] |
| Polydopamine (PDA) | Versatile bio-adhesive layer for anchoring active coatings to inert substrates like metals and polymers. | Enables strong surface bonding via mild oxidative polymerization. [49] |
| Benzophenone | Photo-initiator used for surface-initiated photografting of polymer thin films onto substrates like PDMS. | Generates free radicals upon UV exposure to initiate polymerization. [68] |
| Fluorescently Labeled Fibrinogen | Model protein for quantitative assessment of non-specific protein adsorption onto coated surfaces. | Allows for direct visualization and quantification of fouling using fluorescence microscopy. [68] |
| AIBN (Azoisobutyronitrile) | Thermal radical initiator for conventional free-radical polymerization in solution. | Decomposes upon heating to generate radicals for polymer chain growth. [91] |
| DMEAM (Dimethylaminoethylmethacrylate) | Precursor monomer for the synthesis of sulfobetaine zwitterionic polymers via post-polymerization modification. | The tertiary amine group reacts with 1,3-propanesultone to introduce the sulfonate group. [91] |
Preventing the nonspecific adsorption of proteins, cells, and bacteria—collectively known as biofouling—is a critical challenge in the development of biomedical devices, drug delivery systems, and diagnostic tools [1]. For decades, poly(ethylene glycol) (PEG) has been the prevailing "gold standard" for creating antifouling surfaces through a process known as PEGylation [1]. However, the emergence of zwitterionic polymers as a promising alternative has necessitated a structured framework to guide material selection. This guide provides an objective, data-driven comparison of PEG and zwitterionic polymer coatings, focusing on their antifouling performance across various applications to inform researchers, scientists, and drug development professionals.
Fundamental Antifouling Mechanisms
The antifouling capabilities of both PEG and zwitterionic polymers stem from their ability to form a protective hydration layer that acts as a physical and energetic barrier against fouling agents. However, the molecular nature of their interaction with water differs significantly, leading to distinct performance characteristics.
PEG interacts with water molecules primarily through hydrogen bonding (dipole-dipole interactions) [92]. The ether oxygens in its backbone form hydrogen bonds with water, creating a hydration layer that provides steric repulsion to approaching biomolecules.
Zwitterionic polymers contain both positively and negatively charged groups within the same repeating unit while maintaining overall charge neutrality [1]. They bind water molecules through stronger ionic solvation (ion-dipole interactions) [92], forming a denser, more tightly bound hydration layer than PEG.
The following diagram illustrates these fundamental hydration mechanisms:
Performance Comparison and Experimental Data
Quantitative Antifouling Performance
Direct comparative studies reveal how coating composition, thickness, and environmental conditions influence antifouling efficacy. The following table summarizes key performance metrics from controlled experiments:
Table 1: Direct performance comparison of PEG and zwitterionic coatings
| Coating Material | Test Model | Performance Results | Experimental Conditions | Reference |
|---|---|---|---|---|
| PMEN (Zwitterionic) | Protein adsorption (BSA) | Superior resistance at thin thickness (~1 nm) | Surface Plasma Resonance (SPR) | [6] |
| PEG-OH | Protein adsorption (BSA) | Performance exceeded PMEN at 1.5-3.3 nm thickness | Surface Plasma Resonance (SPR) | [6] |
| PEG-COOH | Protein adsorption | 10-fold increase vs. PEG-OH | Changing end group from -OH to -COOH | [6] |
| pSBMA | Bacterial adhesion | Up to 99% reduction vs. controls | Various substrates | [42] |
| pSBMA | Dengue NS1 biomarker capture | >2-fold signal increase vs. PEG | Wearable microprojection arrays in mouse model | [13] |
Comparative Material Properties
The fundamental differences in chemical structure translate to distinct material properties that influence application suitability:
Table 2: Property comparison between PEG and zwitterionic polymer coatings
| Property | PEG | Zwitterionic Polymers |
|---|---|---|
| Primary Hydration Mechanism | Hydrogen bonding (dipole-dipole) | Ionic solvation (ion-dipole) |
| Hydration Layer Density | Moderate | High |
| Stability in Aqueous Environments | Susceptible to oxidative degradation | Chemically robust |
| Immunogenicity | Can elicit anti-PEG antibodies | Immunologically inert |
| Salt/Solvent Stability | Compromised at high ionic strength | Maintains performance in high ionic strength |
| Structural Stability | EG units can depolymerize | High structural stability |
Experimental Protocols and Methodologies
Coating Fabrication and Optimization
Standardized protocols enable consistent fabrication and performance comparison:
Polydopamine-Assisted Coating Deposition [6]:
- Surface Preparation: Clean substrate (e.g., SPR chip, silicon wafer)
- Polydopamine Adlayer: Immerse in dopamine solution (2 mg/mL in Tris-HCl buffer, pH 8.5) for 30-60 minutes
- Polymer Grafting: Incubate polydopamine-coated substrate with PEG or zwitterionic polymer solution
- Characterization: Measure coating thickness using ellipsometry or SPR
High-Throughput Screening of Zwitterionic Hydrogels [52]:
- Platform: Automated droplet microarray printer
- Droplet Generation: Picoliter-scale dispensing with continuous gradient compositions
- Polymerization: UV-initiated crosslinking of SBMA/HEMA mixtures
- Rapid Assessment: Parallel testing of immersion stability, mechanical properties, and drug-loading capacity
The experimental workflow for comparative analysis is systematic and multifaceted:
Key Research Reagent Solutions
Essential materials and their functions for antifouling coating research:
Table 3: Essential research reagents for antifouling coating development
| Reagent/Category | Function | Specific Examples |
|---|---|---|
| Zwitterionic Monomers | Building blocks for polymerization | SBMA, CBMA, MPC [17] |
| PEG Derivatives | Functionalized PEG for conjugation | PEG-COOH, PEG-NH₂, PEG-SH [6] |
| Surface Initiators | Enable polymer brush growth | Bromine-terminated silanes/thiols, dopamine [42] |
| Polymerization Catalysts | Control living radical polymerization | ATRP catalysts (Cu complexes), RAFT agents [1] |
| Surface Characterization | Quantify coating properties | SPR chips, QCM-D sensors, AFM tips [6] |
| Biofouling Assays | Evaluate antifouling performance | Protein solutions (BSA, Fg), bacterial cultures [42] |
Application-Specific Decision Framework
When to Prefer Zwitterionic Coatings
Implantable Medical Devices: Zwitterionic coatings outperform in blood-contacting applications due to superior hemocompatibility and reduced thrombosis risk [17]. Phosphorylcholine-based polymers particularly mimic the outer surface of cell membranes, making them ideal for cardiovascular implants [32].
Complex Biological Environments: For applications in blood, urine, or other high-ionic-strength fluids, zwitterionic coatings maintain their hydration layer and antifouling properties where PEG performance may deteriorate [92] [17].
Long-term Implantation: The resistance to oxidative degradation makes zwitterionic polymers suitable for devices requiring extended functional longevity, where PEG coatings might deteriorate [1].
Diagnostic Applications: Zwitterionic coatings demonstrate enhanced specific biomarker capture while reducing nonspecific background binding, as evidenced by the superior performance in dengue NS1 detection [13].
When PEG May Remain Preferable
Established Manufacturing Protocols: When working with established production facilities experienced in PEGylation chemistry, continuing with PEG may offer advantages in regulatory compliance and process validation [92].
Low-Risk Short-term Applications: For disposable medical devices or short-duration applications where immunogenicity concerns are minimal, PEG's well-characterized safety profile remains beneficial [92].
Specific Steric Stabilization Needs: PEG's flexible polymer chains provide excellent steric repulsion in nanoparticle systems where conformational flexibility is advantageous [74].
Emerging Hybrid Approaches
Advanced coating systems increasingly combine materials to leverage complementary advantages:
SBMA/HEMA Hydrogels: Incorporating 2-hydroxyethyl methacrylate (HEMA) with sulfobetaine methacrylate (SBMA) creates hybrid hydrogels with improved mechanical stability while maintaining antifouling performance [52].
Zwitterionic-PEG Hybrids: Blending zwitterionic monomers with PEG creates coatings that potentially combine the elastomeric strength and cure versatility of PEG with the superior non-fouling abilities of zwitterions [17].
The selection between PEG and zwitterionic coatings requires careful consideration of application requirements and performance priorities. Zwitterionic polymers generally offer superior antifouling performance in challenging biological environments, enhanced stability, and reduced immunogenicity concerns. PEG remains a viable option for less demanding applications and where manufacturing considerations predominate. The emerging trend toward hybrid materials and high-throughput screening approaches promises to accelerate the development of optimized, application-specific antifouling solutions. Researchers should base their selection on specific performance requirements rather than defaulting to traditional options, with zwitterionic polymers representing the advancing frontier of antifouling surface technology.
Conclusion
The choice between PEG and zwitterionic coatings is not a simple declaration of a universal winner but is instead application-dependent. While PEG remains a well-established 'gold standard' with proven low toxicity, concerns over its immunogenicity and oxidative instability are driving the adoption of zwitterionic alternatives. Zwitterionic polymers demonstrate superior hydration capacity, often leading to enhanced antifouling performance, particularly in complex biological fluids, but can face challenges with mechanical robustness. Future directions will focus on developing next-generation hybrid coatings, improving the long-term in vivo stability of zwitterionic materials, and standardizing immunogenicity assessment protocols. The continued translation of these advanced coatings will critically depend on closing the gap between in vitro performance and long-term clinical efficacy, ultimately enabling safer and more effective biomedical devices and therapies.
References
- 1. Recent advances in zwitterionic polymers-based non- ... [https://www.sciencedirect.com/science/article/abs/pii/S2468519424003380]
- 2. Comparison between polyethylene glycol and zwitterionic ... [https://pubmed.ncbi.nlm.nih.gov/26446192/]
- 3. PEG Hydrophilic Coatings and Their Biomedical Uses [https://hydromer.com/peg-hydrophilic-coatings-biomedical-uses/]
- 4. Interaction Forces and Morphology of a Protein-Resistant ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC1305026/]
- 5. Recent Advances in High-Strength Zwitterionic Polymer ... [https://link.springer.com/article/10.1007/s12209-025-00440-6]
- 6. Quantitative fabrication, performance optimization and ... [https://pubmed.ncbi.nlm.nih.gov/28663144/]
- 7. Steric repulsion forces contributed by PEGylation of ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC6402578/]
- 8. Zwitterionic polymer with minimal reactivity against PEG ... [https://pubs.rsc.org/en/content/articlehtml/2025/bm/d4bm01541j]
- 9. Antifouling Zwitterionic Polymer Coatings for Blood-Bearing ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC11823456/]
- 10. Recent advances of multifunctional zwitterionic polymers ... [https://www.sciencedirect.com/science/article/abs/pii/S1742706124002460]
- 11. Zwitterionic Polymers for Biomedical Applications [https://pubmed.ncbi.nlm.nih.gov/39450830/]
- 12. Experimental and computational techniques to investigate ... [https://pubs.rsc.org/en/content/articlelanding/2025/tb/d4tb01782j]
- 13. Comparison between polyethylene glycol and zwitterionic ... [https://research.monash.edu/en/publications/comparison-between-polyethylene-glycol-and-zwitterionic-polymers-]
- 14. Hydration layer structure modulates superlubrication by ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC10337931/]
- 15. Atomistic insight into the temperature response of ... [https://www.sciencedirect.com/science/article/abs/pii/S2468023025004845]
- 16. Zwitterionic Poly(Carboxybetaine Methacrylate)s in Drug ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC12526207/]
- 17. Hydrophilic Zwitterionic Coatings: Advanced Biomedical ... [https://hydromer.com/hydrophilic-zwitterionic-coatings-biomedical/]
- 18. Surface immobilization of sulfobetaine zwitterionic and ... [https://www.sciencedirect.com/science/article/abs/pii/S0169433224029337]
- 19. Fouling behavior of zwitterionic membranes compared to ... [https://www.sciencedirect.com/science/article/abs/pii/S0011916425010793]
- 20. Quantum Chemical Modeling of Hydrogen Bonding in Ionic ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC5480408/]
- 21. Structure making and breaking effects of ions on ... [https://pubs.rsc.org/en/content/articlehtml/2025/cp/d5cp01180a]
- 22. Solvation Structure and Ion–Solvent Hydrogen Bonding of ... [https://www.mdpi.com/2673-8015/2/4/26]
- 23. Strategies for Enhancing Mechanical Performances of ... [https://www.sciencedirect.com/science/article/abs/pii/S0927776525007398]
- 24. Hydrophobic solvation and liquid water: A theoretical ... [https://www.sciencedirect.com/science/article/pii/S0167732225005756]
- 25. Prediction of the zeta potentials and ionic descriptors of a ... [https://www.sciencedirect.com/science/article/abs/pii/S0003267014013105]
- 26. Grafting of bioactive polymers with various architectures [https://pmc.ncbi.nlm.nih.gov/articles/PMC5800312/]
- 27. Polymer Assembly-assisted Grafting-to Method [https://www.sciencedirect.com/science/article/abs/pii/S2468023025022898]
- 28. Developing a thermal grafting process for zwitterionic ... [https://www.sciencedirect.com/science/article/abs/pii/S1742706118307463]
- 29. Quantitative fabrication, performance optimization and ... [https://www.sciencedirect.com/science/article/pii/S1742706117304166]
- 30. Polydopamine Surface Chemistry: A Decade of Discovery [https://pmc.ncbi.nlm.nih.gov/articles/PMC6320233/]
- 31. Effects of cloud-point grafting, chain length, and density of ... [https://www.sciencedirect.com/science/article/abs/pii/S0142961201003349]
- 32. Zwitteration: Coating Surfaces with Zwitterionic ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC4140545/]
- 33. Durable plasma-mediated zwitterionic grafting on ... [https://www.nature.com/articles/s43246-024-00462-y]
- 34. Critical Length of PEG Grafts on lPEI/DNA Nanoparticles for ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC4829937/]
- 35. Polymer Brushes via Surface-Initiated Electrochemically ... [https://www.mdpi.com/1996-1944/13/16/3559]
- 36. Antifouling Polymer Brushes via Oxygen-Tolerant Surface- ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC7191748/]
- 37. Surface-initiated atom transfer radical polymerization for ... [https://www.nature.com/articles/pj201624]
- 38. Photocontrolled RAFT polymerization: past, present, and future [https://pubs.rsc.org/en/content/articlehtml/2023/cs/d1cs00069a]
- 39. SI-PET-RAFT in flow: improved control over polymer brush ... [https://pubs.rsc.org/en/content/articlehtml/2023/py/d3py00488k]
- 40. Anti-fouling coatings for blood-contacting devices [https://www.sciencedirect.com/science/article/pii/S2590183423000431]
- 41. What Are the Best Biocompatible Materials for ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC12194178/]
- 42. A Review of Recent Progress in Synthetic Polymer Surface ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC12250919/]
- 43. A Universal and Versatile Zwitterionic Coating for Blood ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC12097014/]
- 44. Fabrication of photocurable zwitterionic polymer coatings ... [https://www.sciencedirect.com/science/article/abs/pii/S030094402400540X]
- 45. Biocompatibility and corrosion resistance of drug coatings ... [https://www.sciencedirect.com/science/article/abs/pii/S0927776524004612]
- 46. Comparison of polymer-coated, drug-eluting self- ... [https://pubs.rsc.org/en/content/articlehtml/2025/pm/d4pm00312h]
- 47. Preparation of hydrophilic and antifouling coatings via tannic ... [https://pubs.rsc.org/en/content/articlehtml/2025/ra/d5ra00643k]
- 48. PEGylated PLGA nanoparticles: unlocking advanced ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC12288292/]
- 49. Boosted Drug Sensor Stability With Zwitterionic Coating [https://www.azonano.com/news.aspx?newsID=41545]
- 50. The Versatile Power of Zwitterions: Beyond Water Treatment [https://zwitterco.com/blog/the-versatile-power-of-zwitterions-beyond-water-treatment/]
- 51. Structural and functional analysis of polyethylene glycol ... [https://www.sciencedirect.com/science/article/pii/S0378517325006659]
- 52. High-throughput screening of zwitterion-based coatings ... [https://www.nature.com/articles/s41529-023-00362-5]
- 53. Recent Advances in Zwitterionic Hydrogels: Preparation, ... [https://www.mdpi.com/2310-2861/8/1/46]
- 54. Engineering principles of zwitterionic hydrogels [https://pmc.ncbi.nlm.nih.gov/articles/PMC12284511/]
- 55. Recent Advances in Antifouling Materials for Surface ... [https://www.mdpi.com/2073-4360/13/12/1929]
- 56. From passive response to active interaction and closed ... [https://www.sciencedirect.com/science/article/pii/S2452199X25005031]
- 57. Advances in materials science for ocular diseases induced ... [https://www.frontiersin.org/journals/bioengineering-and-biotechnology/articles/10.3389/fbioe.2025.1618232/full]
- 58. Hydrogel Films in Biomedical Applications - PubMed Central [https://pmc.ncbi.nlm.nih.gov/articles/PMC12652965/]
- 59. Next-Generation Biomaterials for Load-Bearing Tissue ... [https://www.mdpi.com/2079-4983/16/7/232]
- 60. To PEGylate or not to PEGylate - PubMed Central [https://pmc.ncbi.nlm.nih.gov/articles/PMC8899923/]
- 61. How Effective is PEGylation as a Stability Enhancement ... [https://nhsjs.com/2025/how-effective-is-pegylation-as-a-stability-enhancement-method/]
- 62. The Importance of Poly(ethylene glycol) Alternatives for ... [https://www.mdpi.com/2073-4360/12/2/298]
- 63. The curious case of anti-PEG antibodies [https://pubs.rsc.org/en/content/articlelanding/2025/nr/d5nr02301g]
- 64. Anti-PEG Antibodies and Their Biological Impact on ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC12388889/]
- 65. Revealing the mechanism of zwitterionic on ... [https://www.sciencedirect.com/science/article/abs/pii/S0300944025007349]
- 66. Improved Stability and Hyperthermia Efficiency of PEG ... [https://www.sciencedirect.com/science/article/pii/S2666845925002399]
- 67. Strategies for enhancing mechanical performances of long ... [https://pubmed.ncbi.nlm.nih.gov/41187572/]
- 68. Antifouling and Mechanical Properties of Photografted ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC8441969/]
- 69. Microgel reinforced zwitterionic hydrogel coating for blood- ... [https://www.nature.com/articles/s41467-022-33081-7]
- 70. Mechanical training drives structural remodeling of zwitterionic ... [https://pubs.rsc.org/en/content/articlehtml/2025/mh/d5mh00465a]
- 71. Porous hierarchically ordered hydrogels demonstrating ... [https://www.nature.com/articles/s41467-025-59171-w]
- 72. Hydrogel Microspheres as Versatile Platforms for Biomedical ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC12508632/]
- 73. Tuning the Density of Zwitterionic Polymer Brushes on PET ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC7023513/]
- 74. Imidazolium-based zwitterionic liquid-modified PEG –PLGA... [https://pubs.rsc.org/en/content/articlelanding/2024/nr/d3nr06349f]
- 75. Optical fibre long-period grating sensors modified with antifouling ... [https://pubs.rsc.org/en/content/articlelanding/2025/bm/d4bm01447b]
- 76. Stability of Polyethylene Glycol and Zwitterionic Surface ... [https://pubmed.ncbi.nlm.nih.gov/29231737/]
- 77. Recent Advances in Zwitterionic Hydrogels - PubMed Central [https://pmc.ncbi.nlm.nih.gov/articles/PMC8775195/]
- 78. An Improved Method for Quantitatively Measuring ... [https://www.pnnl.gov/publications/improved-method-quantitatively-measuring-antifouling-coating-performance-using-mussel]
- 79. PEGylation technology: addressing concerns, moving ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC12020137/]
- 80. Fiber-reinforced zwitterionic elastomer composites for ... [https://pubs.rsc.org/en/content/articlelanding/2025/tb/d5tb00980d]
- 81. Protein-resistant surface coatings formed by mixed polymer ... [https://www.sciencedirect.com/science/article/abs/pii/S0300944025005090]
- 82. Hostile Environments: Modifying Surfaces to Block ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC12190938/]
- 83. Inhibition of Bacterial Adhesion and Biofilm Formation on ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC5463736/]
- 84. Comparing Zwitterionic and PEG Exteriors of ... [https://www.mdpi.com/1420-3049/25/11/2553]
- 85. Porous silicon biosensors meet zwitterionic peptides [https://pubs.rsc.org/en/content/articlehtml/2025/nh/d5nh00478k]
- 86. Zwitterions in Medical Devices [https://zwitterco.com/blog/zwitterions-in-medical-devices/]
- 87. In vitro and ex vivo systems at the forefront of infection ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC7172914/]
- 88. Using the Traditional Ex Vivo Whole Blood Model to ... [https://www.mdpi.com/2227-9059/12/4/724]
- 89. Evaluation of protein extraction methodologies on bacterial ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC12312612/]
- 90. Polymers for anti-fouling applications: a review - RSC Publishing [https://pubs.rsc.org/en/content/articlehtml/2025/va/d5va00034c]
- 91. Antifouling Performance and Long-Term Efficiency of a ... [https://www.mdpi.com/2079-6412/15/4/462]
- 92. Biodegradable Zwitterionic Polymers as PEG Alternatives ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC11376432/]