Smart Biosensors for Emerging Contaminants: Advanced Detection, Challenges, and Future Directions in Biomedical Research
Emerging contaminants (ECs)—including pharmaceuticals, pesticides, microplastics, and endocrine-disrupting chemicals—pose a significant threat to environmental and human health, driving the need for advanced monitoring solutions.
Smart Biosensors for Emerging Contaminants: Advanced Detection, Challenges, and Future Directions in Biomedical Research
Abstract
Emerging contaminants (ECs)—including pharmaceuticals, pesticides, microplastics, and endocrine-disrupting chemicals—pose a significant threat to environmental and human health, driving the need for advanced monitoring solutions. This article provides a comprehensive overview for researchers and drug development professionals on the application of biosensing technologies for EC detection. It explores the foundational science behind different biosensor types, details cutting-edge methodological advancements and their specific applications, analyzes key performance challenges and optimization strategies, and offers a critical comparison with conventional analytical techniques. By synthesizing current progress and identifying future research trajectories, this review serves as a vital resource for advancing the development and deployment of biosensors in environmental and biomedical fields.
The Unseen Threat: Understanding Emerging Contaminants and the Biosensor Revolution
Emerging contaminants (ECs) represent a diverse group of synthetic or naturally occurring chemicals or microorganisms that are not commonly monitored in the environment but have the potential to cause known or suspected adverse ecological and/or human health effects [1]. The term "emerging" does not necessarily mean newly introduced; it may refer to substances that have been present in the environment for a long time, but their persistence and potential risks were only recently recognized due to advances in analytical techniques that now allow detection at trace levels [1]. These contaminants have attracted growing scientific attention in recent years due to their potential ecological and human health impacts, with global efforts intensifying to monitor and regulate their presence in the environment [2].
The spectrum of ECs is broad and continuously expanding as new chemicals are developed and detection methods improve. These contaminants originate from various anthropogenic activities including industrial discharge, agricultural runoff, wastewater effluents, and modern lifestyle products [2] [1]. A widely accepted classification system categorizes ECs into several major groups: pharmaceuticals and personal care products (PPCPs), per- and polyfluoroalkyl substances (PFAS), endocrine-disrupting chemicals (EDCs), and micro- and nano-plastics (MNPs) [1]. Understanding the fate and distribution of these contaminants is vital for crafting regulatory frameworks and sustainable management strategies to mitigate their environmental impact [1].
Classification and Characterization of Major EC Categories
Pharmaceuticals and Personal Care Products (PPCPs)
Pharmaceuticals and personal care products (PPCPs) constitute a remarkably diverse collection of chemicals employed in human healthcare, veterinary medicine, agricultural practices, and cosmetic applications [1]. This category encompasses pharmaceutical drugs, along with components of everyday personal care products such as soaps, lotions, toothpaste, fragrances, and sunscreens [1]. These compounds are increasingly detected across diverse environmental media because they prove challenging to eliminate through conventional wastewater treatment plants, primarily due to their toxic nature and resistance to standard treatment methods [1].
PPCPs are considered emerging pollutants that could potentially pose risks to both environment and human health, particularly as many act as endocrine disruptors (EDCs) – compounds that alter the normal functions of hormones resulting in a variety of health effects [3]. These contaminants may demonstrate low acute toxicity but cause significant reproductive effects at very low levels of exposure, with impacts that may not be observed until adulthood for aquatic organisms exposed during early life stages [3]. The U.S. Environmental Protection Agency has recognized that traditional toxicity test endpoints may not be sufficiently comprehensive for criteria derivation for these chemicals, as they may have specific modes of action that affect only certain types of aquatic animals [3].
Per- and Polyfluoroalkyl Substances (PFAS)
Per- and polyfluoroalkyl substances (PFAS) are a group of over 9,000 manufactured chemicals that resist heat, oil, stains, grease, and water [4] [5]. Dubbed "forever chemicals" due to their pervasive nature and environmental persistence, PFAS are characterized by carbon-fluorine bonds, among the strongest in organic chemistry, making them resistant to chemical and thermal degradation [6]. These properties make PFAS valuable in numerous industrial processes and consumer products, including clothing, furniture, metal finishing, electroplating, electronic components, adhesives, food packaging, heat-resistant non-stick cooking surfaces, and wire insulation [4].
The semiconductor industry represents a significant source of PFAS emissions, utilizing these chemicals for their distinctive properties such as superacidity, low surface energy, low refractive index, and low dielectric constant, which allow for accurate and reliable production of semiconductors [6]. PFAS exposure has been linked to numerous health concerns including kidney, testicular, and breast cancer; thyroid disease; high cholesterol and high blood pressure; cardiovascular issues; ulcerative colitis; liver damage; weakened immune system; irritable bowel syndrome; and hormone imbalances [5]. In April 2024, the EPA imposed the first national and legally enforceable standard for PFAS in drinking water at 4 parts per trillion, reflecting the significant health risks these compounds pose even at minimal concentrations [5].
Micro- and Nano-Plastics (MNPs)
Microplastics are defined as plastic fragments smaller than 5 mm, while nanoplastics are generally considered to be smaller than 1000 nm (with some researchers defining them between 1-100 nm) [1]. These particles originate from the mismanagement and dumping of domestic and commercial plastic waste, with sources categorized as either primary (manufactured at small sizes) or secondary (resulting from the breakdown of larger plastic items through biological processes, mechanical abrasion, and UV radiation) [1]. Common polymeric constituents found in natural settings include polyvinyl chloride (PVC), polyethylene terephthalate (PET), polypropylene (PP), polyethylene (PE), and both low- and high-density polyethylene (LDPE and HDPE) [1].
The large surface-area-to-volume ratio, bioaccumulative nature, persistence, and potential release of chemical additives used in plastic synthesis pose cascading impacts on living organisms [1]. While microplastics primarily present mechanical risks such as gut blockages, nanoplastics can penetrate tissues and organs, leading to irritation, oxidative damage, digestion impairment, changes in gut microbial communities, impaired fatty acid metabolism, and molecular damage [1]. A recent study published in the New England Journal of Medicine links microplastic exposure to increased risk of heart attack, cardiovascular problems, and strokes, with additional concerns about their role as carriers for harmful bacteria, viruses, and other contaminants [5].
Additional Contaminants of Concern
Beyond the three major categories above, several other ECs warrant significant research attention:
- 6PPD and 6PPD-quinone: 6PPD is an additive to rubber components, most notably in all types of tires, which prevents rubber from breaking down due to reactions with ozone. When 6PPD reacts with ozone, it forms 6PPD-quinone, a transformation product of environmental concern [4].
- Polycyclic Aromatic Hydrocarbons (PAHs): These compounds may have either natural sources (wildfires, volcanic eruptions, degradation of materials within sediments and fossil fuels) or man-made sources (incomplete burning of organic materials, vehicle exhaust, asphalt, coal-tar based pavement sealcoat, and creosote) [4].
- Endocrine Disrupting Chemicals (EDCs): This broad category includes many PPCPs and other industrial chemicals that interfere with hormonal systems, potentially causing reproductive, developmental, and metabolic disorders in both wildlife and humans [1] [3].
Table 1: Classification of Major Emerging Contaminant Categories
| Category | Major Subclasses | Primary Sources | Key Characteristics |
|---|---|---|---|
| PPCPs | Pharmaceuticals, personal care products, fragrances, sunscreen, detergents, preservatives, insect repellents [4] [1] | Wastewater effluent, agricultural runoff, residential use [2] [1] | Biologically active, polar, often optically active, resistant to conventional treatment [1] |
| PFAS | Perfluoroalkyl sulfonates (PFSAs), perfluoroalkyl carboxylates (PFCAs), perfluoroalkyl sulfonamides (FASAs), perfluoroalkyl ether acids (PFEAs) [6] | Industrial discharge, semiconductor manufacturing, consumer products [4] [6] | Thermal/chemical stability, persistent, bioaccumulative, toxic at low concentrations [6] [5] |
| MNPs | PVC, PET, PP, PE, LDPE, HDPE [1] | Plastic waste degradation, personal care products, synthetic textiles [1] [5] | Persistent, bioaccumulative, large surface-area-to-volume ratio, potential for toxin adsorption [1] |
| Other ECs | 6PPD-quinone, PAHs, EDCs [4] [1] [3] | Tire wear, combustion processes, industrial activities [4] | Varying persistence, often transformation products, multiple toxicity mechanisms [4] |
Analytical Methodologies for EC Detection and Quantification
Chromatographic and Spectrometric Techniques
Analytical approaches such as gas chromatography (GC), high-performance liquid chromatography (HPLC), mass spectrometry (MS), and high-resolution tandem techniques (LC-MS/MS) have become central to EC identification and quantification [1]. The selection of appropriate analytical methods depends fundamentally on the chemical structure of the target contaminant, as structure governs key physical-chemical properties including solubility, volatility, and hydrophobicity [6]. For example, volatile PFAS such as fluoroalkanes typically require GC-based approaches, while ionogenic PFAS require LC-based methods [6].
The U.S. Environmental Protection Agency has developed several standardized methods for analyzing PFAS in water, including Method 1633, 537, 537.1, 533, 3512, and 8327, along with ASTM International Method D7979-20 [6]. These methods target specific PFAS compounds for which analytical standards are commercially available, but face challenges with "suspect" PFAS (previously identified but without commercial standards) and "nontarget" PFAS (unidentified compounds) [6]. For comprehensive analysis, workflows incorporating high-resolution mass spectrometry (HRMS) enable suspect screening and nontarget discovery for both volatile and nonvolatile PFAS [6].
Complementary Analytical Approaches
Beyond chromatographic methods, several complementary techniques enhance EC detection capabilities:
- Nonspecific methods for PFAS: These include the total oxidizable precursor assay, total fluorine analyses, and extractable and adsorbable organic fluorine assays, which provide broader screening capabilities beyond specific target compounds [6].
- Nuclear magnetic resonance (NMR) spectroscopy: ¹⁹F NMR offers capabilities for both quantitative and qualitative PFAS analysis in wastewater, providing structural information complementary to mass spectrometric techniques [6].
- Molecular and biochemical tools: Techniques such as enzyme-linked immunosorbent assay (ELISA), polymerase chain reaction (PCR), and biosensors are proving essential in detecting biologically active contaminants and pathogens [1].
Table 2: Analytical Methods for Emerging Contaminant Detection
| Analytical Technique | Target EC Classes | Key Advantages | Detection Limits | Standardized Methods |
|---|---|---|---|---|
| LC-MS/MS | PPCPs, polar PFAS, EDCs [1] | High sensitivity, broad compound coverage, reliable quantification | Low ng/L to pg/L range [6] | EPA 539, ISO 21676 |
| GC-MS | Volatile PFAS, PAHs, fragrances [6] | Excellent separation, robust compound identification, extensive libraries | Low ng/L range [6] | EPA 8270, ASTM D7979-20 |
| HRMS | Suspect and nontarget PFAS, transformation products [6] | Unbiased screening, elemental composition, structure elucidation | Varies by compound [6] | EPA 1633 (draft) |
| ¹⁹F NMR | Fluorinated compounds [6] | Nontarget detection, structural information, quantification without standards | μM range (less sensitive than MS) [6] | None standardized |
| ELISA/Biosensors | Biologically active PPCPs, EDCs [1] | Rapid screening, portability, minimal sample preparation | ng/L to μg/L range [7] | Various commercial kits |
Experimental Protocols for PFAS Analysis in Wastewater
A comprehensive workflow for analyzing PFAS in treated semiconductor wastewater illustrates the complexity of EC analysis [6]:
Sample Collection and Preservation: Water samples must be collected in high-density polyethylene or polypropylene containers, maintained at 4°C, and preserved with ammonium acetate or similar additives to prevent biodegradation and adsorption to container walls. The choice of container material is critical as PFAS can adsorb to glass surfaces [6].
Sample Extraction and Concentration: Solid-phase extraction (SPE) using hydrophilic-lipophilic balance (HLB) or weak anion exchange (WAX) cartridges is employed for nonvolatile PFAS. For volatile PFAS, headspace or purge-and-trap techniques are required to prevent losses during concentration steps [6].
Instrumental Analysis:
- For nonvolatile PFAS: LC separation with C18 or similar columns followed by tandem mass spectrometry with electrospray ionization in negative mode.
- For volatile PFAS: GC separation with specific columns (DB-WAX, DB-624) followed by MS detection with electron capture negative ionization or MS/MS.
- For ultrashort-chain PFAS (C1-C3): Specialized LC columns (such as Ionospher A) or IC columns may be required to retain these highly polar compounds [6].
Quality Assurance/Quality Control: Implementation includes procedural blanks, matrix spikes, duplicate samples, and internal standard quantification (using isotopically labeled PFAS analogs) to account for matrix effects and recovery variations [6].
Advanced Detection Technologies and Biosensing Approaches
Electrochemical Biosensing Platforms
Electrochemical biosensors have emerged as promising solutions for qualitative and quantitative detection of EC targets, with applications in environmental monitoring, toxicology analysis, and therapeutic monitoring [7]. These biosensors typically employ techniques including voltammetry, amperometry, potentiometry, electrochemical impedance spectroscopy, chronocoulometry, and conductometry, often coupled with transduction-based measurements such as electrochemiluminescence and photoelectrochemistry [7]. Recent research has focused on developing miniaturized, portable, flexible, and low-cost electrochemical biosensors that can be deployed for field-based monitoring of ECs [7].
A key advancement in this field includes the development of vanadium MXene-modified disposable screen-printed electrodes for highly sensitive glucose sensing, which demonstrates the potential for cost-effective, reproducible sensor platforms capable of rapid detection [8]. Leveraging the unique electrochemical properties of vanadium-based MXenes, researchers have achieved low detection limits, wide linear ranges, and excellent selectivity – parameters essential for real-world clinical and environmental applications [8]. Such innovations support the growing demand for accessible and accurate monitoring technologies, particularly for managing contaminants like PPCPs in water systems [8].
The Scientist's Toolkit: Essential Research Reagents and Materials
Table 3: Essential Research Materials for EC Analysis and Biosensing
| Research Material | Function/Application | Key Characteristics |
|---|---|---|
| HLB/WAX SPE Cartridges | Extraction and concentration of ionic PFAS and PPCPs from water samples [6] | Hydrophilic-lipophilic balance/weak anion exchange chemistry; suitable for broad compound polarity range |
| C18 LC Columns | Chromatographic separation of nonvolatile PFAS, PPCPs, and EDCs [6] | Reversed-phase chemistry; compatible with aqueous and organic mobile phases |
| Isotopically Labeled Standards | Internal standards for quantification by mass spectrometry [6] | ¹³C, ¹⁵N, or ²H-labeled analogs; identical chemical behavior to native compounds |
| Screen-Printed Electrodes | Disposable platforms for electrochemical biosensing [8] [7] | Cost-effective, reproducible, customizable electrode materials (carbon, gold, platinum) |
| MXene Nanomaterials | Electrode modification for enhanced sensitivity [8] | Two-dimensional transition metal carbides/nitrides; high conductivity, large surface area |
| Molecularly Imprinted Polymers | Synthetic biorecognition elements in biosensors [7] | Artificial receptors with tailor-made binding sites; stable, reusable, cost-effective |
| Antibodies/Aptamers | Biorecognition elements for specific EC detection [7] | High specificity and affinity for target molecules; enable selective detection in complex matrices |
Sensor Integration and System Development
The realization of practical point-of-care electrochemical biosensor systems requires updated designs of electrodes, enhanced sensing matrices, assembly of circuit readout, and improved stability and reproducibility [7]. Strategic design from scratch enables platform technologies that can be adapted across different applications [7]. Key challenges include achieving reproducible biosensor functionalization, where electrode surface modification with nanostructured materials enhances loading efficacy and influences charge transfer characteristics at the electrode-electrolyte interface [7].
Recent developments focus on fully integrated biosensing systems for detecting, prognosing, and continuously monitoring contaminants in environmental matrices [7]. Portable and implantable electrochemical sensors represent emerging frontiers, with innovations in system development, machine learning integration, nanomaterial functionalization, and electrochemiluminescence-based detection advancing the field [7]. For environmental applications, continuous monitoring of pollutants in water bodies requires stable and regenerative biosensors capable of operating in complex matrices [7].
Visualization of EC Classification and Detection Workflows
The workflow diagram above illustrates the comprehensive approach required for emerging contaminant analysis, from sample collection through data processing, highlighting the interconnection between specific EC categories and their corresponding analytical techniques.
The spectrum of emerging contaminants – from PPCPs and PFAS to microplastics – presents complex challenges that require sophisticated analytical approaches and multidisciplinary solutions. Addressing the risks posed by these contaminants necessitates integrating scientific research, technological innovation, and policy reform [2] [9]. Strengthening global regulations, improving wastewater treatment strategies, and increasing public awareness are essential steps toward mitigating EC impacts on both ecosystems and human health [2].
Future research should focus on advancing detection technologies, particularly biosensors for field-deployable monitoring, conducting long-term ecological risk assessments, and developing standardized monitoring guidelines to ensure water security [2] [9] [7]. The continued development of electrochemical biosensors, nanomaterial-based platforms, and integrated sensing systems holds particular promise for transforming EC monitoring capabilities, enabling more comprehensive understanding and effective management of these pervasive environmental contaminants [8] [7]. By implementing these solutions through cross-sectoral collaboration and integration of scientific research into policy-making, we can significantly improve our ability to detect, monitor, and manage emerging contaminants, thereby reducing environmental and public health risks [9].
Emerging Contaminants (ECs) represent a broad class of synthetic or naturally occurring chemicals or biological agents that are now being detected in the environment and for which the risks are not fully understood [10]. This category includes pharmaceuticals and personal care products (PPCPs), per- and poly-fluoroalkyl substances (PFAS), industrial chemicals, micro/nano-plastics, antibiotic resistance genes (ARGs), and various other exogenous substances [10]. The urgency surrounding ECs stems from their continuous introduction into ecosystems, their pseudo-persistent nature due to constant release, and the significant gaps in our understanding of their long-term ecological and health impacts. With the Chemical Abstract Service Registry growing from 20 million chemicals in 2002 to over 204 million by 2023—adding nearly 15,000 new chemicals daily—the scale of the challenge is unprecedented [10]. This technical review examines the multifaceted risks posed by ECs, with particular emphasis on their role in driving antibiotic resistance, and outlines advanced methodological approaches for their study and mitigation.
Global Antibiotic Contamination in Marine Environments
Table 1: Detection Frequency and Concentration Ranges of Major Antibiotic Classes in Marine Environments
| Antibiotic Class | Representative Compounds | Detection Frequency in Seawater | Detection Frequency in Sediment | Detection Frequency in Biota | Peak Concentrations Reported |
|---|---|---|---|---|---|
| Sulfonamides | Sulfamethoxazole (SMX) | 71.1% | 30.4% | 47.6% | 332,440 ng L⁻¹ (seawater) |
| Fluoroquinolones | Ciprofloxacin (CIP), Ofloxacin (OFX) | 34.2% (OFX) | Data Incomplete | Data Incomplete | 1515 ng g⁻¹ (sediment) |
| Macrolides | Clarithromycin (CLM) | 44.8% | Data Incomplete | Data Incomplete | 3341 ng g⁻¹ (biota) |
| Tetracyclines | Various | Data Incomplete | Data Incomplete | Data Incomplete | Data Incomplete |
| Dihydrofolate Reductase Inhibitors | Trimethoprim (TMP) | 65.8% | Data Incomplete | Data Incomplete | 332,440 ng L⁻¹ (seawater) |
Data synthesized from global monitoring studies compiled in [11].
The marine environment serves as a ultimate sink for many ECs, with antibiotics displaying concerning prevalence and persistence [11]. Seawater is the most extensively studied marine compartment, with 72 different antibiotics detected at concentrations ranging from non-detectable to a peak of 332,440 ng L⁻¹ recorded near mariculture zones in Laizhou Bay, China [11]. The most frequently detected antibiotics include sulfamethoxazole (71.1% detection in seawater), trimethoprim (65.8%), and clarithromycin (44.8%) [11]. These quantitative data underscore the significant penetration of antibiotic ECs into aquatic ecosystems, where even low concentrations (ng/L to μg/L) can exert selective pressure on microbial communities and drive resistance development [11].
Global Impact of Antimicrobial Resistance
Table 2: Antimicrobial Resistance Global Burden and Projections
| Metric | 2019 Data | 2050 Projection | Key Contributing Pathogens |
|---|---|---|---|
| Direct AMR-attributable deaths | 1.27 million | 10 million annually | MRSA, MDR-TB, CRE |
| AMR-associated deaths | 4.95 million | Not specified | Multiple resistant bacteria |
| Economic impact (projected cumulative 2025-2050) | Not applicable | $3.4 trillion GDP reduction | N/A |
| Potential lives saved with intervention (2025-2050) | Not applicable | 92 million | N/A |
Antimicrobial resistance (AMR), significantly driven by environmental contamination with antibiotics, already causes substantial mortality worldwide, with 1.27 million deaths directly attributable to AMR in 2019 and nearly 5 million associated deaths [12] [13]. Without effective intervention, annual AMR-attributable deaths are projected to reach 10 million by 2050 [12]. The economic consequences are equally staggering, with projections suggesting AMR could reduce global GDP by $3.4 trillion and drive an additional 24 million people into extreme poverty [13]. Multi-drug resistant pathogens including methicillin-resistant Staphylococcus aureus (MRSA), multidrug-resistant tuberculosis (MDR-TB), and carbapenem-resistant Enterobacterales (CRE) represent particularly concerning threats [12] [14].
Environmental Pathways and Ecological Impacts
ECs enter the environment through multiple pathways, categorized as direct and indirect sources [11]. Direct sources include atmospheric deposition, discharge from mariculture, and offshore sewage [11]. Mariculture represents a significant direct pathway, as antibiotics are frequently incorporated into feed to mitigate disease outbreaks, resulting in their direct release into surrounding marine environments through animal excreta [11]. Indirect sources encompass land-based activities such as domestic sewage, hospital effluent, industrial discharges, and wastewater from livestock and aquaculture [11]. Urban wastewater typically undergoes treatment, but processes often fail to completely remove ECs, while rural areas may lack treatment infrastructure entirely, leading to direct environmental discharge [11].
Industrial activities, particularly pharmaceutical manufacturing, discharge wastewater containing exceptionally high antibiotic concentrations. Studies near pharmaceutical manufacturing hubs have detected ciprofloxacin concentrations exceeding 1 mg/L—30,000 times higher than the minimum selective concentration for resistance development [14]. Similarly, agricultural operations contribute significantly through runoff containing veterinary antibiotics and manure-based fertilizers that introduce antibiotics and resistance genes into soil and water systems [11].
Figure 1: Environmental pathways and ecological impact mechanisms of Emerging Contaminants
Ecological Risk Mechanisms
The ecological impacts of ECs operate through multiple mechanisms. Even at low environmental concentrations, antibiotics can interfere with microbial metabolic activities and alter community structures, threatening ecological integrity [11]. Marine microbial communities are fundamental to ecosystem functioning, serving as the base of the marine food web and contributing to global biogeochemical cycles, including carbon, nitrogen, and sulfur [11]. Sub-inhibitory concentrations of antibiotics present in aquatic environments can exert selection pressure on bacterial populations, triggering stress responses that increase integron activity and enhance acquisition of resistance genes [14].
ECs rarely exist in isolation; they typically occur as complex mixtures that can produce synergistic effects. Co-existing contaminants including heavy metals, microplastics, and disinfectants may enhance AMR impacts through co-selection pressure [11] [14]. Microplastics specifically serve as novel habitats for microbial communities, forming biofilms that facilitate horizontal gene transfer (HGT) between environmental and pathogenic bacteria [14]. This phenomenon enhances the horizontal transmission of resistance traits, with studies demonstrating high-frequency transfer of carbapenem resistance genes (e.g., blaNDM-1) in wastewater biofilms [14].
Antibiotic Resistance: A Silent Pandemic
Mechanisms of Resistance Development and Spread
Table 3: Major Antibiotic Resistance Mechanisms and Examples
| Resistance Mechanism | Functional Principle | Example Genes/Pathogens |
|---|---|---|
| Enzymatic inactivation | Production of enzymes that degrade or modify antibiotics | β-lactamases (blaCTX-M-15), macrolide esterases |
| Target site modification | Alteration of antibiotic binding sites to reduce drug affinity | MRSA (mecA gene), VRE (vanA gene) |
| Efflux pumps | Membrane proteins that actively export antibiotics from cells | Tetracycline efflux pumps (tet genes) |
| Reduced permeability | Modification of cell membrane to limit antibiotic entry | Porin mutations in Gram-negative bacteria |
| Horizontal gene transfer | Transfer of resistance genes between bacteria via mobile genetic elements | Plasmid-borne blaNDM-1, sul1, qnrS |
Antibiotic resistance emerges through natural genetic variations in microbial populations, but this process is dramatically accelerated by anthropogenic factors. Key mechanisms include enzymatic inactivation of antibiotics (e.g., through β-lactamase production), target site modification (e.g., altered penicillin-binding proteins in MRSA), and enhanced efflux pumps that remove antibiotics from bacterial cells [12]. Beyond these mechanistic adaptations, the environmental dimension of AMR spread is particularly concerning. Aquatic environments serve as critical reservoirs and conduits, facilitating the horizontal transfer of resistance genes between environmental and pathogenic bacteria [14].
Horizontal gene transfer (HGT) occurs primarily through three mechanisms: conjugation (direct cell-to-cell transfer via plasmids), transformation (uptake of free DNA from the environment), and transduction (viral-mediated transfer) [14]. Conjugation is particularly significant in aquatic environments, where biofilms provide ideal conditions for plasmid exchange. Studies have documented conjugation rates of carbapenem resistance genes reaching 10² transconjugants per donor cell in wastewater biofilms [14]. Furthermore, extracellular DNA carrying resistance genes can persist in aquatic sediments for extended periods (exceeding 40 days) protected by organic matter and clay particles, creating lasting environmental reservoirs for resistance determinants [14].
Experimental Protocol: Assessing Horizontal Gene Transfer in Aquatic Environments
Protocol Title: Quantifying Conjugative Transfer of Antibiotic Resistance Genes in Wastewater Biofilms
Principle: This method evaluates the frequency of plasmid-mediated horizontal gene transfer between donor and recipient bacterial strains within simulated wastewater biofilm systems, mimicking natural environmental conditions where resistance dissemination occurs.
Materials and Reagents:
- Donor strain: Escherichia coli harboring plasmid-borne resistance markers (e.g., blaNDM-1 on IncX3 plasmid)
- Recipient strain: Antibiotic-susceptible Pseudomonas aeruginosa or environmental Acinetobacter spp.
- Growth media: Luria-Bertani (LB) broth and agar
- Antibiotic selection plates: LB agar supplemented with appropriate antibiotics for selection of transconjugants
- Wastewater matrix: Filter-sterilized primary effluent from municipal wastewater treatment plants
- Biofilm reactor system: Flow-cell chambers or rotating biological contactors
- Microscopy supplies: Fluorescent dyes for viability staining (SYTO 9/propidium iodide)
Procedure:
- Strain Preparation: Grow donor and recipient strains separately in LB broth to mid-exponential phase (OD₆₀₀ ≈ 0.5). Wash cells twice in sterile phosphate-buffered saline (PBS) to remove residual antibiotics.
- Biofilm Establishment: Combine donor and recipient strains at 1:10 ratio in wastewater matrix. Circulate the bacterial suspension through biofilm reactor system for 24-48 hours at ambient temperature (20-25°C) to establish mixed-species biofilms.
- Conjugation Assay: After biofilm establishment, continue circulation with fresh wastewater matrix without antibiotics for 24-48 hours to allow conjugation.
- Biofilm Harvesting: Gently scrape biofilm from reactor surfaces and resuspend in PBS. Disaggregate bacterial clusters by mild sonication (3-5 seconds at 10W) followed by vortexing with glass beads.
- Transconjugant Enumeration: Prepare serial dilutions of the biofilm suspension and plate on selective media containing antibiotics that inhibit donor and recipient strains while allowing growth of transconjugants only. Include appropriate controls to verify selection efficiency and monitor for spontaneous mutation.
- Conjugation Frequency Calculation: After 24-48 hours incubation at 37°C, count transconjugant colonies and calculate conjugation frequency as: Number of transconjugants / Number of donor cells.
Quality Control:
- Include viability counts of donor and recipient populations throughout the experiment
- Verify plasmid stability in transconjugants through replica plating and PCR confirmation
- Perform triparental mating controls to exclude mobilizing factors from the environment
This protocol enables quantitative assessment of environmental resistance gene transfer, providing critical data for evaluating intervention strategies aimed at curbing AMR spread in aquatic ecosystems [14].
The Scientist's Toolkit: Research Reagent Solutions
Table 4: Essential Research Tools for Emerging Contaminant Analysis
| Research Tool Category | Specific Examples | Primary Applications | Technical Considerations |
|---|---|---|---|
| Biosensing Platforms | Electrochemical biosensors, Cell-free transcription-translation systems, Paper-based biochips, Organ-on-chip devices | Detection of antibiotic residues, ARG identification, Toxicity screening | Sensitivity to ng/L levels, Requirement for sample pre-concentration, Matrix effects |
| Molecular Biology Reagents | PCR/qPCR kits for ARG detection, Metagenomic sequencing kits, Plasmid conjugation assay systems, Viability staining dyes | Quantification of resistance genes, Microbial community analysis, HGT studies | Primer specificity for environmental variants, Inhibition by humic substances |
| Analytical Chemistry Standards | Isotope-labeled antibiotic standards, PFAS reference materials, Mixed chemical calibration standards | LC-MS/MS quantification, Quality control, Method validation | Stability in aqueous solution, Cross-reactivity in complex matrices |
| Cell Culture Systems | Bacterial strains with reporter constructs, Human cell lines for toxicity testing, Biofilm reactor systems | Mechanistic studies, Pathogenicity assessment, HGT quantification | Genetic stability of reporter constructs, Physiological relevance |
Data synthesized from [15] [16] [14].
Advanced research tools are essential for investigating the complex dynamics of ECs in environmental and biological systems. Electrochemical biosensors have evolved significantly from the original glucose oxidase-based platforms to incorporate sophisticated recognition elements including aptamers, affibodies, peptide arrays, and molecularly imprinted polymers [15]. Recent innovations include ferroceneboronic acid (FcBA) derivatives for sugar and ion detection, and wireless biosensing systems such as mouth-guard sensors for continuous monitoring of biomarkers like salivary uric acid [15].
For AMR research specifically, cell-free protein synthesis (CFPS) systems enable rapid detection of antibiotic resistance mechanisms without requiring viable bacterial cultures [16]. These systems utilize transcriptional or translational machinery to produce reporter proteins in response to specific resistance markers. Similarly, microelectrode array (MEA) platforms facilitate electric cell-substrate impedance sensing (ECIS) for real-time monitoring of bacterial responses to antimicrobial agents [16]. Metagenomic approaches provide comprehensive assessment of resistance gene reservoirs in environmental samples, though they require specialized bioinformatic tools for data analysis and resistance gene annotation [14].
ECs present multidimensional threats to ecological stability and human health, with antibiotic resistance representing one of the most urgent consequences. The interconnectedness of these challenges necessitates a One Health approach that integrates human, animal, and environmental health perspectives [10] [14]. This review has documented the substantial evidence linking environmental contamination with antibiotics to the development and spread of resistance, creating what the World Health Organization has designated a "planetary health emergency" [14]. The quantitative data presented herein—from antibiotic concentrations in marine ecosystems to AMR mortality statistics—underscore the critical priority of addressing EC contamination through coordinated scientific, regulatory, and technological interventions. Future research must continue to elucidate the complex interactions between multiple classes of ECs, develop more sensitive detection methodologies, and translate scientific findings into effective policies that protect both ecosystem integrity and public health.
In the fields of environmental monitoring, pharmaceutical development, and clinical diagnostics, high-performance liquid chromatography (HPLC), gas chromatography-mass spectrometry (GC-MS), and liquid chromatography-tandem mass spectrometry (LC-MS/MS) represent the gold standard for precise quantitative analysis. These techniques provide unparalleled sensitivity, specificity, and reliability for detecting everything from small drug molecules to endogenous metabolites [17] [18]. However, this analytical precision comes with significant operational burdens that impact research efficiency and accessibility. The interacting triangle of sensitivity, specificity, and throughput presents a fundamental challenge for these platforms, particularly as laboratories face increasing pressure to deliver rapid, cost-effective results without compromising data quality [17]. While these techniques dominate analytical science, with PubMed showing a yearly publication rate of approximately 3,042 for GC-MS and 3,908 for LC-MS in recent decades, their limitations become particularly pronounced in applications requiring rapid screening, field deployment, or analysis of large sample cohorts [19].
This technical guide examines the specific limitations of conventional detection methods, focusing specifically on the high costs and time-intensive processes that constrain their implementation. By quantifying these limitations and presenting detailed experimental protocols, we provide researchers with a comprehensive framework for evaluating analytical approaches within the broader context of emerging contaminant and biosensing technology research.
Quantitative Analysis of Operational Limitations
Financial and Temporal Costs of Conventional Platforms
The operational expenses of maintaining chromatographic and mass spectrometric platforms extend far beyond initial instrument acquisition. Consumable costs, solvent disposal, analyst time, and instrument maintenance create substantial financial burdens that limit accessibility for many laboratories.
Table 1: Operational Cost and Time Analysis for Chromatographic Methods
| Parameter | HPLC | UHPLC | UPLC | GC-MS | LC-MS/MS |
|---|---|---|---|---|---|
| Typical Analysis Time (per sample) | 20-30 min | 10-15 min | 3-7 min | 20-60 min | 10-20 min |
| Mobile Phase Consumption (per injection) | ~1.8 mL | ~0.75 mL | ~0.41 mL | N/A | ~1.0 mL |
| Solvent Cost (per 10,000 injections) | ~$13,500 | ~$2,800 | ~$1,000 | N/A | ~$7,500 |
| Sample Preparation Time | 30-60 min | 30-60 min | 30-60 min | 60-120 min (derivatization) | 30-60 min |
| Method Development Timeline | 2-4 weeks | 2-4 weeks | 2-4 weeks | 3-6 weeks | 4-8 weeks |
| Operator Skill Requirements | High | High | High | Very High | Very High |
As demonstrated in a comprehensive study modernizing a USP monograph method for naproxen sodium tablets, transitioning from conventional HPLC to ultra-performance liquid chromatography (UPLC) resulted in a 13-fold decrease in mobile phase consumption and an 8-fold reduction in analysis time over the column's lifetime (10,000 injections) [20]. This translated to a potential cost savings of approximately $10,000 and a 48-day reduction in testing time, highlighting the significant inefficiencies inherent in older HPLC technologies [20].
Throughput and Accessibility Constraints
Despite their analytical superiority, conventional methods face substantial throughput limitations compared to emerging biosensing technologies. The sample preparation, chromatographic separation, and data analysis steps create significant bottlenecks in high-volume testing scenarios.
Table 2: Throughput Limitations of Conventional Detection Methods
| Limitation Factor | Impact on Analysis | Consequence for Research |
|---|---|---|
| Sample Preparation Complexity | Extensive extraction, purification, and concentration required | Limits sample processing to ~40-50 samples per technologist daily |
| Chromatographic Run Times | Typical LC runs: 10-30 minutes; GC runs: 20-60 minutes | Maximum throughput of 50-100 samples per instrument daily |
| Post-analysis Data Processing | Complex peak integration, calibration curves, quality control | Adds 20-30% additional time to total analysis |
| Matrix Effects | Requires method optimization and standard addition approaches | Redounds to increased development time and validation requirements |
| Regulatory Compliance | Extensive documentation for regulated analyses (GMP/GLP) | Can double the hands-on time per sample batch |
The fundamental architecture of these systems creates inherent throughput restrictions. As noted in evaluation of clinical LC-MS/MS applications, while sample throughput is higher than for conventional HPLC or GC-MS, it "lags behind automated immunoassays" [17]. This throughput limitation becomes particularly problematic in applications requiring rapid results, such as clinical diagnostics or emergency environmental monitoring, where time-sensitive decisions depend on analytical data.
Detailed Experimental Protocols Highlighting Methodological Complexities
Protocol: Comparative Analysis of PPCPs in Surface Water Using LC-MS and GC-MS
This published methodology for analyzing pharmaceuticals and personal care products (PPCPs) in environmental samples illustrates the extensive sample preparation and dual-method requirements that contribute to high analytical costs and extended timelines [21].
Sample Collection and Preservation:
- Collect 1L surface water samples in amber glass bottles pre-rinsed with LC-MS grade methanol.
- Acidify samples to pH 2.5 with hydrochloric acid to preserve analyte integrity.
- Store at 4°C and extract within 48 hours of collection to prevent degradation.
Solid-Phase Extraction (SPE) Procedure:
- Condition ENVI-Disk C18 SPE disks (47mm diameter) with 10mL acetonitrile, 10mL methanol, and 10mL deionized water with 2-minute equilibration between solvents.
- Load 500mL acidified water sample onto conditioned SPE disk at 0.15-0.2mL/min flow rate.
- Dry disks under vacuum for 20 minutes after sample loading.
- Elute analytes with 10mL acetonitrile, allowing 5 minutes residence time before elution at 0.1mL/min.
- Collect eluate in graduated conical tubes and concentrate under nitrogen to <1mL volume.
- Transfer to autosampler vials for instrumental analysis.
Liquid-Liquid Extraction (Alternative Procedure):
- Transfer 2000mL sample to separatory funnel.
- Add 100mL methylene chloride and shake manually for 1 minute.
- Collect organic layer and repeat extraction twice (total 300mL methylene chloride).
- Combine extracts in round-bottom flask and concentrate by rotary evaporation.
- Transfer concentrated extract to graduated tube and adjust to 1mL final volume.
Instrumental Analysis Conditions: HPLC-TOF-MS Parameters:
- Column: Agilent Zorbax Eclipse Plus C18 (150mm × 2.1mm, 3.5μm)
- Mobile Phase: (A) Water with 1% formic acid; (B) Acetonitrile with 1% formic acid
- Gradient: 20-80% B over 20 minutes, hold at 80% B for 5 minutes
- Flow Rate: 0.3mL/min
- Injection Volume: 3μL
- Detection: TOF-MS with ESI positive mode, capillary voltage 3300V
GC-MS Parameters:
- Column: DB-5MS (30m × 0.25mm, 0.5μm film thickness)
- Temperature Program: 150°C (hold 5min), ramp to 300°C at 10°C/min, hold 10min
- Carrier Gas: Helium at 0.8mL/min
- Injection: 1μL splitless mode
- Detection: Electron ionization source, selected ion monitoring (SIM) mode
This comparative study found that HPLC-TOF-MS generally provided lower detection limits than GC-MS for most PPCPs, though optimal detection varied by compound, necessitating both techniques for comprehensive analysis [21]. The requirement for multiple analytical approaches further compounds the cost and time investments in environmental monitoring campaigns.
Protocol: USP Monograph Modernization for Naproxen Sodium Tablets
This pharmaceutical quality control protocol demonstrates how method modernization can alleviate, but not eliminate, the inherent cost and time burdens of chromatographic analysis [20].
Standard and Sample Preparation:
- Crush five naproxen sodium tablets (220mg each) and dissolve in 1L mobile phase.
- Filter through 0.2μm nylon filter and dilute 10:1 with mobile phase to 0.11mg/mL final concentration.
- Prepare reference standard from neat naproxen sodium powder at identical concentration.
HPLC Conditions (USP Monograph Method):
- Column: XBridge BEH C8, 5μm, 4.6 × 150mm
- Mobile Phase: Acetonitrile:water:acetic acid (45:54:1 v/v/v)
- Flow Rate: 1.20mL/min
- Temperature: 30°C
- Detection: UV at 254nm
- Injection Volume: 20μL
- Run Time: 25 minutes
UHPLC Conditions (Modernized Method):
- Column: XBridge BEH C8, 2.5μm, 3.0 × 75mm
- Mobile Phase: Identical to HPLC method
- Flow Rate: 1.02mL/min
- Injection Volume: 4.3μL
- Run Time: 6 minutes (76% reduction)
UPLC Conditions (Optimized Modernization):
- Column: CORTECS UPLC C8, 1.6μm, 2.1 × 50mm
- Mobile Phase: Identical to HPLC method
- Flow Rate: 0.74mL/min
- Injection Volume: 1.4μL
- Run Time: 3 minutes (88% reduction)
This systematic modernization approach maintained chromatographic resolution while dramatically reducing solvent consumption from ~1.8mL per injection (HPLC) to ~0.41mL per injection (UPLC), demonstrating the potential for cost savings through method optimization [20]. Despite these improvements, the fundamental requirement for sophisticated instrumentation and skilled operators remains a barrier to widespread implementation.
Visualization of Methodological Limitations and Relationships
Analytical Limitations in Conventional Detection Methods
This diagram illustrates the interconnected nature of limitations in conventional detection technologies, demonstrating how financial, temporal, and technical constraints collectively impact research applications and resource allocation in analytical laboratories.
The Scientist's Toolkit: Essential Research Reagents and Materials
Table 3: Essential Research Reagents for Conventional Detection Methods
| Reagent/Material | Function | Specific Application Examples |
|---|---|---|
| LC-MS Grade Solvents | Mobile phase composition, sample reconstitution | Acetonitrile with 1% formic acid for positive ESI mode [21] |
| Solid-Phase Extraction Cartridges | Sample clean-up and analyte concentration | ENVI-Disk C18 for PPCP extraction from water samples [21] |
| Chromatographic Columns | Analyte separation by chemical properties | XBridge BEH C8 for pharmaceutical analysis [20] |
| Analytical Standards | Method calibration and quantification | Naproxen sodium reference standard for USP monograph methods [20] |
| Isotope-Labeled Internal Standards | Compensation for matrix effects and recovery variation | Heavy stable isotopes (²H, ¹³C, ¹⁵N) for accurate LC-MS/MS quantification [19] |
| Derivatization Reagents | Analyte functionalization for enhanced detection | Silanation reagents for GC-MS analysis of polar compounds |
| Mobile Phase Additives | Modify selectivity and improve ionization | Formic acid, ammonium formate, acetic acid for LC-MS applications [18] [20] |
The selection of appropriate reagents and materials represents a significant portion of the operational costs for conventional detection methods. LC-MS grade solvents can cost 5-10 times more than standard HPLC-grade solvents, while stable isotope-labeled internal standards represent a substantial recurring expense, with individual compounds often exceeding $500 per milligram [19]. These material requirements create significant financial barriers, particularly for resource-limited laboratories or long-term monitoring studies with extensive sample numbers.
The documented limitations of HPLC, GC-MS, and LC-MS/MS platforms - specifically their substantial operational costs, time-intensive procedures, and technical complexity - highlight the critical need for alternative detection strategies in modern analytical science. While these conventional methods provide exceptional sensitivity and specificity for targeted quantification, their inherent constraints limit application in scenarios requiring rapid analysis, field deployment, or high-throughput screening.
These limitations directly inform the research priorities for emerging biosensing technologies, which aim to maintain analytical performance while dramatically reducing both cost and time requirements. The development of point-of-care testing devices, miniaturized analytical platforms, and automated biosensing systems represents a direct response to the documented constraints of conventional chromatographic and mass spectrometric methods [22] [23]. As the field advances, the comparative assessment of analytical performance must consider not only sensitivity and specificity, but also the practical implementation barriers that ultimately determine technology accessibility and application scope.
Understanding these limitations provides researchers with a framework for selecting appropriate analytical methodologies based on application-specific requirements, resource availability, and operational constraints. This evaluation is particularly relevant in the context of emerging contaminant research, where comprehensive environmental assessment requires analysis of diverse chemical classes across extensive spatial and temporal scales, creating analytical demands that often exceed the practical implementation capabilities of conventional detection platforms alone.
Biosensors have emerged as transformative analytical devices that synergistically combine a biological recognition element with a physicochemical detector, enabling the sensitive and specific detection of a wide range of analytes [24]. These devices are revolutionizing multiple fields, including medical diagnostics, environmental monitoring, food safety, and biodefense, by providing rapid, cost-effective, and decentralized testing capabilities that traditional laboratory methods cannot match [25]. The global biosensors market, valued at $26.75 billion in 2022, is projected to reach $45.95 billion by 2030, reflecting a compound annual growth rate of 7.00% [25].
Within the specific context of monitoring emerging contaminants (ECs) in water environments, biosensors offer a potent technological solution to a pressing global challenge [22]. ECs comprise synthetic or natural chemical compounds from anthropogenic activities that are not routinely monitored or regulated, yet pose significant threats to ecosystems and human health even at trace concentrations [26]. These include pharmaceuticals, endocrine-disrupting agents, pesticides, personal care products, and per-and polyfluoroalkyl substances (PFAS) [22]. The unique capability of biosensors to provide real-time, on-site monitoring positions them as indispensable tools for comprehensive environmental surveillance and protection [26].
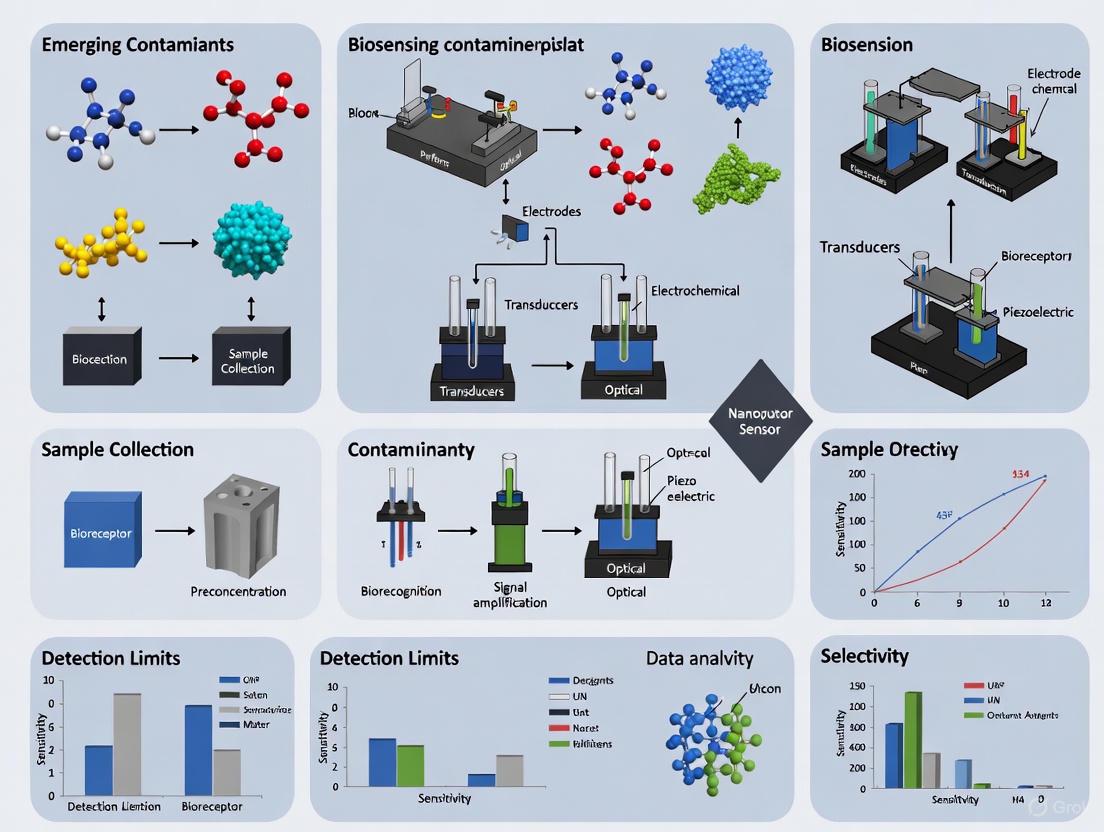
Core Components of Biosensors
All biosensors consist of three fundamental components that work in concert to detect and quantify target analytes: a biological recognition element, a transducer, and a signal processing system [24].
Biological Recognition Elements
The bioreceptor is the specificity-determining component that interacts selectively with the target analyte. The most common types include:
- Enzymes: These biocatalysts recognize substrates through specific binding sites and catalyze reactions that produce measurable products [26]. Enzyme-based biosensors typically operate through three mechanisms: (1) the enzyme metabolizes the analyte; (2) the enzyme is inhibited by the analyte; or (3) the analyte alters enzyme characteristics [26].
- Antibodies: These immunoglobulins provide high-affinity binding to specific antigens through precise molecular complementarity, forming the basis of immunosensors [26].
- Nucleic Acids: Synthetic DNA or RNA aptamers, selected through Systematic Evolution of Ligands by Exponential Enrichment (SELEX), fold into specific three-dimensional structures that bind targets with high affinity and specificity [26].
- Whole Cells: Microorganisms like bacteria, fungi, and algae serve as integrated sensing systems containing both receptors and transducers [26]. These offer advantages of robustness and self-replication capabilities [26].
Transducers
Transducers convert the biological response into a quantifiable electrical signal. The primary transduction mechanisms include:
- Electrochemical Transducers: These measure changes in electrical properties (current, potential, or impedance) resulting from biological recognition events [24]. They dominate the commercial biosensor market due to their simplicity, low cost, and high sensitivity [27].
- Optical Transducers: These detect changes in light properties (absorbance, fluorescence, reflectance, or luminescence) induced by analyte-bioreceptor interactions [24].
- Piezoelectric Transducers: These measure changes in mass or resonance frequency when target molecules bind to the sensor surface [24].
- Thermal Transducers: These detect heat absorbed or released during biochemical reactions [24].
Signal Processing Systems
This component amplifies, processes, and displays the transducer output in a user-readable format, often incorporating data analysis algorithms and user interfaces [24]. Advanced systems now integrate artificial intelligence for enhanced signal interpretation and quantification [28].
Table 1: Core Components of Biosensors and Their Functions
| Component | Subtypes | Primary Function | Examples in Environmental Monitoring |
|---|---|---|---|
| Bioreceptor | Enzymes | Specific catalytic recognition | Laccase for phenol detection [22] |
| Antibodies | High-affinity antigen binding | Immunosensors for atrazine [22] | |
| Nucleic Acids (Aptamers) | Target-selective folding | DNA aptamers for estradiol [26] | |
| Whole Cells | Integrated sensing machinery | Aliivibrio fischeri for toxicity screening [28] | |
| Transducer | Electrochemical | Measure electrical property changes | Heavy metal detection in water [26] |
| Optical | Detect light property alterations | Fluorescence aptasensors for estradiol [22] | |
| Piezoelectric | Sense mass/resonance frequency shifts | Virus and bacteria detection [24] | |
| Thermal | Monitor heat from reactions | Food safety testing [24] | |
| Signal Processor | Hardware/Software | Amplify, process, and display signals | Smartphone apps with AI algorithms [28] |
The Critical Advantage: Real-Time, On-Site Monitoring
The convergence of biosensors with advanced materials science and digital technologies has unlocked their paramount advantage: the capability for real-time, on-site monitoring that transcends the limitations of conventional analytical methods.
Limitations of Conventional Methods
Traditional techniques for detecting environmental contaminants, including high-performance liquid chromatography (HPLC), gas chromatography (GC), and mass spectrometry (MS), require sophisticated instrumentation, complex sample preparation, time-consuming protocols, and skilled personnel [26]. These methods generate historical data rather than real-time information, delaying decision-making and potentially exacerbating contamination problems [22]. Additionally, the high cost and centralized nature of these techniques restrict their application for routine monitoring and rapid screening across multiple locations [26].
The Biosensor Paradigm Shift
Biosensors fundamentally address these limitations by offering:
- Rapid Analysis: Results are typically available within minutes rather than hours or days, enabling immediate intervention when contaminants are detected [26].
- Portability and Field Deployment: Miniaturized biosensors can be deployed directly at sampling sites, eliminating the need for sample transport and preservation [28].
- Continuous Monitoring Capability: With appropriate immobilization strategies, biosensors can provide continuous, real-time data on contaminant levels, enabling dynamic assessment of environmental quality [26].
- User-Friendly Operation: Simplified protocols and integrated data processing make biosensors accessible to non-specialists, potentially enabling citizen science applications [28].
- Cost-Effectiveness: Lower per-test costs and reduced infrastructure requirements make extensive monitoring programs economically feasible [25].
Technological Enablers
Several technological advancements have been crucial for realizing the potential of biosensors for real-time, on-site monitoring:
- Miniaturization and Microfluidics: The development of lab-on-a-chip and microfluidic systems has enabled the creation of compact, portable biosensing platforms that require minimal sample volumes [27].
- IoT Integration: The convergence of biosensors with Internet of Things (IoT) technology allows wireless data transmission to cloud platforms, facilitating remote monitoring and centralized data management [29].
- Advanced Materials: Nanomaterials, including metal-organic frameworks (MOFs) and gold nanoparticles, enhance sensitivity and stability while enabling novel immobilization approaches [22].
- Artificial Intelligence: AI algorithms improve signal processing, enable multiplexed detection, and enhance quantification accuracy, as demonstrated in smartphone-based biosensors [28].
Table 2: Comparison of Biosensor Performance for Detecting Emerging Contaminants in Water
| Contaminant Category | Biosensor Type | Detection Mechanism | Detection Limit | Analysis Time | Reference |
|---|---|---|---|---|---|
| Cyanotoxin (Microcystin-LR) | Whole-cell (A. fischeri) | Bioluminescence inhibition | 0.23 ppb | 15 minutes | [28] |
| Heavy Metals (Cobalt) | Whole-cell (Engineered bacteria) | Fluorescence (eGFP) | Low concentrations in complex food matrices | Real-time | [30] |
| Endocrine Disruptors (17β-estradiol) | Aptasensor | Fluorescence | Not specified | Real-time capability | [22] |
| Antibiotics (Ciprofloxacin) | Immunosensor | Impedimetric | 10 pg/mL | Rapid | [26] |
| Pesticides (Atrazine) | Immunosensor | Electrochemical | Not specified | Rapid | [22] |
| General Toxicity | Whole-cell (A. fischeri) | Bioluminescence inhibition | Various toxicants | 15 minutes | [28] |
Experimental Protocols in Biosensing
To illustrate the practical implementation of biosensors for environmental monitoring, two representative experimental methodologies are detailed below.
Whole-Cell Biosensor for Cobalt Detection in Food Matrices
A recent study developed a whole-cell bacterial system to detect cobalt contamination along the pasta production chain [30].
Protocol:
- Bacterial Engineering: E. coli cells were engineered with a plasmid containing the enhanced green fluorescent protein (eGFP) gene under the control of the UspA (universal stress protein) promoter.
- Response Testing: The responsiveness of four stress-responsive gene promoters (DnaK, GroE, UspA, and ZntA) to cobalt was evaluated, with the UspA promoter demonstrating optimal activation.
- Sample Exposure: Food matrices derived from durum wheat seeds were exposed to the engineered bacterial biosensors with and without exogenous cobalt addition.
- Signal Detection: Fluorescence emission was measured following cobalt exposure, indicating the presence of contamination.
Key Findings: The system successfully detected low cobalt concentrations within complex food matrices. The fluorescence signal was predominantly observed in bran and fine bran samples, confirming these seed components as primary accumulation sites for contaminants [30].
Smartphone-Based Paper Biosensor for Water Toxicity Monitoring
An innovative all-in-one paper biosensor was developed for comprehensive water toxicity assessment using bioluminescent bacteria [28].
Protocol:
- Bacteria Immobilization: Aliivibrio fischeri cells were entrapped in a 0.5% w/v agarose hydrogel matrix within predefined hydrophilic wells on wax-printed paper.
- Sensor Design: A circular flower-like paper sensor (30mm diameter) was created with seven hydrophilic wells - six for calibration standards and one for the test sample.
- Assay Procedure:
- 30μL of standard solutions or samples were dispensed into appropriate wells.
- Incubation proceeded for 15 minutes at room temperature.
- The sensor was placed in a cardboard dark box to eliminate ambient light interference.
- Imaging was performed using a smartphone camera with 30-second integration time at ISO1600.
- Data Analysis: Images were analyzed using a custom Android application ("Scentinel") incorporating artificial intelligence algorithms to convert bioluminescence signals into quantitative toxicity measurements.
Key Findings: The biosensor detected microcystin-LR at 0.23 ppb and demonstrated effectiveness with various smartphone models, highlighting its potential for widespread citizen science applications [28].
Visualization of Biosensor Principles
The fundamental working principle of a biosensor, from analyte recognition to signal output, can be visualized through the following workflow:
Diagram 1: Fundamental biosensor operational workflow showing the sequence from sample introduction to result generation.
The specific mechanism of an enzyme-based biosensor, one of the most common configurations for environmental monitoring, can be represented as follows:
Diagram 2: Enzyme-based biosensor mechanism showing the catalytic conversion of analyte to measurable product.
The Researcher's Toolkit: Essential Reagents and Materials
Successful development and implementation of biosensors for environmental monitoring requires specific research reagents and materials. The following table details essential components from representative studies:
Table 3: Essential Research Reagents and Materials for Biosensor Development
| Reagent/Material | Function/Application | Example in Context | Reference |
|---|---|---|---|
| Aliivibrio fischeri | Bioluminescent whole-cell bioreporter for toxicity assessment | Water toxicity screening via luminescence inhibition | [28] |
| Agarose Hydrogel | Immobilization matrix for biological components | Entrapment of A. fischeri in paper biosensor | [28] |
| Wax-Printed Paper | Low-cost, sustainable sensor substrate | Creation of hydrophilic wells for sample containment | [28] |
| Engineered Bacterial Strains | Customizable whole-cell biosensors with specific promoters | E. coli with UspA promoter for cobalt detection | [30] |
| Aptamers | Synthetic nucleic acid recognition elements | Selective binding of estradiol and other contaminants | [22] [26] |
| Gold Nanoparticles | Signal amplification and immobilization platform | Enhanced sensitivity in electrochemical immunosensors | [22] |
| Antibodies | High-specificity immunorecognition elements | Immunosensors for antibiotics like ciprofloxacin | [26] |
| Nanomaterials (MOFs, Graphene) | Enhanced sensitivity and stability | Covalent organic frameworks for reticular electrochemiluminescence | [30] |
Biosensors represent a paradigm shift in environmental monitoring, particularly for addressing the global challenge of emerging contaminants in water systems. Their core architecture—integrating biological recognition elements with physical transducers and signal processors—enables unprecedented capabilities for real-time, on-site detection that traditional laboratory methods cannot match. The experimental protocols and essential research tools detailed in this review provide a foundation for researchers developing next-generation biosensing platforms.
While challenges remain in sensor stability, standardization, and integration, ongoing advancements in nanotechnology, materials science, artificial intelligence, and IoT connectivity are rapidly addressing these limitations. The future trajectory of biosensor technology points toward increasingly sophisticated, multiplexed, and autonomous systems that will fundamentally transform how we monitor and protect environmental and public health. As these technologies continue to mature, they will play an indispensable role in global efforts to ensure water safety, sanitation, and sustainability.
Engineered Detection: A Deep Dive into Biosensor Types and Their Real-World Applications
The increasing presence of emerging contaminants (ECs)—including pharmaceuticals, personal care products, pesticides, and industrial chemicals—in global water resources represents a significant threat to environmental and human health [31] [23]. These compounds persist in the environment at low concentrations, exhibiting complex chemical structures and resistance to traditional degradation processes, making them challenging to detect using conventional analytical methods [32]. Traditional detection techniques such as high-performance liquid chromatography (HPLC) and mass spectrometry (MS) offer sensitivity but suffer from significant limitations including high costs, complex sample preparation, time-consuming protocols, and labor-intensive processes that hinder real-time, on-site monitoring [31] [22].
Biosensors have emerged as powerful analytical tools that address these limitations by combining a biological recognition element (bioreceptor) with a transducer that converts the biological response into a quantifiable signal [23]. The selection of an appropriate bioreceptor is paramount, as it dictates the specificity, sensitivity, and overall performance of the biosensing system [33]. Within the context of detecting ECs, four primary bioreceptor classes have demonstrated significant utility: enzyme-based systems, antibody-based immunosensors, aptamer-based aptasensors, and whole-cell biosensors [31]. Each category offers distinct advantages and limitations, making them suitable for different applications within environmental monitoring, clinical diagnostics, and food safety [34]. This technical guide provides an in-depth examination of these bioreceptor classes, focusing on their operational mechanisms, experimental protocols, and applications in detecting emerging contaminants to advance biosensing technology research.
Bioreceptor Types: Mechanisms and Characteristics
Enzyme-Based Biosensors
Enzyme-based biosensors utilize the specific catalytic activity of enzymes to recognize and convert target analytes into measurable products [31]. The high specificity of enzymes for their substrates makes them ideal for detecting specific classes of ECs, particularly pesticides and phenolic compounds [22]. The operational mechanism relies on three principal biorecognition strategies: (1) the direct metabolism of the target analyte by the enzyme, with the analyte concentration correlating to the rate of catalytic transformation; (2) enzyme inhibition by the analyte, where the analyte concentration correlates with reduced product formation; and (3) modulation of enzyme characteristics by the analyte, where measurable changes in enzyme properties enable quantification [31].
The catalytic reaction in enzyme-based biosensors typically generates electrical, optical, or thermal signals that can be transduced for quantification [31]. For instance, hydrolytic enzymes are frequently employed in colorimetric biosensing platforms for EC detection, where enzyme-catalyzed reactions produce measurable changes in color or absorbance [22]. A significant advancement in this field involves the development of nanobiohybrid systems, which integrate enzymes with nanomaterials like metal-organic frameworks or gold nanoparticles to enhance enzyme stability, loading capacity, and detection sensitivity within complex water matrices [22]. Despite these advancements, enzyme-based biosensors face challenges related to enzyme instability under varying environmental conditions, high purification costs, and limited reusability [34].
Antibody-Based Biosensors (Immunosensors)
Antibody-based biosensors, or immunosensors, exploit the high specificity and affinity of antibodies for target antigen recognition [31] [35]. These systems primarily employ immunoglobulin G (IgG) and its derivatives, including antigen-binding fragments (Fab, Fab'), single-chain variable fragments (scFv), and single-chain antibodies (scAb) [35]. The design of immunosensors critically depends on antibody size and immobilization strategy to ensure proper orientation and maximize antigen-binding site accessibility [35].
Immunosensors typically operate in direct, sandwich, or competitive assay formats [35]. Direct immunosensors measure the physical changes (e.g., impedance, refractive index, mass) resulting from antigen-antibody binding events [31]. For example, an impedimetric immunosensor developed for ciprofloxacin (CIP) antibiotic detection achieved a detection limit of 10 pg/mL by measuring impedance changes upon antibody-antigen complex formation [31]. Sandwich immunosensors, offering heightened sensitivity and specificity, require the target analyte to possess multiple epitopes for simultaneous binding by capture and detection antibodies [35]. Competitive formats are often employed for small molecules with single epitopes, where labeled and unlabeled antigens compete for limited antibody-binding sites [22]. While immunosensors provide exceptional specificity, they face limitations including antibody sensitivity to denaturation, batch-to-batch variability, and the inability to develop antibodies against non-immunogenic or toxic targets [35] [36].
Aptamer-Based Biosensors (Aptasensors)
Aptasensors utilize synthetic single-stranded DNA or RNA oligonucleotides as recognition elements, selected through Systematic Evolution of Ligands by Exponential Enrichment (SELEX) for high-affinity binding to specific targets [31] [36]. Aptamers fold into unique three-dimensional structures that recognize diverse analytes, from small molecules to entire cells, through mechanisms involving π-π stacking, van der Waals forces, and hydrogen bonding [31]. Their chemical synthesis, thermal stability, and ability to renature after denaturation offer significant advantages over antibodies, including minimal batch-to-batch variation and reduced production costs [36].
Aptasensors employ various signal transduction mechanisms, including optical, electrochemical, and piezoelectric techniques [31]. Colorimetric aptasensors frequently utilize gold nanoparticle (AuNP) aggregation or enzyme-mimicking DNAzymes for signal generation [36]. In AuNP-based systems, aptamer binding prevents or induces nanoparticle aggregation in salt solutions, resulting in visible color changes from red to blue [36]. DNAzymes, such as hemin-binding G-quadruplexes, provide peroxidase-like activity without requiring natural enzymes, enhancing biosensor stability and cost-effectiveness [36] [37]. Aptasensors can be further engineered into aptazymes, where aptamer-ligand interactions allosterically regulate DNAzyme activity, creating switchable biosensing platforms [37]. Despite their promise, aptasensors require further development to improve performance in complex biological fluids and achieve widespread commercial adoption [36] [37].
Whole-Cell Biosensors
Whole-cell biosensors employ microorganisms (e.g., bacteria, fungi, algae) as integrated sensing elements that contain both recognition and transduction components [31]. These systems leverage inherent biological processes such as metabolic activity, stress responses, gene expression regulation, and detoxification pathways to detect target contaminants [31]. A key advantage lies in their ability to self-replicate, potentially lowering costs and enabling long-term monitoring through self-regeneration of biorecognition elements [31].
Microbial cells can be genetically engineered through genomic editing or plasmid introduction to tailor sensing specificity and response mechanisms [31]. For example, researchers have developed Escherichia coli-based biosensors incorporating quorum-sensing systems that express reporter proteins (e.g., green fluorescent protein) upon detecting specific bacterial pathogens or pyrethroid insecticides, achieving detection limits as low as 3 ng/mL [31] [23]. Whole-cell biosensors demonstrate particular utility in environmental monitoring for detecting heavy metals, pesticides, and organic contaminants, offering advantages in robustness across variable conditions and simplified handling requirements compared to other biosensor types [31].
Table 1: Comparative Analysis of Biosensor Bioreceptor Types
| Parameter | Enzyme-Based | Antibody-Based | Aptamer-Based | Whole-Cell Based |
|---|---|---|---|---|
| Bioreceptor | Enzymes | Antibodies/Fragments | DNA/RNA Aptamers | Microorganisms |
| Selection Method | Natural/Engineering | Hybridoma/Recombinant | SELEX | Genetic Engineering |
| Affinity (Kd) | Variable | 0.1-100 nM [36] | 0.1-100 nM [36] | Variable |
| Production Cost | High | High | Low | Moderate |
| Stability | Moderate; sensitive to conditions | Low; sensitive to denaturation | High; thermal renaturation | Moderate; requires viability |
| Development Time | Weeks-months | Months | Weeks (SELEX) | Weeks-months |
| Key Advantage | Catalytic amplification | High specificity & affinity | Chemical stability & design flexibility | Functional response & self-replication |
| Primary Limitation | Limited target scope | Batch variability, immunogenicity required | Complex sample interference | Longer response time, viability maintenance |
| Example Detection Limit | Varies by enzyme | Ciprofloxacin: 10 pg/mL [31] | VEGF165: 0.3 pM [36] | Pyrethroid: 3 ng/mL [31] |
Experimental Protocols for Biosensor Development
Enzyme-Based Biosensor Protocol for Pesticide Detection
Objective: Detect organophosphorus pesticides via acetylcholinesterase (AChE) inhibition.
Materials:
- Acetylcholinesterase (AChE): Primary bioreceptor that catalyzes substrate hydrolysis.
- Acetylthiocholine (ATCh): Enzyme substrate.
- 5,5'-Dithio-bis-(2-nitrobenzoic acid) (DTNB): Chromogenic agent producing yellow product with thiocholine.
- Electrochemical transducer: For amperometric detection of enzymatic products.
- Phosphate buffer (0.1 M, pH 7.4): Optimal pH for enzyme activity.
- Nanobiohybrid material (e.g., AuNPs/MOFs): Enhance enzyme stability and signal amplification.
Procedure:
- Enzyme Immobilization: Co-immobilize AChE with nanobiohybrid material on electrode surface using glutaraldehyde crosslinking or physical adsorption.
- Baseline Measurement: Incubate biosensor with ATCh substrate in DTNB-containing buffer. Measure initial enzymatic activity via amperometric current (oxidation of thiocholine) or absorbance at 412 nm.
- Inhibition Phase: Expose biosensor to sample containing target pesticide for 10-15 minutes. Organophosphorus compounds irreversibly inhibit AChE, reducing enzyme activity.
- Post-Inhibition Measurement: Re-introduce ATCh/DTNB solution. Measure residual enzymatic activity.
- Quantification: Calculate percentage inhibition relative to baseline. Correlate inhibition percentage to pesticide concentration using calibration curve.
Validation: Compare results with GC-MS reference method for accuracy verification.
Aptamer-Based Colorimetric Biosensor Protocol for VEGF165 Detection
Objective: Detect vascular endothelial growth factor (VEGF165) using gold nanoparticle (AuNP) aggregation.
Materials:
- DNA Aptamer: Specific to VEGF165 target protein.
- Gold Nanoparticles (13 nm): Signal generation via color change.
- Sodium Chloride (NaCl): Induces AuNP aggregation.
- Phosphate buffer (10 mM, pH 7.4): Reaction medium.
- VEGF165 Standards: For calibration curve generation.
Procedure:
- Aptamer Functionalization: Incubate AuNPs with VEGF165-specific aptamers (30 min, room temperature). Aptamers adsorb onto AuNP surface via non-specific interactions.
- Salt-Induced Aggregation Optimization: Titrate NaCl concentration to determine minimal salt concentration causing controlled aggregation of unmodified AuNPs (red to blue color shift).
- Sample Incubation: Mix aptamer-functionalized AuNPs with sample/standard containing VEGF165 (15 min incubation). Target binding induces aptamer conformation change, protecting AuNPs from salt-induced aggregation.
- Colorimetric Reaction: Add optimized NaCl concentration to reaction mixture. Incubate 5 min.
- Signal Measurement: Record UV-Vis spectra (450-650 nm) or capture image for digital color analysis. Calculate absorbance ratio (A520/A620). Increased ratio indicates greater target concentration.
- Quantification: Generate calibration curve with VEGF165 standards (0.1-100 pM). Apply linear regression for unknown sample quantification.
Validation: Assess specificity against non-target proteins (e.g., bovine serum albumin).
Table 2: Research Reagent Solutions for Featured Experiments
| Reagent/Chemical | Function in Experiment | Specifications/Alternatives |
|---|---|---|
| Acetylcholinesterase (AChE) | Primary bioreceptor for pesticide detection | Source: Electric eel; Alternatives: Human recombinant AChE |
| DNA Aptamer (VEGF165-specific) | Molecular recognition element for target protein | Sequence: Custom synthesized; Modification: 5'-thiol for AuNP conjugation |
| Gold Nanoparticles (AuNPs) | Colorimetric signal transducer | Size: 13-20 nm; Characterization: UV-Vis λmax ~520 nm |
| 5,5'-Dithio-bis-(2-nitrobenzoic acid) (DTNB) | Chromogenic agent for thiocholine detection | Working concentration: 0.3-0.5 mM in buffer |
| Acetylthiocholine (ATCh) | Enzyme substrate for AChE | Alternatives: Acetylcholine with choline oxidase |
| Nanobiohybrid Materials | Enzyme stabilization & signal enhancement | Types: Metal-organic frameworks (MOFs), graphene oxide |
| Phosphate Buffered Saline (PBS) | Reaction medium for biological components | Concentration: 10-100 mM; pH: 7.2-7.4 |
Bioreceptor diversity provides a robust foundation for developing advanced biosensing platforms capable of addressing the complex challenges associated with emerging contaminant detection. Each bioreceptor class—enzyme-based, antibody-based, aptamer-based, and whole-cell based—offers unique advantages that can be leveraged for specific applications in environmental monitoring, clinical diagnostics, and food safety [31] [34]. The ongoing refinement of these biological recognition elements, coupled with advancements in transducer technologies and nanomaterial integration, continues to push the boundaries of biosensor performance, particularly in sensitivity, specificity, and operational stability [22] [23].
Future research directions should focus on several key areas to advance the field. The development of hybrid biosensing systems that combine multiple bioreceptor types could capitalize on their complementary advantages, such as the enzymatic signal amplification with aptamer stability [35] [37]. Additionally, the integration of artificial intelligence and machine learning with biosensor data analysis holds significant potential for improving detection accuracy, enabling multiplexed analysis, and facilitating real-time monitoring in complex sample matrices [34]. The continued evolution of point-of-care testing (POCT) devices, including paper-based and microfluidic platforms, will enhance the portability and accessibility of biosensing technologies for on-site environmental monitoring [33] [22]. As these technologies mature, standardization and rigorous validation against established reference methods will be crucial for their widespread adoption and implementation in regulatory frameworks aimed at safeguarding environmental and public health [32] [23].
The rapid and sensitive detection of emerging contaminants (ECs)—including pharmaceuticals, pesticides, microplastics, and endocrine-disrupting chemicals—is a critical challenge in environmental monitoring and public health protection. Biosensors, which integrate a biological recognition element with a transducer, have emerged as powerful analytical tools that offer a viable alternative to traditional laboratory-based methods such as chromatography and mass spectrometry. The performance and applicability of these biosensors are fundamentally determined by their signal transduction mechanism. This whitepaper provides an in-depth technical examination of the three principal transducer platforms—electrochemical, optical, and piezoelectric—detailing their underlying operational principles, specific experimental protocols, and key performance metrics in the context of EC detection. By synthesizing recent advancements and presenting a curated toolkit of research reagents and materials, this review serves as a comprehensive resource for researchers and industry professionals working to develop next-generation biosensing technologies for environmental surveillance.
Emerging contaminants (ECs) represent a diverse class of unregulated or inadequately monitored synthetic or naturally occurring chemicals that pose a potential threat to ecosystems and human health. Common categories include pharmaceuticals and personal care products (PPCPs), endocrine-disrupting chemicals (EDCs), per- and polyfluoroalkyl substances (PFAS), pesticides, and micro- and nano-plastics (MNPs) [1]. Their persistent occurrence in aquatic environments, even at trace concentrations (ng/L to µg/L), coupled with their complex toxicological profiles, necessitates the development of rapid, sensitive, and on-site monitoring capabilities [31] [9].
Traditional analytical techniques, such as high-performance liquid chromatography (HPLC) and gas chromatography-mass spectrometry (GC-MS), are considered gold standards for EC quantification due to their high sensitivity and accuracy. However, their extensive use for routine monitoring is constrained by high instrumental costs, complex sample preparation, time-consuming protocols, and the need for specialized laboratory facilities and personnel [31] [22] [23]. These limitations hinder real-time, in-situ decision-making, potentially exacerbating contamination events.
Biosensors are analytical devices that integrate a biological recognition element (bioreceptor) with a physicochemical transducer, which converts the biorecognition event into a quantifiable signal [23]. The bioreceptor—which can be an enzyme, antibody, nucleic acid (aptamer), or whole cell—confers high specificity for the target analyte [31]. The transducer, the focus of this whitepaper, is the component that defines the primary readout modality of the biosensor. The integration of advanced materials, particularly nanomaterials, and microfluidic systems has significantly enhanced biosensor performance, leading to improvements in sensitivity, specificity, response time, and portability for on-site detection [38] [23] [39].
Electrochemical Transduction Platforms
Electrochemical biosensors are among the most extensively developed transducers for environmental monitoring, prized for their high sensitivity, portability, low cost, and capacity for miniaturization and real-time analysis [31] [38]. Their operation is based on the detection of electrical changes arising from biochemical reactions occurring at the surface of an electrode.
Fundamental Mechanisms
Electrochemical transducers can be broadly categorized based on the specific electrical parameter measured:
- Amperometry measures the current generated by the oxidation or reduction of an electroactive species during a biochemical reaction at a constant applied potential. The magnitude of the current is directly proportional to the concentration of the target analyte [38].
- Potentiometry measures the potential difference (voltage) between a working electrode and a reference electrode under conditions of zero current. This potential change results from the accumulation of charge or alteration of ion concentration at the electrode surface due to a recognition event [38].
- Impedimetry (or Conductometry) measures changes in the impedance (resistance to alternating current) or conductivity of a solution resulting from the binding of a target analyte to the bioreceptor on the electrode surface. This label-free approach is particularly suitable for monitoring binding events in real-time without the need for redox probes [31] [22].
Experimental Protocol: Impedimetric Immunosensor for Antibiotic Detection
The following protocol outlines the development of an impedimetric immunosensor for the detection of ciprofloxacin (CIP), an antibiotic EC [31].
- Electrode Modification: A gold disk electrode is sequentially cleaned and modified with a self-assembled monolayer (SAM) of a suitable linker molecule (e.g., 4-aminothiophenol) to create a functionalized surface.
- Antibody Immobilization: Anti-ciprofloxacin antibodies are covalently immobilized onto the SAM-functionalized electrode surface using a cross-linking agent such as EDC/NHS (1-Ethyl-3-(3-dimethylaminopropyl)carbodiimide / N-Hydroxysuccinimide).
- Blocking: The remaining active sites on the electrode are "blocked" with a non-reactive protein (e.g., Bovine Serum Albumin, BSA) to prevent non-specific adsorption of other molecules.
- Measurement Setup: The modified electrode is connected to an electrochemical impedance spectrometer and immersed in a redox probe solution, typically 5 mM K₃[Fe(CN)₆]/K₄[Fe(CN)₆] in a suitable buffer (e.g., PBS).
- Baseline Measurement: Electrochemical Impedance Spectroscopy (EIS) is performed over a specified frequency range (e.g., 0.1 Hz to 100 kHz) at a fixed DC potential to record the charge transfer resistance (Rₐ) of the bare, modified electrode.
- Analyte Incubation: The electrode is incubated with the sample containing the target CIP antibiotic for a fixed duration (e.g., 20-30 minutes).
- Target Measurement: The electrode is washed, re-immersed in the redox probe solution, and the EIS measurement is repeated.
- Data Analysis: The increase in Rₐ after analyte incubation, caused by the antibody-antigen binding event hindering electron transfer to the electrode surface, is calculated. The ΔRₐ is plotted against the logarithm of CIP concentration to generate a calibration curve, achieving detection limits as low as 10 pg/mL [31].
Performance Metrics for Electrochemical Biosensors
Table 1: Performance metrics of electrochemical biosensors for detecting various ECs.
| Target EC | Bioreceptor | Electrochemical Method | Linear Range | Detection Limit | Reference |
|---|---|---|---|---|---|
| Ciprofloxacin (Antibiotic) | Antibody | Impedimetry | Not Specified | 10 pg/mL | [31] |
| Polybrominated Diphenyl Ethers (PBDE) | Enzyme (Glucose Oxidase) | Amperometry | Not Specified | 0.014 μg/L | [38] |
| Atrazine (Pesticide) | Antibody | Voltammetry | Not Specified | ~nM range | [22] |
| Heavy Metals (e.g., Hg²⁺, Pb²⁺) | Aptamer / Whole Cell | Potentiometry / Amperometry | 0.5–500 nM (Hg²⁺) | 0.5 nM (Hg²⁺) | [38] |
Diagram 1: Experimental workflow for an impedimetric immunosensor.
Optical Transduction Platforms
Optical biosensors measure changes in the properties of light resulting from the interaction between the bioreceptor and the target analyte. They are highly versatile and can be extremely sensitive, enabling the detection of low-abundance ECs.
Fundamental Mechanisms
Key optical transduction mechanisms include:
- Fluorescence: This method detects the light emitted by a fluorescent label (e.g., a dye, quantum dot) when it is excited by a specific wavelength of light. The presence or concentration of the analyte can modulate the intensity, polarization, or lifetime of the fluorescence. For instance, quantum dot-based fluorescence immunoassays have been developed for multiplex antibiotic detection [31] [34].
- Surface Plasmon Resonance (SPR): SPR sensors detect changes in the refractive index at the surface of a thin metal film (usually gold). When biomolecular binding occurs on the functionalized metal surface, it causes a shift in the resonance angle of reflected light, allowing for real-time, label-free monitoring of binding kinetics [34].
- Colorimetry: This technique measures changes in the absorption of light, often visible as a color change. Enzyme-catalyzed reactions that produce colored products (e.g., horseradish peroxidase with TMB substrate) are commonly used. Colorimetric sensors are simple, low-cost, and well-suited for point-of-care testing (POCT) and paper-based microfluidic devices (μPADs) [22] [39].
- Surface-Enhanced Raman Scattering (SERS): SERS utilizes nanostructured metal surfaces to greatly enhance the weak Raman scattering signal from molecules adsorbed on them. This provides a unique vibrational "fingerprint" of the target analyte, enabling highly sensitive and specific detection [34].
Experimental Protocol: Fluorescent Aptasensor for Mycotoxin Detection
This protocol details the construction of a fluorescent aptasensor for Aflatoxin B1 (AFB1) using a microfluidic platform [39].
- Chip Fabrication: A polydimethylsiloxane (PDMS) microfluidic chip is fabricated using standard soft lithography techniques, creating microchannels for fluid control.
- Surface Functionalization: The internal surfaces of the microchannels are activated with oxygen plasma and functionalized with amine or carboxyl groups to facilitate aptamer immobilization.
- Aptamer Immobilization: Fluorescently-labeled DNA or RNA aptamers specific to AFB1 are covalently immobilized onto the functionalized channel surfaces.
- Baseline Measurement: The chip is mounted on a fluorescence microscope equipped with a CCD camera. A buffer solution is flowed through the chip, and the baseline fluorescence intensity of the immobilized aptamers is recorded.
- Sample Introduction and Incubation: The sample solution, potentially containing AFB1, is introduced into the microchannel and allowed to incubate statically for a predetermined time (e.g., 10-15 minutes).
- Washing and Measurement: Unbound molecules are washed away with buffer. The fluorescence intensity is measured again. Upon binding to the target mycotoxin, the aptamer may undergo a conformational change (e.g., folding), leading to a measurable change (quenching or enhancement) in fluorescence signal.
- Data Analysis: The change in fluorescence intensity (ΔF) is calculated and correlated with the concentration of AFB1 in the sample. Integrated with microfluidics, such sensors can achieve detection limits surpassing those of conventional methods, suitable for on-site screening.
Performance Metrics for Optical Biosensors
Table 2: Performance metrics of optical biosensors for detecting various ECs.
| Target EC | Bioreceptor | Optical Method | Linear Range | Detection Limit | Reference |
|---|---|---|---|---|---|
| Multiple Antibiotics | Antibody | Fluorescence (Quantum Dots) | Not Specified | Low ng/mL range | [31] |
| 17β-Estradiol (Hormone) | Aptamer | Fluorescence | Not Specified | ~nM range | [22] |
| Mycotoxins (e.g., Aflatoxin) | Aptamer/Antibody | Fluorescence / Colorimetry | Varies by target | < 0.05 µg/kg | [39] |
| Phenolic Compounds | Enzyme (Laccase) | Colorimetry | µM to mM | ~nM range | [22] [38] |
Diagram 2: Core principles and signal pathways in optical biosensing.
Piezoelectric Transduction Platforms
Piezoelectric biosensors are mass-sensitive devices that detect the binding of an analyte to a bioreceptor-coated surface through a change in the resonant frequency of a piezoelectric crystal.
Fundamental Mechanisms
The primary transducer in this category is the Quartz Crystal Microbalance (QCM). A QCM sensor typically consists of a thin disk of quartz crystal sandwiched between two metal electrodes. When an alternating voltage is applied, the crystal oscillates at a characteristic resonant frequency. This frequency is highly sensitive to the mass bound to the crystal surface. According to the Sauerbrey equation, the change in frequency (Δf) is directly proportional to the mass change (Δm) on the electrode surface. Therefore, when target analytes bind to the immobilized bioreceptors, the increase in mass causes a decrease in the resonant frequency, enabling label-free and real-time monitoring of the binding event [31]. While less explicitly detailed for ECs in the provided results compared to electrochemical and optical methods, aptasensors utilizing piezoelectric transduction are noted as a viable platform [31].
The Scientist's Toolkit: Research Reagent Solutions
The development and operation of high-performance biosensors rely on a suite of specialized reagents and materials.
Table 3: Essential research reagents and materials for biosensor development.
| Item Category | Specific Examples | Function in Biosensor Development |
|---|---|---|
| Bioreceptors | Anti-ciprofloxacin IgG; DNA/RNA aptamers for AFB1, 17β-Estradiol; Laccase enzyme; Engineered E. coli cells. | Provides high specificity and selectivity for the target emerging contaminant. The choice dictates the sensor's molecular recognition capability [31] [34]. |
| Nanomaterials | Gold nanoparticles (AuNPs); Quantum Dots (QDs); Graphene; Metal-Organic Frameworks (MOFs). | Enhances signal transduction, increases electrode surface area, and improves bioreceptor immobilization density. Used for signal amplification in optical and electrochemical sensors [22] [38] [23]. |
| Immobilization/Chemistry | EDC/NHS crosslinker; Self-Assembled Monolayers (SAMs, e.g., 4-aminothiophenol); Bovine Serum Albumin (BSA). | Enables stable and oriented covalent attachment of bioreceptors to the transducer surface. BSA is used to block non-specific binding sites [31] [23]. |
| Transducer Substrates | Screen-printed Carbon Electrodes (SPCEs); Gold disk electrodes; PDMS or PMMA microfluidic chips; Paper-based substrates (μPADs). | Forms the physical platform for the biosensor assembly. SPCEs and paper enable low-cost, disposable sensors, while gold and PDMS are used in more robust or reusable configurations [38] [39]. |
| Signal Detection Reagents | Redox probes (e.g., [Fe(CN)₆]³⁻/⁴⁻); Enzyme substrates (e.g., TMB for HRP); Fluorescent dyes (e.g., FITC). | Generates or contributes to the measurable signal in electrochemical (redox probes) and optical (enzymatic color development, fluorescence) biosensors [22] [38]. |
Electrochemical, optical, and piezoelectric transduction platforms each offer distinct advantages for the detection of emerging contaminants. The choice of transducer is contingent on the specific application requirements, including desired sensitivity, need for portability, cost constraints, and the complexity of the sample matrix. The ongoing integration of these platforms with nanomaterials, microfluidics, and portable electronics (including smartphones) is steadily advancing the field towards highly sophisticated, multiplexed, and connected sensing systems. By providing detailed mechanistic insights, experimental workflows, and a catalog of essential research tools, this whitepaper aims to equip scientists and engineers with the foundational knowledge to innovate and deploy these critical technologies in the global effort to monitor and mitigate the impact of environmental pollutants.
The convergence of nanomaterials, microfluidics, and paper-based substrates is driving a revolution in the design of analytical devices, particularly for biosensing applications. This integration creates platforms that are not only highly sensitive and specific but also affordable, portable, and accessible, making them ideal for point-of-care (POC) diagnostics, environmental monitoring, and food safety testing [40] [41]. Paper, as a foundational substrate, brings unique advantages to this synergy, including its inherent porosity, capillary action, flexibility, biodegradability, and low cost [40] [42]. Microfluidic paper-based analytical devices (µPADs), first pioneered by the Whitesides group in 2007, leverage these properties to manipulate small fluid volumes without the need for external pumps [43] [44].
The performance of these devices is significantly enhanced by the strategic incorporation of nanomaterials. Metal nanoparticles, carbon-based nanomaterials, and metal-organic frameworks (MOFs) introduce unique optical, electrical, and catalytic properties that drastically improve sensor capabilities [40] [45] [46]. When combined with the lab-on-a-chip functionality of microfluidics, these nanomaterial-enabled paper-based devices meet the critical ASSURED criteria (Affordable, Sensitive, Specific, User-friendly, Rapid and Robust, Equipment-free, and Deliverable to end-users) set by the World Health Organization for ideal diagnostic tools [46]. This technical guide explores the core innovations in the design of these integrated systems, providing a detailed examination of fabrication techniques, sensing mechanisms, and experimental protocols, framed within the context of detecting emerging contaminants and biomarkers.
Fabrication Techniques for Paper-Based Microfluidic Devices
The fabrication of microfluidic paper-based analytical devices (µPADs) centers on creating well-defined hydrophobic barriers that form hydrophilic channels and reaction zones on paper substrates. The chosen fabrication method directly influences the device's resolution, cost, scalability, and overall performance [40].
Primary Fabrication Methods
- Wax Printing: This is one of the most common and cost-effective methods. It involves printing a wax pattern onto paper using a solid-ink printer, which is then heated to allow the wax to permeate the paper thickness, creating hydrophobic barriers. Key optimized parameters include a nozzle temperature of 64–85 °C and a print speed of 40–60 mm/s to achieve consistent patterns [40].
- Screen Printing: This technique is widely used for mass-producing electrodes and conductive features on paper. Conductive inks (e.g., carbon, silver/silver chloride) are forced through a fine mesh screen onto the paper substrate. Factors such as mesh density, squeegee pressure and angle, ink viscosity, and curing conditions must be tightly controlled to ensure high conductivity and feature resolution as fine as 40–50 µm [40].
- Photolithography: This method, used in the original µPADs, involves coating paper with a photoresist (e.g., SU-8). The photoresist is then exposed to UV light through a photomask, and the unexposed regions are washed away, leaving a patterned hydrophobic polymer barrier. While it offers high resolution, it requires more specialized equipment and chemicals [44].
- Pen/Pencil-on-Paper: For ultra-low-cost, rapid prototyping, the pen-on-paper (PoP) technique uses commercial ink pens to manually draw electrodes and channels. Similarly, the pencil-drawing method uses graphite pencils to create conductive traces. This method has been successfully used to detect pathogens like Escherichia coli, demonstrating its potential for field diagnostics [40].
Table 1: Comparison of Fabrication Techniques for Paper-Based Microfluidic Devices
| Fabrication Method | Resolution | Cost | Scalability | Key Advantages | Key Limitations |
|---|---|---|---|---|---|
| Wax Printing | ~100-500 µm | Very Low | High for batch production | Low cost, rapid, simple process | Limited resolution, wax can spread |
| Screen Printing | ~40-100 µm | Low | Very High | High throughput, good for electrodes | Requires screen fabrication, parameter sensitivity |
| Photolithography | ~10-100 µm | High | Medium | High resolution, well-defined barriers | Complex process, requires toxic chemicals |
| Pen/Pencil-on-Paper | ~500-1000 µm | Extremely Low | Low | On-demand fabrication, no equipment needed | Low resolution, poor reproducibility |
Integration of Nanomaterials and Signaling Mechanisms
The functionalization of µPADs with nanomaterials is a critical step for enhancing their analytical performance. Nanomaterials act as signal reporters, catalysts, and immobilization platforms, enabling highly sensitive and specific detection of target analytes.
Classification and Functionalization of Nanomaterials
- Metal Nanoparticles (MNPs): Gold and silver nanoparticles (AuNPs and AgNPs) are extensively used due to their unique localized surface plasmon resonance (LSPR). This property causes intense light absorption and scattering, making them ideal for colorimetric assays where the aggregation or dispersion of nanoparticles leads to a visible color change [46]. Beyond their plasmonic role, MNPs also enhance conductivity in electrochemical sensors and possess catalytic properties [46].
- Carbon-Based Nanomaterials: Materials like graphene oxide, carbon nanotubes, and their derivatives are prized for their high electrical conductivity and large surface area. They are often used to modify electrodes in electrochemical µPADs, facilitating electron transfer and increasing the loading capacity for biorecognition elements [40] [45].
- Metal-Organic Frameworks (MOFs): MOFs are porous crystalline materials with an extremely high surface area.当他们 integrated into paper matrices, they improve sensor stability, selectivity, and preconcentrate target analytes, thereby enhancing the signal transduction efficiency [40]. They have shown particular promise in the detection of heavy metals and gases [43].
Detection and Signaling Modalities
The integration of nanomaterials enables a diverse range of detection modalities on µPADs, each with its own advantages.
- Electrochemical Detection: This is one of the most sensitive methods. Nanomaterial-modified working electrodes transduce a biological recognition event (e.g., antibody-antigen binding) into a measurable electrical signal (current, potential, or impedance). The use of nanomaterials like reduced graphene oxide and AuNPs has been shown to allow for the precise detection of low-concentration biomarkers [40]. For example, an aptamer sensor integrated with two-dimensional nanomaterials achieved ketamine detection with a sensitivity at the 10-ppb level [43].
- Optical Detection:
- Colorimetric: The most common and user-friendly optical method. It relies on visual color changes, often amplified by LSPR from AuNPs or AgNPs, which can be quantified with a smartphone camera [46] [44].
- Fluorescence: Fluorescent dyes or quantum dots are used as labels. The paper background provides a low-autofluorescence environment, enhancing the signal-to-noise ratio. Yuan et al. developed a portable multi-channel fluorescence µPAD for simultaneously detecting four heavy metals (Pb²⁺, Hg²⁺, Cd²⁺, and As³⁺) in food samples [43].
- Surface-Enhanced Raman Scattering (SERS): This powerful technique relies on the dramatic enhancement of Raman signals when analyte molecules are adsorbed onto roughened noble metal surfaces (e.g., Au or Ag nanostructures). SERS-based µPADs offer rapid, sensitive, and fingerprint-like identification of molecules, with growing potential due to advances in portable Raman instrumentation [46].
- Chemiluminescence (CL) and Electrochemiluminescence (ECL): These methods detect light emission generated from a chemical or electrochemical reaction, respectively. MNPs can act as catalysts to amplify the light signal in these systems, leading to highly sensitive detection without the need for an external light source [46].
Experimental Protocols and Workflows
This section provides detailed methodologies for key experimental procedures in the development and operation of nanomaterial-integrated µPADs.
Protocol 1: Fabrication of a Wax-Printed µPAD
Objective: To create a patterned microfluidic paper-based device with hydrophilic channels and hydrophobic barriers using wax printing.
Materials:
- Whatman No. 1 chromatography paper
- Solid-ink wax printer (e.g., Xerox ColorQube)
- Hot plate or oven
- Design software (e.g., Adobe Illustrator, CAD)
Procedure:
- Design: Create the desired pattern of microfluidic channels and detection zones using the design software. The design should consist of solid lines that will form the hydrophobic barriers.
- Printing: Load the paper into the wax printer and print the design. Ensure the printer is set to the highest possible density to deposit sufficient wax.
- Heating: Place the printed paper on a hot plate preheated to 150 °C for 120 seconds. This allows the wax to melt and permeate through the entire thickness of the paper, forming complete hydrophobic barriers.
- Cooling: Allow the device to cool to room temperature. The hydrophilic channels are now clearly defined and ready for functionalization.
Protocol 2: Functionalization with Gold Nanoparticles for Colorimetric Detection
Objective: To immobilize citrate-capped AuNPs on a paper-based detection zone for a colorimetric assay.
Materials:
- Fabricated µPAD (from Protocol 1)
- Citrate-capped AuNP solution (13 nm diameter, OD~1)
- Phosphate Buffered Saline (PBS), pH 7.4
- Micropipettes
Procedure:
- Preparation: Spot 1-2 µL of the AuNP solution onto the center of the predefined detection zone on the µPAD.
- Drying: Allow the device to dry completely at room temperature for 30-60 minutes. The AuNPs will be physically adsorbed onto the cellulose fibers.
- Blocking (Optional): To reduce non-specific binding, a blocking agent like bovine serum albumin (BSA) or poly(ethylene glycol) (PEG) can be applied to the zone after the AuNPs have dried.
- Assay Execution: Introduce the liquid sample to the sample inlet. The fluid will wick through the channel via capillary action and reach the AuNP-functionalized detection zone.
- Readout: A positive result is indicated by a visible color shift from red (dispersed NPs) to blue/purple (aggregated NPs), which can be quantified using a smartphone scanner or a desktop scanner.
Protocol 3: Electrochemical Detection of a Biomarker using a Screen-Printed ePAD
Objective: To perform an electrochemical sandwich immunoassay for the detection of a specific biomarker (e.g., Carcinoembryonic Antigen, CEA) on a paper-based electrode.
Materials:
- Screen-printed carbon electrode (SPCE) on paper substrate
- Primary anti-CEA antibody
- Secondary anti-CEA antibody conjugated with a redox label (e.g., Horseradish Peroxidase - HRP)
- Electrochemical reader (potentiostat)
- Wash buffer (PBS with Tween-20)
Procedure:
- Electrode Modification: Drop-cast a solution of the primary anti-CEA antibody onto the working electrode and allow it to immobilize overnight at 4 °C.
- Blocking: Apply a blocking buffer (e.g., 1% BSA) to the electrode for 1 hour to cover any non-specific binding sites. Rinse with wash buffer.
- Sample Incubation: Introduce the sample containing the CEA antigen to the electrode. Incubate for 20 minutes at room temperature to form the antibody-antigen complex. Rinse thoroughly.
- Label Binding: Introduce the HRP-conjugated secondary antibody. Incubate for 20 minutes to complete the sandwich complex. Perform a final rinse.
- Electrochemical Measurement: Add an electrochemical substrate (e.g., TMB/H₂O₂) and apply the appropriate potential. Measure the amperometric current. The current magnitude is proportional to the concentration of CEA captured in the sandwich assay. A reported molecularly imprinted polymer (MIP)-facilitated sensor achieved a limit of detection (LOD) of 0.32 ng/mL for CEA using this principle [47].
The Scientist's Toolkit: Essential Research Reagents and Materials
Successful development of nanomaterial-integrated µPADs relies on a core set of reagents and materials. The table below details these essential components and their functions.
Table 2: Key Research Reagent Solutions for µPAD Development
| Category/Item | Specific Examples | Function/Purpose | Key Characteristics |
|---|---|---|---|
| Paper Substrates | Whatman No. 1 Filter Paper, Whatman Chr1, Nitrocellulose Membrane | Microfluidic substrate; provides capillary flow and platform for reagent immobilization. | High porosity, consistent flow, biocompatibility, protein binding (nitrocellulose) [40] [44]. |
| Conductive Inks | Carbon/Graphite ink, Silver/Silver Chloride (Ag/AgCl) ink | Fabrication of electrodes for electrochemical sensing (working, reference, counter). | High electrical conductivity, stability, adhesion to paper substrate [40] [42]. |
| Nanomaterials | Gold Nanoparticles (AuNPs), Silver Nanoparticles (AgNPs), Reduced Graphene Oxide (rGO) | Signal generation (colorimetric, SERS), signal amplification, enhanced electron transfer, increased surface area. | Tunable LSPR, high catalytic activity, excellent conductivity [40] [45] [46]. |
| Biorecognition Elements | Antibodies, Aptamers, Molecularly Imprinted Polymers (MIPs) | Provide specificity by binding to the target analyte (e.g., biomarker, contaminant). | High affinity and selectivity, stability on paper, compatibility with assay conditions [45] [47]. |
| Signal Generation Reagents | Horseradish Peroxidase (HRP), TMB/H₂O₂ substrate, Fluorescent dyes (e.g., FITC) | Generate measurable signals (color, light, current) upon target recognition. | High turnover rate, stable signal output, low background noise. |
| Buffers & Blockers | Phosphate Buffered Saline (PBS), Bovine Serum Albumin (BSA), Tween-20 | Maintain pH and ionic strength; block non-specific binding sites to reduce background signal. | Biocompatible, effective blocking, no interference with biorecognition [47]. |
The innovative integration of nanomaterials and microfluidic principles into paper-based devices has created a powerful and versatile technological platform. This guide has detailed the core aspects of this integration, from fabrication and functionalization to detection mechanisms and experimental protocols. These devices represent a paradigm shift in biosensing, moving complex laboratory analyses into portable, affordable, and user-friendly formats. For researchers focused on emerging contaminants and drug development, these systems offer a promising path toward rapid, on-site screening and monitoring. Future advancements will likely focus on improving multiplexing capabilities, reagent stability for long-term storage, seamless integration with digital health platforms, and streamlining mass production to fully realize the potential of this transformative technology in global health and environmental protection.
The pervasive contamination of water resources by pesticides, pharmaceuticals, and pathogens presents a critical challenge to global health and ecosystem integrity. These contaminants of emerging concern (CECs) are often not completely removed by conventional water treatment processes and can exhibit detrimental effects even at trace concentrations [22] [26]. Traditional analytical methods, such as gas chromatography (GC) and high-performance liquid chromatography coupled with mass spectrometry (HPLC-MS/MS), provide high accuracy but are constrained by high costs, complex sample preparation, lengthy analysis times, and lack of portability for real-time, on-site monitoring [38] [48] [26].
Biosensing technologies have emerged as a powerful alternative, integrating biological recognition elements with transducers to create devices that are highly sensitive, selective, rapid, and amenable to miniaturization for field deployment [26] [31]. This whitepaper provides an in-depth technical examination of biosensor applications across three critical domains: pesticide detection in water, pharmaceutical monitoring in wastewater, and pathogen identification. By synthesizing recent advancements and presenting structured performance data, experimental protocols, and visual frameworks, this review aims to equip researchers and development professionals with a comprehensive toolkit for advancing environmental monitoring capabilities.
Biosensor Fundamentals and Classification
Biosensors are analytical devices that combine a biological sensing element with a transducer to produce a measurable signal proportional to the concentration of a target analyte. Their operation hinges on the specific interaction between the biorecognition element and the analyte, which is then converted into an electrical, optical, or other quantifiable signal [26] [31].
Table 1: Classification of Biosensors by Biorecognition Element and Transduction Mechanism
| Biosensor Type | Biorecognition Element | Transduction Mechanism | Key Features | Example Application |
|---|---|---|---|---|
| Enzyme-Based | Enzymes (e.g., acetylcholinesterase, laccase) | Electrochemical, Optical, Thermal | High specificity and catalytic activity; can be based on inhibition or metabolism. | Organophosphorus pesticide detection [49] |
| Antibody-Based (Immunosensor) | Antibodies (IgG, IgM, etc.) | Electrochemical, Optical (SPR, Fluorescence) | Exceptional specificity and affinity; can be label-free or labeled. | Ciprofloxacin detection [26] [31] |
| Nucleic Acid-Based (Aptasensor) | DNA or RNA aptamers | Electrochemical, Optical, Piezoelectric | High stability, in vitro synthesis; target variety via SELEX. | Oxytetracycline, heavy metal detection [22] [26] |
| Whole Cell-Based | Bacteria, fungi, algae | Optical (Bioluminescence), Electrochemical | Robustness, self-replication, can report on bioavailability and toxicity. | Pyrethroid insecticide detection [31] |
| Nanozyme-Based | Nanomaterials with enzyme-like activity | Colorimetric, Electrochemical, Fluorescent | High stability, tunable activity, cost-effective. | Pesticide residue detection [50] |
The following diagram illustrates the logical relationship between the biorecognition element, the transduction event, and the final output in a generalized biosensor.
Diagram 1: Core Biosensor Operation Logic
Technical Application Spotlights
Detection of Pesticides in Water
Pesticides, including insecticides, herbicides, and fungicides, are among the most prevalent CECs in water bodies, originating primarily from agricultural runoff [26] [31]. Their toxicity necessitates monitoring at very low concentrations.
Performance Metrics of Pesticide Biosensors
Table 2: Analytical Performance of Representative Biosensors for Pesticide Detection
| Target Pesticide | Biosensor Type | Biorecognition Element | Transduction Method | Linear Range | Limit of Detection (LOD) |
|---|---|---|---|---|---|
| Organophosphorus | Enzyme-based [49] | Acetylcholinesterase (AChE) | Electrochemical | Not Specified | Low detection limit potential |
| Various Pesticides | Nanozyme-based [50] | Nanomaterial (e.g., MOFs, Graphene) | Colorimetric / Electrochemical | Not Specified | Demonstrated high sensitivity |
| Pyrethroids | Whole Cell-based [31] | Engineered E. coli | Optical (Label-free) | Not Specified | 3 ng/mL |
| Atrazine | Immunosensor [22] | Antibody | Electrochemical | Not Specified | Demonstrated in real samples |
Detailed Experimental Protocol: Enzyme-Inhibition Based Electrochemical Biosensor for Organophosphorus Pesticides
This protocol is adapted from methodologies described in recent literature on biomaterials and biosensing technologies [49].
Principle: The assay is based on the inhibition of the enzyme acetylcholinesterase (AChE). AChE normally hydrolyzes the substrate acetylthiocholine (ATCh), producing thiocholine, which generates an electrochemical signal (e.g., a measurable current). The presence of organophosphorus pesticides inhibits AChE activity, leading to a reduction in the output signal, which is proportional to the pesticide concentration.
Materials:
- Acetylcholinesterase (AChE): Biological recognition element.
- Acetylthiocholine (ATCh): Enzyme substrate.
- Electrochemical Transducer: Screen-printed carbon electrode (SPCE) or gold electrode.
- Nanomaterial Modifiers: Graphene oxide (GO) or gold nanoparticles (AuNPs) to enhance electrode surface area and electron transfer.
- Phosphate Buffered Saline (PBS): For preparing solutions and as an assay buffer.
- Standard Solutions: Of target organophosphorus pesticides (e.g., parathion, chlorpyrifos).
Procedure:
- Electrode Modification: Deposit a suspension of graphene oxide or AuNPs onto the surface of the working electrode. Allow to dry under ambient conditions. This step increases sensitivity.
- Enzyme Immobilization: Drop-cast a solution of AChE onto the modified electrode surface. Allow the enzyme to physically adsorb or chemically cross-link (e.g., using glutaraldehyde) onto the nanomaterial layer. Rinse gently with PBS to remove unbound enzyme.
- Baseline Measurement: Place the biosensor in an electrochemical cell containing PBS and a fixed concentration of ATCh. Perform amperometric measurement at a constant potential (e.g., +0.5 V vs. Ag/AgCl) and record the steady-state current (I₀) generated by the enzymatic production of thiocholine.
- Inhibition (Sample Exposure): Incubate the biosensor in a sample solution (or standard pesticide solution) for a fixed period (e.g., 10-15 minutes). Rinse gently with PBS.
- Post-Inhibition Measurement: Re-immerse the biosensor in the PBS/ATCh solution and record the new steady-state current (I₁).
- Data Analysis: Calculate the percentage of enzyme inhibition using the formula: Inhibition (%) = [(I₀ - I₁) / I₀] × 100. The pesticide concentration in the unknown sample is determined by interpolating the inhibition value against a calibration curve constructed with standard solutions.
Detection of Pharmaceuticals in Wastewater
Pharmaceutical residues, including antibiotics, endocrine disruptors, and non-steroidal anti-inflammatory drugs, are increasingly detected in wastewater and surface waters, contributing to the development of antimicrobial resistance and endocrine disruption in aquatic life [26].
Performance Metrics of Pharmaceutical Biosensors
Table 3: Analytical Performance of Representative Biosensors for Pharmaceutical Detection
| Target Pharmaceutical | Biosensor Type | Biorecognition Element | Transduction Method | Linear Range | Limit of Detection (LOD) |
|---|---|---|---|---|---|
| Ciprofloxacin (Antibiotic) | Immunosensor [26] [31] | Anti-ciprofloxacin Antibody | Impedimetric (Label-free) | Not Specified | 10 pg/mL |
| 17β-Estradiol (Hormone) | Aptasensor [22] | DNA Aptamer | Fluorescence | Not Specified | Demonstrated high sensitivity |
| Multiple Antibiotics | Immunosensor [26] [31] | Antibodies | Quantum Dot Fluorescence | Not Specified | Demonstrated for milk |
| Diclofenac (Anti-inflammatory) | Immunosensor [22] | Antibody | Electrochemical | Not Specified | Demonstrated in urine/water |
Detailed Experimental Protocol: Aptamer-Based Fluorescent Biosensor for Antibiotics
This protocol is based on the application of nucleic acid aptamers for the detection of contaminants of emerging concern [22] [26].
Principle: A DNA or RNA aptamer, specific to the target pharmaceutical, is labeled with a fluorescent dye. The binding of the target molecule to the aptamer induces a conformational change (or proximity to a quencher), which alters the fluorescence intensity, enabling quantification.
Materials:
- Fluorescently-Labeled Aptamer: Synthesized single-stranded DNA with a fluorophore (e.g., FAM) at the 5' end.
- Quencher Molecule or Graphene Oxide (GO): For signal modulation. GO is a common nanomaterial that quenches the fluorescence of nearby fluorophores via energy transfer.
- Buffer Solution: Typically Tris-EDTA or HEPES buffer with optimized ionic strength and pH.
- Standard Solutions: Of the target antibiotic (e.g., oxytetracycline, ampicillin).
Procedure:
- Probe Preparation: Dilute the fluorescently-labeled aptamer in the appropriate assay buffer to a working concentration.
- Signal Quenching (for GO-based systems): Add a calculated amount of GO dispersion to the aptamer solution and incubate for a few minutes. The aptamer adsorbs onto the GO surface, bringing the fluorophore close enough for its fluorescence to be quenched. The solution will exhibit low background fluorescence.
- Sample Introduction and Incubation: Mix the quenched aptamer/GO complex with the sample solution containing the target pharmaceutical. Incubate for 15-20 minutes to allow the binding reaction to occur.
- Signal Recovery: The binding of the target to the aptamer induces a conformational change that causes the aptamer to detach from the GO surface, or it moves the fluorophore away from a quencher. This restores fluorescence.
- Fluorescence Measurement: Transfer the solution to a cuvette or microplate reader. Measure the fluorescence intensity at the excitation/emission wavelengths specific to the fluorophore (e.g., 494/521 nm for FAM).
- Data Analysis: Plot the measured fluorescence intensity against the logarithm of the antibiotic concentration. The intensity is directly proportional to the target concentration in the sample.
Detection of Pathogens
Rapid detection of foodborne and waterborne pathogenic bacteria, such as E. coli and Salmonella, is crucial for public health protection. Fluorescent biosensors offer a rapid and sensitive alternative to traditional culture methods [51].
Detailed Experimental Protocol: Fluorescent Biosensor for Foodborne Pathogenic Bacteria
This protocol summarizes the advanced strategies outlined in reviews on fluorescent biosensors for pathogens [51].
Principle: The protocol utilizes an antibody-based recognition system coupled with a signal amplification strategy, such as the use of quantum dots (QDs), for highly sensitive detection.
Materials:
- Capture Antibody: Specific to the target pathogen's surface antigen.
- Detection Antibody: Conjugated to a quantum dot (QD) or another fluorescent nanomaterial.
- Magnetic Beads: Coated with streptavidin for easy separation if a biotinylated capture antibody is used.
- Blocking Buffer: Bovine serum albumin (BSA) or milk protein to prevent non-specific binding.
- Wash Buffer: PBS containing a mild detergent (e.g., Tween-20).
Procedure:
- Capture Probe Formation: Immobilize the biotinylated capture antibody onto streptavidin-coated magnetic beads. Incubate with rotation, then wash to remove excess antibody.
- Blocking: Incubate the beads with a blocking buffer to cover any non-specific binding sites on the bead surface. Wash again.
- Pathogen Capture: Incubate the prepared capture beads with the sample (e.g., food homogenate or water sample). The target bacteria will bind to the capture antibodies.
- Separation and Washing: Use a magnetic rack to separate the beads (now with captured bacteria) from the sample matrix. Wash thoroughly to remove unbound material.
- Fluorescent Labeling: Resuspend the bead-bacteria complex in a solution containing the QD-conjugated detection antibody. This creates a "sandwich" immunocomplex on the bead surface.
- Final Wash and Measurement: After incubation, magnetically separate the beads and perform a final wash. Resuspend the beads in buffer and measure the fluorescence intensity of the supernatant or the re-suspended beads. The fluorescence signal is directly correlated with the number of captured pathogen cells.
The workflow for this sandwich immunoassay is detailed in the diagram below.
Diagram 2: Pathogen Detection via Sandwich Immunoassay
The Scientist's Toolkit: Essential Research Reagents and Materials
The development and deployment of advanced biosensors rely on a suite of specialized reagents and materials that enhance sensitivity, stability, and specificity.
Table 4: Key Research Reagent Solutions for Biosensor Development
| Reagent/Material | Function/Application | Specific Examples |
|---|---|---|
| Gold Nanoparticles (AuNPs) | Signal amplification; electrode modification; colorimetric reporting. | Citrate-capped AuNPs for antibody conjugation [22] [49]. |
| Graphene Oxide (GO) | Electrode modifier; fluorescence quencher in aptasensors. | Sheets used in electrochemical and fluorescent platforms [49]. |
| Metal-Organic Frameworks (MOFs) | Nanozyme activity; high surface area for biomolecule immobilization. | Zeolitic imidazolate frameworks (ZIFs) for pesticide detection [50] [49]. |
| Quantum Dots (QDs) | Fluorescent labels for optical biosensors; multiplexed detection. | CdSe/ZnS QDs for immunoassays [26]. |
| Molecularly Imprinted Polymers (MIPs) | Synthetic, stable antibody mimics for molecular recognition. | MIPs for pesticides or antibiotics as recognition elements [49]. |
| Circularly Permutated GFP (cpsfGFP) | Genetically encoded reporter for transporter-based biosensors. | Used in SweetTrac1 for sugar transport monitoring [52]. |
| DNA/RNA Aptamers | Synthetic recognition elements from SELEX; high stability. | Aptamers for oxytetracycline, estradiol, or heavy metals [22] [26]. |
The field of biosensing is rapidly evolving, driven by interdisciplinary innovations. Key future trends include the integration of artificial intelligence (AI) and Internet of Things (IoT) technologies for data analysis and the development of smart, adaptive environmental monitoring networks [38]. The creation of multifunctional and portable platforms, such as paper-based microfluidic devices (μPADs), will further enable rapid, on-site detection, moving analysis from the central laboratory directly to the point of need [38] [22] [48]. Despite the significant progress, challenges remain in achieving long-term stability of biological elements in harsh environments, ensuring selectivity in complex matrices like wastewater, and reducing costs for widespread deployment [38] [26] [49]. Ongoing research focused on novel biorecognition elements, advanced nanomaterials, and integrated systems will be crucial in overcoming these hurdles.
In conclusion, biosensors represent a transformative technological paradigm for monitoring pesticides, pharmaceuticals, and pathogens in aquatic environments. Their exceptional sensitivity, potential for miniaturization, and capability for real-time analysis offer a robust solution to the limitations of conventional methods. As these technologies mature, they are poised to play an indispensable role in safeguarding water quality, protecting public health, and enabling a more sustainable future.
Navigating Development Hurdles: Key Challenges and Optimization Strategies for Robust Biosensors
The accurate detection of emerging contaminants—such as pharmaceuticals, personal care products, and per- and polyfluoroalkyl substances (PFAS)—in environmental and biological matrices demands analytical tools with exceptional performance. Biosensors, as analytical devices, convert a biological response into an quantifiable electrical, optical, or other signal, serving as a cornerstone for this purpose [53] [54]. Their efficacy in monitoring programs and research aimed at implementing stricter environmental legislation hinges on the rigorous assessment of four critical performance metrics: sensitivity, specificity, dynamic range, and response time [54]. This whitepaper provides an in-depth technical guide for researchers, scientists, and drug development professionals on the methodologies for evaluating these core metrics, framed within the context of advancing biosensing technologies for the detection of trace-level emerging contaminants. We summarize quantitative data in structured tables, detail experimental protocols, and provide visualizations of workflows and signaling pathways to serve as a foundational resource in this rapidly evolving field.
Emerging contaminants present a unique challenge for environmental and clinical monitoring due to their low environmental concentrations and potential for significant ecological and human health impacts, even at part-per-trillion levels. Conventional analytical methods, such as liquid or gas chromatography coupled with mass spectrometry, while highly accurate, are often expensive, time-consuming, and require highly trained personnel and centralized laboratory facilities, making them unsuitable for rapid, on-site screening [53] [54]. In this context, biosensors have emerged as a powerful alternative or complementary analytical technology. The International Union of Pure and Applied Chemistry (IUPAC) defines a biosensor as a self-contained integrated device that provides specific quantitative or semi-quantitative analytical information using a biological recognition element in direct spatial contact with a transduction element [54].
The fundamental principle of a biosensor involves the specific interaction between a biological recognition element (e.g., enzyme, antibody, DNA, whole cell) and the target analyte, which produces a physicochemical change that is converted by a transducer into a measurable signal [54]. The growing need for disposable, cost-effective, and rapid systems for environmental and clinical applications has accelerated the development of sophisticated biosensing platforms [55]. Key challenges in the field, particularly for detecting elusive emerging contaminants and circulating biomarkers, include enhancing sensitivity through signal amplification, constructing multi-functional sensing platforms, enabling robust performance in complex systems, and simplifying operational procedures [55]. Addressing these challenges requires a systematic and critical evaluation of a biosensor's analytical performance, which is the focus of this technical guide.
Core Performance Metrics: Definitions and Quantitative Assessment
The performance of a biosensor is quantitatively characterized by several key parameters. The following sections define these metrics and provide a framework for their measurement.
Sensitivity
Sensitivity measures the ability of a biosensor to produce a significant signal change in response to a minimal change in analyte concentration. In practical terms, it is often derived from the slope of the calibration curve (signal vs. analyte concentration) within the linear range. A steeper slope indicates higher sensitivity. The limit of detection (LOD), a closely related parameter, is the lowest analyte concentration that can be reliably distinguished from a blank. It is typically calculated as three times the standard deviation of the blank signal divided by the slope of the calibration curve.
Specificity
Specificity (or selectivity) refers to the biosensor's ability to respond exclusively to the target analyte in the presence of potential interferents that may coexist in a sample matrix (e.g., serum, wastewater). It is a function of the biological recognition element's affinity. High specificity is crucial for avoiding false-positive or false-negative results in complex samples.
Dynamic Range
The dynamic range is the span of analyte concentrations over which the biosensor provides a quantifiable response. It is bounded at the lower end by the LOD and at the upper end by the point where the signal plateaus or becomes non-linear (saturation). The linear dynamic range is the concentration interval where the response is linearly proportional to the concentration.
Response Time
The response time is the time required for the biosensor to reach a stable, measurable signal (e.g., 90% or 95% of the maximum steady-state signal) after exposure to the analyte. This metric is critical for applications requiring real-time or near-real-time monitoring.
Table 1: Key Performance Metrics for Biosensors Targeting Emerging Contaminants
| Performance Metric | Definition | Typical Target for Emerging Contaminants | Experimental Determination |
|---|---|---|---|
| Sensitivity (LOD) | Lowest detectable concentration | Low ng/L to µg/L (ppb-ppt) | Calibration curve (3σ/slope) |
| Specificity | Discrimination against interferents | >90% signal retention in complex matrices | Cross-reactivity assays |
| Dynamic Range | Concentration range of quantifiable response | 3-6 orders of magnitude | Calibration curve (LOD to saturation) |
| Response Time | Time to reach stable signal | Seconds to minutes | Real-time signal monitoring |
Table 2: Exemplary Performance of Select Biosensor Types
| Biosensor Type | Target Analyte | Sensitivity (LOD) | Dynamic Range | Response Time | Reference |
|---|---|---|---|---|---|
| Immunosensor | E. coli (pathogen) | -- | -- | -- | [53] |
| DNA Biosensor | Specific gene sequences | -- | -- | -- | [53] [55] |
| Enzyme-based | Organophosphate Pesticides | -- | -- | -- | [53] |
| Whole-cell | Heavy Metals (e.g., Pb, As) | -- | -- | -- | [54] |
Experimental Protocols for Metric Assessment
Standardized experimental protocols are essential for the reliable and comparable assessment of biosensor performance.
Protocol for Sensitivity and Dynamic Range Determination
- Preparation of Standard Solutions: Prepare a series of standard solutions of the target analyte in a relevant buffer (e.g., phosphate-buffered saline) or a simulated matrix. The concentration range should be sufficiently wide to cover from below the expected LOD to well above the expected saturation point (e.g., from 0.001 to 1000 nM).
- Biosensor Measurement: For each standard solution, introduce the sample to the biosensor and record the steady-state signal (e.g., current for electrochemical, fluorescence intensity for optical). Between measurements, regenerate the biosensor surface according to the established protocol (e.g., with a glycine-HCl buffer for antibody-based sensors) to ensure a consistent baseline.
- Data Analysis: Plot the signal (y-axis) against the analyte concentration (x-axis) to generate a calibration curve. Fit the data within the linear region using linear regression (y = mx + c). The slope (m) represents the sensitivity.
- Calculation of LOD and LOQ: Calculate the LOD as 3σ/m, where σ is the standard deviation of the signal from at least ten blank measurements. The limit of quantification (LOQ) can be calculated as 10σ/m.
Protocol for Specificity Assessment
- Selection of Interferents: Identify a panel of potential interferents that are structurally similar to the target analyte or are commonly found in the sample matrix of interest (e.g., other pharmaceuticals, natural organic matter, ions, proteins).
- Cross-Reactivity Test: Measure the biosensor's response to solutions containing each potential interferent at a concentration significantly higher (e.g., 10-100x) than the expected concentration of the target analyte.
- Signal Comparison: Compare the signal generated by the interferent to the signal generated by the target analyte at its relevant concentration. The percentage cross-reactivity is calculated as (Signal from Interferent / Signal from Target Analyte) × 100%. A well-designed biosensor should exhibit minimal cross-reactivity (<5-10%).
Protocol for Response Time Measurement
- Setup: Place the biosensor in a flow cell or a static measurement chamber with continuous signal monitoring.
- Stimulus Introduction: Rapidly introduce a solution containing the target analyte at a known concentration (e.g., within the mid-range of the dynamic range).
- Signal Acquisition: Continuously record the output signal at a high frequency (e.g., 10-100 Hz) from the moment of analyte introduction.
- Time Calculation: Determine the time taken for the signal to rise from the baseline to 90% (t90) or 95% (t95) of its final maximum steady-state value. This is the response time.
Visualization of Biosensor Workflows and Signaling
The following diagrams, generated using Graphviz DOT language, illustrate core concepts and experimental workflows in biosensing. The color palette and contrast adhere to the specified guidelines for accessibility.
Diagram 1: Core Biosensor Mechanism.
Diagram 2: Sensitivity Workflow.
Diagram 3: FRET Biosensor Pathway.
The Scientist's Toolkit: Essential Research Reagents and Materials
The development and operation of high-performance biosensors rely on a suite of specialized reagents and materials.
Table 3: Essential Research Reagent Solutions for Biosensor Development
| Reagent/Material | Function and Role in Biosensing |
|---|---|
| Biological Recognition Elements | Provides specificity. Includes enzymes (e.g., glucose oxidase), antibodies, DNA/RNA aptamers, and whole cells (e.g., engineered bacteria for toxicity screening) [53] [54]. |
| Transducer Materials | Converts biological event to measurable signal. Includes screen-printed carbon electrodes (electrochemical), optical fibers, gold films for SPR, and piezoelectric crystals [53] [55]. |
| Immobilization Matrices | Stabilizes and confines the biorecognition element to the transducer surface. Includes hydrogels, self-assembled monolayers (SAMs), sol-gels, and nanomaterials (e.g., graphene oxide, carbon nanotubes) [54]. |
| Signal Amplification Agents | Enhances sensitivity. Includes enzyme labels (e.g., Horseradish Peroxidase), catalytic nanomaterials (e.g., gold nanoparticles), and DNA amplification reagents for strategies like rolling circle amplification [55]. |
| Blocking Agents | Reduces non-specific binding (NSB) of non-target molecules to the sensor surface, thereby improving specificity. Common agents include bovine serum albumin (BSA) or casein. |
| Regeneration Buffers | Allows for the reversible binding and reuse of biosensors (particularly affinity-based sensors like immunosensors) by breaking the analyte-bioreceptor bond without damaging the bioreceptor (e.g., low-pH glycine buffer). |
The accurate and reliable detection of emerging contaminants (ECs)—including pharmaceuticals, pesticides, and endocrine-disrupting chemicals—is critical for safeguarding water safety and public health [31] [9]. Biosensing technologies present a promising alternative to conventional analytical techniques, offering advantages in portability, cost, and potential for real-time monitoring [31] [23]. However, the transition of biosensors from laboratory prototypes to robust field-deployable tools is hindered by significant challenges related to their stability and reproducibility in complex sample matrices [56] [57].
This technical guide examines the three predominant factors compromising biosensor performance: temperature fluctuations, pH variations, and surface fouling. These factors can induce physical changes in biorecognition elements, alter electrochemical kinetics, and promote non-specific adsorption, collectively leading to signal drift, reduced sensitivity, and unreliable data [58] [59] [56]. Framed within the broader context of ECs research, this document provides a detailed analysis of these challenges and offers evidence-based strategies and standardized experimental protocols to mitigate them, aiming to enhance the robustness and credibility of biosensing data for researchers and drug development professionals.
Core Challenges and Quantitative Impacts
Environmental factors and sample complexity introduce significant variability in biosensor function. The data below quantify these effects on sensor performance.
Table 1: Impact of Temperature on Sensor and Biological System Performance
| System / Parameter | Test Condition 1 | Test Condition 2 | Observed Impact on Performance |
|---|---|---|---|
| Anaerobic Membrane Bioreactor (AnMBR) Biofouling [58] | 25°C | 35°C | Rapid TMP increase in 5 ± 2 days at 25°C vs. 18 ± 2 days at 35°C; more adhesive but less viscoelastic EPS layer at 25°C, exacerbating fouling. |
| Oral Temperature Sensor Equilibrium [60] | 34°C / 38.5°C / 43°C (Water Bath) | N/A | Median time to reach steady-state temperature was 380 seconds (range: 130 - 690 seconds), varying with temperature. |
| Electrochemical Aptamer Sensor Longevity [56] | 37°C (Bovine Serum) | 4°C (Buffer) | Achieved week-long operation at 37°C; stability challenges are significantly more pronounced at body temperature compared to refrigerated buffer. |
Table 2: Impact of Fouling and Strategies for Its Mitigation
| Strategy / Material | Experimental Context | Key Outcome / Performance |
|---|---|---|
| pH-triggered Polymer Coating (Eudragit S100) [59] | Implantable electrochemical sensor in biological media. | Delayed sensor activation and significant improvement in anti-biofouling characteristics compared to uncoated electrodes. |
| Zwitterionic Materials & Membranes [56] | Electrochemical aptamer sensor in raw serum at 37°C. | Critical for achieving week-long operation by protecting the surface from fouling by albumin and other solutes. |
| Polyethylene Glycol (PEG) & Derivatives [61] | Electrochemical immunosensor for tumor markers in serum. | Forms a hydration layer that acts as a physical barrier to protein adsorption, effectively reducing non-specific adsorption. |
| Increased Alkylthiolate Chain Length [56] | Self-assembled monolayer (SAM) on gold electrodes in serum. | Increased van der Waals interactions between monolayer molecules, reducing the rate of thermal desorption. |
Table 3: Reagent Toolkit for Enhancing Biosensor Stability
| Research Reagent / Material | Primary Function in Biosensing | Key Mechanism of Action |
|---|---|---|
| Eudragit S100 [59] | pH-responsive dissolvable coating for delayed sensor activation. | Dissolves at pH > 7, physically protecting the sensor during the initial inflammatory response and preventing biofouling. |
| Zwitterionic Polymers [56] [61] | Anti-fouling surface modification for electrodes and membranes. | Forms a strong electro-neutral hydration layer via ionic solvation, resisting non-specific protein adsorption. |
| Polyethylene Glycol (PEG) [61] | Anti-fouling surface coating to resist non-specific adsorption. | Its chains form a dense hydration layer via hydrogen bonding, creating a physical and energetic barrier to fouling agents. |
| C16 Alkylthiolates [56] | Passivating molecule in self-assembled monolayers (SAMs) on gold. | Longer carbon chains increase van der Waals interactions, enhancing SAM density and stability against thermal desorption. |
Mechanisms and Interrelationships: A Systems View
The challenges of temperature, pH, and fouling are not isolated; they form a complex, interconnected network that collectively dictates the stability of a biosensor in a complex environment. The following diagram maps these critical relationships and their impact on the final sensor output.
Experimental Protocols for Stability Assessment
To systematically evaluate and mitigate the impacts of these challenges, researchers should incorporate the following standardized protocols.
Protocol for Temperature Stability Profiling
This protocol is adapted from methods used to validate intra-oral temperature sensors and assess long-term aptamer sensor function [60] [56].
- Objective: To quantify the impact of temperature on sensor signal drift, response time, and operational longevity.
- Materials:
- Biosensor prototype.
- Thermostatic water bath with calibrated heater (accuracy ±0.1°C).
- Certified reference thermometer (e.g., PT1000 Wired Digital Thermometer).
- Data logging system.
- Procedure:
- Set the water bath to a target temperature relevant to the application (e.g., 25°C for ambient water, 37°C for body temperature).
- Allow the bath to stabilize for at least 30 minutes before measurement.
- Submerge the biosensor and reference thermometer in the bath, ensuring proximity.
- Log the output from both sensors simultaneously at 10-second intervals for a minimum of 30 minutes or until a steady-state is confirmed.
- Repeat the experiment across a range of temperatures (e.g., 4°C, 25°C, 35°C, 45°C).
- Data Analysis:
- Calculate the mean absolute error and root mean squared error between the biosensor and the reference.
- Determine the time to reach steady-state (defined as, for example, 60 seconds of continuous data with variation < 0.02°C) [60].
- For longevity tests, operate the sensor continuously at 37°C in a relevant complex fluid like serum, monitoring the decay of the signal-to-noise ratio over days [56].
Protocol for Evaluating pH and Fouling Resistance
This protocol is based on anti-fouling research for implantable sensors and electrochemical immunosensors [59] [61].
- Objective: To validate the efficacy of anti-fouling coatings and the sensor's resilience to pH variations.
- Materials:
- Coated and uncoated (control) biosensors.
- Complex biological media (e.g., undiluted bovine serum, fetal bovine serum (FBS), artificial wastewater).
- Buffer solutions spanning a physiologically relevant pH range (e.g., pH 4.0, 7.4, 9.0).
- Electrochemical workstation (e.g., potentiostat).
- Procedure:
- pH Activation Test: Immerse a polymer-coated sensor (e.g., Eudragit S100) in a buffer at the trigger pH (e.g., >7). Monitor the dissolution of the coating and the subsequent activation of the sensor signal [59].
- Fouling Resistance Test: a. Incubate coated and uncoated sensors in the chosen complex medium (e.g., 100% FBS) at a constant temperature (e.g., 37°C). b. At predetermined time intervals (e.g., 1, 6, 24, 72 hours), remove the sensors, rinse gently with buffer, and perform electrochemical characterization. c. Use techniques such as Electrochemical Impedance Spectroscopy (EIS) or Cyclic Voltammetry (CV) in a standard redox probe solution (e.g., Ferri/Ferrocyanide, [Fe(CN)₆]³⁻/⁴⁻). An increase in charge transfer resistance (Rₑₜ) or a decrease in peak current indicates fouling.
- Data Analysis:
The experimental workflow for assessing pH and fouling resistance, from sensor preparation to data interpretation, is outlined below.
The path to reliable biosensing for emerging contaminants in real-world samples runs directly through the core challenges of temperature, pH, and fouling. This guide has detailed how these factors quantitatively impact sensor performance and has provided actionable experimental protocols for their assessment. By adopting a rigorous, standardized approach to stability testing—incorporating the profiling and validation methods outlined herein—researchers can significantly enhance the reproducibility and reliability of their data. The integration of advanced materials, such as zwitterionic polymers and smart pH-responsive coatings, presents a promising frontier. Moving forward, the systematic implementation of these strategies is paramount for translating the considerable promise of biosensing technologies into robust, field-deployable solutions that can effectively safeguard environmental and public health.
This technical guide outlines advanced engineering strategies for enhancing the performance of biosensors, with a specific focus on applications for detecting emerging contaminants (ECs) in water environments. ECs, which include pharmaceuticals, pesticides, and industrial chemicals, pose a significant threat to environmental and human health, often at trace concentrations [26]. Biosensors, which combine a biological recognition element with a transducer, offer a promising alternative to conventional analytical methods due to their potential for sensitivity, specificity, and on-site deployment [22]. This whitepaper details the methodologies, experimental protocols, and material solutions for developing next-generation biosensors through three core engineering approaches: directed evolution of biomolecules, synthetic promoter engineering, and the integration of hybrid nanomaterials.
Advanced Biosensor Engineering Strategies
Directed Evolution of Biosensor Components
Directed evolution mimics natural selection in the laboratory to engineer biomolecules with enhanced properties, such as increased sensitivity, altered specificity, or improved stability. This process involves iterative cycles of genetic diversification and screening or selection for desired traits [62].
Key Methodologies and Workflow: The general workflow begins with the creation of a diverse library of gene variants. This is followed by a high-throughput screening or selection process to identify improved variants, which are then isolated and amplified for the next round of evolution [62].
Table 1: Common Techniques for Genetic Diversification in Directed Evolution
| Technique | Purpose | Key Advantages | Key Limitations |
|---|---|---|---|
| Error-prone PCR | Introduction of random point mutations across the entire gene sequence. | Easy to perform; does not require prior structural knowledge. | Biased mutagenesis spectrum; limited sampling of sequence space. |
| DNA Shuffling | Random recombination of sequences from multiple parent genes. | Recombines beneficial mutations; can lead to dramatic functional leaps. | Requires high sequence homology between parent genes. |
| Site-Saturation Mutagenesis | Targeted mutagenesis of specific amino acid positions to all possible variants. | In-depth exploration of key residues; enables "smart" library design. | Libraries can become very large; only a few positions are mutated. |
| RAISE | Insertion of random short insertions and deletions (indels). | Enables exploration of length diversity; mimics indels in natural evolution. | Can introduce frameshifts; indels are limited to a few nucleotides. |
A prime example is the directed evolution of the transcription factor CaiF, a biosensor for l-carnitine. The wild-type biosensor had a restricted detection range. Researchers used a Functional Diversity-Oriented Volume-Conservative Substitution Strategy on key amino acid sites, which were first identified using computer-aided design and alanine scanning. This approach led to the development of a CaiF variant (CaiFY47W/R89A) with a dramatically expanded dynamic range. The evolved biosensor responded to l-carnitine concentrations from 10⁻⁴ mM to 10 mM, a 1000-fold wider range, and produced a 3.3-fold higher output signal compared to the wild-type control [63].
Experimental Protocol: Directed Evolution via Error-Prone PCR and Screening This protocol outlines a standard method for evolving a protein-based bioreceptor.
Library Generation:
- Set up error-prone PCR reactions to amplify the target gene. Reaction conditions are optimized to introduce a low mutation rate (e.g., 0.5-2 mutations per gene) by adjusting parameters such as Mn²⁺ concentration or nucleotide imbalance.
- Clone the mutated PCR products into an appropriate expression vector and transform into a host cell line (e.g., E. coli).
Screening for Desired Phenotype:
- Plate the transformed cells to form isolated colonies.
- For biosensors, a common screening method is a colorimetric or fluorimetric assay. Colonies are grown in multi-well plates with the target analyte (e.g., an EC) and a reporter system that produces a measurable signal (e.g., fluorescence). This allows for high-throughput screening of thousands of variants [62].
- Using automated plate readers, identify clones that exhibit a significantly enhanced signal output or a shifted operational range compared to the parent biosensor.
Variant Isolation and Iteration:
- Isolate the plasmid DNA from the top-performing clones and sequence the gene to identify the beneficial mutations.
- Use these improved variants as the template for subsequent rounds of evolution to accumulate further enhancements.
Promoter Engineering and Regulatory Circuit Design
Promoter engineering involves the design and construction of synthetic genetic circuits to control the expression of reporter genes in whole-cell biosensors. This approach allows for the fine-tuning of sensitivity, dynamic range, and reduction of background noise.
Key Methodologies and Workflow: Whole-cell biosensors utilize engineered microorganisms, such as E. coli, that contain a genetic circuit where a promoter is activated by the target contaminant, leading to the expression of a detectable reporter protein (e.g., GFP, mRFP1) [64]. Advanced circuits incorporate regulatory elements like negative feedback loops to enhance performance.
Table 2: Performance of Engineered Whole-Cell Biosensors for Cadmium Detection
| Biosensor Name | Circuit Structure Key Features | Detection Limit for Cd²⁺ | Key Performance Outcome |
|---|---|---|---|
| CP100 | Single-input promoter (PT7-cadO) | Not specified (Met WHO requirement) | Baseline sensor with low response and sensitivity. |
| LC100 | Dual-input promoter (PT7-cadO-lacO-cadO) with signal amplifier | Not specified | Significantly improved response and sensitivity vs. CP100. |
| LC100-2 | Dual-input promoter with LacI negative feedback loop (LCPM-2 circuit) | 0.00001 - 0.02 nM | 3748-fold sensitivity increase vs. CP100; ultra-trace detection. |
A case study in optimizing a cadmium (Cd²⁺) biosensor demonstrates this approach. The initial sensor, CP100, used a synthetic promoter (PT7-cadO) controlled by the cadmium-binding protein CadR. While its detection limit met WHO requirements, its response was low. To amplify the signal, researchers introduced the lacI gene and lacO operator, creating a dual-input promoter and the LC100 biosensor. This improved sensitivity but introduced high background leakage. The final design, LC100-2, employed an autoregulatory negative feedback circuit (LCPM-2), where LacI protein is co-expressed with the reporter. This circuit simultaneously enhanced the response to Cd²⁺ and suppressed background leakage, achieving an ultra-trace detection limit of 0.00001 nM and a 3748-fold sensitivity increase over the original CP100 [64].
Experimental Protocol: Construction of a Whole-Cell Biosensor with a Feedback Circuit This protocol details the construction of an advanced biosensor with a regulatory circuit.
Circuit Design and Plasmid Construction:
- Design the genetic circuit, for example, the LCPM-2 circuit: "CadR-PJ23100-PT7-cadO-lacO-cadO-mRFP1-LacI" [64].
- Amplify all genetic components (e.g., cadR, cadO, lacI, lacO, promoter PJ23100, reporter gene mrfp1) via PCR.
- Assemble these components stepwise into a relaxed-low-copy-number plasmid vector (e.g., pET-28a+) using seamless cloning techniques. Verify the final plasmid sequence by sequencing.
Biosensor Strain Preparation and Testing:
- Transform the constructed plasmid into an appropriate expression host, such as E. coli BL21(DE3).
- Inoculate the sensor strain into a medium with antibiotic and grow overnight.
- Dilute the culture and transfer it to a fresh medium in a multi-well plate.
- Expose the sensor cells to a concentration gradient of the target analyte (e.g., CdCl₂) and incubate.
- Measure the output signal (e.g., fluorescence or optical density) using a microplate reader.
Data Analysis and Characterization:
- Calculate the response intensity based on the output signal. Generate a dose-response curve by plotting the response against the analyte concentration.
- Determine key performance parameters: limit of detection (LOD), dynamic range, sensitivity, and specificity against other potential interferents.
Integration of Hybrid Nanomaterials
Hybrid nanomaterials combine the unique physical and chemical properties of nanomaterials with the specific recognition capabilities of biological elements. This synergy results in significant signal amplification, improved stability, and lower detection limits.
Key Methodologies and Workflow: Nanomaterials such as graphene, gold nanoparticles (AuNPs), and quantum dots (QDs) are integrated into biosensor platforms as transducers, carriers, or signal amplifiers. The choice of nanomaterial depends on the required properties, such as high electrical conductivity, plasmonic behavior, or fluorescence [65].
Table 3: Performance of Nanomaterial-Enhanced Biosensors for Environmental Contaminants
| Nanomaterial | Biosensor Type / Target | Detection Limit | Key Advantage / Mechanism |
|---|---|---|---|
| Graphene | Immunosensor for Lead Ions (Pb²⁺) | 0.01 ppb | High resistivity and stability [65]. |
| Gold Nanoparticles (AuNPs) | Sensor for Mercury Ions (Hg²⁺) | 0.005 ppb | High sensitivity and plasmonic properties [65]. |
| Graphene-QD Hybrid | FET Biosensor for Biotin-Streptavidin | 0.1 fM (femtomolar) | Dual-mode (electrical/optical) detection via charge transfer [66]. |
| Albumin Nanoparticles | Immunoassay loaded with Europium complexes | Not specified (Applied to IgG detection) | Strong, long-lived luminescence for time-resolved detection [30]. |
The enhancement mechanisms are diverse. For electrochemical sensors, nanomaterials like AuNPs and MoS₂ can be used to modify electrodes, increasing the electroactive surface area and electron transfer kinetics, leading to higher sensitivity. For example, an electrochemical immunosensor for the cancer biomarker BRCA-1 used a nanocomposite of AuNPs, MoS₂, and chitosan on a pencil graphite electrode, achieving a low detection limit of 0.04 ng/mL [66]. In optical sensors, a graphene-quantum dot hybrid functions via a charge transfer mechanism. The photoluminescence of QDs is quenched when in close contact with graphene. Upon binding of the target analyte, this interaction is disturbed, leading to a recovery of fluorescence, enabling detection down to 0.1 fM [66]. Furthermore, nanomaterials can stabilize enzymes, enhancing the biosensor's operational lifespan, particularly in complex environmental matrices [22].
Experimental Protocol: Fabrication of a Nanocomposite-Modified Electrochemical Immunosensor This protocol describes the creation of a sensor with a nanomaterial-enhanced electrode.
Electrode Modification:
- Prepare a dispersion of the nanocomposite (e.g., AuNPs-MoS₂ in chitosan solution).
- Drop-cast a precise volume of the nanocomposite dispersion onto the surface of a clean working electrode (e.g., glassy carbon or pencil graphite).
- Allow the modified electrode to dry under ambient conditions or in an incubator to form a stable film.
Bioreceptor Immobilization:
- Incubate the modified electrode with a solution containing the specific capture bioreceptor (e.g., an antibody or aptamer against the target EC).
- Wash the electrode thoroughly with a buffer solution to remove any unbound bioreceptors.
- Block the remaining active sites on the electrode surface with a blocking agent (e.g., Bovine Serum Albumin - BSA) to prevent non-specific binding.
Electrochemical Detection and Analysis:
- Incubate the functionalized electrode with the sample containing the target analyte.
- After washing, perform electrochemical measurements (e.g., electrochemical impedance spectroscopy (EIS) or differential pulse voltammetry (DPV)) in a suitable redox probe solution (e.g., [Fe(CN)₆]³⁻/⁴⁻).
- Quantify the analyte concentration by correlating the change in the electrochemical signal (e.g., charge transfer resistance or peak current) to a calibration curve.
The Scientist's Toolkit: Essential Research Reagents and Materials
Table 4: Key Reagents and Materials for Biosensor Engineering and Development
| Item | Function / Application | Specific Examples |
|---|---|---|
| Error-Prone PCR Kit | Library generation for directed evolution. | Commercial kits from suppliers like Thermo Fisher or Takara. |
| pET-28a(+) Plasmid | A common relaxed-low-copy-number vector for genetic circuit construction in E. coli. | Used in the construction of Cd²⁺ WCBs [64]. |
| Synthetic Oligonucleotides | For gene synthesis, PCR amplification, and assembly of genetic circuits. | Used to create synthetic promoters (e.g., PT7-cadO) [64]. |
| Gold Nanoparticles (AuNPs) | Enhance electrochemical signal and provide a surface for bioreceptor immobilization. | Used in BRCA-1 immunosensor and Hg²⁺ detection [65] [66]. |
| Molybdenum Disulfide (MoS₂) | A 2D nanomaterial that provides a high surface area and catalytic properties for sensing. | Component of the nanocomposite in the BRCA-1 immunosensor [66]. |
| Chitosan | A biopolymer used to form stable films and facilitate the attachment of nanomaterials to electrodes. | Used in the BRCA-1 sensor matrix [66]. |
| Restriction Enzymes & Cloning Kits | For molecular biology work in plasmid construction. | e.g., from New England Biolabs or TransGen Biotech [64]. |
| Microplate Reader | For high-throughput screening of whole-cell biosensors or immunoassays. | Measures fluorescence/absorbance in 96-well or 384-well formats. |
The monitoring of emerging contaminants (ECs)—including pharmaceuticals, pesticides, personal care products, and perfluoroalkyl substances—is critical for safeguarding water safety and public health [22] [23]. These compounds, often unregulated and not routinely monitored, can pose significant risks to ecosystems and human health even at trace concentrations [31]. Traditional analytical methods, such as liquid chromatography-mass spectrometry (LC-MS) and gas chromatography-mass spectrometry (GC-MS), provide high sensitivity and accuracy but are constrained by their high cost, complex operation, need for professional personnel, and lack of real-time monitoring capabilities [22] [31]. These limitations render them unsuitable for rapid, on-site decision-making in resource-limited settings.
Biosensing technologies have emerged as a potent alternative, leveraging the specificity of biological recognition elements such as enzymes, antibodies, aptamers, and whole cells [22] [31]. The current frontier in this field involves transforming these laboratory-based biosensors into portable, disposable, and cost-effective systems for field deployment. This transition is driven by the growing demand for point-of-care testing (POCT) that enables rapid, on-site analysis, facilitating immediate intervention and continuous environmental monitoring [22] [67]. This technical guide outlines the core design principles, material selections, and integration strategies essential for developing next-generation portable biosensing systems.
Core Design Principles for Portability and Low Cost
The development of portable biosensing platforms rests on three foundational pillars: the strategic use of disposable materials, the integration of miniaturized components, and the simplification of user operation.
Disposability and Low-Cost Fabrication: The primary goal is to create single-use devices that prevent cross-contamination and eliminate the need for complex regeneration protocols. Materials such as polydimethylsiloxane (PDMS), polymethylmethacrylate (PMMA), and, most notably, paper substrates are widely employed [39]. Paper-based microfluidic analytical devices (μPADs) leverage capillary action to fluid movement, removing the requirement for external pumps. Their porous nature allows for easy modification with chemical and biological reagents, and they can be manufactured using simple techniques like inkjet printing or wax patterning, making them exceptionally cost-effective [39].
Miniaturization and System Integration: Miniaturization consolidates multiple analytical steps—including sample preparation, reaction, separation, and detection—onto a single, compact chip or device, often referred to as a Micro Total Analysis System (μTAS) [39]. This approach significantly reduces the consumption of samples and reagents (volumes in the microliter range or lower) and shortens analysis times [68] [39]. A key design innovation is the creation of instrument-free operation mechanisms. For example, manual squeezing of a dropper bulb can induce vortex flow for rapid mixing and incubation, while embedded magnetic elements can facilitate separation without external power [68].
User-Centric Design for Field Use: For successful deployment in non-laboratory environments, devices must be designed for simplicity. This involves minimizing the number of operational steps and integrating intuitive, smartphone-based readout systems. Smartphones, with their advanced cameras, processing power, and connectivity, serve as powerful tools for capturing analytical signals (e.g., colorimetric changes) and performing quantitative analysis with dedicated applications, making them ideal for point-of-need testing [67] [68].
Material Selection and Fabrication Techniques
The choice of material is paramount, as it dictates the fabrication method, cost, performance, and applicability of the biosensor.
Table 1: Comparison of Common Materials for Disposable Microfluidic Biosensors
| Material | Key Advantages | Key Limitations | Common Fabrication Methods | Example Application |
|---|---|---|---|---|
| Paper | Very low cost, disposable, capillary-driven flow (pump-free), easy functionalization. | Limited structural integrity, can be sensitive to environmental conditions. | Wax printing, inkjet printing, screen printing. | Lateral flow assays for pathogens [39]. |
| Polydimethylsiloxane (PDMS) | Optically transparent, flexible, gas-permeable, biocompatible. | Hydrophobic, prone to nonspecific adsorption of molecules. | Soft lithography, replica molding. | Organ-on-a-chip models, microfluidic channels [39]. |
| Polymethylmethacrylate (PMMA) | Good optical clarity, rigid, low cost. | Requires high temperatures for thermoforming. | Laser cutting, hot embossing, injection molding. | Optical detection chips for mycotoxins [39]. |
| Polyethylene (Dropper) | Extremely low cost, integrates sample handling and detection, disposable. | Limited to simpler fluidic designs. | Commercial sourcing, 3D printing integration. | Integrated biosensor for Salmonella [68]. |
Beyond the base substrate, the functionalization of these materials with biorecognition elements is critical. Aptamers (synthetic single-stranded DNA or RNA) are particularly advantageous for portable systems due to their thermal stability, ease of chemical synthesis, and ability to be selected for a wide range of targets via the SELEX (Systematic Evolution of Ligands by Exponential Enrichment) process [22] [31]. Antibodies offer high specificity and are well-suited for immunosensors [69] [68], while whole cells can provide self-replicating sensing elements for monitoring toxicity or specific metabolic compounds [30] [31].
Detection Mechanisms and Signal Transduction
The transduction mechanism converts the biological recognition event into a quantifiable signal. For field-portable devices, the mechanism must be compatible with miniaturization and simple readout.
Colorimetric Detection: This is one of the most widely used methods for POC devices due to its simplicity. The output—a visible color change—can often be assessed by the naked eye or quantified using a smartphone camera. Gold nanoparticles (AuNPs) are frequently used as colorimetric reporters; a red-to-blue color shift occurs upon nanoparticle aggregation induced by target binding [67]. Nanozymes (nanomaterials with enzyme-like activity) are also gaining traction as stable and cost-effective signal amplifiers for colorimetric reactions [68].
Electrochemical Detection: Electrochemical biosensors are highly attractive for portability due to their high sensitivity, low power requirements, and ease of miniaturization with screen-printed electrodes [22] [69]. Techniques such as amperometry and impedimetry can detect changes in current or impedance resulting from the binding of a target to a bioreceptor on an electrode surface. For instance, a Bioelectric Recognition Assay (BERA) uses the electrophysiological response of membrane-engineered cells as a sensitive readout for fungicides like boscalid [69].
Fluorescence Detection: While often requiring more complex optics, fluorescence-based detection offers high sensitivity. Smartphone-based systems can be adapted for fluorescence readout by using the camera with an additional light source and optical filters [30]. For example, whole-cell biosensors can be engineered to express a green fluorescent protein (eGFP) in the presence of a target like cobalt [30].
The following diagram illustrates a generalized workflow integrating these elements into a portable biosensing system.
Integrated Experimental Protocols
This section provides a detailed methodological breakdown for two representative portable biosensor types: a dropper-based immunoassay and a paper-based nucleic acid sensor.
Protocol: Dropper-Based Immunomagnetic Biosensor for Pathogen Detection
This protocol details the construction and operation of a fully integrated dropper-based biosensor for detecting Salmonella typhimurium [68].
Research Reagent Solutions:
- Carboxylated Magnetic Beads (180 nm): Serve as a solid support for immunomagnetic separation.
- Anti-S. typhimurium Polyclonal/Monoclonal Antibodies: Provide specific recognition for the target pathogen.
- Au@Pt Core-Shell Nanozymes: Act as a catalytic label for signal amplification, mimicking peroxidase activity.
- TMB (3,3',5,5'-Tetramethylbenzidine) Substrate: A chromogenic substrate that produces a blue color in the presence of the nanozyme and H₂O₂.
Step-by-Step Procedure:
- Functionalization of Magnetic Beads: Conjugate anti-Salmonella antibodies to the surface of carboxylated magnetic beads using a standard EDC/NHS cross-linking chemistry protocol. Purify the conjugate and resuspend in a suitable buffer.
- Assembly of the Detection Complex:
- Aspirate the food sample (e.g., milk, juice) into the disposable plastic dropper.
- Add the antibody-functionalized magnetic beads and Au@Pt nanozymes into the dropper.
- Manual Incubation and Mixing: Repeatedly squeeze and release the dropper's bulb for approximately 5-10 minutes. This manual action creates a vortex flow, efficiently mixing the contents and facilitating the formation of the "bead-bacteria-nanozyme" sandwich complex.
- Magnetic Separation: Place the dropper tip into a custom 3D-printed magnetic field generator. The magnetic field immobilizes the bead-complexes on the inner wall of the dropper tip. Expel the unbound reagents and washing buffer by squeezing the bulb, thereby purifying the captured target.
- Colorimetric Detection: Add the TMB substrate solution into the dropper. The Au@Pt nanozymes catalyze the oxidation of TMB, producing a blue color. The intensity of the color is directly proportional to the concentration of the captured pathogen.
- Smartphone Quantification: Capture an image of the dropper's tip using a smartphone. Use a dedicated color analysis application (e.g., a RGB color value detector) to quantify the color intensity, which is correlated with the target concentration via a pre-established calibration curve.
Protocol: On-Chip Colorimetric Aptasensor for RNA Detection
This protocol describes a chip-based, label-free method for detecting pathogen-specific RNA, such as from Cryptosporidium [67].
Research Reagent Solutions:
- Gold Nanoparticles (AuNPs, 20 nm): Function as the colorimetric reporting element.
- Thiolated Oligonucleotide Probes: Single-stranded DNA sequences complementary to adjacent regions on the target RNA; modified with a thiol group for covalent attachment to AuNPs.
- Saline Solution (NaCl): Used to induce aggregation of unhybridized AuNPs.
Step-by-Step Procedure:
- Functionalization of AuNPs: Incubate the thiolated oligonucleotide probes with the citrate-capped AuNPs. The thiol groups form a stable bond with the gold surface, creating a layer of DNA that stabilizes the nanoparticles against aggregation in saline solution.
- Chip-Based Assay Assembly: Pipette the functionalized AuNP solution onto a microfabricated chip, often housed within a 3D-printed holder for alignment and stability.
- Sample Hybridization: Introduce the extracted RNA sample directly to the AuNP solution on the chip. If the target RNA is present, it hybridizes with the two adjacent probes on different AuNPs, forming a cross-linked network.
- Induction of Aggregation and Color Shift: Add a controlled volume of saline solution. The cross-linked AuNPs aggregate due to the neutralization of surface charge, resulting in a visible color change from red to blue. Unhybridized, probe-coated AuNPs remain dispersed and red due to their protection from salt-induced aggregation.
- Smartphone Imaging and Analysis: Use a smartphone mounted on the 3D-printed holder to capture an image of the chip. Analyze the image using a dedicated app to measure the color shift quantitatively, providing a concentration of the target RNA.
The Scientist's Toolkit: Essential Research Reagents
Table 2: Key Research Reagent Solutions for Portable Biosensor Development
| Reagent / Material | Function | Example in Use |
|---|---|---|
| Gold Nanoparticles (AuNPs) | Colorimetric reporter; color shifts upon aggregation from target binding. | Detection of Cryptosporidium RNA [67]. |
| Au@Pt Core-Shell Nanozymes | Signal amplification; catalyzes chromogenic reaction for enhanced sensitivity. | Detection of S. typhimurium in a dropper biosensor [68]. |
| Carboxylated Magnetic Beads | Solid support for immunomagnetic separation and concentration of target. | Isolation of S. typhimurium from food samples [68]. |
| Thiolated DNA/Oligonucleotide Aptamers | Biorecognition element; binds specific targets (ions, molecules, cells). | Aptasensors for pharmaceuticals and toxins [22] [67]. |
| Screen-Printed Electrodes (SPEs) | Disposable electrochemical transducer for signal measurement. | Portable electrochemical immunosensors [69]. |
| TMB (3,3',5,5'-Tetramethylbenzidine) | Chromogenic substrate for peroxidase-like enzymes/nanozymes. | Generating blue color for optical detection of pathogens [68]. |
| Membrane-Engineered Cells | Living biorecognition element; responds to analytes with electric potential change. | Detection of boscalid fungicide via BERA [69]. |
The path to effective field-deployable biosensors is paved with strategic design choices that prioritize disposability, cost-effectiveness, and user-friendliness without sacrificing analytical performance. The integration of novel materials like paper and polymers, innovative fluidic handling such as finger-actuated droppers, and the ubiquitous power of the smartphone creates a powerful toolkit for addressing the global challenge of monitoring emerging contaminants.
Future advancements will likely involve the deeper integration of artificial intelligence (AI) to enhance signal interpretation, reduce false positives, and enable predictive analytics [70]. Furthermore, the development of multiplexed platforms capable of simultaneously detecting dozens of contaminants in a single run will be crucial for comprehensive environmental monitoring. As these technologies mature and standardize, they hold the promise of transforming environmental and food safety monitoring, shifting the paradigm from centralized laboratories to distributed, real-time, on-site surveillance networks.
Benchmarking Performance: Validation Against Gold Standards and Future Market Trends
The accurate and efficient detection of analytes, from disease biomarkers to environmental contaminants, is a cornerstone of modern scientific research and public health. For decades, conventional analytical methods have been the reference standard in laboratories. However, the emergence of complex challenges, such as monitoring trace levels of emerging contaminants (ECs) and the need for rapid, on-site diagnostics, has highlighted the limitations of these traditional techniques [26]. In response, biosensing technologies have evolved as a powerful alternative, offering the potential for decentralized, real-time analysis [71]. This technical guide provides a direct, detailed comparison between biosensors and conventional analytics, focusing on the critical performance parameters of limit of detection (LOD), cost, speed, and operational workflow, with specific application to the analysis of emerging contaminants.
The core distinction lies in their fundamental operation. Conventional methods typically rely on sophisticated physical separation and detection of analytes, requiring extensive sample preparation and centralized laboratories [72] [26]. In contrast, a biosensor is an integrated analytical device that uses a biological recognition element (e.g., enzyme, antibody, aptamer) in direct spatial contact with a transducer that converts the biorecognition event into a measurable signal [73]. This direct coupling is key to the advantages biosensors offer in point-of-care and environmental field testing.
Performance Comparison: Quantitative Data
The following tables summarize a direct comparison of key performance indicators between conventional analytical techniques and biosensors.
Table 1: Comparison of Analytical Performance and Operational Workflow
| Parameter | Conventional Analytical Methods | Biosensors |
|---|---|---|
| Limit of Detection (LOD) | Extremely low (e.g., ng/L to pg/L for LC-MS/MS) [26] | Ranges from ng/L to g/L; generally higher than conventional methods but continually improving with nanotechnology [26] [74] |
| Sensitivity | High, driven by powerful detectors (e.g., MS) [26] | High and enhanced by nanomaterials (e.g., Au nanoparticles, CNTs) which increase surface area and electron transfer [74] [71] |
| Selectivity/Specificity | Achieved through physical separation (e.g., chromatography) [26] | Inherent from biorecognition elements (antibody-antigen, enzyme-substrate) [74] [22] |
| Analysis Speed | Minutes to hours per sample; includes lengthy preparation [72] [26] | Seconds to minutes; rapid response due to direct measurement [71] [22] |
| Sample Throughput | High for automated systems in batch analysis | Typically lower, but ideal for single or sequential rapid tests |
| Sample Volume | Relatively large volumes often required | Minimal sample volume required (microliters) [71] |
| Sample Preparation | Complex, multi-step processes (extraction, purification, enrichment) often required [72] [26] | Minimal to no preparation; often capable of analyzing complex matrices directly [71] |
Table 2: Comparison of Cost, Usability, and Application
| Parameter | Conventional Analytical Methods | Biosensors |
|---|---|---|
| Equipment Cost | Very high (e.g., HPLC, GC-MS, ICP-MS) [26] | Low to moderate; portable readers are inexpensive [26] [75] |
| Operational Cost | High (skilled labor, maintenance, expensive reagents) | Low, especially for disposable sensors [75] |
| Per-Test Cost | High | Very low, driven by disposable test strips (e.g., glucose strips) [75] |
| Portability | Non-existent; confined to laboratory settings | High; devices are handheld and battery-operated for field use [26] [71] |
| Ease of Use | Requires highly trained and skilled personnel [72] [26] | Simple; can be operated by non-specialists [71] |
| Real-Time Monitoring | Not suitable; provides historical data [22] | Excellent capability for continuous, real-time, and in-situ monitoring [74] [22] |
| Primary Applications | Laboratory-based reference analysis, multi-analyte profiling | Point-of-care diagnostics, environmental field monitoring, food safety screening [72] [26] [71] |
Underlying Mechanisms and Experimental Protocols
The stark differences in performance and workflow stem from the fundamental mechanisms employed by each approach.
Conventional Analytical Methodology
The gold standard for detecting emerging contaminants, such as pharmaceuticals and pesticides, often involves liquid chromatography coupled with tandem mass spectrometry (LC-MS/MS).
- Principle: This technique separates compounds in a mixture based on their interaction with a stationary and a mobile phase (chromatography) and then identifies and quantifies each separated compound based on its mass-to-charge ratio (mass spectrometry) [26].
- Detailed Protocol for Water Analysis of ECs:
- Sample Collection and Preservation: Water samples are collected in chemically inert bottles, often acidified, and stored at 4°C to prevent degradation.
- Sample Pre-concentration and Extraction: Due to low analyte concentrations (ng/L), solid-phase extraction (SPE) is used. The sample is passed through a cartridge containing a sorbent material (e.g., C18), which traps the target contaminants.
- Clean-up and Elution: Interfering matrix components are washed off with a mild solvent. The trapped analytes are then released ("eluted") using a small volume of a strong organic solvent (e.g., methanol).
- Concentration and Reconstitution: The eluent is evaporated to dryness under a gentle stream of nitrogen to concentrate the analytes. The residue is then re-dissolved in a solvent compatible with the LC mobile phase.
- Instrumental Analysis (LC-MS/MS): The extract is injected into the LC system. Separated analytes elute at different retention times and are introduced into the MS/MS. Here, they are ionized (e.g., by electrospray ionization), and specific ion fragments are monitored for highly selective and sensitive quantification.
- Data Analysis: The concentration in the sample is determined by comparing the analyte's signal intensity to a calibration curve prepared from standard solutions [26].
Biosensor Signaling Pathways and Workflows
Biosensors function via a biorecognition event coupled to a signal transduction pathway. A generalized workflow and specific examples are detailed below.
Figure 1: Generalized biosensor workflow.
Experimental Protocol: Developing an Electrochemical Aptasensor for an Emerging Contaminant
- Transducer Modification: A working electrode (e.g., glassy carbon or gold) is polished and cleaned. To enhance sensitivity, it is modified with a nanomaterial, such as a dispersion of graphene oxide or gold nanoparticles, and dried [74] [22].
- Aptamer Immobilization: A solution containing a specific DNA or RNA aptamer (bioreceptor) selected for the target EC (e.g., Bisphenol A) is drop-cast onto the modified electrode. The electrode is incubated in a humid chamber to allow the aptamers to covalently or physically adsorb onto the surface.
- Blocking: The electrode is rinsed and treated with a blocking agent (e.g., bovine serum albumin or ethanolamine) to cover any remaining non-specific binding sites, thus minimizing background signal.
- Measurement (Calibration and Analysis):
- The functionalized electrode is connected to a potentiostat.
- For a calibration curve, the electrode is exposed to standard solutions with known concentrations of the target analyte.
- The binding of the target to the immobilized aptamer causes a measurable change in an electrochemical parameter (e.g., a decrease in current in voltammetry or an increase in charge transfer resistance in impedimetry) [22].
- This signal change is recorded and plotted against the analyte concentration to create the calibration curve.
- An unknown sample is then tested, and its concentration is determined from the calibration curve.
Table 3: Research Reagent Solutions for Biosensor Development
| Reagent / Material | Function in Experimental Protocol |
|---|---|
| Aptamers / Antibodies | Acts as the biorecognition element that specifically binds to the target analyte [26] [22]. |
| Gold Nanoparticles (AuNPs) | Nanomaterial used to modify the transducer surface, enhancing electrical conductivity and surface area for greater signal amplification [74] [22]. |
| Graphene Oxide / Carbon Nanotubes (CNTs) | Nanomaterials used in electrode modification to improve electron transfer kinetics and provide a high surface area for bioreceptor immobilization [74] [71]. |
| Bovine Serum Albumin (BSA) | A common blocking agent used to passivate the sensor surface and prevent non-specific binding of other molecules, which reduces false signals [22]. |
| Electrochemical Cell & Potentiostat | The platform and instrument used to apply electrical potentials and measure the current or impedance response of the biosensor [72]. |
Discussion and Future Perspectives
The data clearly illustrates a trade-off. Conventional analytics remain unparalleled for achieving the lowest possible LODs and providing definitive, multi-analyte validation in a controlled lab setting. Their primary drawbacks are cost, speed, and lack of portability. Biosensors excel in providing rapid, low-cost, and on-site analysis with sufficient sensitivity for a vast range of applications, from clinical diagnostics to environmental early-warning systems [72] [26] [71].
The future of biosensing lies in overcoming current challenges, such as the stability of biological elements and reproducibility in complex real-world samples. Key research directions include:
- Integration of Nanomaterials: The use of advanced materials like metal-organic frameworks (MOFs) and carbon nano-onions (CNOs) will continue to push the boundaries of sensitivity and stability [71] [22].
- Point-of-Care (POC) Platforms: The development of paper-based microfluidic devices and other POC formats will further simplify testing and expand accessibility [72] [22].
- Artificial Intelligence (AI): AI is being integrated with biosensors to enhance data processing, improve pattern recognition for complex samples, and even enable predictive diagnostics, though attention must be paid to mitigating potential false results from both the sensor and the algorithm [73].
- Multiplexing: A significant frontier is the development of biosensors capable of simultaneously detecting multiple contaminants or biomarkers in a single test [71].
In conclusion, the choice between biosensors and conventional analytics is not a matter of superiority but of application-specific suitability. For routine, high-precision, multi-analyte lab work, conventional methods are indispensable. For rapid, decentralized, and cost-effective monitoring, biosensors represent a transformative technological leap. The ongoing research and development in biosensing technologies promise to further narrow the performance gap while unlocking entirely new capabilities in analytical science.
The accurate detection of emerging contaminants (ECs) in complex environmental matrices represents a significant challenge in environmental monitoring and public health protection. Complex matrices—such as wastewater, surface water, and biological samples—contain numerous interfering substances that can compromise analytical accuracy. Validation ensures that analytical methods produce reliable, reproducible data that can withstand scientific and regulatory scrutiny, particularly for contaminants present at trace concentrations (ng/L to μg/L) amidst complex background interference [76] [31]. In the context of a broader thesis on emerging contaminants and biosensing technologies, understanding validation principles becomes paramount for translating laboratory research into field-deployable solutions for environmental surveillance.
The critical importance of rigorous validation is underscored by the environmental and health risks posed by ECs. These compounds, including pharmaceuticals, endocrine disruptors, and pesticides, can exert adverse effects such as endocrine disruption, mutagenesis, and carcinogenesis even at exceptionally low concentrations [31]. As regulatory bodies move toward establishing stricter controls for these contaminants, the demand for properly validated methods that can reliably quantify ECs in complex matrices has intensified, bridging the gap between analytical chemistry, environmental science, and public health policy.
Key Validation Parameters and Performance Criteria
Method validation requires demonstrating that an analytical procedure meets predefined acceptance criteria for multiple parameters. The International Council for Harmonisation (ICH) guidelines Q2(R2) provide a framework for validating analytical procedures, emphasizing specificity, linearity, accuracy, precision, and sensitivity [76].
Specificity refers to the method's ability to unequivocally assess the analyte in the presence of potential interferents. In complex matrices, this is particularly challenging as matrix components may co-elute or produce similar signals to the target analytes. For chromatographic methods, specificity is often demonstrated through chromatographic resolution between analytes and potential interferents [76]. For biosensors, specificity depends on the biorecognition element's affinity for the target analyte compared to structurally similar compounds [22] [31].
Linearity demonstrates the method's ability to produce results directly proportional to analyte concentration. It is established across a specified range and evaluated using statistical methods such as correlation coefficients, with values ≥0.999 representing excellent linearity [76].
Accuracy reflects the closeness of measured values to the true value, typically assessed through recovery studies where known amounts of analyte are spiked into the matrix. Recovery rates between 70-120% are generally considered acceptable, though wider ranges may be justified for complex matrices [76].
Precision expresses the degree of scatter between a series of measurements, usually evaluated at repeatability (within-day) and intermediate precision (between-day) levels. Precision is expressed as relative standard deviation (RSD%), with values <5.0% typically indicating acceptable method performance [76].
Sensitivity is defined through the limit of detection (LOD) and limit of quantification (LOQ). The LOD represents the lowest detectable concentration, while the LOQ is the lowest concentration that can be quantified with acceptable accuracy and precision [76].
Table 1: Validation Parameters and Performance Criteria for Analytical Methods in Complex Matrices
| Parameter | Definition | Performance Criteria | Assessment Method |
|---|---|---|---|
| Specificity | Ability to measure analyte unequivocally in matrix | No interference from matrix components | Chromatographic resolution, signal differentiation |
| Linearity | Proportionality of response to concentration | Correlation coefficient ≥0.999 | Regression analysis |
| Accuracy | Closeness to true value | Recovery rates 70-120% | Spike recovery studies |
| Precision | Degree of measurement scatter | RSD <5.0% | Repeated measurements |
| LOD | Lowest detectable concentration | Signal-to-noise ≥3:1 | Statistical evaluation |
| LOQ | Lowest quantifiable concentration | Signal-to-noise ≥10:1 | Statistical evaluation |
Validation Approaches for Different Analytical Techniques
Chromatographic Methods (UHPLC-MS/MS)
Liquid chromatography coupled with tandem mass spectrometry represents the gold standard for pharmaceutical analysis in complex matrices due to its high sensitivity and selectivity [76]. A validated UHPLC-MS/MS method for detecting carbamazepine, caffeine, and ibuprofen in water matrices demonstrates key validation parameters, with LODs of 100 ng/L, 300 ng/L, and 200 ng/L respectively, and LOQs of 300 ng/L, 1000 ng/L, and 600 ng/L [76]. The method exhibited excellent precision (RSD <5.0%) and accuracy (recovery rates 77-160%), with the latter range highlighting how complex matrices can sometimes yield recovery values outside ideal ranges due to matrix effects [76].
A significant innovation in green analytical chemistry involves modifying traditional sample preparation approaches. The omission of the evaporation step after solid-phase extraction (SPE) not only reduces solvent consumption and environmental impact but also demonstrates how validation can confirm the effectiveness of streamlined methodologies without compromising analytical performance [76].
Biosensing Platforms
Biosensors represent promising alternatives to conventional analytical techniques, offering potential for real-time, on-site monitoring of ECs [22] [31]. Validation of biosensors presents unique challenges due to the incorporation of biological recognition elements whose stability and activity may vary.
Enzyme-based biosensors employ enzymes as bioreceptors that catalyze reactions with the target analyte, producing detectable signals [31]. Validation must establish the sensor's performance under various environmental conditions and demonstrate specificity against potential interferents. These biosensors can operate through different mechanisms: the enzyme may metabolize the analyte, be inhibited by the analyte, or undergo characteristic changes in the presence of the analyte [31].
Aptasensors utilize synthetic single-stranded DNA or RNA aptamers as recognition elements, selected through Systematic Evolution of Ligands by Exponential Enrichment (SELEX) [31]. Validation includes demonstrating binding affinity through various electrostatic and intramolecular mechanisms, including π-π stacking, van der Waals forces, and hydrogen bonding [31]. Signal transduction in aptasensors typically relies on optical, electrochemical, and piezoelectric techniques.
Immunosensors (antibody-based biosensors) leverage the high specificity of antigen-antibody interactions [31]. They are categorized into label-free systems (detecting physical changes from binding events) and labeled systems (utilizing secondary molecules to generate signals) [31]. Validation must demonstrate minimal cross-reactivity with structurally similar compounds and consistent performance across matrix types.
Whole cell-based biosensors utilize microbial cells as integrated sensing systems [31]. A unique advantage is their ability to self-replicate, potentially enhancing signal detection over time [31]. Validation includes assessing robustness across application conditions and confirming sensing mechanisms based on metabolic activity, stress responses, or gene expression regulation.
Table 2: Comparison of Biosensing Platforms for Emerging Contaminant Detection
| Biosensor Type | Biorecognition Element | Detection Mechanism | Advantages | Validation Challenges |
|---|---|---|---|---|
| Enzyme-based | Enzymes | Catalytic transformation of analyte | High specificity, fast response | Enzyme stability, inhibition by matrix components |
| Aptasensors | DNA/RNA aptamers | Folding upon target binding | Chemical stability, design flexibility | Non-specific binding, reproducibility |
| Immunosensors | Antibodies | Antigen-antibody binding | Exceptional specificity, high affinity | Antibody stability, cross-reactivity |
| Whole cell-based | Microbial cells | Cellular response mechanisms | Robustness, self-regeneration | Response time, viability maintenance |
Experimental Protocols for Method Validation
UHPLC-MS/MS Method Validation for Pharmaceutical Compounds
Sample Preparation Protocol:
- Collect water/wastewater samples in clean glass containers
- Perform solid-phase extraction (SPE) without subsequent evaporation step [76]
- Reconstitute in mobile phase compatible solvent
- Inject into UHPLC-MS/MS system
Chromatographic Conditions:
- Analysis Time: 10 minutes [76]
- Stationary Phase: C18 column or equivalent
- Mobile Phase: Gradient elution with aqueous and organic phases
- Flow Rate: Optimized for separation (typically 0.2-0.5 mL/min)
Mass Spectrometric Detection:
- Ionization: Electrospray ionization (ESI) in positive or negative mode
- Detection: Multiple Reaction Monitoring (MRM) for selective detection
- Quantification: Internal standard method preferred for improved accuracy
Validation Experiments:
- Specificity: Analyze blank matrix samples to confirm no interference at retention times of target analytes [76]
- Linearity: Prepare calibration standards at 5-7 concentration levels across the expected range; calculate correlation coefficient [76]
- Accuracy: Spike samples with known concentrations of analytes at low, medium, and high levels (n=6 each); calculate percent recovery [76]
- Precision: Analyze replicated samples (n=6) at low, medium, and high concentrations within the same day (repeatability) and on different days (intermediate precision) [76]
- LOD/LOQ: Determine based on signal-to-noise ratio of 3:1 and 10:1, respectively, or using statistical methods [76]
Biosensor Validation Protocol
Sensor Preparation:
- Immobilize biorecognition element (enzyme, antibody, aptamer, cells) on transducer surface
- Block non-specific binding sites with appropriate blocking agent
- Characterize immobilization efficiency through appropriate methods
Calibration Procedure:
- Expose sensor to standard solutions of known concentrations
- Record response signal for each concentration
- Generate calibration curve relating signal intensity to analyte concentration
- Determine linear range and sensitivity from calibration data
Validation Experiments:
- Selectivity: Test sensor response to structurally similar compounds and potential interferents present in the matrix [31]
- Stability: Monitor sensor response over time under storage and operational conditions [31]
- Reproducibility: Evaluate response variability between different sensor batches [31]
- Matrix Effects: Compare sensor performance in buffer versus real samples; implement standard addition method if significant matrix effects are observed [31]
- Recovery: Spike real samples with known analyte concentrations; calculate recovery percentage [31]
Workflow Visualization
Validation Workflow for Analytical Methods
Statistical Approaches to Data Validation
Statistical analysis forms the foundation of objective method validation, providing quantitative measures of method performance. Exploratory Factor Analysis (EFA) serves as a valuable statistical technique for psychometric analysis in validation studies, particularly when developing new instruments or when a robust theoretical model is unavailable [77]. EFA helps identify a more limited set of variables or factors that explain the variability observed in the measured variables, thereby assessing construct validity by quantifying the extent to which items measure the intended constructs [77].
Key decisions in statistical validation include determining sample size, addressing missing values, assessing data factorability, deciding on the number of factors and items to retain, and evaluating scale reliability [77]. For analytical method validation, statistical approaches should include:
- Regression Analysis for establishing linearity, calculating correlation coefficients, and determining confidence intervals for slope and intercept
- Analysis of Variance (ANOVA) for evaluating precision data and identifying sources of variability
- Statistical Tests for Outliers to identify and address anomalous data points
- Calculation of Confidence Intervals for accuracy (recovery) and precision data
Principal Component Analysis (PCA) represents another powerful statistical tool for validation, as demonstrated in a study assessing surface water quality near an urban landfill, where PCA identified five principal components accounting for 92.16% of the Water Quality Index variation [78].
Case Studies and Applications
Pharmaceutical Monitoring in Water Matrices
A green/blue UHPLC-MS/MS method was developed and validated for trace pharmaceutical monitoring in water and wastewater, focusing on carbamazepine, caffeine, and ibuprofen as indicator compounds [76]. The method demonstrated exceptional sensitivity with LODs of 100 ng/L for carbamazepine, 300 ng/L for caffeine, and 200 ng/L for ibuprofen, with a short analysis time of 10 minutes [76]. This case study highlights the importance of selecting appropriate indicator compounds that represent different therapeutic classes and environmental behaviors.
The validation data confirmed method reliability across different water matrices, with the method applied in a case study from the Kraków area, demonstrating practical utility for environmental monitoring programs [76]. The success of this validation underscores how properly validated methods can support regulatory decision-making and pollution management strategies.
Surface Water Quality Assessment Near Urban Landfill
Advanced statistical techniques validated surface water quality assessment around an urban landfill in Dhaka, Bangladesh, where 19 physicochemical parameters were analyzed [78]. Cluster analysis revealed that samples collected nearer to the landfill exhibited excessive chemical oxygen demand (COD), biochemical oxygen demand (BOD), and total suspended solids (TSS), while samples from farther locations demonstrated superior water quality [78].
The Water Quality Index (WQI) categorized three samples as "very bad" (WQI <31) and seven as "bad" (WQI between 31 and 51.9), with the lowest value of 1.85 recorded from a sewer [78]. This case study illustrates how validated methods and statistical approaches can identify contamination gradients and support targeted intervention strategies.
The Scientist's Toolkit: Essential Research Reagents and Materials
Table 3: Essential Research Reagents and Materials for Analytical Method Validation
| Item | Function | Application Notes |
|---|---|---|
| Certified Reference Materials | Accuracy verification | Provides traceable standards for quantification |
| Internal Standards | Correction for variability | Isotope-labeled analogs for mass spectrometry |
| Solid-Phase Extraction Cartridges | Sample clean-up and concentration | C18, mixed-mode, or selective sorbents for different analyte classes |
| UHPLC Columns | Chromatographic separation | C18 or specialized stationary phases for specific separations |
| Mass Spectrometry Solvents | Mobile phase preparation | HPLC-grade solvents with low background interference |
| Biorecognition Elements | Target capture in biosensors | Enzymes, antibodies, aptamers with demonstrated affinity |
| Immobilization Matrices | Sensor surface preparation | Gold, carbon, or polymer surfaces for bioreceptor attachment |
| Blocking Agents | Reduction of non-specific binding | BSA, casein, or specialized commercial blockers |
| Signal Transduction Reagents | Signal generation in biosensors | Enzymatic substrates, electrochemical mediators, fluorescent dyes |
| Matrix-Matched Calibrants | Compensation for matrix effects | Standards prepared in analyte-free matrix |
Comprehensive validation of analytical methods for complex matrices requires a systematic, multi-parameter approach that addresses the unique challenges posed by each matrix type. The convergence of traditional chromatographic techniques with emerging biosensing platforms offers complementary approaches for detecting emerging contaminants across different concentration ranges and application scenarios. As regulatory requirements for emerging contaminants continue to evolve, properly validated methods will play an increasingly critical role in environmental monitoring, exposure assessment, and public health protection. The validation frameworks and experimental protocols outlined in this technical guide provide researchers with structured approaches for demonstrating method reliability, thereby supporting the generation of defensible data for scientific and regulatory decision-making.
The continuous production and release of new chemical and biological agents present a formidable challenge to global health and environmental sustainability [10]. Emerging contaminants (ECs)—including pharmaceuticals, personal care products, endocrine disruptors, per- and polyfluoroalkyl substances (PFAS), and microplastics—are detected in water environments at concentrations ranging from ng/L to μg/L, yet even at trace levels can induce harmful ecological impacts [9] [79]. Conventional analytical methods such as liquid chromatography-tandem mass spectrometry (LC-MS/MS) and gas chromatography-mass spectrometry (GC-MS), while highly accurate, are often impractical for large-scale monitoring due to their time-consuming procedures, expensive equipment requirements, and need for professional operators [39] [80]. This analytical gap has accelerated the adoption of tiered assessment approaches that strategically combine rapid screening with confirmatory analysis.
The tiered assessment paradigm operates on an iterative principle where each evaluation level aims to "strike a balance between the costs of adding detail and refinement to an assessment and the benefits associated with that additional refinement" [81]. In this framework, biosensors serve as powerful screening tools in initial tiers, using readily available data and conservative assumptions to estimate high-end exposure potential for sensitive receptors [81]. Subsequent tiers then employ more resource-intensive, refined assessments for contaminants that exceed screening thresholds. This systematic approach allows researchers and regulatory agencies to prioritize resources effectively, focusing sophisticated analytical capabilities on the most pressing contamination issues while efficiently screening out negligible risks.
Biosensing Technologies: Principles and Applications in Screening
Fundamental Biosensor Architecture and Operating Principles
A biosensor is an analytical device that integrates a biological recognition element with a transducer to produce a measurable signal proportional to the concentration of a target analyte [39]. The core components include: (1) a biometric element (e.g., antibody, aptamer, enzyme, molecularly imprinted polymer) that selectively binds to the target contaminant; (2) a signal transduction element (e.g., electrochemical, optical, piezoelectric) that converts the recognition event into a quantifiable signal; and (3) a display system for data recording and interpretation [39]. This integrated design enables rapid, sensitive detection that is particularly suitable for on-site screening applications in environmental monitoring.
The operational principle begins with the specific interaction between the biological recognition element and the target EC. This binding event produces a physicochemical change (e.g., mass, electrical charge, optical properties) that the transducer detects and converts into an electrical signal. For instance, in electrochemical biosensors, binding may alter interfacial properties measurable through impedance or capacitance changes, while optical biosensors may detect fluorescence, luminescence, or refractive index variations [39]. The resulting signal is then processed to quantify analyte concentration, often within minutes rather than the hours required by conventional methods.
Advanced Biosensor Platforms for Environmental Screening
Recent technological advances have significantly enhanced biosensor capabilities for EC detection. Microfluidic biosensors, also known as Lab-on-a-Chip or Micro Total Analysis Systems (μTAS), enable precise manipulation of small fluid volumes (10⁻⁶–10⁻¹⁵ L) in microfabricated channels, allowing for minimal reagent consumption, high-throughput analysis, and portable on-site detection [39]. These systems can be constructed from various materials including silicon, glass, polydimethylsiloxane (PDMS), polymethylmethacrylate (PMMA), and paper substrates, each offering distinct advantages for specific applications [39].
Paper-based microfluidic analytical devices (μPADs) represent particularly promising platforms for field-deployable screening. Their porous structure creates capillary action that eliminates the need for external pumps, while their low cost and simple manufacturing processes make them suitable for widespread deployment [39]. These devices can be fabricated using techniques such as screen printing, inkjet printing, photolithography, or embossing to form hydrophilic channels separated by hydrophobic barriers [39]. When functionalized with appropriate recognition elements, μPADs enable rapid, low-cost screening of ECs in remote or resource-limited settings.
The Tiered Assessment Model: Integrating Biosensors with Confirmatory Methods
Tiered Assessment Framework and Decision Logic
The tiered assessment model follows a sequential approach where initial screening tiers use conservative assumptions to identify potential hazards, while subsequent tiers apply increasingly refined methods to characterize risks more accurately [81]. This iterative process continues until the assessment achieves sufficient confidence for decision-making, resources are depleted, or additional data cannot be reasonably obtained [81]. The systematic progression through assessment tiers ensures efficient resource allocation while maintaining scientific rigor.
Table 1: Characteristics of Screening-Level and Refined Assessments in Tiered Analysis
| Assessment Component | Screening-Level Assessment | Refined Assessment |
|---|---|---|
| Input Data | Readily available data, conservative/default assumptions, point estimates | Site- or scenario-specific data, realistic assumptions, distributions of data |
| Tools and Methods | Simple models and equations, deterministic approach, biosensors | Complex models, probabilistic approach, advanced instrumentation |
| Results and Outputs | Conservative exposure estimate, useful for prioritization, greater uncertainty | Realistic exposure estimate, variability and uncertainty better characterized |
| Resource Requirements | Relatively inexpensive and quick to carry out | Resource-intensive, time-consuming |
| Primary Applications | Prioritization, comparing multiple sites, ruling out exposure pathways | Informed decision-making, regulatory actions, risk quantification |
The decision logic governing transitions between assessment tiers involves evaluating whether "this level of detail or degree of confidence is good enough to achieve the purpose of the assessment" after each iteration [81]. Screening-level assessments using biosensors typically employ a deterministic approach with single value (point estimate) inputs to generate conservative exposure estimates [81]. If these initial screenings indicate potential risks exceeding predetermined thresholds, the assessment progresses to higher tiers incorporating more sophisticated analytical approaches.
Table 2: Analytical Techniques in Tiered Assessment of Emerging Contaminants
| Analytical Technique | Limit of Detection | Analysis Time | Cost per Sample | Required Expertise | Primary Tier |
|---|---|---|---|---|---|
| Biosensors | Variable (pg/mL-ng/mL) | Minutes to hours | Low | Moderate | Screening (Tier 1) |
| Immunoassays | ng/mL range | 1-2 hours | Low to moderate | Moderate | Screening (Tier 1) |
| High-Performance Liquid Chromatography (HPLC) | ng/mL range | 10-30 minutes | Moderate | High | Confirmation (Tier 2) |
| Gas Chromatography-Mass Spectrometry (GC-MS) | ng/mL range | 30-60 minutes | High | High | Confirmation (Tier 2) |
| Liquid Chromatography-Tandem Mass Spectrometry (LC-MS/MS) | pg/mL range | 20-40 minutes | Very high | Very high | Definitive (Tier 3) |
Experimental Protocol for Tiered Contaminant Assessment
Tier 1: Biosensor-Based Screening Protocol
Sample Collection and Preparation: Collect water samples from monitoring sites using appropriate containers. For biosensor analysis, minimal pretreatment is typically required, though filtration (0.45 μm) may be necessary to remove particulate matter that could interfere with detection.
Biosensor Calibration: Prepare standard solutions of target analytes at known concentrations (e.g., 0, 0.1, 1, 10, 100 ng/mL). Measure biosensor response for each standard to establish a calibration curve. For optical biosensors, record signal intensity (fluorescence, absorbance); for electrochemical biosensors, measure current, potential, or impedance changes.
Limit of Detection (LoD) Determination: Following IUPAC protocols, perform replicate measurements (n≥10) of a blank sample (without analyte) to determine the mean blank signal (yB) and standard deviation (sB) [82]. Calculate the critical value (yC) of the signal using the formula: yC = yB + ksB, where k is a numerical factor chosen according to the desired confidence level (typically k=3, corresponding to 99.7% confidence) [82]. The concentration at the LoD (CLoD) is then determined from the calibration curve sensitivity (slope, a): CLoD = (yC - yB)/a = ksB/a [82].
Sample Analysis: Apply prepared samples to the biosensor platform following manufacturer protocols. For microfluidic devices, introduce samples via capillary action or controlled flow rates. Record signals and interpolate concentrations from the calibration curve. Include quality control samples (blanks, spikes) to validate analysis.
Data Interpretation and Tier Transition Decision: Compare detected concentrations with pre-established threshold levels (e.g., regulatory limits, risk-based screening values). If contaminant levels exceed thresholds, proceed to Tier 2 for confirmation. If below thresholds, no further action may be necessary.
Tier 2: Confirmatory Analysis Protocol Using Chromatographic Methods
Sample Extraction and Cleanup: For LC-MS/MS analysis, perform solid-phase extraction (SPE) to concentrate target analytes and remove matrix interferents. Condition SPE cartridges (e.g., C18, Oasis HLB) with methanol and water. Load samples, wash with appropriate solvents, and elute analytes with organic solvents (e.g., acetonitrile, methanol). Evaporate eluents under gentle nitrogen stream and reconstitute in mobile phase compatible solvents.
Instrumental Analysis:
- Chromatographic Separation: Utilize reverse-phase C18 columns with gradient elution (water/acetonitrile or water/methanol, both with 0.1% formic acid) to separate analytes.
- Mass Spectrometric Detection: Employ tandem mass spectrometry with electrospray ionization (ESI) in multiple reaction monitoring (MRM) mode. Optimize MRM transitions for each target compound (precursor ion > product ion). Use internal standards (isotope-labeled analogs when available) for quantification.
Quality Assurance/Quality Control: Include procedural blanks, matrix spikes, duplicate samples, and continuing calibration verification throughout the analysis batch. Ensure quantitation accuracy within ±20% of true values and precision ≤20% relative standard deviation.
Data Analysis: Integrate chromatographic peaks and calculate analyte concentrations using internal standard calibration curves with weighted (1/x or 1/x²) linear regression. Apply recovery corrections to field samples based on matrix spike results.
Tier 3: Refined Assessment Protocol
Site-Specific Exposure Modeling: Develop scenario-specific exposure models incorporating site measurement data, population characteristics, and exposure factors. Utilize probabilistic approaches to characterize variability and uncertainty in exposure estimates [81].
Advanced Analytical Techniques: For complex situations, employ non-targeted analysis using high-resolution mass spectrometry to identify unknown transformation products or previously unmonitored contaminants [83].
Risk Characterization: Integrate exposure estimates with toxicity data to quantify potential risks. Conduct sensitivity analysis to identify parameters with greatest influence on risk estimates and guide further data collection needs.
Critical Performance Parameters in Biosensor Design for Screening Applications
Limit of Detection (LoD) and the "LOD Paradox"
The limit of detection represents a fundamental performance parameter for biosensors, defined as the smallest analyte concentration that can be reliably distinguished from analytical noise [82]. Following IUPAC guidelines, LoD is mathematically determined as CLoD = k*sB/a, where sB is the standard deviation of blank measurements, a is the analytical sensitivity (calibration curve slope), and k is a numerical factor (typically 3) corresponding to the desired confidence level [82]. This statistical approach ensures reliable detection while minimizing false positives.
However, an intense focus on achieving ultra-low LoDs has created a "LOD paradox" in biosensor research [84]. While technological advances enabling picomolar or femtomolar sensitivity represent impressive scientific achievements, such extreme sensitivity may not always translate to practical utility. For many environmental applications, detecting contaminants well below their biologically relevant concentrations provides diminishing returns while potentially compromising other critical performance aspects such as detection range, robustness, cost-effectiveness, and user-friendliness [84]. A biosensor capable of detecting picomolar concentrations of a biomarker represents a technical feat, but if the biomarker's clinical or environmental relevance occurs in the nanomolar range, such excessive sensitivity becomes redundant while potentially increasing complexity and cost [84].
Comprehensive Analytical Validation for Biosensors
Robust validation of biosensor performance extends beyond LoD determination to include multiple analytical figures of merit:
Selectivity/Specificity: Evaluate cross-reactivity with structurally similar compounds that may be present in environmental samples. Determine percentage cross-reactivity by comparing dose-response curves for target analyte versus interferents.
Accuracy and Precision: Assess accuracy through spike-recovery experiments at low, medium, and high concentrations within the dynamic range. Determine precision by calculating intra-assay and inter-assay coefficients of variation (CV), with acceptable CV typically <15%.
Dynamic Range and Linearity: Establish the concentration interval over which the biosensor response maintains a linear relationship with analyte concentration. The upper limit of quantification (LoQ) represents the highest concentration measurable with acceptable accuracy and precision, while the lower LoQ is typically set at 3-10 times the LoD [82].
Robustness and Stability: Evaluate performance under varying environmental conditions (temperature, pH, ionic strength) that may be encountered during field deployment. Determine shelf-life by monitoring signal stability over time under appropriate storage conditions.
The Research Toolkit: Essential Reagents and Materials
Table 3: Research Reagent Solutions for Biosensor Development and Application
| Reagent/Material | Function | Examples/Specifications |
|---|---|---|
| Biological Recognition Elements | Target capture and specific binding | Antibodies, aptamers, molecularly imprinted polymers (MIPs), enzymes, whole cells |
| Transduction Materials | Signal generation and conversion | Electrodes (gold, carbon, screen-printed), fluorophores, quantum dots, electrochemical reporters (ferrocene, methylene blue) |
| Microfluidic Substrates | Device fabrication and fluid handling | PDMS, PMMA, paper, glass, silicon; selection based on optical properties, biocompatibility, and fabrication requirements |
| Signal Amplification Reagents | Enhanced sensitivity | Enzymes (HRP, ALP), nanomaterials (gold nanoparticles, graphene, carbon nanotubes), dendrimers |
| Sample Preparation Materials | Matrix cleanup and analyte enrichment | Solid-phase extraction cartridges (C18, HLB, ion-exchange), filters (0.22μm, 0.45μm), dilution buffers |
| Blocking and Stabilization Agents | Reduce non-specific binding and improve stability | BSA, casein, sucrose, trehalose, surfactants (Tween-20) |
Advanced Integration: Machine Learning and Future Directions
The integration of machine learning (ML) with biosensing technologies represents a promising frontier in tiered contaminant assessment [83]. ML algorithms can enhance biosensor performance through advanced signal processing, pattern recognition in complex environmental mixtures, and prediction of contaminant sources. The workflow for ML-assisted analysis involves four key stages: (1) sample treatment and extraction, (2) data generation and acquisition, (3) ML-oriented data processing and analysis, and (4) result validation [83].
ML techniques particularly excel at identifying latent patterns in high-dimensional data generated by biosensor arrays. For instance, supervised learning algorithms such as Random Forest, Support Vector Classifier, and Logistic Regression can classify contamination sources based on biosensor response patterns with balanced accuracy ranging from 85.5% to 99.5% in controlled studies [83]. Dimensionality reduction techniques like principal component analysis (PCA) and t-distributed stochastic neighbor embedding (t-SNE) simplify complex datasets for visualization and interpretation, while clustering methods (hierarchical cluster analysis, k-means) group samples by chemical similarity [83].
Future developments in biosensor technology will likely focus on multiplexed detection platforms capable of simultaneously monitoring multiple EC classes, connectivity for real-time data transmission, and improved robustness for long-term deployment in environmental monitoring networks. Additionally, the integration of biosensors with complementary techniques such as passive sampling and automated extraction systems will enable more comprehensive spatial and temporal assessment of contaminant distributions. As these technologies mature, biosensors will play an increasingly central role in tiered assessment frameworks, providing critical decision-support data for environmental management and public health protection.
The tiered assessment model, with biosensors as rapid screening tools complemented by confirmatory methods, represents a scientifically robust and resource-efficient strategy for monitoring emerging contaminants in environmental matrices. By leveraging the strengths of each analytical approach—the speed, portability, and cost-effectiveness of biosensors in initial tiers, combined with the precision, accuracy, and comprehensiveness of chromatographic and spectrometric techniques in higher tiers—this framework enables effective prioritization of limited analytical resources. As biosensing technologies continue to advance through integration with microfluidics, nanomaterials, and machine learning, their role in environmental assessment will expand, providing researchers and regulatory agencies with powerful tools to address the complex challenge of contaminant monitoring in an increasingly chemical-intensive world.
The monitoring of emerging contaminants (ECs)—such as pharmaceuticals, personal care products, and endocrine-disrupting compounds—is a critical challenge for global water safety and public health. These contaminants, often present at trace concentrations but possessing high toxicity, threaten ecosystems and human health, necessitating the development of advanced detection technologies [22] [31]. Traditional analytical methods, including liquid chromatography-mass spectrometry (LC-MS), are sensitive but suffer from significant limitations: they are laboratory-bound, require complex sample preparation, involve high operational costs, and are incapable of providing real-time, on-site data [22] [31]. These shortcomings hinder effective routine monitoring and rapid response to contamination events.
Biosensing technologies have emerged as a powerful alternative, offering the potential for sensitive, selective, and rapid analysis. Recent advancements have led to the development of enzyme-based biosensors, aptasensors, and electrochemical immunosensors that are increasingly portable and cost-effective [22] [31]. The integration of artificial intelligence (AI) is now poised to trigger a paradigm shift, transforming these biosensors from simple analytical tools into intelligent, adaptive systems capable of processing complex data, improving detection accuracy, and enabling real-time decision-making [85] [86]. This whitepaper examines the market adoption and regulatory landscape for these AI-powered biosensing systems, analyzing the key drivers of their growth, the experimental protocols underpinning their functionality, and the evolving regulatory frameworks that will govern their widespread deployment.
Market Adoption Landscape
The adoption of AI technologies is accelerating across industries, creating a fertile environment for AI-enhanced biosensors. A 2025 global survey reveals that 88% of organizations are now regularly using AI in at least one business function, a significant increase from 78% just a year prior [87]. This broad-based adoption provides the foundational infrastructure and acceptance necessary for specialized applications like intelligent environmental monitoring.
Quantitative Adoption Metrics
The table below summarizes key quantitative data on the adoption of AI and related technologies, illustrating the current state of the market.
Table 1: Key Metrics for AI and Biosensor Adoption
| Metric | Reported Value/Status | Context and Implications |
|---|---|---|
| Organizational AI Use | 88% of organizations [87] | Indicates widespread familiarity and infrastructure for AI integration. |
| AI Scaling Phase | ~33% of organizations [87] | Most organizations are still experimenting or piloting, showing significant growth potential. |
| AI Agent Experimentation | 62% of organizations [87] | Highlights growing interest in autonomous AI systems, relevant for independent field sensors. |
| Private AI Investment (U.S., 2024) | $109.1 billion [88] | Demonstrates substantial financial backing for AI development and application. |
| FDA-Approved AI Medical Devices | 223 in 2023 (vs. 6 in 2015) [88] | Serves as a proxy for regulatory acceptance of AI in sensitive, life-science applications. |
Key Growth Drivers
Several interconnected factors are propelling the development and adoption of AI-powered biosensing systems for environmental monitoring:
Demand for Real-Time, On-Site Monitoring: The limitations of conventional laboratory analysis have created a pressing need for technologies that can provide immediate results at the point of need. Point-of-care testing (POCT) devices, including paper-based and microfluidic systems, are gaining substantial attention for their ability to conduct rapid, on-site analysis of ECs, enabling prompt decision-making [22] [31]. AI integration enhances these platforms by enabling the real-time processing of sensor data directly in the field.
Enhanced Performance through AI: AI algorithms, particularly machine learning (ML) and deep learning (DL), dramatically improve biosensor performance. They enhance sensitivity and specificity by filtering out undesirable noise and signals, leading to more accurate and reliable measurements [85]. Furthermore, AI enables predictive modeling and adaptive sensing capabilities, allowing systems to identify complex patterns and weak trends in data that would be imperceptible to human analysts or traditional algorithms [85] [89].
Technological Convergence: The power of AI-powered biosensors stems from the synergy of multiple advanced fields. The development of novel nanomaterials (e.g., graphene, metal nanoparticles, metal-organic frameworks) enhances sensor sensitivity, functionality, and miniaturization [85] [86]. Concurrently, the rise of the Internet of Things (IoT) allows individual sensors to be networked, creating distributed monitoring systems that provide a comprehensive picture of environmental contamination [85].
Technical Framework: AI-Enhanced Biosensing
Fundamental Biosensing Modalities
AI-powered systems build upon established biosensing frameworks. The following table outlines the primary biosensor types used for detecting emerging contaminants.
Table 2: Core Biosensing Techniques for Emerging Contaminants
| Biosensor Type | Biorecognition Element | Transduction Mechanism | Example Application |
|---|---|---|---|
| Enzyme-Based | Enzymes (e.g., hydrolytic, oxidative) [22] | Electrochemical, Colorimetric [22] [31] | Detection of pesticides via enzyme inhibition [31]. |
| Aptasensor | Synthetic DNA/RNA aptamers [22] [31] | Optical, Electrochemical [22] | Real-time monitoring of contaminants like antibiotics [22]. |
| Electrochemical Immunosensor | Antibodies [22] [31] | Impedimetric, Voltammetric [22] | Sensitive detection of antibiotics like Ciprofloxacin [31]. |
| Whole Cell-Based | Microbial cells (e.g., bacteria) [31] | Optical, Electrochemical [31] | Monitoring pyrethroid insecticides using E. coli [31]. |
The AI Integration Workflow
The integration of AI transforms the traditional biosensing workflow into an intelligent, closed-loop system. The diagram below illustrates this enhanced process, from sensor design to final data interpretation.
Diagram 1: AI-powered biosensing workflow.
Experimental Protocol: Aptasensor Development with AI
The following is a detailed methodology for developing an AI-enhanced aptasensor for the detection of a specific environmental contaminant, such as an antibiotic or hormone.
Objective: To develop a highly sensitive and specific electrochemical aptasensor for the detection of a target EC (e.g., 17β-estradiol) in water samples, with AI-driven signal calibration and data analysis.
Materials and Reagents: Table 3: Research Reagent Solutions for Aptasensor Development
| Reagent/Material | Function | Specific Example |
|---|---|---|
| DNA Aptamer | Biorecognition element; binds target with high affinity and specificity. | Synthetic ssDNA sequence selected via SELEX for 17β-estradiol [22] [31]. |
| Gold Nanoparticles (AuNPs) | Nanomaterial for electrode modification; increases surface area and enhances electron transfer. | Colloidal AuNP solution, ~20nm diameter [22] [86]. |
| Electrochemical Probe | Generates measurable electrical signal upon binding event. | Methylene Blue or Ferricyanide solution [90]. |
| Buffer Solutions | Maintain optimal pH and ionic strength for biorecognition. | Phosphate Buffered Saline (PBS), Tris-EDTA (TE) buffer [31]. |
| Target Analytic Standard | Used for sensor calibration and validation. | Pure 17β-estradiol powder dissolved in appropriate solvent [31]. |
Step-by-Step Procedure:
Aptamer Selection and Preparation:
- Utilize a pre-identified aptamer sequence specific to the target EC. AI can be employed in this initial stage to analyze SELEX (Systematic Evolution of Ligands by EXponential enrichment) data and predict high-affinity aptamer sequences, accelerating discovery [86].
- Prepare the aptamer solution in a suitable buffer (e.g., TE buffer) and denature it at 95°C for 5 minutes, then allow it to cool slowly to room temperature to ensure proper folding.
Electrode Modification and Biosensor Fabrication:
- Clean the working electrode (e.g., glassy carbon or gold electrode) thoroughly with alumina slurry and sonicate in ethanol and deionized water.
- Electrodeposit or drop-cast a suspension of gold nanoparticles (AuNPs) onto the electrode surface and dry. This creates a high-surface-area platform for aptamer immobilization [86].
- Immobilize the thiolated aptamer onto the AuNP-modified electrode via self-assembled monolayer formation, incubating for several hours. Passivate any remaining bare gold sites with a mercaptohexanol solution to minimize non-specific binding.
Electrochemical Measurement and Data Acquisition:
- Use an electrochemical workstation to perform techniques such as Differential Pulse Voltammetry (DPV) or Electrochemical Impedance Spectroscopy (EIS) in a solution containing the electrochemical probe [90].
- Record the baseline signal from the aptamer-functionalized electrode.
- Incubate the electrode with samples containing varying concentrations of the target EC.
- After incubation and washing, measure the electrochemical signal again. The binding of the target to the aptamer causes a measurable change in current (DPV) or charge transfer resistance (EIS).
AI-Powered Data Processing and Analysis:
- Signal Pre-processing: Input the raw electrochemical data (e.g., full voltammogram curves) into an ML pipeline. Apply filtering algorithms (e.g., Savitzky-Golay) to smooth the data and attenuate high-frequency noise [85].
- Model Training and Prediction: Train a machine learning model, such as a Support Vector Machine (SVM) or Random Forest (RF), on a dataset of signals corresponding to known concentrations of the target EC [85]. The model learns the complex relationship between the signal features and the analyte concentration.
- Concentration Prediction: Use the trained model to predict the concentration of the target EC in unknown environmental samples based on their electrochemical signatures. This AI-driven calibration can correct for sensor drift and matrix effects, improving accuracy over traditional calibration curves [89].
Regulatory Outlook and Challenges
As AI-powered biosensing systems advance, they encounter a dynamic and complex regulatory landscape. The transition from laboratory research to commercially deployed products requires navigating challenges related to performance validation, data reliability, and ethical use.
Evolving Regulatory Frameworks
Global regulatory bodies are intensifying their focus on AI. In 2024, U.S. federal agencies introduced 59 AI-related regulations—more than double the number in 2023—indicating a rapid move towards formal governance [88]. Legislative mentions of AI have seen a ninefold increase since 2016 across 75 countries [88]. For AI-powered biosensors, regulations will likely center on:
- Performance Validation: Demonstrating consistent sensitivity, specificity, and accuracy across diverse and complex environmental samples [31] [90].
- Data Integrity and Algorithm Transparency: Ensuring the reliability of training data and providing some level of explainability for the AI's decisions, a challenge known as the "black box" problem [85] [86].
- Standardization: Developing universal benchmarks and testing protocols, such as the emerging benchmarks HELM Safety and AIR-Bench for AI safety and factuality [88].
Critical Adoption Barriers
Despite the promising outlook, several significant barriers must be overcome for widespread adoption, as identified in industry analyses:
Table 4: Key Challenges in Adopting Advanced AI Systems
| Challenge Category | Specific Barriers | Relevance to AI-Powered Biosensing |
|---|---|---|
| Technical & Operational | Integration with legacy systems; Data reliability in complex matrices; Sensor fouling and operational stability [91]. | Environmental samples contain interferents that can degrade sensor performance and confuse AI models [22] [90]. |
| Workforce & Expertise | Lack of in-house technical expertise in both biosensing and AI [91]. | Creates a talent gap, slowing development and deployment and increasing vendor dependence [87] [91]. |
| Risk & Compliance | Unclear regulatory pathways; Data privacy and security; Liability for autonomous decisions [91]. | Stifles investment and complicates the path to market for new biosensing products [91]. |
The integration of artificial intelligence with advanced biosensing platforms represents a transformative leap in our ability to monitor and manage emerging environmental contaminants. The market for these systems is poised for significant growth, driven by the convergence of nanotechnology, IoT connectivity, and intelligent algorithms. While the path to widespread commercialization is fraught with challenges—including technical robustness, regulatory ambiguity, and a shortage of skilled personnel—the ongoing efforts in standards development and the clear surge in AI investment and adoption are positive indicators. For researchers and drug development professionals, engaging with this interdisciplinary field—by contributing to robust validation studies, exploring explainable AI models, and participating in the development of open standards—will be crucial to unlocking the full potential of AI-powered biosensing for ensuring environmental and public health safety.
Conclusion
Biosensors represent a transformative technological shift for monitoring emerging contaminants, offering a powerful combination of sensitivity, portability, and real-time analysis that traditional lab-based methods cannot match. The synthesis of insights from this review confirms that while challenges in long-term stability and reproducibility persist, ongoing innovations in material science, nanotechnology, and microfluidic integration are rapidly overcoming these barriers. For biomedical and clinical research, the implications are profound. The ability to conduct rapid, on-site screening of contaminants in water sources is crucial for drug safety, understanding environmental triggers of disease, and managing antibiotic resistance. Future efforts should focus on developing multifunctional and multiplexed biosensor platforms, establishing standardized validation protocols, and further miniaturizing systems for seamless integration into healthcare and environmental surveillance networks, ultimately contributing to safer pharmaceuticals and improved public health outcomes.
References
- 1. A comprehensive review of emerging environmental ... [https://link.springer.com/article/10.1007/s44274-025-00259-x]
- 2. Understanding emerging contaminants in water and ... [https://www.sciencedirect.com/science/article/pii/S2772416625001664]
- 3. Contaminants of emerging concern (CECs), including ... [https://www.epa.gov/wqc/contaminants-emerging-concern-including-pharmaceuticals-and-personal-care-products]
- 4. Stormwater Pollutants of Emerging Concern [https://www.epa.gov/npdes/stormwater-pollutants-emerging-concern]
- 5. Top 10 Emerging Contaminants in 2025 | Fluoride Filter ... [https://clearlyfiltered.com/articles/top-10-emerging-contaminants-in-2025]
- 6. Practical Guidance on Selecting Analytical Methods for ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC12371593/]
- 7. Electrochemical Biosensors: A Prospective Insight to ... [https://www.sciencedirect.com/science/article/abs/pii/S2451910325001358]
- 8. ECS Sensors Plus Spotlight: Vanadium MXene-Modified ... [https://www.electrochem.org/ecsnews/ecssp-spotlight-sensitive-glucose-sensing]
- 9. Comprehensive review of emerging contaminants [https://www.sciencedirect.com/science/article/pii/S0147651324004962]
- 10. Emerging contaminants: A One Health perspective - PMC [https://pmc.ncbi.nlm.nih.gov/articles/PMC11096751/]
- 11. A systematic review of sources, occurrence, behavior and ... [https://www.nature.com/articles/s44454-025-00015-z]
- 12. Antimicrobial Resistance: A Growing Serious Threat for ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC10340576/]
- 13. What is antimicrobial resistance and how can we tackle it? [https://www.weforum.org/stories/2025/11/antimicrobial-resistance-superbugs-antibiotics/]
- 14. The Role of the Environment (Water, Air, Soil) in the ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC12382627/]
- 15. Recent Advances in Biosensor Technology for Potential ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC4754454/]
- 16. Future advances of artificial biosensor technology in ... [https://www.sciencedirect.com/science/article/pii/S2666831924000158]
- 17. LC-MS/MS in the Clinical Laboratory – Where to From Here? [https://pmc.ncbi.nlm.nih.gov/articles/PMC3052391/]
- 18. The Essence of Modern HPLC: Advantages, Limitations ... [https://www.chromatographyonline.com/view/essence-modern-hplc-advantages-limitations-fundamentals-and-opportunities]
- 19. Perspectives of Quantitative GC-MS, LC-MS, and ICP-MS in ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC11641993/]
- 20. Reductions in Cost and Time by Modernizing a USP ... [https://www.waters.com/nextgen/us/en/library/application-notes/2021/reductions-in-cost-and-time-by-modernizing-a-usp-monograph-method-from-hplc-to-uhplc-and-uplc-instrumentation-and-columns.html?srsltid=AfmBOorzlnWmIXv3xKvo5ydI-VS2jduN6YE52GufL7hctqF7qlyIUK-f]
- 21. Comparison of LC–MS and GC–MS for the Analysis ... [https://www.chromatographyonline.com/view/comparison-lc-ms-and-gc-ms-analysis-pharmaceuticals-and-personal-care-products-surface-water-and-t-0]
- 22. Biosensing contaminants of emerging concerns [https://www.sciencedirect.com/science/article/abs/pii/S2468584425000583]
- 23. Biosensor Technologies for Water Quality: Detection of ... [https://www.mdpi.com/2079-6374/15/3/189]
- 24. The Rising Role of Biosensors in Modern Technology [https://www.delveinsight.com/blog/rising-role-of-biosensors]
- 25. Biosensors Market: Comprehensive Analysis and Forecast ... [https://www.pharmiweb.com/press-release/2025-02-13/biosensors-market-comprehensive-analysis-and-forecast-2025-2035]
- 26. A Review on the Application of Biosensors for Monitoring ... [https://www.mdpi.com/1424-8220/25/16/4945]
- 27. Biosensors Market 2025 To 2030 : Future, Product, ... [https://www.linkedin.com/pulse/biosensors-market-2025-2030-future-product-wearables-p-virkhe-xtmqf]
- 28. An All-in-One Sustainable Smartphone Paper Biosensor for ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC12392259/]
- 29. IoT-enabled biosensors for real-time monitoring and early ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC11811615/]
- 30. Biosensors, Volume 15, Issue 11 (November 2025) [https://www.mdpi.com/2079-6374/15/11]
- 31. A Review on the Application of Biosensors for Monitoring ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC12390133/]
- 32. Biosensors for detection of emerging contaminants [https://www.sciencedirect.com/science/article/pii/B9780443342707000158]
- 33. Bioreceptors as the key components for electrochemical ... [https://www.sciencedirect.com/science/article/pii/S2666386424000213]
- 34. Emerging applications of biorecognition elements-based ... [https://link.springer.com/article/10.1007/s44397-025-00003-3]
- 35. Aptasensors versus immunosensors—Which will prevail? [https://pmc.ncbi.nlm.nih.gov/articles/PMC8961048/]
- 36. Aptamer-Based Biosensors for the Colorimetric Detection of ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC9313021/]
- 37. Electrochemical biosensors combining aptamers and ... [https://www.sciencedirect.com/science/article/abs/pii/S0013468621011531]
- 38. Advances in Smart Biosensors for Real-Time Pollutant ... [https://www.sciencedirect.com/science/article/abs/pii/S0165993625003516]
- 39. a promising analytical tool for on-site detection of mycotoxins [https://www.nature.com/articles/s41538-025-00444-5]
- 40. Emerging Trends in Paper-Based Electrochemical ... [https://www.mdpi.com/2673-4591/106/1/8]
- 41. Microfluidics-Based Nanobiosensors for Healthcare ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC10173227/]
- 42. Paper-based flexible electronic devices: processing, ... [https://www.nature.com/articles/s41528-025-00446-z]
- 43. Microfluidic Paper-Based Devices [https://www.mdpi.com/2072-666X/16/3/307]
- 44. Advances in paper-based analytical devices relying on ... [https://link.springer.com/article/10.1007/s42114-025-01333-x]
- 45. Nanomaterials-driven paper-based biosensors for food ... [https://www.sciencedirect.com/science/article/abs/pii/S0010854525000827]
- 46. Plasmonic sensing in microfluidic paper-based analytical ... [https://pubs.rsc.org/en/content/articlehtml/2025/ra/d5ra01972a?page=search]
- 47. Microfluidic paper–based analytical extraction devices ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC12228674/]
- 48. Research Progress of Biosensors in the Detection ... [https://www.mdpi.com/2079-6374/15/12/778]
- 49. Biomaterials and biosensing technologies in the detection ... [https://www.sciencedirect.com/science/article/abs/pii/S0010854525006800]
- 50. Application of nanozyme-based biosensors in pesticide ... [https://pubs.rsc.org/en/content/articlelanding/2025/ma/d5ma00800j]
- 51. Fluorescent biosensors for the detection of foodborne ... [https://pubs.rsc.org/en/content/articlelanding/2025/ay/d5ay01025j]
- 52. Development and quantitative analysis of a biosensor ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC8794804/]
- 53. and their Biosensors – A review - PMC applications [https://pmc.ncbi.nlm.nih.gov/articles/PMC4862100/]
- 54. of Applications : Future... | PharmaTutor Biosensors technology [https://www.pharmatutor.org/articles/applications-of-biosensors-technology-future-trends-development-and-new-intervation-in-biotechnology]
- 55. Journal of Materials Chemistry B Editor's choice web collection... [https://pubs.rsc.org/en/content/articlehtml/2018/tb/c8tb90055h]
- 56. Week-long operation of electrochemical aptamer sensors [https://pmc.ncbi.nlm.nih.gov/articles/PMC10443649/]
- 57. Recent advances in surface modification and antifouling ... [https://www.sciencedirect.com/science/article/pii/S2451910323001126]
- 58. The effect of temperature on fouling in anaerobic ... [https://www.sciencedirect.com/science/article/pii/S0043135424007681]
- 59. pH-Activated Dissolvable Polymeric Coatings to Reduce ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC10298981/]
- 60. Determining the performance of a temperature sensor ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC9343656/]
- 61. Anti-Fouling Strategies of Electrochemical Sensors for ... [https://www.mdpi.com/1424-8220/23/11/5202]
- 62. A primer to directed evolution: current methodologies and ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC10074555/]
- 63. Extending dynamic and operational range of the biosensor ... [https://www.sciencedirect.com/science/article/pii/S2405805X25000596]
- 64. Whole-cell biosensors with regulatory circuits based on a ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC12509908/]
- 65. Next-generation nanomaterials-based biosensors: Real- ... [https://www.sciencedirect.com/science/article/pii/S2589234724004044]
- 66. Biosensors, Volume 15, Issue 5 (May 2025) – 69 articles [https://www.mdpi.com/2079-6374/15/5]
- 67. Portable on-chip colorimetric biosensing platform ... [https://www.nature.com/articles/s41598-021-02580-w]
- 68. Development of a Portable Dropper-based Biosensor for ... [https://www.sciencedirect.com/science/article/abs/pii/S0003267025011298]
- 69. Development of a Portable Cell-Based Biosensor for the ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC11201857/]
- 70. Integration of Artificial Intelligence in Biosensors for Enhanced ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC12564411/]
- 71. Recent trends and technical advancements in biosensors ... [https://www.sciencedirect.com/science/article/abs/pii/S2212429222001547]
- 72. A comparison of conventional and advanced electroanalytical ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC9270057/]
- 73. Biosensors, Artificial Intelligence Biosensors, False Results ... [https://www.mdpi.com/2075-4418/15/8/1037]
- 74. Critical overview on the application of sensors and biosensors ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC7112812/]
- 75. Market Size and Economics for Biosensors - PMC [https://pmc.ncbi.nlm.nih.gov/articles/PMC7152385/]
- 76. Development and validation of a green/blue UHPLC-MS ... [https://www.nature.com/articles/s41598-025-15614-4]
- 77. A statistical approach to quantitative data validation focused ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC3795879/]
- 78. Assessment of surface water quality using advanced ... [https://www.sciencedirect.com/science/article/pii/S2590252025000510]
- 79. Current Status of Emerging Contaminant Models and Their ... [https://www.mdpi.com/2073-4441/17/1/85]
- 80. A Review on Analytical Techniques for Quantitative ... [https://www.mdpi.com/2227-9040/12/12/274]
- 81. Exposure Assessment Tools by Tiers and Types [https://www.epa.gov/expobox/exposure-assessment-tools-tiers-and-types-screening-level-and-refined]
- 82. On the Determination of Uncertainty and Limit of Detection in ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC6068557/]
- 83. Integrating non-target analysis and machine learning [https://www.nature.com/articles/s41545-025-00504-z]
- 84. When lower isn't always better in biosensor research and ... [https://www.sciencedirect.com/science/article/abs/pii/S0956566324006766]
- 85. The Role of Artificial Intelligence in Advancing Biosensor ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC12392873/]
- 86. Artificial Intelligence-Assisted Nanosensors for Clinical ... [https://www.mdpi.com/2079-6374/15/10/656]
- 87. The State of AI: Global Survey 2025 [https://www.mckinsey.com/capabilities/quantumblack/our-insights/the-state-of-ai]
- 88. The 2025 AI Index Report | Stanford HAI [https://hai.stanford.edu/ai-index/2025-ai-index-report]
- 89. Unlocking next-generation biosensing with AI [https://www.sciencedirect.com/science/article/abs/pii/S0165993625000767]
- 90. Recent progress of biosensing techniques for ... [https://www.sciencedirect.com/science/article/abs/pii/S0165993625002481]
- 91. AI trends 2025: Adoption barriers and updated predictions [https://www.deloitte.com/us/en/services/consulting/blogs/ai-adoption-challenges-ai-trends.html]