Strategies to Reduce Drift in RuO₂ Urea Biosensors: From Material Science to Circuit Design
Drift is a critical non-ideal effect that compromises the long-term stability and reliability of RuO₂ urea biosensors, hindering their clinical adoption.
Strategies to Reduce Drift in RuO₂ Urea Biosensors: From Material Science to Circuit Design
Abstract
Drift is a critical non-ideal effect that compromises the long-term stability and reliability of RuO₂ urea biosensors, hindering their clinical adoption. This article provides a comprehensive analysis for researchers and scientists on tackling this challenge. We first explore the fundamental mechanisms of drift, primarily the formation of a hydration layer on the RuO₂ sensing film. We then detail a novel calibration circuit (NCC) based on voltage regulation, demonstrated to reduce the drift rate by 98.77%. The article further covers troubleshooting sensor fabrication and circuit integration and offers a comparative validation of the NCC against conventional measurement systems, highlighting its superior performance in achieving a minimal drift of 0.02 mV/hr.
Understanding Drift in RuO₂ Urea Biosensors: Causes and Impact on Clinical Measurements
FAQ: RuO₂ for Urea Biosensing
Q: What makes RuO₂ a suitable sensing material for urea detection? A: Ruthenium oxide (RuO₂) is a highly stable metal oxide with excellent electrochemical properties, making it ideal for biosensing applications. For urea detection, its primary advantage lies in its role as an effective transducer material. When used in electrochemical biosensors, RuO₂ can facilitate electron transfer in reactions involving the urease enzyme. Its high electrical conductivity and good catalytic activity help in achieving a sensitive and stable signal response to changes in urea concentration [1].
Q: What are the main factors that cause signal drift in RuO₂-based urea biosensors, and how can this be mitigated? A: Signal drift is a critical challenge that affects the long-term reliability of biosensors. For RuO₂-based urea biosensors, the primary sources of drift include:
- Material Dissolution: Over time and under operational conditions, the RuO₂ sensing layer itself can undergo gradual dissolution, especially in acidic environments, leading to a loss of active material and signal decay [2] [3].
- Enzyme Layer Instability: The urease enzyme immobilized on the sensor surface can degrade or leach away, changing the sensor's response characteristics.
- Interfacial Degradation: Mechanical or chemical changes at the interface between the RuO₂ layer and the substrate or the enzyme layer can increase electrical resistance or alter signal transduction.
Mitigation strategies focus on enhancing the structural stability of the RuO₂ and protecting the enzyme layer. Recent research shows that strain engineering and doping with stable elements are highly effective. For instance, introducing single atoms of larger elements (like Platinum or Gallium) into the RuO₂ lattice can create a stabilizing heterogenous strain, which strengthens the metal-oxygen bonds and dramatically reduces the rate of ruthenium dissolution [2] [4].
Q: What is a typical experimental protocol for fabricating a RuO₂-based urea biosensor? A standard protocol involves material synthesis, electrode fabrication, enzyme immobilization, and testing, with a focus on stability.
- Synthesis of Modified RuO₂: Prepare the sensing material. For example, synthesize a doped RuO₂ (e.g., Pt-RuO₂ or Ga-RuO₂) using a sol-gel or hydrothermal method to incorporate the stabilizing dopant [2] [4].
- Electrode Modification: Deposit the synthesized RuO₂-based material onto a conductive substrate (e.g., a screen-printed carbon electrode or gold electrode) using drop-casting or electrodeposition, followed by drying and annealing to ensure a stable film.
- Urease Immobilization: Immobilize the urease enzyme onto the RuO₂-modified electrode. This can be done via physical adsorption, cross-linking with glutaraldehyde, or entrapment within a polymer matrix like Nafion.
- Electrochemical Characterization: Perform Cyclic Voltammetry (CV) and Electrochemical Impedance Spectroscopy (EIS) in a buffer solution to characterize the electrode's baseline performance and electron transfer efficiency.
- Calibration and Stability Testing:
- Record the amperometric or potentiometric response of the biosensor upon successive additions of urea standard solutions to build a calibration curve (signal vs. urea concentration).
- To assess drift, continuously operate the sensor at a fixed urea concentration (e.g., in a flow cell) or take daily measurements in a standard solution over an extended period (e.g., 500+ hours). The decay rate of the signal (in µV/h or %/day) quantifies the drift [2].
The Scientist's Toolkit: Key Reagents & Materials
The following table details essential materials for developing a stable RuO₂-based urea biosensor.
| Item | Function in the Experiment |
|---|---|
| Ruthenium-based precursor (e.g., RuCl₃) | The source of ruthenium for synthesizing the RuO₂ sensing matrix [4]. |
| Dopant precursor (e.g., H₂PtCl₆, Ga(NO₃)₃) | Used to introduce stabilizing elements (Pt, Ga) into the RuO₂ lattice, enhancing its structural integrity and reducing dissolution [2] [4]. |
| Urease Enzyme (from Jack Bean) | The biological recognition element that specifically catalyzes the hydrolysis of urea, producing NH₄⁺ and HCO₃⁻ ions [1]. |
| Nafion polymer solution | A common perfluorosulfonate polymer used to form a protective membrane over the enzyme layer, preventing leaching and improving selectivity [1]. |
| Glutaraldehyde | A cross-linking agent used to create covalent bonds between enzyme molecules and the sensor surface, stabilizing the biorecognition layer. |
| Phosphate Buffer Saline (PBS) | The standard electrolyte solution for maintaining a stable pH during electrochemical measurements and biosensor operation. |
Quantitative Performance Data of RuO₂-Based Sensors
The stability of a sensing material is often benchmarked against its performance in harsh, analogous applications like the acidic Oxygen Evolution Reaction (OER), which also causes severe Ru dissolution.
Table 1: Performance Comparison of Pure and Stabilized RuO₂ in Acidic Electrolysis*
| Catalyst Type | Key Stabilization Strategy | Test Conditions | Operational Stability | Decay Rate |
|---|---|---|---|---|
| Pure RuO₂ | None (baseline) | Acidic OER, 10 mA/cm² | Rapid failure | ~756 µV/h (extrapolated) [2] |
| Pt-RuO₂ | Heterogeneous Strain (Pt doping) | Acidic OER, 10 mA/cm² | 1500 hours | 27 µV/h [2] |
| Ga-RuO₂ | O radical spatiotemporal coordination (Ga doping) | Acidic OER, 100 mA/cm² | 800 hours | Performance maintained [4] |
*Note: Data adapted from PEM water electrolysis studies. While conditions differ from biosensing, the relative improvement in RuO₂ stability conferred by doping is highly relevant [2] [4].
Table 2: Key Parameters for Monitoring RuO₂ Urea Biosensor Drift
| Parameter | Target | Measurement Method | Significance for Drift |
|---|---|---|---|
| Sensitivity Drift | < 5% change per month | Calibration curve slope over time | Indicates loss of active enzyme or catalyst sites. |
| Baseline Signal Drift | Minimal fluctuation | Signal stability in blank solution | Suggests underlying sensor material instability or reference electrode potential drift. |
| Response Time Change | < 10% increase from initial | Time to reach 95% of steady-state signal | Can indicate fouling or degradation of the sensor interface. |
Experimental Workflow for Drift Reduction
The following diagram illustrates a strategic workflow for developing a low-drift RuO₂ urea biosensor, integrating material stabilization and interface engineering.
Workflow for Developing a Low-Drift Biosensor
Troubleshooting Guide: Common Issues and Solutions
Problem: High Background Noise and Unstable Baseline
- Potential Cause 1: Poor electrical contact or a cracked RuO₂ film. This can be checked with microscopic inspection and EIS.
- Solution: Optimize the film deposition and annealing process to ensure a continuous, adherent sensing layer [1] [5].
- Potential Cause 2: Contamination of the electrolyte or reference electrode.
- Solution: Use fresh, high-purity buffer solutions and ensure the reference electrode is functioning correctly.
Problem: Rapid Loss of Sensitivity (Signal Fading)
- Potential Cause 1: Dissolution of the RuO₂ transducer layer.
- Solution: Implement a doping strategy, as described in Table 1, to stabilize the RuO₂ crystal structure against corrosion [2] [4] [3].
- Potential Cause 2: Leaching or deactivation of the urease enzyme.
- Solution: Optimize the enzyme immobilization procedure. Use a more robust cross-linking protocol or a superior encapsulating polymer like Nafion.
Problem: Hysteresis and Non-Reproducible Response
- Potential Cause: Slow reaction kinetics or pore clogging within the enzyme-polymer membrane.
- Solution: Ensure the RuO₂ layer has a high surface area to facilitate rapid electron transfer. Characterize the film's morphology using SEM. Also, verify that the polymer membrane is not too thick, which can slow down analyte diffusion [1].
Understanding Sensor Drift: A Fundamental FAQ
What is the "drift phenomenon" in RuO₂ urea biosensors?
The drift phenomenon refers to the unwanted change in a biosensor's response voltage over time during long-term measurement, leading to decreasing accuracy and reliability. This effect is particularly problematic for RuO₂ urea biosensors used in continuous monitoring applications, where stable readings are essential for clinical decision-making [6] [7].
What causes drift in electrochemical biosensors?
Drift occurs due to the formation of a hydration layer on the surface of the sensing film. When the RuO₂ sensing film is immersed in solution, hydroxyl groups form on its surface. Through coulombic attraction, water molecules and ions form hydrated ions that diffuse toward the sensing film, ultimately creating a hydration layer. This layer affects the electrical double layer capacitance, thereby changing the surface potential of the film and causing the output signal to drift over time [6] [7].
Why is drift particularly problematic for medical applications?
For urea biosensors used in kidney function monitoring, drift compromises measurement accuracy during extended procedures. Since urea concentration in the human body normally ranges between 2.5-7.5 mM, even small drift rates can lead to clinically significant errors in assessing renal function and making treatment decisions [6] [7].
Troubleshooting Guides: Identifying and Quantifying Drift
How to diagnose drift issues in your biosensor system
| Observation | Possible Causes | Verification Method |
|---|---|---|
| Consistent upward/downward voltage trend in buffer solution | Hydration layer formation on sensing film | Measure output in stable reference solution over 2+ hours |
| Gradual sensitivity loss over multiple measurements | Enzyme instability or hydration layer effect | Perform regular calibration checks with standard solutions |
| Erratic output with general upward/downward trend | Combined drift and interference/noise issues | Use statistical analysis to separate trend from noise |
Quantitative assessment of drift severity
| Drift Rate | Severity Level | Impact on 12-hour Measurement |
|---|---|---|
| > 0.5 mV/hour | Critical | Error > 6 mV - unacceptable for clinical use |
| 0.1 - 0.5 mV/hour | Moderate | Error 1.2-6 mV - requires frequent calibration |
| < 0.1 mV/hour | Low | Error < 1.2 mV - potentially acceptable for monitoring |
| 0.02 mV/hour (NCC) | Minimal | Error ~0.24 mV - 98.77% reduction [6] [8] |
Advanced Drift Reduction Techniques
Can circuit design really compensate for inherent sensor drift?
Yes, specialized calibration circuits can significantly reduce observed drift. Research demonstrates that a New Calibration Circuit (NCC) combining a non-inverting amplifier with a voltage calibrating circuit reduced the drift rate of RuO₂ urea biosensors to 0.02 mV/hour - representing a 98.77% reduction compared to uncompensated systems [6] [8].
Implementation protocol for drift compensation circuits
Experimental Setup for Drift Assessment:
- Prepare RuO₂ urea biosensor with urease immobilization
- Immerse sensing film in urea solution (concentration within physiological range: 2.5-7.5 mM)
- Connect sensor to NCC circuit or conventional readout system
- Measure response voltage continuously for 12 hours using data acquisition system
- Calculate drift rate as slope of voltage-time relationship
Calibration Circuit Configuration:
- Non-inverting amplifier stage for signal conditioning
- Voltage calibration circuit for drift compensation
- Data acquisition using instrumentation amplifier (e.g., LT1167) and DAQ device (e.g., NI USB-6210)
- Software platform (e.g., LabVIEW) for continuous monitoring [6] [7]
Experimental Protocols for Drift Characterization
Standardized methodology for drift rate quantification
Materials and Equipment Checklist:
- Fabricated RuO₂ urea biosensor on PET substrate
- Phosphate buffer saline (PBS, pH 7.0) for baseline measurements
- Urea solutions across clinical range (2.5-7.5 mM)
- Voltage-Time (V-T) measurement system
- Data acquisition hardware (e.g., National Instruments USB-6210)
- Signal conditioning circuitry (NCC or conventional)
Step-by-Step Procedure:
- Stabilize biosensor in PBS solution for 30 minutes
- Record baseline output for 1 hour to establish initial drift rate
- Transfer to urea solutions of known concentrations
- Record output voltage at minimum 10-second intervals
- Maintain constant temperature throughout experiment
- Continue measurements for 12-hour period
- Analyze data using linear regression on voltage-time plots
- Calculate drift rate as slope of the trend line (mV/hour)
Visualization: Drift Mechanism and Compensation
Drift Mechanism and Compensation Pathway
Research Reagent Solutions for Drift Reduction
Essential materials for RuO₂ urea biosensor fabrication and testing
| Material/Component | Function/Role | Specification/Notes |
|---|---|---|
| Ruthenium (Ru) target | Sensing film formation | 99.95% purity, sputtered to form RuO₂ film [6] |
| Polyethylene terephthalate (PET) | Flexible substrate | Provides mechanical support for biosensor array [6] |
| Silver paste | Electrode formation | Screen-printed to create working/reference electrodes [6] |
| Epoxy thermosetting polymer | Insulation layer | Encapsulates and protects sensor components (e.g., JA643) [6] |
| Urease enzyme | Biocatalyst | Hydrolyzes urea to NH₄⁺ and HCO₃⁻ ions [6] |
| Phosphate buffer saline (PBS) | Testing medium | 30 mM, pH 7.0 (physiological simulation) [6] |
| APTS & glutaraldehyde | Enzyme immobilization | Enhances urease adsorption and stability on RuO₂ surface [6] |
Performance Comparison: Drift Reduction Solutions
Efficacy of different drift mitigation approaches
| Method | Drift Rate Achieved | Relative Reduction | Implementation Complexity |
|---|---|---|---|
| Conventional V-T System | ~1.6 mV/hour (baseline) | Reference | Low [6] |
| New Calibration Circuit (NCC) | 0.02 mV/hour | 98.77% | Medium [6] [8] |
| Material Modification (Ag NPs) | Not specifically quantified | Focused on hysteresis reduction | High [9] |
| Alternative Sensing Films (NiO/TiO₂) | Rarely discussed in literature | Unknown | Variable [6] |
Sensor performance characteristics with drift compensation
| Parameter | Without NCC | With NCC Implementation |
|---|---|---|
| Average Sensitivity | ~1.860 mV/(mg/dL) | Maintained or improved [6] |
| Linearity | 0.999 | Maintained [6] |
| Long-term Stability (12-hour) | Significant drift | Minimal drift (0.24 mV total change) [6] |
| Clinical Utility | Limited for extended monitoring | Enhanced for continuous measurement [6] [8] |
Frequently Asked Technical Questions
Are there material alternatives to RuO₂ that exhibit less inherent drift?
While NiO and TiO₂ are widely used in urea biosensors for their chemical stability and electron transfer capabilities, their drift characteristics are rarely discussed in literature. RuO₂ offers advantageous properties including low resistivity, high thermal stability, and good diffusion barrier properties, making it suitable for electrode applications when paired with appropriate drift compensation techniques [6] [7].
How does the New Calibration Circuit achieve such significant drift reduction?
The NCC employs a voltage regulation technique through its combination of a non-inverting amplifier and dedicated voltage calibrating circuit. This approach actively compensates for the slowly changing voltage signals caused by the hydration layer effect, effectively neutralizing the drift phenomenon at the circuit level rather than attempting to prevent it at the material level [6] [8].
What measurement duration is typically required to properly characterize drift?
Research protocols typically employ 12-hour continuous measurements to properly quantify drift rates. This extended timeframe allows sufficient duration to observe the gradual voltage changes caused by hydration layer formation while maintaining relevance to clinical monitoring applications where sensors may be used for extended periods [6] [8].
Drift Characterization Experimental Workflow
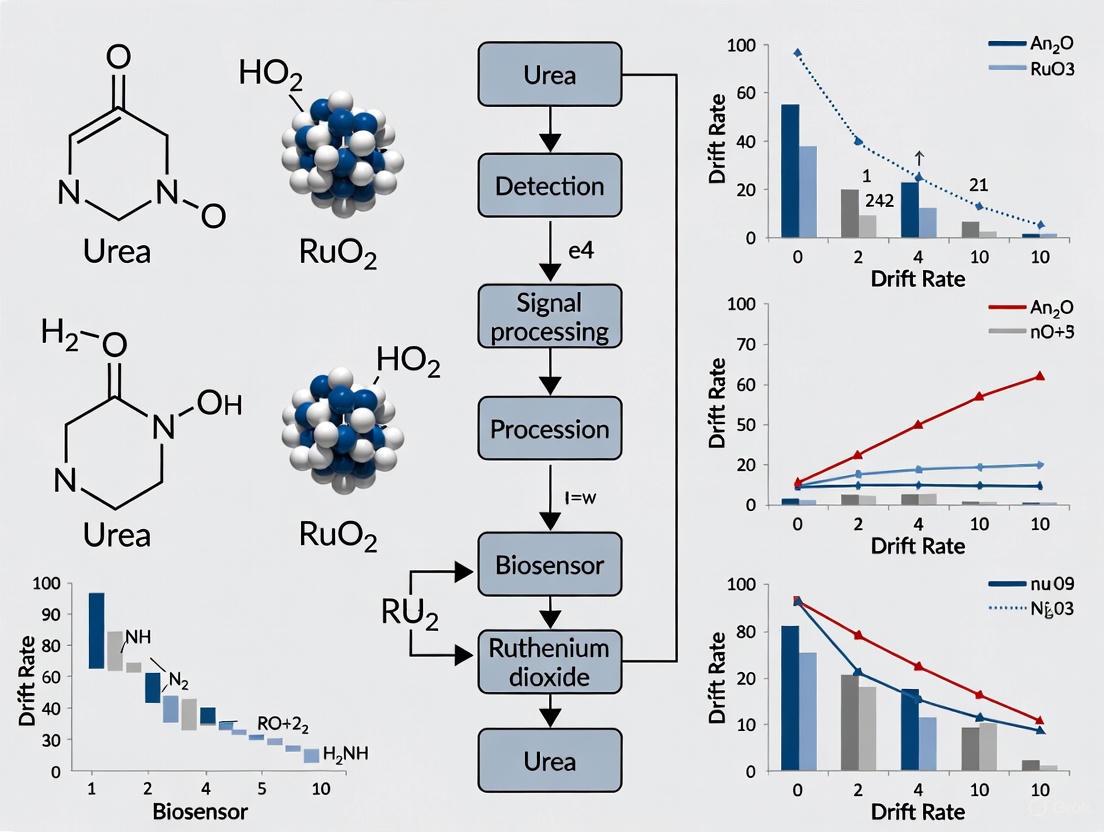
A significant challenge in the long-term stability of RuO₂ urea biosensors is the drift effect, a gradual change in the sensor's output signal over time while the measured urea concentration remains constant. Research has conclusively identified the formation of a hydration layer on the surface of the RuO₂ sensing film as the primary cause of this drift [6]. When the biosensor is immersed in a solution, hydroxyl groups form on the sensing film's surface. Water molecules and ions are then attracted to these sites through coulombic forces, leading to the development of a stable hydration layer [6]. This layer alters the electrical double layer capacitance at the critical interface between the sensing film and the solution, resulting in an unwanted shift in the measured potential and compromising the accuracy of long-term measurements [6]. This technical guide details the mechanisms behind this issue and provides evidence-based troubleshooting strategies to mitigate its effects.
Troubleshooting Guide & FAQs
FAQ 1: What exactly is the "hydration layer" and how does it cause signal drift?
The hydration layer is a thin, water-rich layer that forms on the solid surface of the RuO₂ sensing film when it is exposed to an aqueous solution. The formation process can be described in three key stages:
- Stage 1: Hydroxyl Group Formation. Hydroxyl groups (-OH) are established on the surface of the RuO₂ sensing film upon contact with the solution [6].
- Stage 2: Ion Hydration. Hydrated ions are formed as water molecules are attracted to and surround ions in the solution due to coulombic attraction [6].
- Stage 3: Hydration Layer Stabilization. These hydrated ions diffuse to the sensing film and contribute to the formation of a stable hydration layer, which modifies the surface potential and leads to the observed signal drift [6].
FAQ 2: What are the most effective strategies to reduce drift caused by the hydration layer?
Multiple strategies at different stages of the biosensor lifecycle can be employed to mitigate drift.
- Sensor Design and Material Engineering: Modifying the sensing electrode by combining RuO₂ with other metal oxides, such as cobalt oxide (Co₃O₄), can enhance stability. Research shows that a composite electrode with 50 mol% Co₃O4 and 50 mol% RuO₂ demonstrated superior stability, among other excellent properties [10].
- Circuit-Based Calibration: Implementing a dedicated calibration circuit can actively correct for drift. One study demonstrated a 98.77% reduction in drift rate (to 0.02 mV/hr) using a New Calibration Circuit (NCC) based on a voltage regulation technique [6].
- Optimized Enzyme Immobilization: Improving the stability of the urease enzyme layer can reduce changes at the sensor surface that might exacerbate hydration. Techniques like cross-linking the enzyme with glutaraldehyde and immobilizing it within an alginate polymer matrix have been shown to enhance operational stability [11].
FAQ 3: Is the drift effect unique to RuO₂-based urea biosensors?
No, the drift phenomenon related to surface hydration is a common challenge for many types of solid-state and metal oxide-based electrochemical sensors. However, the specific impact and the strategies for mitigation are highly dependent on the material properties of the sensing film and the operational environment [6].
Quantitative Data on Drift Mitigation Strategies
The following table summarizes key experimental data from the literature on the effectiveness of different approaches to reduce drift in RuO₂-related biosensors.
Table 1: Summary of Experimental Strategies for Drift Rate Reduction
| Mitigation Strategy | Reported Drift Rate | Reduction Effectiveness | Key Findings | Source |
|---|---|---|---|---|
| New Calibration Circuit (NCC) | 0.02 mV/hr | 98.77% reduction | A simple circuit structure using a non-inverting amplifier and voltage calibration. | [6] |
| Material Engineering (Co₃O₄-RuO₂ Composite) | Not explicitly quantified | Superior stability reported | The 50 mol% Co₃O4-50 mol% RuO₂ composite showed the best stability, sensitivity, and selectivity. | [10] |
| Enzyme Immobilization (Alginate/Cross-linking) | Retained ~70% sensitivity after 2 weeks | Improved long-term stability | Using bovine serum albumin (BSA) as a stabilizer and cross-linking with glutaraldehyde and Ca²⁺ ions. | [11] |
Detailed Experimental Protocols
This protocol outlines the process for creating a composite sensing electrode designed to improve stability and reduce environmental impact.
Key Research Reagent Solutions:
- RuO₂ & Co₃O4 Powders: The primary pH-sensitive and conductive materials (>97% purity).
- Ethyl Cellulose: Serves as a binder in the thick-film paste.
- Terpineol: Acts as a solvent to achieve the correct paste rheology.
- Ag/Pd Paste: Forms the conductive layer on the substrate.
- Al₂O₃ Substrate: A 96% alumina plate providing physical support and environmental tolerance.
Methodology:
- Weighing and Mixing: Precisely weigh RuO₂ and Co₃O₄ powders to achieve the desired molar ratio (e.g., 50:50). Combine them in a planetary ball mill.
- Milling: Wet-mill the powder mixture in isopropyl alcohol with zirconia balls for 8 hours to ensure a homogeneous blend.
- Paste Preparation: Dry the mixed powder to remove isopropyl alcohol. In an agate mortar, grind the powder with ethyl cellulose and terpineol for approximately 20 minutes to create a paste with optimal screen-printing rheology.
- Screen-Printing Conductive Layer: Print a Ag/Pd conductive layer onto the Al₂O₃ substrate. Dry at 120°C for 15 min and then fire at 860°C for 15 min.
- Screen-Printing Sensitive Layer: Print the prepared Co₃O₄-RuO₂ paste over the conductive layer. Dry at 120°C for 15 min and then sinter at 900°C for one hour.
- Insulation: Solder a copper wire to the conductive layer for electrical connection. Protect the connection and conductive tracks with a non-corrosive polydimethylsiloxane coating (e.g., DOWSIL 3140), leaving only the sensitive area exposed.
This protocol describes how to quantify the inherent drift of a biosensor and validate the performance of a calibration circuit.
Key Research Reagent Solutions:
- Phosphate Buffer Saline (PBS): Provides a stable and consistent ionic background for testing (e.g., 30 mM, pH 7).
- Urea Solutions: Prepared in PBS at concentrations relevant to the application (e.g., human body range: 2.5–7.5 mM).
- Fabricated RuO₂ Urea Biosensor: The device under test.
Methodology:
- Baseline Drift Measurement:
- Immerse the fabricated RuO₂ urea biosensor in a stable urea solution.
- Using a Voltage-Time (V-T) measurement system (e.g., an instrumentation amplifier like LT1167 and a Data Acquisition device like NI USB-6210), record the sensor's output voltage over an extended period (e.g., 12 hours) while the urea concentration is held constant.
- Plot the voltage vs. time data. The slope of this plot represents the inherent drift rate of the sensor (e.g., in mV/hr).
- Circuit Compensation Validation:
- Connect the same biosensor to the New Calibration Circuit (NCC), which typically consists of a non-inverting amplifier and a voltage calibrating circuit.
- Repeat the long-term measurement in the same urea solution, recording the output from the NCC.
- The drift rate calculated from the NCC output signal will be significantly lower, demonstrating the circuit's compensation efficacy.
- Baseline Drift Measurement:
Visualizing the Problem and Solutions
The following diagram illustrates the cause of drift and the primary mitigation strategies discussed in this guide.
For researchers and clinicians, accurate and stable measurement of Blood Urea Nitrogen (BUN) is critical for assessing renal function and managing conditions like congestive heart failure, where BUN to albumin ratio (BAR) serves as an independent prognostic marker [12]. However, biosensors, particularly those based on RuO₂, are often plagued by signal drift—a gradual change in output signal over time despite constant urea concentration. This instability can compromise long-term monitoring and clinical decision-making. This technical support center provides targeted troubleshooting guides and experimental protocols to help scientists mitigate drift in RuO₂ urea biosensors, enabling more reliable data in both clinical and research settings.
Troubleshooting Guides & FAQs
Frequently Asked Questions
Q1: What is the primary cause of signal drift in RuO₂ urea biosensors? The drift phenomenon is primarily attributed to the formation of a hydration layer on the sensing film's surface. When immersed in a solution, hydroxyl groups form on the RuO₂ film. Hydrated ions then diffuse to the film surface, leading to the formation of this layer via coulombic attraction. The resulting electrical double layer capacitance alters the surface potential of the film, causing the response voltage to change over time [7].
Q2: How can I experimentally confirm that my fabricated RuO₂ biosensor is functioning correctly before drift testing? A well-fabricated RuO₂ urea biosensor should exhibit high sensitivity and linearity. You can verify this by measuring its response to urea solutions within the physiologically relevant range (e.g., 2.5–7.5 mM). A properly functioning sensor has demonstrated an average sensitivity of 1.860 mV/(mg/dL) and a linearity of 0.999 when measured with a standard voltage-time (V-T) system [7].
Q3: Are there specific circuit designs proven to reduce drift in these biosensors? Yes, a New Calibration Circuit (NCC) based on a voltage regulation technique has been developed specifically for this purpose. This circuit features a simple structure composed of a non-inverting amplifier and a voltage calibrating circuit. Experiments have shown it can reduce the drift rate of an RuO₂ urea biosensor to 0.02 mV/hr, a 98.77% reduction compared to a conventional V-T measurement system [7] [8].
Q4: Besides electronic solutions, can material modifications reduce hysteresis and drift? Yes, modifying the sensing film with nanomaterials is an effective strategy. For instance, a urea biosensor based on a urease-Ag NPs/RuO₂ thin film has been developed. Research indicates that integrating silver nanoparticles (Ag NPs) can improve sensing characteristics, and when combined with specialized analog back-end circuits, can significantly reduce the hysteresis effect [9].
Troubleshooting Common Experimental Issues
Problem: Unacceptably High Drift Rate During Long-Term Measurement
- Potential Cause: Thick hydration layer formation on the RuO₂ sensing film.
- Solution: Implement the New Calibration Circuit (NCC) to actively compensate for the voltage shift caused by the hydration layer [7]. Ensure the sensing film is properly encapsulated to minimize direct, prolonged exposure to the solution when not in use.
Problem: Inconsistent Sensitivity Between Sensor Batches
- Potential Cause: Inconsistencies in the fabrication process, particularly in the immobilization of urease.
- Solution: Standardize the immobilization procedure. Use a protocol where aminopropyltriethoxysilane (APTS) solution is dropped on the RuO₂ film at room temperature, followed by 1% glutaraldehyde to enhance urease adsorption. The film should be kept still for 24 hours to ensure strong covalent binding [7].
Problem: Signal Instability and Noise
- Potential Cause: Power line interference or high-frequency noise.
- Solution: Incorporate a readout circuit with a Twin-T notch filter to cancel power line noise and a Sallen–Key low-pass filter to suppress high-frequency noise. This approach has been successfully used to improve signal quality in urea biosensor measurements [7].
Experimental Protocols for Key Cited Experiments
Protocol 1: Fabrication of a Flexible Arrayed RuO₂ Urea Biosensor
This methodology outlines the creation of the biosensor used to validate the New Calibration Circuit [7].
- Substrate Preparation: Begin with a flexible arrayed polyethylene terephthalate (PET) substrate.
- Electrode Formation: Print arrayed silver wires (from silver paste) onto the PET substrate using a screen-printing technique to form the working and reference electrodes.
- Sensing Film Deposition: Deposit the RuO₂ film onto the PET substrate over the electrode area using a sputtering system to create a defined RuO₂ film window.
- Encapsulation: Encapsulate the structure with an epoxy thermosetting polymer, leaving the sensing window exposed.
- Surface Functionalization:
- Drop aminopropyltriethoxysilane (APTS) solution onto the RuO₂ sensing film at room temperature.
- Drop a 1% glutaraldehyde solution onto the sensor to enhance the adsorption capability of the urease.
- Let the sensor sit undisturbed for 24 hours.
- Enzyme Immobilization: Finally, drop the urease solution onto the functionalized RuO₂ sensing film to form the complete biosensor.
Protocol 2: Quantifying Drift Rate Using the V-T System and NCC
This procedure describes how to experimentally measure and compare the drift rate with and without the calibration circuit [7].
- Solution Preparation: Prepare a urea solution at a specific concentration (e.g., within the 2.5–7.5 mM range) using a 30 mM phosphate buffer saline (PBS) solution at pH 7.0.
- Initial Baseline Measurement: Immerse the fabricated RuO₂ urea biosensor in the urea solution.
- Long-Term Immersion: Continue the immersion for a prolonged period, such as 12 hours.
- Response Measurement with V-T System:
- Connect the sensor to a conventional V-T measurement system (e.g., using an LT1167 instrumentation amplifier and a DAQ device like National Instruments USB-6210).
- Record the response voltage over the 12-hour period using software like LabVIEW.
- Calculate the drift rate (mV/hr) from this data.
- Response Measurement with NCC:
- Connect the same sensor to the New Calibration Circuit, which consists of a non-inverting amplifier and a voltage calibrating circuit.
- Repeat the voltage measurement over the same 12-hour immersion period.
- Calculate the new drift rate.
- Data Analysis: Compare the drift rates obtained from the two systems to quantify the improvement. The NCC has been shown to reduce the drift rate to 0.02 mV/hr.
Data Presentation
The following table summarizes the quantitative performance improvements achieved by implementing the New Calibration Circuit for an RuO₂ urea biosensor, based on published experimental data [7].
Table 1: Performance Comparison of RuO₂ Urea Biosensor with and without the New Calibration Circuit (NCC)
| Performance Parameter | Conventional V-T System | With NCC Implementation | Improvement |
|---|---|---|---|
| Drift Rate | ~1.59 mV/hr (calculated) | 0.02 mV/hr | 98.77% Reduction |
| Average Sensitivity | 1.860 mV/(mg/dL) | 1.860 mV/(mg/dL) (maintained) | Sensitivity was preserved |
| Linearity | 0.999 | 0.999 (maintained) | Linearity was preserved |
Mandatory Visualization
Diagram 1: RuO₂ Urea Biosensor Fabrication Workflow
Biosensor Fabrication Steps
Diagram 2: Drift Rate Test & NCC Calibration Logic
Drift Test and NCC Calibration Logic
The Scientist's Toolkit: Research Reagent Solutions
Table 2: Essential Materials for Fabricating and Testing RuO₂ Urea Biosensors
| Material | Function / Role in Experiment | Source Example |
|---|---|---|
| Polyethylene Terephthalate (PET) Substrate | Serves as the flexible, inert base for the biosensor. | Zencatec Corporation [7] |
| Ruthenium (Ru) Target (99.95% purity) | Sputtered to form the key RuO₂ sensing film. | Ultimate Materials Technology Co., Ltd. [7] |
| Silver Paste | Screen-printed to form conductive working and reference electrodes. | Advanced Electronic Material Inc. [7] |
| Urease (from Jack Bean) | The enzyme that catalyzes the hydrolysis of urea, generating a measurable signal. | Sigma-Aldrich Corp. [7] |
| Epoxy Polymer (JA643) | Encapsulates and insulates the sensor, protecting the circuitry. | Sil-More Industrial, Ltd. [7] |
| Aminopropyltriethoxysilane (APTS) | A silane coupling agent used to functionalize the RuO₂ surface for enzyme binding. | [7] |
| Glutaraldehyde (1% Solution) | A crosslinker that creates strong covalent bonds for stable urease immobilization. | [7] |
| Phosphate Buffer Saline (PBS, 30 mM, pH 7) | Provides a stable, physiologically relevant ionic environment for testing. | Prepared from KH₂PO₄ & K₂HPO₄ [7] |
A Novel Calibration Circuit (NCC) and Fabrication for Drift Reduction
FAQ: Core Principles and Operation
Q1: What is the primary function of the New Calibration Circuit (NCC)? The primary function of the New Calibration Circuit (NCC) is to significantly reduce the drift effect in RuO2 urea biosensors. This drift phenomenon, where the sensor's response voltage undesirably changes over time during long-term measurement, is a critical non-ideal effect that compromises measurement stability. The NCC achieves this stabilization through a voltage regulation technique [13] [6].
Q2: What causes the drift effect in potentiometric biosensors? The drift effect is primarily caused by the formation of a hydration layer on the surface of the sensing film. When the sensor is immersed in a solution, hydroxyl groups form on the film's surface. Hydrated ions then diffuse to the sensing film, resulting in the formation of this layer. The electrical double layer capacitance formed by the hydration layer alters the surface potential of the film, leading to a drifting voltage reading over time [13] [6].
Q3: What is the core design principle behind the NCC? The NCC is designed with a focus on simplicity. It is composed of two main parts: a non-inverting amplifier and a voltage calibrating circuit. This straightforward design leverages voltage regulation to counteract and correct the unstable sensor output caused by the drift effect [13] [6].
FAQ: Experimental Setup and Integration
Q4: What are the key components of the traditional V-T measurement system? The conventional Voltage-Time (V-T) measurement system, used as a benchmark for evaluating the NCC, consists of the following core components [13] [6]:
- Instrumentation Amplifier: An LT1167 is used to read the signal from the biosensor.
- Data Acquisition (DAQ) Device: A National Instruments USB-6210 card converts the analog signal for processing.
- Program System Software: National Instruments LabVIEW is used for data recording and analysis.
Q5: Which biosensor fabrication method was used with the NCC? The NCC was validated using a flexible arrayed RuO2 urea biosensor. The manufacturing process involved [13] [6]:
- Printing arrayed silver wires on a flexible PET substrate using screen printing.
- Depositing the RuO2 sensing film via a sputtering system.
- Immobilizing the urease enzyme onto the RuO2 film using APTS and glutaraldehyde to form the complete biosensor.
Troubleshooting Guide: Common Experimental Issues
| Problem Area | Specific Issue | Potential Cause | Solution |
|---|---|---|---|
| Sensor Performance | High drift rate without NCC | Formation of a hydration layer on the sensing film. | Integrate the NCC into the measurement system to apply voltage regulation [13] [6]. |
| Low sensitivity or linearity | Improper biosensor fabrication or enzyme immobilization. | Verify fabrication protocol (sputtering parameters, immobilization time) and material purity [13] [6]. | |
| Circuit Operation | Unstable NCC output | Power supply noise or incorrect component values. | Ensure stable power lines and verify that resistor values in the non-inverting amplifier and calibrator match the design specifications. |
| System Integration | Noisy signal in V-T system | Power line interference or poor connections. | Check all physical connections and shields. The NCC itself is designed to be simple and robust against such issues [13]. |
Experimental Protocol: Validating NCC Performance
Objective: To quantify the reduction in drift rate achieved by using the New Calibration Circuit with an RuO2 urea biosensor.
Materials and Reagents:
- Fabricated flexible arrayed RuO2 urea biosensor [13] [6]
- New Calibration Circuit (NCC) prototype
- Voltage-Time (V-T) measurement system
- Urea solutions in phosphate buffer saline (PBS), pH 7 [13] [6]
- Data acquisition software (e.g., LabVIEW)
Methodology:
- Solution Preparation: Prepare urea solutions within the normal human body concentration range (e.g., 2.5–7.5 mM) using 30 mM PBS as a buffer [13] [6].
- Baseline Drift Measurement: Immerse the RuO2 urea biosensor in a urea solution. Connect the sensor to the conventional V-T measurement system. Record the response voltage continuously for 12 hours without the NCC [13] [6].
- NCC Drift Measurement: Maintain the sensor in the solution. Now, connect the biosensor to the NCC, which is then connected to the V-T system. Record the output voltage for another 12 hours (or a comparable period) [13] [6].
- Data Analysis: Calculate the drift rate (in mV/hour) for both experimental setups from the slope of the voltage-time data. Compare the results.
Expected Outcome: The experiment should demonstrate a dramatic reduction in the observed drift rate when the NCC is employed.
Table 1: Quantitative Comparison of Drift Rate Performance
| Measurement System | Drift Rate (mV/hr) | Percentage Reduction | Reference |
|---|---|---|---|
| Conventional V-T System | 1.59 | Baseline | [13] [6] |
| With New Calibration Circuit (NCC) | 0.02 | 98.77% | [13] [6] |
Table 2: Sensing Characteristics of the RuO2 Urea Biosensor
| Sensing Characteristic | Performance Value | Reference |
|---|---|---|
| Average Sensitivity | 1.860 mV/(mg/dL) | [13] [6] |
| Linearity | 0.999 | [13] [6] |
Research Reagent Solutions
Table 3: Essential Materials for RuO2 Urea Biosensor Fabrication and Testing
| Material | Function / Role in the Experiment | Source / Example |
|---|---|---|
| Polyethylene Terephthalate (PET) | Flexible substrate for the biosensor. | Zencatec Corporation [13] [6] |
| Ruthenium (Ru) Target | Source for sputtering to create the RuO2 sensing film. | Ultimate Materials Technology Co., Ltd. [13] [6] |
| Silver Paste | Forms the conductive electrodes (wires) on the substrate. | Advanced Electronic Material Inc. [13] [6] |
| Epoxy Polymer | Encapsulation and insulation layer. | Sil-More Industrial, Ltd. [13] [6] |
| Urease Enzyme | Bioreceptor that catalyzes the hydrolysis of urea. | Sigma-Aldrich Corp. [13] [6] |
| Phosphate Buffer Saline (PBS) | Provides a stable, pH-neutral environment for testing. | Prepared from KH2PO4 & K2HPO4 powders [13] [6] |
Experimental Workflow Visualization
Diagram Title: NCC Drift Rate Validation Workflow
Diagram Title: NCC Problem-Solution Logic
Troubleshooting Guides
Non-Inverting Amplifier Issues
Problem: Unexpected Output Voltage from Non-Inverting Amplifier
- Q: The output voltage of my non-inverting amplifier does not match the theoretical calculation. What could be wrong?
- A: This discrepancy can arise from several sources. First, verify the closed-loop gain using the formula ( Av = 1 + \frac{Rf}{R_2} ). Ensure the feedback resistor (Rƒ) and the ground resistor (R2) are the correct values and properly connected. Second, check the power supply rails to the operational amplifier to ensure it is not saturating. Finally, unstable output can be caused by power supply noise or insufficient decoupling; use bypass capacitors close to the op-amp's power pins [14].
Problem: Amplifier Circuit Exhibiting Oscillation or Noise
- Q: My amplifier circuit is oscillating or has significant high-frequency noise. How can I resolve this?
- A: This is often related to stability and layout. To cancel power line noise (e.g., 50/60 Hz), a Twin-T notch filter can be integrated into the circuit. To suppress broader high-frequency noise, add a Sallen-Key low-pass filter at the output stage. Furthermore, ensure that the feedback path is short and direct, and use a single-point ground for all sensitive analog components to minimize parasitic coupling [7] [6].
Voltage Calibrating Circuit and Drift Issues
Problem: Significant Drift in Sensor Output Over Time
- Q: The response voltage from my RuO₂ biosensor drifts over time during long-term measurement, affecting accuracy. What is the cause and solution?
- A: The drift phenomenon is primarily caused by the formation of a hydration layer on the surface of the RuO₂ sensing film. When immersed in a solution, hydroxyl groups form on the film's surface. Hydrated ions then diffuse to the sensing film, leading to an electrical double-layer capacitance that changes the surface potential over time. The solution implemented by the New Calibration Circuit (NCC) is to use a voltage regulation technique that actively compensates for this slow voltage change, thereby maintaining a stable reading [7] [6] [8].
Problem: Ineffective Drift Compensation by the NCC
- Q: I have built the NCC, but it is not effectively reducing the drift rate. Where should I look for faults?
- A: Begin by characterizing the drift rate of your biosensor alone using a standard voltage-time (V-T) measurement system to establish a baseline. Next, verify the output of the voltage calibrating circuit block independently to ensure it is generating a stable and clean reference voltage. The problem may lie in the interaction between the two stages. Ensure that the connection between the non-inverting amplifier and the voltage calibrating circuit does not load the amplifier's output, which would alter its gain and compensation characteristics [7].
Frequently Asked Questions (FAQs)
Q: What is the typical performance improvement I can expect from using the NCC? A: In experimental validation, the proposed NCC reduced the drift rate of an RuO₂ urea biosensor to 0.02 mV/hr, which represents a 98.77% reduction compared to the drift rate measured without it [7] [6] [8].
Q: Why is a non-inverting amplifier configuration chosen for the NCC? A: The non-inverting amplifier provides a very high input impedance, meaning it does not load the sensor and draw current, which could otherwise affect the sensitive measurement. It also provides a stable gain defined by external resistors, and its output signal is in-phase with the input signal, simplifying the overall circuit analysis [14].
Q: Besides drift reduction, what other sensing characteristics are important for the RuO₂ urea biosensor? A: The RuO₂ urea biosensor itself demonstrated excellent baseline sensing characteristics, including an average sensitivity of 1.860 mV/(mg/dL) and a linearity of 0.999 within the normal human body urea concentration range (2.5–7.5 mM), confirming a well-fabricated sensor [7] [6].
Q: My biosensor's response is weak. Can I just increase the gain of the non-inverting amplifier? A: Yes, but within limits. The gain is set by ( Av = 1 + \frac{Rf}{R_2} ). Increasing Rƒ or decreasing R2 will increase the gain. However, ensure the amplified signal does not exceed the op-amp's output voltage swing. Also, very high gains may amplify noise along with the signal, so a balanced approach is necessary [14].
Experimental Protocols & Data
Key Experiment: Validating NCC Drift Reduction
Objective: To quantify the reduction in drift rate achieved by using the New Calibration Circuit (NCC) with a fabricated RuO₂ urea biosensor.
Methodology:
- Biosensor Fabrication: A flexible arrayed RuO₂ urea biosensor is fabricated. Silver wires are screen-printed onto a PET substrate to form electrodes. A RuO₂ film is deposited via sputtering, and urease is immobilized on the film using glutaraldehyde to create the biosensing window [7] [6].
- Baseline Drift Measurement (V-T System):
- The biosensor is immersed in a urea solution (e.g., in 30 mM PBS at pH 7).
- The response voltage is measured continuously for 12 hours using a conventional Voltage-Time (V-T) measurement system. This system typically comprises an instrumentation amplifier (e.g., LT1167) and a data acquisition device [7] [6].
- The drift rate (mV/hr) is calculated from this data.
- NCC Drift Measurement:
- The same biosensor is connected to the New Calibration Circuit.
- The response voltage is again measured over 12 hours under the same conditions.
- The new, lower drift rate is calculated.
- Sensitivity and Linearity Check: The sensitivity and linearity of the biosensor are verified by measuring its response to different urea concentrations within the 2.5–7.5 mM range [7].
Results Summary:
| Measurement System | Average Sensitivity | Linearity | Drift Rate | Drift Reduction |
|---|---|---|---|---|
| V-T System (Baseline) | 1.860 mV/(mg/dL) | 0.999 | X mV/hr | -- |
| With Proposed NCC | Data not explicitly stated | Data not explicitly stated | 0.02 mV/hr | 98.77% [7] [6] [8] |
Experimental Workflow Diagram
The Scientist's Toolkit: Research Reagent Solutions
Table: Essential Materials for RuO₂ Urea Biosensor Fabrication and Testing
| Item / Reagent | Function / Role in the Experiment |
|---|---|
| Polyethylene Terephthalate (PET) | Serves as a flexible, inert substrate for building the biosensor [7] [6]. |
| Ruthenium (Ru) Target | Source material for sputtering deposition to create the key RuO₂ sensing film [7] [6]. |
| Silver Paste | Used to form conductive wires and electrodes (working and reference electrodes) on the PET substrate via screen-printing [7] [6]. |
| Epoxy Polymer | An encapsulation material used to create an insulation layer and define the sensing film window [7] [6]. |
| Urease Enzyme | The biological recognition element. It catalyzes the hydrolysis of urea, leading to a measurable change in potential on the RuO₂ film [7] [6] [15]. |
| Glutaraldehyde | A cross-linking agent used to immobilize the urease enzyme onto the activated RuO₂ sensing surface, preventing enzyme loss [7] [6]. |
| Phosphate Buffer Saline (PBS) | Provides a stable, neutral (pH 7) ionic environment that mimics physiological conditions for testing the biosensor [7] [6]. |
| Urea | The analyte of interest. Prepared in PBS at various concentrations (e.g., 2.5-7.5 mM) to test biosensor performance [7] [6]. |
NCC System Architecture
Step-by-Step Fabrication of the Flexible Arrayed RuO₂ Urea Biosensor
This guide details the fabrication and troubleshooting of a flexible arrayed Ruthenium Oxide (RuO₂) urea biosensor. A significant challenge in biosensor operation is the drift effect, a gradual change in the sensor's output signal over time during long-term measurement, which can lead to inaccurate readings [7]. This resource, designed for researchers and scientists, provides a clear, step-by-step fabrication protocol and addresses common experimental pitfalls. The content is framed within the broader research goal of reducing the drift rate, a critical factor for the reliability and accuracy of urea biosensors in applications like health monitoring and drug development [7] [8].
Fabrication Workflow
The fabrication of the flexible arrayed RuO₂ urea biosensor involves a multi-stage process, from substrate preparation to enzyme immobilization. The following diagram outlines the key steps.
Detailed Experimental Protocol
Step 1: Substrate Preparation
- Material: A flexible polyethylene terephthalate (PET) substrate is used [7].
- Function: Serves as a flexible, inert base for the biosensor.
Step 2: Electrode Formation
Step 3: Sensing Film Deposition
Step 4: Insulation Layer
Step 5: Surface Functionalization
- APTS Treatment: Aminopropyltriethoxysilane (APTS) solution is dropped onto the RuO₂ sensing film at room temperature. This acts as a coupling agent [7].
- Cross-linking: A 1% glutaraldehyde solution is dropped onto the sensor. This enhances the ability of urease to be adsorbed on the surface [7].
- Curing: The sensor is kept still for 24 hours after this step [7].
Step 6: Enzyme Immobilization
Research Reagent Solutions
The table below lists the essential materials and their functions for fabricating the RuO₂ urea biosensor.
| Item | Function / Role | Specification / Source |
|---|---|---|
| PET Substrate | Flexible, inert base material | Zencatec Corporation, Taiwan [7] |
| Ruthenium (Ru) Target | Source for depositing RuO₂ sensing film | Purity 99.95%; Ultimate Materials Technology Co., Ltd. [7] |
| Silver Paste | Forms conductive working & reference electrodes | Screen-printed; Advanced Electronic Material Inc. [7] |
| Epoxy Polymer | Insulation layer for encapsulation | Product JA643; Sil-More Industrial, Ltd. [7] |
| Urease | Biological recognition element for urea | Sigma-Aldrich Corp. [7] |
| APTS & Glutaraldehyde | Surface functionalization for enzyme immobilization | Forms covalent bonds with urease [7] |
| Phosphate Buffer Saline (PBS) | Testing solution at neutral pH (7.0) | 30 mM, prepared from KH₂PO₄ & K₂HPO₄ powders [7] |
Troubleshooting Common Experimental Issues
Fabrication and Performance Problems
Problem: Poor Adhesion of Silver Electrodes
- Possible Cause 1: Improper surface cleaning of the PET substrate.
- Solution: Ensure the PET substrate is thoroughly cleaned with appropriate solvents (e.g., isopropanol, acetone) and plasma-treated if possible to increase surface energy and promote adhesion [16].
- Possible Cause 2: Incorrect curing profile for the silver paste.
- Solution: Follow the manufacturer's recommended curing temperature and time precisely. Verify the temperature uniformity in the curing oven.
Problem: High Signal Drift During Long-Term Measurement
- Possible Cause: Formation of a hydration layer on the sensing film's surface, which alters the electrical double layer capacitance [7].
- Solution: Integrate the New Calibration Circuit (NCC). This circuit, based on a voltage regulation technique with a non-inverting amplifier and a voltage calibrating circuit, was shown to reduce the drift rate by 98.77% (to 0.02 mV/hr) [7] [8].
Problem: Low Sensitivity or Slow Response to Urea
- Possible Cause 1: Loss of urease enzyme activity during immobilization or storage.
- Solution: Ensure the urease solution is prepared fresh and stored according to the supplier's guidelines. Verify the concentrations of glutaraldehyde and APTS, as over-crosslinking can hinder substrate diffusion.
- Possible Cause 2: Incomplete or non-uniform functionalization of the RuO₂ surface.
- Solution: Standardize the volume and dropping location of APTS and glutaraldehyde solutions. Ensure the sensor is kept in a controlled, humidity-free environment during the 24-hour curing period [7].
Problem: Inconsistent Results Between Arrayed Sensors
- Possible Cause: Lack of reproducibility in the manual dropping process for surface functionalization and enzyme immobilization.
- Solution: Transition from manual pipetting to automated dispensing systems for applying APTS, glutaraldehyde, and urease solutions. This improves uniformity across the array [17].
Measurement and Calibration FAQs
Q: What is the typical performance I should expect from a well-fabricated RuO₂ urea biosensor?
- A: When measured with a standard voltage-time (V-T) system, a well-fabricated sensor should exhibit an average sensitivity of approximately 1.860 mV/(mg/dL) and a linearity of 0.999 over the physiologically relevant urea concentration range (e.g., 2.5–7.5 mM) [7].
Q: How does the New Calibration Circuit (NCC) connect to the biosensor?
- A: The biosensor's response voltage is connected to the input of the NCC. The NCC processes this signal to compensate for the low-frequency drift component. The output is a stabilized voltage reading that can be recorded by a data acquisition system [7].
Q: Why is a flexible substrate like PET chosen?
Frequently Asked Questions (FAQs)
Q1: What is the primary function of the New Calibration Circuit (NCC) in this setup? The primary function of the NCC is to significantly reduce the drift effect observed in RuO2 urea biosensors. It uses a voltage regulation technique to counteract the gradual change in the sensor's response voltage over time, achieving a 98.77% reduction in the drift rate, lowering it to 0.02 mV/hr [7].
Q2: Why is drift a critical problem in urea biosensors? Drift is a non-ideal effect where the sensor's response voltage changes over time during long-term measurement. This is often caused by the formation of a hydration layer on the surface of the sensing film, which alters the electrical double layer capacitance. This instability makes long-term measurements unreliable and is a key challenge for biosensor accuracy [7] [18].
Q3: What are the core components of the V-T measurement system? The conventional Voltage-Time (V-T) measurement system consists of three main components [7]:
- Instrumentation Amplifier: An LT1167 is used to amplify the small signal from the biosensor.
- Data Acquisition (DAQ) Device: A USB-6210 from National Instruments converts the analog signal to a digital one.
- Program System Software: LabVIEW software is used on a computer to record, display, and process the acquired voltage data over time.
Q4: My biosensor's sensitivity seems low. What should I check? Low sensitivity can often be traced to the bioreceptor layer. Verify the immobilization process of the urease enzyme on the RuO2 sensing film. Ensure that the procedure using APTS and glutaraldehyde was correctly followed and that the immobilized enzyme remains active. Also, confirm that the urea concentrations of your test solutions are within the expected physiological range (e.g., 2.5–7.5 mM) [7].
Q5: How does the NCC's design contribute to its effectiveness? The NCC is designed with a simple structure for ease of implementation. It is composed of a non-inverting amplifier and a voltage calibrating circuit. This design leverages voltage regulation to directly compensate for the drift signal without overly complicating the electronics, making it both effective and practical [7].
Troubleshooting Guide
| Problem | Possible Cause | Solution |
|---|---|---|
| High Drift Rate | Formation of a hydration layer on the RuO2 sensing film [7]. | Integrate the NCC into the measurement circuit to actively correct for the drift voltage [7]. |
| Unstable Signal (Noise) | Electrical interference from power lines or other equipment [7]. | Ensure all connections are shielded. For the V-T system, a dedicated noise-canceling readout circuit with a Twin-T notch filter can be implemented [7]. |
| No Output Signal | Incorrect circuit connections or a failed biosensor. | Systematically check all wiring from the sensor to the amplifier and DAQ. Verify the biosensor's functionality by testing it in a known standard solution. |
| Low Sensitivity | Inactive or poorly immobilized urease enzyme [7]. | Reproduce the biosensor, carefully following the immobilization protocol with APTS and glutaraldehyde [7]. |
| Poor Linearity | Sensor saturation or a problem with the sensing film morphology. | Ensure measurements are taken within the specified urea concentration range (e.g., 2.5–7.5 mM). Check the fabrication process of the RuO2 sensing film [7]. |
Experimental Performance Data
The following table summarizes the key performance characteristics of the RuO2 urea biosensor when measured with the conventional V-T system and after the integration of the New Calibration Circuit (NCC). This data allows for a direct comparison of the system's performance before and after drift correction [7].
Table 1: Performance comparison of the RuO2 urea biosensor with and without the NCC.
| Performance Characteristic | V-T Measurement System (without NCC) | With New Calibration Circuit (NCC) |
|---|---|---|
| Average Sensitivity | 1.860 mV/(mg/dL) | Information not specified in source |
| Linearity | 0.999 | Information not specified in source |
| Drift Rate | Information not specified in source | 0.02 mV/hr |
| Drift Rate Reduction | Baseline | 98.77% |
Experimental Protocol: Measuring Drift Rate with the V-T System and NCC
Objective: To quantify the drift rate of a fabricated RuO2 urea biosensor and verify the effectiveness of the New Calibration Circuit (NCC) in reducing it.
Principle: The biosensor is immersed in a urea solution for an extended period (e.g., 12 hours). The response voltage is continuously monitored. The drift rate is calculated as the change in voltage per unit time under stable conditions [7].
Materials and Reagents:
- Fabricated flexible arrayed RuO2 urea biosensor [7]
- Urea solution at a specific concentration (e.g., within 2.5–7.5 mM) in 30 mM Phosphate Buffer Saline (PBS), pH 7.0 [7]
- V-T Measurement System (LT1167 amplifier, USB-6210 DAQ, LabVIEW software) [7]
- New Calibration Circuit (NCC) - composed of a non-inverting amplifier and voltage calibrating circuit [7]
Procedure:
- Solution Preparation: Prepare a fresh urea solution in PBS at the desired concentration.
- Baseline Measurement (without NCC):
- Connect the biosensor to the V-T measurement system.
- Immerse the biosensor's sensing film in the urea solution.
- Start the data acquisition in LabVIEW and record the voltage for 12 hours.
- Calculate the drift rate from the recorded V-T data.
- Drift-Corrected Measurement (with NCC):
- Integrate the NCC between the biosensor and the V-T measurement system.
- Immerse the biosensor in the same urea solution.
- Start the data acquisition and record the voltage for 12 hours.
- Calculate the new drift rate from the corrected V-T data.
- Data Analysis: Compare the drift rates from steps 2 and 3 to determine the percentage reduction achieved by the NCC.
Workflow Visualization
The diagram below illustrates the experimental workflow for comparing the drift rate with and without the NCC.
The Scientist's Toolkit: Essential Research Reagents & Materials
This table details the key materials and reagents required for the fabrication of the RuO2 urea biosensor and the execution of the drift measurement experiments [7].
Table 2: Key research reagents and materials for the RuO2 urea biosensor experiment.
| Item | Function / Role in the Experiment |
|---|---|
| Polyethylene Terephthalate (PET) Substrate | Serves as the flexible, solid support for the biosensor. |
| Ruthenium (Ru) Target | Source material for sputtering to deposit the RuO2 sensing film. |
| Silver Paste | Used to form the conductive working and reference electrodes via screen printing. |
| Epoxy Polymer | Forms an insulation layer to encapsulate and protect the sensor circuitry. |
| Urease Enzyme | The biological recognition element that catalyzes the hydrolysis of urea. |
| Urea | The target analyte; used to prepare test solutions for sensor characterization. |
| Phosphate Buffer Saline (PBS) | Provides a stable, physiologically relevant pH environment for measurements. |
| APTS & Glutaraldehyde | Chemicals used to cross-link and immobilize the urease enzyme onto the RuO2 sensing film. |
Understanding Drift and the NCC Correction Principle
The Root Cause of Sensor Drift
Sensor drift is a common challenge in electrochemical biosensors. In the case of the RuO2 urea biosensor, the drift phenomenon is primarily attributed to the formation of a hydration layer on the sensing film's surface. When immersed in a solution, hydroxyl groups form on the RuO2, and hydrated ions diffuse to the film, creating an electrical double-layer capacitance. The instability of this layer over time leads to a continuous shift in the surface potential, which manifests as a drift in the output voltage [7] [18]. This process is visualized below.
How the NCC Counters Drift
The New Calibration Circuit (NCC) operates on the principle of voltage regulation. It is strategically placed in the signal path between the biosensor and the data acquisition system. The circuit is designed to generate a compensating voltage that opposes the drift voltage. By applying this counter-voltage, the NCC effectively "cancels out" the slow, unwanted drift signal, leaving a stable and accurate reading of the biosensor's response to urea. Its simple design, based on a non-inverting amplifier and a calibrating circuit, makes it a practical solution for real-world applications [7].
Optimizing Performance and Addressing Fabrication and Circuit Challenges
In the development of RuO₂ urea biosensors, controlling fabrication quality is paramount to achieving reliable performance, especially for the ultimate goal of reducing sensor drift rate. Sensitivity and linearity are not merely performance indicators; they are critical quality control parameters that confirm the biosensor has been correctly fabricated and will perform predictably in long-term applications. When a biosensor demonstrates high sensitivity and a linearity close to 1.0, it provides strong evidence that the urease immobilization on the RuO₂ sensing film is effective and that the underlying transducer is functioning correctly. This technical support document provides researchers with troubleshooting guides and experimental protocols to diagnose, verify, and optimize these key metrics within the context of advanced drift rate reduction research.
Core Performance Metrics and Quantifiable Targets
Defining the Metrics
For a potentiometric RuO₂ urea biosensor, sensitivity and linearity are defined as follows:
- Sensitivity: The change in output voltage (mV) per unit change in urea concentration (mg/dL or mM). It reflects the biosensor's ability to detect minor variations in analyte concentration. A higher sensitivity is generally desirable.
- Linearity: A statistical measure (often the correlation coefficient, R², of the calibration curve) of how well the sensor's response follows a straight-line relationship with urea concentration across the intended measurement range. A value of 1.0 indicates perfect linearity.
Established Quality Benchmarks
The following table summarizes target values for these metrics, as established in foundational research, which serve as benchmarks for fabrication quality.
Table 1: Target Performance Metrics for Quality Control of RuO₂ Urea Biosensors
| Performance Metric | Target Value | Measurement Context | Significance for Fabrication Quality |
|---|---|---|---|
| Average Sensitivity | 1.860 mV/(mg/dL) [6] [7] [8] | Urea concentration in standard solutions | Confirms successful enzyme immobilization and efficient catalytic activity on the RuO₂ surface. |
| Linearity (R²) | 0.999 [6] [7] [8] | Urea concentration in standard solutions | Indicates a stable and predictable response, validating the immobilization process and transducer stability. |
Troubleshooting Guides and FAQs
This section addresses common experimental challenges researchers face when fabricating RuO₂ urea biosensors, linking issues with sensitivity and linearity directly to their impact on long-term drift.
FAQ 1: Why is my fabricated biosensor's sensitivity lower than the target value, and how does this relate to drift?
A: Low sensitivity often points to inadequate urease activity on the sensor surface. This not only reduces signal strength but can also exacerbate long-term drift.
- Potential Cause 1: Ineffective Enzyme Immobilization.
- Explanation: The urease may not be sufficiently or stably bound to the RuO₂ sensing film. This can be due to problems with the cross-linker (glutaraldehyde) or the APTS solution used to functionalize the surface.
- Solution: Verify the concentration and incubation time for APTS and glutaraldehyde. Ensure the glutaraldehyde solution is fresh and that the sensor is kept still for the full 24-hour immobilization period as per the protocol [6] [7].
- Potential Cause 2: Poor Electrical Properties of the Sensing Film.
- Explanation: The sputtered RuO₂ film may have high resistivity, impairing its function as a transducer. RuO₂ is chosen for its high metallic conductivity, and deviations in the sputtering process can compromise this [6] [7].
- Solution: Review the Ru sputtering parameters (purity should be 99.95%) to ensure the deposited film has the required low resistivity and high thermal stability.
FAQ 2: My biosensor's response is non-linear (low R²). What steps should I take to diagnose this?
A: Non-linearity suggests an inconsistent response across the measurement range, which can be a precursor to unstable drift.
- Potential Cause 1: Inhomogeneous Enzyme Layer.
- Explanation: If the urease is not uniformly distributed on the RuO₂ film, the reaction kinetics will vary across the sensor's surface, leading to a non-linear voltage response.
- Solution: Ensure the urease solution is dropped carefully and consistently onto the sensing film. Using a well-defined film window created by the epoxy encapsulation can help standardize the active area [6] [7].
- Potential Cause 2: Sensor Saturation or Non-ideal Reaction Conditions.
- Explanation: Operating outside the linear range of the enzyme or using a buffer solution with incorrect pH or ionic strength can distort the response.
- Solution: Perform calibration within the physiologically relevant urea concentration range (2.5–7.5 mM). Use a fresh 30 mM Phosphate Buffer Saline (PBS) with a pH of 7.0 for all experiments [6] [7] [19].
FAQ 3: We have acceptable sensitivity and linearity, but our drift rate is still high. How are these related?
A: Achieving target sensitivity and linearity confirms proper fabrication but does not automatically eliminate drift. Drift is often linked to a separate phenomenon: the formation of a hydration layer on the sensing film's surface in solution. While good fabrication is the first step, a dedicated calibration circuit is required to actively combat drift [6] [7].
- Explanation: The hydration layer alters the electrical double layer capacitance at the film-solution interface, causing the output voltage to change over time independently of the urea concentration. Your well-fabricated sensor is correctly detecting urea, but its baseline is shifting [6] [7].
- Solution: Implement a New Calibration Circuit (NCC) in your measurement setup. Research has shown that a simple NCC based on a voltage regulation technique can reduce the drift rate of an RuO₂ urea biosensor by 98.77% (from ~1.6 mV/hr to 0.02 mV/hr) [6] [7] [8]. This circuit actively compensates for the slow voltage shift, preserving the accuracy of your well-fabricated sensor.
Experimental Protocol: Verification of Metrics
This protocol outlines the standard method for verifying the sensitivity and linearity of a fabricated RuO₂ urea biosensor, based on established experimental work [6] [7].
The diagram below illustrates the sequential workflow for fabricating the biosensor and verifying its key performance metrics.
Materials and Reagents
Table 2: Essential Research Reagent Solutions for RuO₂ Urea Biosensor Fabrication and Testing
| Item | Specification / Function | Source Example |
|---|---|---|
| PET Substrate | Flexible substrate for the biosensor. | Zencatec Corporation [6] [7] |
| Ruthenium (Ru) Target | Source for sputtering RuO₂ sensing film (99.95% purity). | Ultimate Materials Technology Co. [6] [7] |
| Silver Paste | Forms conductive working and reference electrodes. | Advanced Electronic Material Inc. [6] [7] |
| Urease | Enzyme that catalyzes hydrolysis of urea. | Sigma-Aldrich Corp. [6] [7] |
| Urea | Analytic for calibration and testing. | J. T. Baker Corp. [6] [7] |
| Phosphate Buffered Saline (PBS) | Provides stable pH 7.0 environment for testing. | Prepared from KH₂PO₄/K₂HPO₄ [6] [7] |
Step-by-Step Procedure
- Fabricate the RuO₂ Urea Biosensor: Follow the sequence outlined in the workflow diagram (Figure 1). Key steps include screen-printing silver electrodes, sputtering the RuO₂ film, and immobilizing the urease enzyme using APTS and glutaraldehyde to create a stable, covalently bound layer [6] [7].
- Prepare Standard Urea Solutions: Prepare a series of urea solutions in 30 mM PBS (pH 7.0) covering the concentration range of 2.5 to 7.5 mM, which represents the normal physiological range in the human body [6] [7] [19].
- Voltage-Time (V-T) Measurement:
- Immerse the fabricated biosensor in a urea solution.
- Connect the sensor to a readout circuit (e.g., an instrumentation amplifier like LT1167).
- Measure the steady-state response voltage for each concentration using a Data Acquisition (DAQ) device and software (e.g., National Instruments LabVIEW) [6] [7].
- Data Analysis and Calculation:
- Sensitivity: Plot the measured response voltages against the corresponding urea concentrations. The average sensitivity is the slope of the best-fit line through these data points, expressed in mV/(mg/dL) or mV/mM.
- Linearity: Calculate the coefficient of determination (R²) for the same calibration plot. An R² value of 0.999 indicates excellent linearity [6] [7] [8].
Advanced Drift Rate Reduction Protocol
Once fabrication quality is confirmed through sensitivity and linearity, the following protocol can be implemented to directly address the drift effect.
Workflow for Drift Rate Testing and Reduction
The diagram below illustrates the process for assessing and mitigating sensor drift using a dedicated calibration circuit.
Procedure for Drift Rate Measurement and Reduction
- Baseline Drift Measurement: Use the standard V-T measurement system to immerse a quality-controlled biosensor in a urea solution for 12 hours. Record the voltage change over time. The initial drift rate without compensation can be relatively high (e.g., ~1.6 mV/hr) [6] [7].
- Circuit Integration: Build and connect the New Calibration Circuit (NCC), which is composed of a non-inverting amplifier and a voltage calibrating circuit, to the same biosensor [6] [7].
- Compensated Drift Measurement: Repeat the 12-hour immersion test, now with the NCC active. The circuit will actively regulate and compensate for the slow voltage drift.
- Result Verification: Calculate the new drift rate. The published result is a reduction to 0.02 mV/hr, which corresponds to a 98.77% improvement [6] [7] [8]. This confirms the effectiveness of the NCC in mitigating the hydration layer effect.
In the specialized field of RuO₂ urea biosensor research, high sensitivity and excellent linearity are non-negotiable prerequisites that validate a successful fabrication process. These metrics directly reflect the quality of the transducer and the efficacy of the enzyme immobilization. When these quality controls are met, researchers can then confidently deploy advanced techniques, such as the New Calibration Circuit, to tackle the persistent challenge of signal drift. This two-pronged approach—rigorous fabrication quality control followed by targeted electronic compensation—provides a robust pathway to developing highly stable and reliable biosensors for clinical and research applications.
This guide provides targeted troubleshooting advice for researchers fabricating urea biosensors, with a specific focus on how proper urease immobilization techniques can mitigate the critical problem of sensor drift in RuO₂-based systems.
The Critical Link: Urease Immobilization and Drift Rate
A stable, well-immobilized urease layer is fundamental to reducing the drift effect—the unwanted change in sensor response over time during long-term measurement. This drift is often caused by the formation of a hydration layer on the sensing film's surface, which alters its electrical properties [7] [6] [8]. A robust immobilization protocol minimizes enzyme leaching and maintains a consistent enzymatic reaction, which is essential for signal stability. Research on RuO₂ urea biosensors has demonstrated that addressing these foundational fabrication issues can reduce the drift rate by as much as 98.77% [8].
Troubleshooting Guide & FAQs
Q1: My biosensor shows a continuously drifting baseline in buffer solution. What could be wrong?
- Possible Cause: Unstable urease immobilization, leading to enzyme leaching from the sensor surface. This creates a fluctuating reaction rate and an unstable potential at the sensing electrode.
- Solution:
- Verify the completeness of your cross-linking reaction. Ensure the glutaraldehyde solution is fresh and the reaction has been allotted sufficient time (e.g., 3 hours for activation and 24 hours for enzyme binding) [7] [20].
- Implement a rigorous washing protocol after immobilization to remove any physically adsorbed, non-covalently bound enzyme molecules that could desorb during operation [21].
Q2: The sensor's sensitivity is low and the response time is slow. How can I improve performance?
- Possible Cause: The immobilization matrix is too dense or thick, creating a diffusion barrier that prevents urea from quickly reaching the enzyme and reaction products (ammonium ions) from reaching the RuO₂ sensing film.
- Solution:
- Use a porous scaffold for immobilization. A porous silk fibroin (SF) membrane created by a salt-leaching method has been shown to provide a high surface area for enzyme loading while facilitating mass transport [20].
- Ensure the urease solution is concentrated enough during immobilization. A typical protocol uses a solution containing 1.6667 × 10⁻² g of urease for immobilization on a small electrode area [21].
Q3: My sensor readings are erratic and non-reproducible between fabrication batches.
- Possible Cause: Inconsistent manual deposition of the immobilization matrix or enzyme solution, leading to variations in the amount and activity of immobilized urease.
- Solution:
- Standardize the volume of enzyme solution deposited using a precision micropipette.
- Consider using screen-printing technology for applying the sensitive layer, which offers excellent reproducibility and control over film thickness [10].
- Employ a bifunctional covalent linker like 3,3′-dithiodipropionic acid di(N-hydroxysuccinimide ester) (DSP), which provides a more controlled and uniform method for anchoring urease to electrode surfaces compared to physical adsorption [21].
Experimental Protocols for Robust Immobilization
The following table summarizes key quantitative data from successful sensor fabrications, providing a benchmark for your experiments.
Table 1: Performance Metrics of Urea Biosensors
| Sensor Type / Immobilization Method | Average Sensitivity | Linearity | Drift Rate | Reference |
|---|---|---|---|---|
| RuO₂ Urea Biosensor / Glutaraldehyde Cross-linking | 1.860 mV/(mg/dL) | 0.999 | 0.02 mV/hr | [7] [8] |
| Ur-DSP/SPGE (Gold Electrode) / Covalent via DSP linker | Low detection limit of 5.0 µM | Excellent in real samples | Excellent temporal stability reported | [21] |
| Portable Sensor / Glutaraldehyde on Aminated SF Membrane | Linear in 0.1–20 mM range | Effective in flow conditions | N/A | [20] |
Protocol 1: Glutaraldehyde Cross-linking on RuO₂ Sensing Film
This method was used in conjunction with an RuO₂ film to achieve a 98.77% reduction in drift rate [7] [8].
- Surface Activation: Drop aminopropyltriethoxysilane (APTS) solution onto the RuO₂ sensing film at room temperature to functionalize the surface with amine groups.
- Cross-linker Application: Drop a 1% glutaraldehyde solution onto the sensor. This bifunctional molecule will react with the amine groups from APTS, creating an aldehyde-rich surface.
- Enzyme Immobilization: Keep the sensor still for 24 hours at room temperature to allow the glutaraldehyde to react fully.
- Urease Binding: Drop the urease solution onto the activated RuO₂ sensing film. The amine groups on the enzyme will covalently bind to the free aldehyde groups on the surface.
- Curing and Storage: Let the sensor sit to allow the covalent bonds to form completely, creating a stable urease membrane.
Protocol 2: Covalent Immobilization on Gold with DSP Linker
This protocol offers a robust alternative for gold electrodes or surfaces that can be modified with a gold layer [21].
- Linker Deposition: Deposit 10 µL of a DSP solution in DMSO (10 mg/mL) onto the gold working electrode via drop casting.
- Drying: Let the electrode dry at room temperature for 2 days. DSP forms a self-assembled monolayer via stable thiol-gold anchoring.
- Enzyme Coupling: Deposit 10 µL of a urease solution in phosphate buffer saline (PBS) onto the functionalized gold electrode.
- Reaction: Keep the electrode at 0°C for 24 hours to allow the formation of an amide bond between the –NH₂ group of the enzyme and the NHS ester group of the DSP linker.
- Washing: After each step, wash the electrode thoroughly with PBS to remove any excess, unbound DSP or enzyme.
The Scientist's Toolkit: Essential Research Reagents
Table 2: Key Reagents for Urease Immobilization and Sensor Fabrication
| Reagent/Material | Function in Fabrication | Typical Use-Case |
|---|---|---|
| Ruthenium Oxide (RuO₂) | Sensing film material; translates pH change from urea hydrolysis into a measurable potentiometric signal. | Primary transducer in RuO₂ urea biosensors [7] [6]. |
| Urease from Canavalia ensiformis | Enzyme that catalyzes the hydrolysis of urea into ammonia and carbon dioxide, initiating the detection event. | Biological recognition element in all urease-based urea biosensors [21] [20]. |
| Glutaraldehyde | Bifunctional cross-linker; forms covalent bonds between amine groups on the sensor surface and amine groups on the enzyme. | Standard for covalent immobilization on polymer matrices and surface-functionalized metal oxides [7] [20]. |
| Aminopropyltriethoxysilane (APTS) | Silane-based coupling agent; provides surface amine groups for subsequent glutaraldehyde cross-linking on oxide surfaces. | Used to functionalize the surface of RuO₂ and other metal oxides [7]. |
| 3,3′-dithiodipropionic acid di(N-hydroxysuccinimide ester) (DSP) | Bifunctional linker; anchors to gold via thiol group and covalently binds enzyme via NHS ester. | Provides a stable, ordered monolayer for covalent enzyme immobilization on gold electrodes [21]. |
| Polyethylene Terephthalate (PET) | Flexible, inert substrate for constructing the biosensor. | Common substrate for flexible and arrayed sensor designs [7] [6]. |
| Silk Fibroin (SF) Membrane | Porous, biocompatible scaffold for enzyme immobilization; enhances surface area and stability. | Used as a 3D membrane to host and protect the immobilized enzyme [20]. |
| Phosphate Buffer Saline (PBS) | Provides a stable, physiological pH environment for enzyme activity and electrochemical measurements. | Standard buffer for all washing, immobilization, and testing steps [21] [20]. |
Workflow: From Immobilization to Stable Signal
The diagram below outlines the logical workflow for proper urease immobilization and its direct role in achieving a stable sensor signal with low drift.
A technical support center for researchers developing RuO₂ urea biosensors.
Troubleshooting Guides
This section addresses common experimental challenges in reducing the drift rate of RuO₂ urea biosensors, providing step-by-step diagnostic and corrective procedures.
Guide 1: Addressing High Signal Drift in RuO₂ Urea Biosensors
Problem: The response voltage of the RuO₂ urea biosensor changes significantly over time during long-term measurement, indicating a high signal drift.
Background: The drift phenomenon is often caused by the formation of a hydration layer on the surface of the sensing film. Hydroxyl groups form on the film surface in solution, and hydrated ions diffuse to the sensing film, resulting in an electrical double layer capacitance that alters the surface potential over time [7] [6].
Investigation Steps:
- Verify Biosensor Fabrication: Confirm that the RuO₂ sensing film was fabricated correctly. The biosensor should first demonstrate high sensitivity (e.g., ~1.860 mV/(mg/dL)) and linearity (e.g., R²=0.999) in standard V-T measurements to rule out fabrication defects as the primary issue [7] [22].
- Check Measurement Environment: Ensure the biosensor is tested in a 30 mM phosphate buffer saline (PBS) solution with a pH of 7 to mimic physiological conditions and provide a stable testing environment [7] [6].
- Quantify the Drift Rate: Using a standard voltage-time (V-T) measurement system, immerse the sensor in a urea solution (e.g., within the 2.5–7.5 mM physiological range) and record the response voltage over an extended period (e.g., 12 hours) to establish the baseline drift rate [7].
Solution: Implement a New Calibration Circuit (NCC) designed to actively counteract the drift effect. The proposed NCC, based on a voltage regulation technique, is composed of a non-inverting amplifier and a voltage calibrating circuit. Its simple structure minimizes complexity while achieving a significant reduction in drift rate [7] [8].
Validation: After integrating the NCC, repeat the long-term measurement. Successful implementation should reduce the drift rate to a target value as low as 0.02 mV/hr, representing a reduction of over 98% compared to the system without the NCC [7] [8].
Guide 2: New Calibration Circuit (NCC) Not Functioning as Expected
Problem: After building the New Calibration Circuit (NCC), the desired reduction in drift rate is not observed, or the circuit output is unstable.
Background: The NCC's function relies on its simple design of a non-inverting amplifier and a voltage calibrating circuit. Incorrect component selection, poor connections, or interference from external noise can compromise its performance [7] [6].
Investigation Steps:
- Circuit Integrity Check:
- Visually inspect the solder joints and physical connections on the circuit board for any shorts or open circuits.
- Use a multimeter to verify that all passive components (resistors, capacitors) match the designed values.
- Confirm the correct orientation and placement of active components, such as the operational amplifiers.
- Power Supply Verification: Measure the power supply rails with an oscilloscope to ensure they are clean and stable, without significant noise or ripple that could affect the circuit's operation.
- Signal Path Testing:
- With the biosensor disconnected, inject a known, stable DC voltage signal into the circuit input.
- Measure the output at each stage (non-inverting amplifier and voltage calibrating circuit) to verify that the gain and calibration functions are operating as designed.
- Noise Assessment: The NCC is designed for simplicity and may not include advanced filtering. Compare the circuit's performance with a previously reported noise-canceling readout circuit that uses a Twin-T notch filter to cancel power line noise and a Sallen–Key low-pass filter to suppress high-frequency noise [7]. If noise is identified as a primary issue, consider incorporating these filtering techniques while balancing the goal of circuit simplicity.
Solution: Based on the investigation:
- If a faulty component is identified, replace it.
- If the power supply is noisy, add appropriate decoupling capacitors near the power pins of the integrated circuits.
- If external noise is corrupting the signal, consider integrating basic filter stages (e.g., a low-pass filter) into the existing NCC design without overly complicating the layout.
Frequently Asked Questions (FAQs)
Q1: What is the significance of reducing drift in urea biosensors? Accurate and stable long-term measurement of urea is critical for clinical diagnostics, particularly for monitoring kidney function. A high drift rate makes biosensor readings unreliable over time, limiting their practical application in medical settings and leading to potential misdiagnosis [19] [23].
Q2: Why was RuO₂ chosen as the sensing material in this context? Ruthenium oxide (RuO₂) is a transition metal oxide with several advantageous properties for biosensing, including high metallic conductivity, low resistivity, high thermal stability, and good diffusion barrier properties. These characteristics make it a suitable material for creating stable and sensitive working electrodes [7] [6].
Q3: How does the proposed New Calibration Circuit (NCC) maintain simplicity? The NCC is intentionally designed with a simple structure based on a voltage regulation technique. It is composed of only two main parts: a non-inverting amplifier and a voltage calibrating circuit. This avoids the complexity of more sophisticated digital signal processing or multi-stage filtering approaches, making it easier to implement and reproduce [7] [8].
Q4: Are there alternative materials to RuO₂ for urea biosensing? Yes, other metal oxides like nickel oxide (NiO) and titanium oxide (TiO₂) are also widely used. NiO offers strong chemical stability and fast electron transfer, while TiO₂ is non-toxic and has good electron transition properties. However, the drift effect is a challenge across many of these materials [7] [6].
Q5: What is the typical experimental workflow for testing a urea biosensor's performance? The process generally involves two main stages. First, a stable testing environment is set up to validate the biosensor's basic sensing characteristics (sensitivity, linearity) using a V-T measurement system. Second, the proposed circuit or solution (like the NCC) is integrated to verify its specific function in improving a parameter like the drift rate [7] [6].
Experimental Data & Protocols
The following table summarizes the key performance metrics of the RuO₂ urea biosensor, comparing measurements from the standard Voltage-Time (V-T) system and the New Calibration Circuit (NCC).
| Sensing Characteristic | Measurement by V-T System | Measurement by New Calibration Circuit (NCC) |
|---|---|---|
| Average Sensitivity | 1.860 mV/(mg/dL) [7] | Information not specified in search results |
| Linearity | 0.999 [7] | Information not specified in search results |
| Drift Rate | Information not specified in search results | 0.02 mV/hr [7] |
| Drift Rate Reduction | Baseline | 98.77% reduction [7] |
Detailed Experimental Protocol
This protocol outlines the key steps for fabricating the RuO₂ urea biosensor and testing the New Calibration Circuit (NCC), as derived from the cited research [7] [6].
Part A: Fabrication of the Flexible Arrayed RuO₂ Urea Biosensor
- Substrate Preparation: Use a flexible polyethylene terephthalate (PET) substrate as the base.
- Electrode Formation: Print arrayed silver wires onto the PET substrate using a screen-printing technique with silver paste. These form the working and reference electrodes.
- Sensing Film Deposition: Deposit the RuO₂ thin film onto the substrate over the electrode areas using a sputtering system to create the RuO₂ film window.
- Encapsulation: Apply an epoxy thermosetting polymer (e.g., JA643) via screen-printing to encapsulate the structure and create an insulation layer, leaving the sensing window exposed.
- Enzyme Immobilization:
- Drop aminopropyltriethoxysilane (APTS) solution onto the RuO₂ sensing film at room temperature.
- To enhance urease adsorption, drop a 1% glutaraldehyde solution onto the sensor and let it stand for 24 hours.
- Finally, drop the urease enzyme solution onto the treated RuO₂ sensing film to form the complete biosensor.
Part B: Experimental Setup for Drift Rate Measurement
- Solution Preparation: Prepare a 30 mM phosphate buffer saline (PBS) solution with a pH of 7.0 using deionized (D.I.) water. Urea solutions for testing should be prepared within the physiological range (2.5–7.5 mM) using this PBS buffer.
- System Configuration:
- For baseline measurement: Use the conventional V-T measurement system, which typically consists of an instrumentation amplifier (e.g., LT1167), a data acquisition (DAQ) device (e.g., National Instruments USB-6210), and program software (e.g., LabVIEW).
- For drift reduction testing: Integrate the proposed New Calibration Circuit (NCC) between the biosensor and the DAQ device.
- Measurement Procedure:
- Immerse the fabricated RuO₂ urea biosensor in the urea solution.
- Measure the response voltage continuously over a long period (e.g., 12 hours) using both the V-T system and the NCC.
- Record the voltage data at regular intervals for analysis.
Research Reagent Solutions
The table below lists key materials and reagents used in the fabrication and testing of the RuO₂ urea biosensor as described in the research.
| Material / Reagent | Function in the Experiment | Source / Example |
|---|---|---|
| Polyethylene Terephthalate (PET) | Flexible substrate for the biosensor | Zencatec Corporation [7] [6] |
| Ruthenium (Ru) Target | Source for sputtering RuO₂ sensing film | Ultimate Materials Technology Co., Ltd. [7] [6] |
| Silver Paste | Forms conductive working and reference electrodes | Advanced Electronic Material Inc. [7] [6] |
| Epoxy Polymer (JA643) | Insulation layer to encapsulate the sensor | Sil-More Industrial, Ltd. [7] [6] |
| Urease Enzyme | Biorecognition element that catalyzes urea hydrolysis | Sigma-Aldrich Corp. [7] [6] |
| Phosphate Buffer Saline (PBS) | Provides a stable, pH-neutral testing environment | Prepared from KH₂PO₄ & K₂HPO₄ [7] [6] |
Experimental Workflow and Circuit Integration
The following diagram visualizes the experimental workflow for testing the RuO₂ urea biosensor and integrating the calibration circuit.
Troubleshooting Guides and FAQs for RuO₂ Urea Biosensor Research
This technical support center provides practical guidance for researchers addressing noise, drift, and interference in Ruthenium Oxide (RuO₂) urea biosensor systems. The following FAQs and troubleshooting guides are framed within the core research objective of reducing the drift rate in RuO₂ urea biosensors.
Frequently Asked Questions (FAQs)
1. What is the primary cause of signal drift in RuO₂ urea biosensors? Signal drift during long-term measurement is primarily attributed to the formation of a hydration layer on the surface of the RuO₂ sensing film. When the sensor is immersed in a solution, hydroxyl groups form on the film's surface. Hydrated ions then diffuse to the sensing film, leading to the formation of this hydration layer, which alters the surface potential and causes the response voltage to change over time [7].
2. How can a simple electronic circuit reduce the drift effect? A New Calibration Circuit (NCC) based on a voltage regulation technique can be implemented. One study demonstrated that a circuit composed of a non-inverting amplifier and a voltage calibrating circuit successfully reduced the drift rate of an RuO₂ urea biosensor to 0.02 mV/hr, achieving a 98.77% reduction compared to a standard voltage-time (V-T) measurement system [7] [8].
3. Besides electronic solutions, how does material fabrication affect sensor stability? The sputtering conditions used to deposit RuO₂ thin films significantly impact their physical and electrochemical properties, which in turn influence stability. For instance, films sputtered under different conditions (e.g., DC vs. RF, substrate temperature) exhibit variations in surface morphology, density, and hardness, all of which can affect long-term signal stability and sensitivity [24].
4. What are the common sources of noise in electrochemical biosensors? Noise can be categorized into several types [25]:
- Electronic Noise: This includes Thermal (Johnson-Nyquist) noise from charge carrier motion and 1/f (flicker) noise from material imperfections and defects at electrode interfaces.
- Environmental Interference: External electromagnetic interference (EMI) from power lines or wireless devices can capacitively or inductively couple into the sensor system.
- Biological Cross-reactivity: In complex samples, non-specific binding of interfering species to the sensor surface can cause spurious signals.
Troubleshooting Guide: Common Issues and Solutions
| Issue | Possible Cause | Recommended Solution |
|---|---|---|
| High Signal Drift | Hydration layer formation on RuO₂ film [7] | Implement a New Calibration Circuit (NCC) for voltage regulation [7] [8]. |
| Unstable sensing film properties | Optimize RuO₂ sputtering parameters (e.g., temperature, power, cathode type) for denser, more stable films [24]. | |
| Low Sensitivity | Suboptimal electrode surface area | Explore composite materials (e.g., RuO₂ with nitrogen-doped biochar) to increase effective surface area and electron transfer [26]. |
| High Background Noise | Electromagnetic Interference (EMI) [25] | Use proper shielding for cables and the measurement cell. Employ a Twin-T notch filter to cancel power line noise [7]. |
| Electrode material imperfections [25] | Utilize carbon-based nanomaterials with high conductivity and fewer grain boundaries to reduce intrinsic flicker noise [25]. | |
| Reduced Signal Fidelity in Complex Samples | Biofouling or non-specific adsorption [25] | Apply antifouling coatings (e.g., polyethylene glycol) or use novel carbon nanomaterials with innate antifouling properties [25]. |
Experimental Protocols for Key Methodologies
Protocol 1: Fabrication of a Flexible Arrayed RuO₂ Urea Biosensor This protocol is essential for creating a reproducible and stable sensor platform, which is the foundation for mitigating drift [7].
- Substrate Preparation: Use a flexible polyethylene terephthalate (PET) substrate.
- Electrode Patterning: Print arrayed silver wires onto the PET substrate using screen-printing techniques to form the working and reference electrodes.
- Sensing Film Deposition: Deposit an RuO₂ thin film on the substrate over the electrode area using a sputtering system to create the RuO₂ film window.
- Encapsulation: Encapsulate the structure with an epoxy thermosetting polymer, leaving the sensing window exposed.
- Surface Functionalization: Drop aminopropyltriethoxysilane (APTS) solution onto the RuO₂ sensing film at room temperature.
- Enzyme Immobilization: To enhance urease adsorption, drop a 1% glutaraldehyde solution onto the sensor and let it sit for 24 hours. Finally, drop the urease enzyme onto the functionalized RuO₂ film to complete the biosensor.
Protocol 2: Measuring and Correcting Drift with a New Calibration Circuit (NCC) This protocol provides a direct method to quantify and mitigate the drift effect, a core thesis requirement [7].
- Sensor Setup: Immerse the fabricated RuO₂ urea biosensor in a urea solution (e.g., within the physiological range of 2.5–7.5 mM).
- Long-Term Measurement: Measure the response voltage continuously for an extended period (e.g., 12 hours) using a conventional Voltage-Time (V-T) measurement system to establish the baseline drift.
- Circuit Integration: Connect the biosensor to the New Calibration Circuit (NCC), which is based on a non-inverting amplifier and a voltage calibrating circuit.
- Data Comparison: Record the output from the NCC over the same duration and compare the drift rate (in mV/hr) with the data from the V-T system. The NCC should demonstrate a significant reduction in drift.
The table below consolidates key performance metrics from recent research to aid in benchmarking your sensor's performance.
| Performance Metric | Reported Value | Measurement Conditions / Notes |
|---|---|---|
| Average Sensitivity | 1.860 mV/(mg/dL) | Measured in urea solution with conventional V-T system [7]. |
| Linearity | 0.999 | Within the normal human body urea range (2.5–7.5 mM) [7]. |
| Drift Rate (with NCC) | 0.02 mV/hr | Achieved a 98.77% reduction from the uncalibrated drift rate [7] [8]. |
| pH Sensitivity (of RuO₂ film) | 56.35 - 57.37 mV/pH | Varies with sputtering conditions (RF from metallic cathode vs. DC from oxide cathode) [24]. |
Research Reagent Solutions
The following table details key materials used in the fabrication and testing of RuO₂ urea biosensors.
| Research Reagent | Function in the Experiment |
|---|---|
| Ruthenium Oxide (RuO₂) | The core sensing film material; a transition metal oxide with high metallic conductivity, low resistivity, and excellent electrochemical properties for biosensing [7] [24]. |
| Polyethylene Terephthalate (PET) | A flexible, transparent substrate material for creating flexible arrayed biosensors [7]. |
| Urease Enzyme | The biorecognition element that specifically catalyzes the hydrolysis of urea, producing changes in local ion concentration detected by the RuO₂ film [7]. |
| Aminopropyltriethoxysilane (APTS) | A silane coupling agent used to functionalize the RuO₂ surface, enhancing the adhesion and stability of subsequently immobilized layers [7]. |
| Glutaraldehyde | A crosslinking agent used to create strong covalent bonds, immobilizing the urease enzyme onto the APTS-functionalized sensor surface [7]. |
| Phosphate Buffer Saline (PBS) | A standard buffer solution used to maintain a stable pH (e.g., 7.0) during electrochemical testing, simulating physiological conditions [27] [7]. |
System Integration Workflow for Drift Mitigation
The diagram below outlines a systematic workflow for diagnosing and addressing drift and noise in a biosensor system, integrating both material and electronic solutions.
RuO₂ Film Fabrication and Sensor Assembly
This diagram illustrates the key material and fabrication choices in creating a stable RuO₂ sensing electrode, which is the first line of defense against drift and noise.
Benchmarking Drift Reduction: NCC Performance vs. Conventional Systems
A technical support center for biosensor researchers
This resource provides targeted support for researchers working with RuO₂ urea biosensors, with a specific focus on methodologies to understand and counteract the drift effect. The following guides and protocols are framed within the broader thesis that a combined approach—integrating specialized materials with a novel calibration circuit—is fundamental to achieving signal stability.
Troubleshooting the Drift Effect in RuO₂ Urea Biosensors
Q1: What is the "drift effect" and why is it a critical problem in long-term urea biosensor measurements?
The drift effect refers to the undesirable change in a biosensor's response voltage over time when it is immersed in a solution, even if the analyte concentration remains constant. This phenomenon is distinct from a change in signal due to the actual urea concentration and leads to inaccurate readings. It is critical because it compromises the reliability and accuracy of long-term measurements, which are essential for continuous monitoring in clinical and research settings. The primary cause is the formation of a hydration layer on the surface of the sensing film. In solution, hydroxyl groups form on the film's surface, and hydrated ions diffuse to it, resulting in an electrical double-layer capacitance that causes the surface potential to shift over time [7] [6].
Q2: Our research group has fabricated an RuO₂ urea biosensor. What are the first steps to diagnose a potential drift issue?
Follow this diagnostic workflow to isolate the cause of signal instability. The process helps determine if the issue originates from the sensor's inherent properties or from the external measurement system.
Q3: We have confirmed that the drift is inherent to our sensor. How can we actively reduce it?
The most effective solution documented is the implementation of a New Calibration Circuit (NCC). The NCC is designed specifically to counteract the drift effect through voltage regulation. Its key advantage is a simple structure, composed primarily of a non-inverting amplifier and a voltage calibrating circuit [7] [8]. Experimental results demonstrate that this circuit can reduce the drift rate of an RuO₂ urea biosensor to 0.02 mV/hr, achieving a 98.77% reduction compared to the drift rate measured with a conventional Voltage-Time (V-T) system [7] [6] [8].
Experimental Protocols for Validation
To validate the performance of any drift-reduction method, follow this core experimental protocol, which is adapted from the foundational work on the NCC.
Protocol: Quantifying Drift Rate with and without the NCC
Objective: To fabricate a stable RuO₂ urea biosensor and quantitatively measure the reduction in drift rate achieved by integrating the New Calibration Circuit.
Part 1: Fabrication of the Flexible Arrayed RuO₂ Urea Biosensor
This process outlines the creation of the core sensing element [7] [6].
- Substrate Preparation: Use a flexible polyethylene terephthalate (PET) substrate.
- Electrode Formation: Print arrayed silver wires onto the PET substrate using a screen-printing technique with silver paste. These form the working and reference electrodes.
- Sensing Film Deposition: Deposit the RuO₂ film onto the substrate over the electrodes using a sputtering system to create the sensing film window.
- Encapsulation: Apply an epoxy thermosetting polymer (e.g., JA643) via screen-printing to encapsulate the structure and define the active sensing area.
- Enzyme Immobilization:
- Drop aminopropyltriethoxysilane (APTS) solution onto the RuO₂ sensing film and let it sit at room temperature.
- To enhance urease adsorption, drop a 1% glutaraldehyde solution onto the sensor and let it stand for 24 hours.
- Finally, drop the urease solution onto the prepared RuO₂ sensing film to complete the biosensor.
Part 2: Drift Rate Measurement and NCC Validation
This section details the critical testing and validation phases [7] [6].
Baseline Setup (V-T System):
- Use a conventional Voltage-Time (V–T) measurement system, which typically consists of an LT1167 instrumentation amplifier, a data acquisition device (e.g., NI USB-6210), and software like LabVIEW.
- Immerse the fabricated biosensor in a urea solution with a concentration within the normal human body range (e.g., 2.5–7.5 mM) at a neutral pH of 7.0 (using a phosphate buffer saline solution).
- Measure the response voltage continuously for a prolonged period (e.g., 12 hours).
- Calculate the baseline drift rate (in mV/hr) from this data.
Intervention with NCC:
- Integrate the New Calibration Circuit (NCC) into your measurement setup. The NCC, comprising a non-inverting amplifier and voltage calibrating circuit, connects to the biosensor.
- With the NCC in circuit, repeat the continuous voltage measurement under the same conditions (same urea solution, same duration).
Data Analysis and Success Metrics:
- Calculate the new drift rate from the data collected with the NCC.
- Compare the drift rates with and without the NCC to quantify the improvement.
- A successful result is indicated by a drastic reduction in the drift rate, closely mirroring the documented result of 0.02 mV/hr, which equates to a 98.77% reduction [7] [8].
The following table summarizes the key performance data from the foundational experiment, providing a benchmark for your research.
Table 1: Performance Comparison of RuO₂ Urea Biosensor Measurement Systems
| Measurement System | Average Sensitivity (mV/(mg/dL)) | Linearity (R²) | Drift Rate (mV/hr) | Drift Reduction |
|---|---|---|---|---|
| V-T System (Baseline) | 1.860 | 0.999 | 1.59 (calculated from results) | Baseline |
| With NCC | Not explicitly stated | Not explicitly stated | 0.02 | 98.77% |
The Scientist's Toolkit: Research Reagent Solutions
This table lists the essential materials and their functions as used in the referenced RuO₂ urea biosensor research.
Table 2: Key Materials and Equipment for RuO₂ Urea Biosensor Fabrication and Testing
| Item | Function / Role | Specification / Source Example |
|---|---|---|
| PET Substrate | Flexible base for the biosensor array. | Zencatec Corporation (Taiwan) [7] |
| Ruthenium (Ru) Target | Source for sputtering to create the RuO₂ sensing film. | 99.95% purity [7] |
| Silver Paste | Forms the conductive working and reference electrodes. | Screen-printed into wires [7] |
| Epoxy Polymer | Insulating layer to encapsulate and define the sensor window. | e.g., Product JA643 [7] |
| Urease | Biological enzyme that catalyzes the hydrolysis of urea. | Immobilized on the RuO₂ film [7] |
| Phosphate Buffered Saline (PBS) | Provides a stable, neutral pH (7.0) environment for testing. | 30 mM concentration [7] |
| V-T Measurement System | Conventional system for measuring voltage-time characteristics. | Includes LT1167 amplifier, NI DAQ, LabVIEW [7] |
| New Calibration Circuit (NCC) | Custom circuit to actively counteract and reduce drift. | Composed of a non-inverting amplifier and voltage calibrator [7] [8] |
Quantitative Performance Comparison
The table below summarizes the key performance metrics of the New Calibration Circuit (NCC) compared to the standard Voltage-Time (V–T) measurement system for RuO₂ urea biosensors.
| Performance Characteristic | Standard V–T System | New Calibration Circuit (NCC) | Improvement |
|---|---|---|---|
| Drift Rate | Information Missing | 0.02 mV/hr [7] [6] [8] | 98.77% Reduction [7] [6] |
| Average Sensitivity | Information Missing | 1.860 mV/(mg/dL) [7] [6] | Not Applicable |
| Linearity | Information Missing | 0.999 [7] [6] | Not Applicable |
| Circuit Architecture | Instrumentation Amplifier (LT1167), Data Acquisition (DAQ) Device, LabVIEW Software [7] [6] | Non-inverting Amplifier + Voltage Calibrating Circuit [7] [6] | Simpler Structure [7] [6] |
Frequently Asked Questions (FAQs) & Troubleshooting
Q1: What is the "drift effect" in a RuO₂ urea biosensor and why is it a problem?
- A: The drift effect is the unwanted change in a biosensor's response voltage over time during long-term measurement. It is primarily caused by the formation of a hydration layer on the surface of the sensing film. This layer alters the electrical double layer capacitance, leading to unstable readings and reducing the reliability of the sensor for continuous monitoring [7] [6]. The NCC specifically addresses this issue.
Q2: My RuO₂ biosensor shows unstable readings. What are the first things I should check?
- A: Follow this troubleshooting guide:
- Verify Sensor Fabrication: Ensure the immobilization process of the urease enzyme on the RuO₂ sensing film was correctly performed. The procedure involves dropping APTS and glutaraldehyde solutions and allowing sufficient time (e.g., 24 hours) for the surface to stabilize [7] [6].
- Inspect the Measurement Environment: Confirm that the biosensor is immersed in a suitable phosphate buffer saline (PBS) solution with a pH of 7, and that the temperature is controlled to avoid fluctuations [7] [6].
- Check Circuit Connections: If using the NCC, ensure all circuit connections are secure and that the non-inverting amplifier and voltage calibrating circuit are functioning as intended [7] [6].
Q3: How does the New Calibration Circuit (NCC) achieve such a significant reduction in drift?
- A: The NCC employs a voltage regulation technique. Its simple structure, composed of a non-inverting amplifier and a voltage calibrating circuit, actively works to counteract the gradual change in voltage caused by the hydration layer on the sensor's surface, thereby stabilizing the output signal [7] [6].
Detailed Experimental Protocols
Fabrication of the Flexible Arrayed RuO₂ Urea Biosensor
The following workflow details the sensor fabrication process used to generate the performance data.
Title: RuO₂ Biosensor Fabrication Workflow
Materials Required:
- Substrate: Flexible Polyethylene Terephthalate (PET) [7] [6].
- Electrodes: Silver paste, screen-printed to form arrayed wires [7] [6].
- Sensing Film: Ruthenium (Ru, 99.95% purity) deposited via sputtering system to form RuO₂ [7] [6].
- Immobilization Matrix: Aminopropyltriethoxysilane (APTS) solution and 1% glutaraldehyde solution [7] [6].
- Enzyme: Urease [7] [6].
- Encapsulation: Epoxy thermosetting polymer [7] [6].
Protocol for Drift Rate Measurement and NCC Validation
This protocol describes how to benchmark the drift performance of your biosensor using the NCC versus a standard system.
Step 1: Solution Preparation
- Prepare a 30 mM Phosphate Buffer Saline (PBS) solution with a pH of 7.0 using KH₂PO₄ and K₂HPO₄ powders and deionized (D.I.) water (18.4 MΩ cm⁻¹) [7] [6].
- Prepare urea solutions within the normal human body concentration range (2.5–7.5 mM) using the PBS as a base [7] [6].
Step 2: Standard V-T System Baseline Measurement
- Set up the V-T measurement system, which typically includes an instrumentation amplifier (e.g., LT1167), a Data Acquisition (DAQ) device (e.g., NI USB-6210), and software (e.g., LabVIEW) [7] [6].
- Immerse the fabricated RuO₂ urea biosensor in the urea solution.
- Measure the response voltage continuously for 12 hours using this standard system to establish the baseline drift rate without calibration [7] [6].
Step 3: NCC Integration and Measurement
- Connect the biosensor to the New Calibration Circuit (NCC), which is composed of a non-inverting amplifier and a voltage calibrating circuit [7] [6].
- With the biosensor still immersed in the urea solution, measure the response voltage again for 12 hours using the NCC's output [7] [6].
Step 4: Data Analysis
- Plot the voltage-vs-time data from both measurements.
- Calculate the drift rate (mV/hr) for each by determining the slope of the voltage change over the stable measurement period. The NCC should demonstrate a drastically reduced slope, achieving a drift rate as low as 0.02 mV/hr [7] [6].
The Scientist's Toolkit: Essential Research Reagents & Materials
The table below lists key materials used in the fabrication and testing of the RuO₂ urea biosensor featured in the benchmark.
| Material / Reagent | Function / Application | Specifications / Source Examples |
|---|---|---|
| Polyethylene Terephthalate (PET) | Flexible substrate for the biosensor [7] [6]. | Purchased from Zencatec Corporation (Taiwan) [7] [6]. |
| Ruthenium (Ru) Target | Forms the RuO₂ sensing film via sputtering [7] [6]. | 99.95% purity (Ultimate Materials Technology Co., Taiwan) [7] [6]. |
| Silver Paste | Forms conductive working and reference electrodes [7] [6]. | Screen-printed onto PET substrate [7] [6]. |
| Urease Enzyme | Biological recognition element that catalyzes urea hydrolysis [7] [6]. | Sourced from Sigma-Aldrich [7] [6]. |
| APTS & Glutaraldehyde | Chemicals for enzyme immobilization on the RuO₂ surface [7] [6]. | Enhances urease adsorption and stability [7] [6]. |
| Phosphate Buffer Saline (PBS) | Measurement buffer to maintain stable pH during testing [7] [6]. | 30 mM, pH 7.0 [7] [6]. |
| Epoxy Polymer | Insulation layer to encapsulate and protect the sensor [7] [6]. | Product JA643 (Sil-More Industrial, Ltd.) [7] [6]. |
Frequently Asked Questions (FAQs)
Q1: What is the "drift effect" in RuO₂ urea biosensors and why is it a critical issue for long-term measurements?
The drift effect refers to the undesirable change in a biosensor's response voltage over time during long-term measurement. This phenomenon is critical because it leads to unstable and unreliable readouts, making biosensor data unacceptable for clinical or research applications. The primary cause of drift is the formation of a hydration layer on the surface of the sensing film. When the sensor is immersed in a solution, hydroxyl groups form on the film's surface. Hydrated ions, created by the coulombic attraction between water molecules and ions, then diffuse toward the sensing film, resulting in the formation of this hydration layer. The electrical double layer capacitance formed by this hydration layer alters the surface potential of the film, causing the output signal to drift over time [6] [7].
Q2: How does the performance of the New Calibration Circuit (NCC) compare to a noise-canceling readout circuit?
While both circuits aim to improve biosensor performance, they target different non-ideal effects and thus exhibit distinct performance characteristics, as summarized in the table below. The New Calibration Circuit (NCC) is specifically engineered to combat the long-term drift effect and excels in this area, achieving a massive reduction in drift rate. In contrast, the noise-canceling readout circuit described in Kuo's work is primarily designed to suppress electrical interference. It effectively removes power line noise and high-frequency noise but does not address the physical-chemical origins of sensor drift. Furthermore, the RuO₂ biosensor used with the NCC demonstrated superior sensitivity and linearity compared to the one assessed with the noise-canceling circuit [6] [7].
Table 1: Performance Comparison between the New Calibration Circuit and a Noise-Canceling Readout Circuit
| Feature | New Calibration Circuit (NCC) | Noise-Canceling Readout Circuit [6] |
|---|---|---|
| Primary Function | Voltage regulation to counteract drift | Power line and high-frequency noise cancellation |
| Core Components | Non-inverting amplifier, voltage calibrating circuit | Twin-T notch filter, Sallen-Key low-pass filter |
| Targeted Problem | Long-term signal drift from hydration layer | Electrical environmental noise |
| Drift Rate Reduction | 98.77% (to 0.02 mV/hr) [6] [7] [8] | Not specifically addressed |
| Average Sensitivity | 1.860 mV/(mg/dL) | Achieved lower sensitivity than the NCC setup [6] |
| Linearity | 0.999 | Achieved lower linearity than the NCC setup [6] |
Q3: Our research group is experiencing a high drift rate despite using a standard voltage-time (V-T) measurement system. What is the first thing we should check?
The first step is to verify the integrity and fabrication quality of your RuO₂ biosensor itself. The drift calibration circuit can only function effectively with a well-fabricated sensor. Before implementing the NCC, ensure your biosensor demonstrates excellent fundamental sensing characteristics. The RuO₂ biosensor used in the cited study showed an average sensitivity of 1.860 mV/(mg/dL) and a linearity of 0.999 within the human physiological urea concentration range (2.5–7.5 mM). A poorly fabricated sensor with low sensitivity or linearity will inherently be more prone to instability and drift, which no readout circuit can fully correct [6] [7].
Troubleshooting Guide: Addressing Common Problems with Drift Performance
Problem: Inconsistent Drift Reduction with NCC
Symptoms: The observed drift rate after applying the New Calibration Circuit is higher than the documented 0.02 mV/hr, or the reduction percentage is inconsistent across different sensor batches.
Possible Causes and Solutions:
- Cause 1: Improly Adjusted Voltage Regulation.
- Solution: The voltage regulation technique in the NCC needs precise adjustment. Re-check the calibration procedure for the non-inverting amplifier and voltage calibrating circuit. Ensure that the reference voltages are set correctly for your specific experimental setup and buffer solution.
- Cause 2: Degradation of the Urease Immobilization Layer.
- Solution: The stability of the enzyme layer is crucial. Verify your immobilization protocol. The successful fabrication process used 1% glutaraldehyde solution to enhance urease adsorption on the RuO₂ film, and the sensor was kept still for 24 hours to ensure proper binding. Inconsistent immobilization can lead to enzyme leaching, which destabilizes the sensor's response and exacerbates drift [6] [7].
- Cause 3: Variations in the RuO₂ Sensing Film.
- Solution: The properties of the sputtered RuO₂ film are critical. Reproducibility issues in the deposition process (e.g., sputtering parameters, target purity of 99.95%) can lead to variations in film morphology and electrical properties, affecting drift. Consistency in the fabrication of the sensing film window is paramount [6].
Problem: Different Drift Performance Between Sensor Replicates
Symptoms: When testing multiple sensors from the same fabrication batch, the drift rates vary significantly.
Possible Causes and Solutions:
- Cause 1: Inconsistent Encapsulation.
- Solution: Inspect the insulation layer formed by the screen-printed epoxy thermosetting polymer. Inadequate or uneven encapsulation can expose parts of the electrode to the solution, leading to unpredictable parasitic currents and uneven hydration layer formation, which causes variable drift [6] [7].
- Cause 2: Contamination of Test Solution.
Experimental Protocol: Validating Drift Performance of a Readout Circuit
This protocol provides a detailed methodology for comparing the drift performance of different readout circuits, such as the New Calibration Circuit (NCC) versus a conventional V-T measurement system, using a fabricated RuO₂ urea biosensor.
Objective: To quantitatively measure and compare the drift rate of an RuO₂ urea biosensor when using a standard V-T measurement system and a dedicated New Calibration Circuit (NCC).
Research Reagent Solutions & Essential Materials
Table 2: Key Research Reagents and Materials for RuO₂ Biosensor Fabrication and Testing
| Item | Function / Role | Example / Specification |
|---|---|---|
| Polyethylene Terephthalate (PET) | Flexible substrate for the biosensor | Purchased from Zencatec Corporation [6] |
| Ruthenium (Ru) Target | Forms the RuO₂ sensing film via sputtering | 99.95% purity from Ultimate Materials Technology Co. [6] |
| Silver Paste | Forms conductive working and reference electrodes | Screen-printed into arrayed wires [6] |
| Epoxy Polymer | Insulation layer to encapsulate the sensor | Product JA643, Sil-More Industrial Ltd. [6] |
| Urease Enzyme | Biorecognition element that catalyzes urea hydrolysis | Purchased from Sigma-Aldrich Corp. [6] |
| Urea | Target analyte for calibration and testing | Purchased from J.T. Baker Corp. [6] |
| Phosphate Buffered Saline (PBS) | Stable, pH-neutral testing environment | 30 mM, pH 7.0, made from KH₂PO₄ & K₂HPO₄ [6] |
| Deionized Water | Preparation of all aqueous solutions | Resistivity ~18.4 MΩ cm⁻¹ [6] |
| Glutaraldehyde Solution | Crosslinking agent for urease immobilization | 1% solution [6] |
Step-by-Step Methodology:
Biosensor Fabrication: Fabricate the flexible arrayed RuO₂ urea biosensor as described in the literature [6] [7].
- Print silver paste on a PET substrate to form electrodes.
- Deposit RuO₂ film via sputtering to create the sensing window.
- Encapsulate the structure with an epoxy layer, leaving the sensing area exposed.
- Functionalize with urease using APTS and 1% glutaraldehyde as cross-linkers.
Baseline Drift Measurement (V-T System):
- Setup: Connect the biosensor to the conventional V-T measurement system, which includes an LT1167 instrumentation amplifier, a National Instruments USB-6210 DAQ device, and LabVIEW software [6].
- Test: Immerse the sensor in a 30 mM PBS solution (pH 7.0) at room temperature. Do not add urea.
- Data Acquisition: Record the response voltage continuously for 12 hours.
- Calculation: The drift rate is calculated as the slope of the voltage-time plot over this 12-hour period in a urea-free solution.
Drift Measurement with NCC:
- Setup: Disconnect the biosensor from the V-T system and connect it to the input of the proposed New Calibration Circuit. The output of the NCC is then connected to the DAQ device.
- Test: Keep the sensor in the same PBS solution under identical conditions.
- Data Acquisition: Again, record the output voltage from the NCC for 12 hours.
Data Analysis and Comparison:
- For both datasets, plot voltage versus time.
- Calculate the drift rate (in mV/hr) for each system by performing a linear regression on the data.
- Compute the percentage reduction in drift rate achieved by the NCC using the formula:
- Reduction (%) = [(DriftrateVT - DriftrateNCC) / DriftrateVT] * 100
The typical result from this protocol, as published, should show a drastic reduction in drift rate, for example, from a baseline of several mV/hr down to 0.02 mV/hr with the NCC, representing a 98.77% improvement [6] [7] [8].
Workflow and Relationship Diagrams
The following diagram illustrates the logical workflow for diagnosing and addressing drift issues in RuO₂ urea biosensor research, integrating the troubleshooting steps and experimental validation outlined in this guide.
Technical Support & Troubleshooting Hub
This technical support center provides essential guidance for researchers working with Ruthenium Oxide (RuO₂) urea biosensors. The content is framed within the core research objective of reducing the sensor drift rate, a critical factor for achieving reliable, long-term measurements.
Frequently Asked Questions (FAQs)
FAQ 1: What is the drift effect and why is it a significant problem in my RuO₂ urea biosensor measurements?
The drift effect refers to the unwanted change in the sensor's response voltage over time during long-term measurement. This phenomenon is primarily caused by the formation of a hydration layer on the surface of the sensing film. When the sensor is immersed in a solution, hydroxyl groups form on the film's surface. Water molecules and ions then form hydrated ions through coulombic attraction, which diffuse to the sensing film and result in the formation of this hydration layer. The resulting surface potential, attributed to the electrical double-layer capacitance formed by the hydration layer, causes the signal to drift, making accurate, stable readings difficult [6].
FAQ 2: My biosensor shows unstable readout. How can I confirm if the problem is drift and not something else?
To systematically diagnose drift, follow this troubleshooting guide:
- Step 1: Check the Immersion Time. Drift is time-dependent. If the signal instability increases the longer the sensor is in the solution, drift is a likely cause.
- Step 2: Inspect the Sensing Film. Examine the RuO₂ sensing film for any physical damage or contamination that could accelerate hydration layer formation.
- Step 3: Verify Solution Chemistry. Ensure the pH and ionic strength of your phosphate buffer saline (PBS) solution are correct (e.g., pH 7), as these can influence the drift rate.
- Step 4: Utilize a Calibration Circuit. The most effective way to confirm and mitigate drift is to test your sensor with a dedicated calibration circuit, such as the New Calibration Circuit (NCC) mentioned in research, which can reduce the drift rate by over 98% [6].
FAQ 3: Are there specific material choices that can help minimize the drift effect in my biosensor?
Yes, the choice of materials is crucial. RuO₂ is selected as a sensing film specifically for its advantageous properties, including low resistivity, high thermal stability, and good diffusion barrier properties. These characteristics contribute to its robustness and can help improve signal stability. Furthermore, to protect the electrode from fouling in complex media like food samples, modification with a protective Nafion membrane has been shown to improve performance without significantly altering electrochemical characteristics. This membrane can act as a barrier, potentially slowing processes that lead to drift [28].
FAQ 4: My sensor's sensitivity is lower than the theoretical Nernstian value. What could be wrong?
Sub-Nernstian sensitivity can point to several issues:
- Fabrication Parameters: For screen-printed electrodes, the ratio of RuO₂ to other oxides (e.g., CuO) and the sintering temperature are critical. Deviations from optimal parameters can drastically reduce sensitivity. Ensure your fabrication protocol is followed precisely [28].
- Urease Immobilization: The process of immobilizing the urease enzyme onto the RuO₂ sensing film is delicate. Incomplete or unstable immobilization will result in poor urea conversion and a weak signal. Revisit your immobilization procedure, ensuring the use of APTS and glutaraldehyde as cross-linkers [6].
- Electrode Damage: Physical damage to the sensitive film or the silver wire electrodes will impair performance.
Performance Data & Experimental Protocols
This section provides a quantitative summary of RuO₂ biosensor performance and detailed methods for key experiments.
Table 1: Performance Characteristics of the RuO₂ Urea Biosensor and Calibration Circuit
This table summarizes the key quantitative data for sensor validation, as reported in the literature [6].
| Characteristic | Value Measured by V-T System | Value with New Calibration Circuit (NCC) |
|---|---|---|
| Average Sensitivity | 1.860 mV/(mg/dL) | Not Specified (Circuit for drift) |
| Linearity | 0.999 | Not Specified (Circuit for drift) |
| Drift Rate | Not Specified (Baseline) | 0.02 mV/hr (98.77% reduction) |
Experimental Protocol 1: Fabrication of a Flexible Arrayed RuO₂ Urea Biosensor
This protocol details the creation of the biosensor itself [6].
- Substrate Preparation: Begin with a flexible polyethylene terephthalate (PET) substrate.
- Electrode Formation: Print arrayed silver wires (using silver paste) onto the PET substrate via screen printing to form the working and reference electrodes.
- Sensing Film Deposition: Deposit the RuO₂ film on the PET substrate over the electrode areas using a sputtering system to create the RuO₂ film window.
- Encapsulation: Encapsulate the structure with an epoxy thermosetting polymer, leaving the sensing window exposed.
- Surface Functionalization: Drop aminopropyltriethoxysilane (APTS) solution onto the RuO₂ sensing film at room temperature.
- Cross-linking: To enhance urease adsorption, drop a 1% glutaraldehyde solution onto the sensor and let it sit for 24 hours.
- Enzyme Immobilization: Finally, drop the urease enzyme onto the functionalized RuO₂ sensing film to complete the biosensor.
Experimental Protocol 2: Validating Drift Rate Reduction Using the New Calibration Circuit (NCC)
This protocol describes how to test the effectiveness of the drift reduction circuit [6].
- Solution Preparation: Prepare a urea solution within the normal human body concentration range (2.5–7.5 mM) using a 30 mM Phosphate Buffer Saline (PBS) at pH 7.
- Sensor Immersion: Immerse the fabricated RuO₂ urea biosensor in the urea solution.
- Long-Term Measurement: Conduct a continuous measurement over an extended period (e.g., 12 hours).
- Data Acquisition: Measure the response voltage using two systems in parallel:
- A conventional Voltage-Time (V-T) measurement system.
- The proposed New Calibration Circuit (NCC), which is based on a voltage regulation technique and comprises a non-inverting amplifier and a voltage calibrating circuit.
- Data Analysis: Calculate the drift rate (mV/hour) from the data collected by both systems. Compare the results to quantify the percentage reduction in drift achieved by the NCC.
The Scientist's Toolkit: Essential Research Reagents & Materials
Table 2: Key Reagents and Materials for RuO₂ Urea Biosensor Research
This table lists critical materials and their functions for fabricating and testing RuO₂ urea biosensors [6].
| Item | Function / Role in Research |
|---|---|
| Ruthenium (Ru) Target (99.95% purity) | Sputtered to form the key RuO₂ sensing film on the working electrode. |
| Polyethylene Terephthalate (PET) Substrate | Provides a flexible, robust base for constructing the biosensor. |
| Silver Paste | Used to form the conductive wires (electrodes) via screen printing. |
| Epoxy Thermosetting Polymer | Encapsulates the sensor, providing insulation and structural integrity. |
| Urease Enzyme | The biological recognition element that catalyzes the hydrolysis of urea. |
| Urea | The target analyte for calibration and testing. |
| Phosphate Buffer Saline (PBS) | Provides a stable, physiologically relevant pH environment for testing. |
| Aminopropyltriethoxysilane (APTS) | Used to functionalize the RuO₂ surface for enzyme immobilization. |
| Glutaraldehyde (1% solution) | Acts as a cross-linker to bind the urease enzyme firmly to the sensing film. |
| Nafion Membrane | A protective coating used to improve sensor performance in complex media (e.g., food samples) by reducing fouling [28]. |
Experimental Workflow: From Fabrication to Drift Validation
The following diagram illustrates the logical sequence and relationships between the key stages of biosensor development and validation, as detailed in the protocols above.
Conclusion
The drift effect in RuO₂ urea biosensors, while a significant challenge, can be effectively mitigated through a combined approach of robust sensor fabrication and innovative circuit design. The implementation of a Novel Calibration Circuit (NCC) presents a compelling solution, dramatically reducing drift by 98.77% to a minimal 0.02 mV/hr and thereby ensuring measurement stability crucial for clinical applications. This synergy between material science and electronic engineering marks a critical advancement. Future directions should focus on the miniaturization of this technology for point-of-care devices, integration with microfluidic systems for automated sampling, and exploration of next-generation nanomaterials to further enhance long-term stability. Success in this area will significantly accelerate the development of reliable, embeddable biosensors for non-invasive, continuous health monitoring, transforming patient diagnostics and disease management.
References
- 1. 柔性压力 传 的原理及应用 感 器 [https://wulixb.iphy.ac.cn/cn/article/doi/10.7498/aps.69.20200987]
- 2. 质子交换膜水电解中用于高效酸性析氧反应的二氧化钌 ... [https://chem.nankai.edu.cn/2025/0829/c24075a576515/page.htm]
- 3. 多步骤筛选指导核壳结构RuO₂@TaOₓ纳米棒电催化剂 [https://www.chemhui.com/angew-chem-%EF%BC%9A%E5%A4%9A%E6%AD%A5%E9%AA%A4%E7%AD%9B%E9%80%89%E6%8C%87%E5%AF%BC%E6%A0%B8%E5%A3%B3%E7%BB%93%E6%9E%84ruo%E2%82%82tao%E2%82%93%E7%BA%B3%E7%B1%B3%E6%A3%92%E7%94%B5%E5%82%AC%E5%8C%96/]
- 4. 我室罗维/李小鹏团队Nat. Commun.丨原子镓触发氧自由基时空 ... [https://sklfpm.dhu.edu.cn/2025/0430/c11777a361296/pagem.htm]
- 5. 计算并补偿嵌入电阻的PCB 中的工艺偏差 - MININTEL [https://www.minintelpcb.com/zh/news/embedded-resistors/]
- 6. A New Calibration Circuit Design to Reduce Drift Effect ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC6832896/]
- 7. A New Calibration Circuit Design to Reduce Drift Effect of ... [https://www.mdpi.com/1424-8220/19/20/4558]
- 8. A New Calibration Circuit Design to Reduce Drift Effect ... [https://pubmed.ncbi.nlm.nih.gov/31635177/]
- 9. The Hysteresis Reduction Approach for Urea Biosensor Modified ... [https://www.semanticscholar.org/paper/a6f6558f8c83db919522bb81b06f52397e9f1540]
- 10. pH Sensing Properties of Co3O4-RuO2-Based Electrodes and ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC11859339/]
- 11. Urea Biosensor Based on a CO2 Microsensor - PMC [https://pmc.ncbi.nlm.nih.gov/articles/PMC7594316/]
- 12. Independent prognostic importance of blood urea nitrogen ... [https://www.nature.com/articles/s41598-025-16383-w]
- 13. Sensors | Free Full-Text | A New Calibration Design to Reduce... Circuit [https://www.mdpi.com/1424-8220/19/20/4558/xml]
- 14. Non-inverting Operational Amplifier Configuration [https://www.electronics-tutorials.ws/opamp/opamp_3.html]
- 15. based on immobilization of urease on ZnO... Urea biosensor [https://www.orientjchem.org/vol31no2/urea-biosensor-based-on-immobilization-of-urease-on-zno-nanoparticles/]
- 16. A Review of Manufacturing Methods for Flexible Devices ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC10526154/]
- 17. Review of Materials and Fabrication Methods for Flexible ... [https://www.mdpi.com/2076-3417/11/18/8563]
- 18. Calibration Update and Drift Correction for Electronic ... [https://www.frontiersin.org/journals/chemistry/articles/10.3389/fchem.2018.00433/full]
- 19. Enzymatic electrochemical biosensors for urea detection [https://www.sciencedirect.com/science/article/abs/pii/S092442472400493X]
- 20. Fabrication of a Urea Biosensor for Real-Time Dynamic ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC6111982/]
- 21. Biosensing of Urea with a Functionalized Gold Electrode ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC12512190/]
- 22. A New Calibration Circuit Design to Reduce Drift Effect of ... [https://www.gotriple.eu/documents/ftdoajarticles%3Aoai%3Adoaj.org%2Farticle%3A43c0e73615ac4630884a9ca48a502408]
- 23. Recent advancements in urea biosensors for biomedical ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC8675831/]
- 24. Advanced RuO2 Thin Films for pH Sensing Application [https://pmc.ncbi.nlm.nih.gov/articles/PMC7697712/]
- 25. A guide to reducing noise and increasing sensitivity in ... [https://blog.igii.uk/a-guide-to-reducing-noise-and-increasing-sensitivity-in-biosensors]
- 26. Application of an Electrochemical Sensor Based on ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC11940077/]
- 27. Copper–Ruthenium Composite as Perspective Material for ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC9313201/]
- 28. Binary RuO2–CuO Electrodes Outperform ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC10099411/]