Systematic Optimization of Biosensors Using Central Composite Design: From Foundational Principles to Clinical Applications
This article provides a comprehensive overview of the application of Central Composite Design (CCD), a powerful response surface methodology, in the optimization of biosensors.
Systematic Optimization of Biosensors Using Central Composite Design: From Foundational Principles to Clinical Applications
Abstract
This article provides a comprehensive overview of the application of Central Composite Design (CCD), a powerful response surface methodology, in the optimization of biosensors. Tailored for researchers, scientists, and drug development professionals, it covers foundational principles, practical methodologies, troubleshooting strategies, and comparative validation. The content explores how CCD overcomes the limitations of one-variable-at-a-time approaches by efficiently modeling complex interactions and nonlinear effects, leading to enhanced biosensor sensitivity, specificity, and reproducibility. Real-world case studies from electrochemical and optical biosensor development illustrate its transformative potential for creating reliable point-of-care diagnostics and other biomedical applications.
Understanding Central Composite Design: A Foundational Tool for Efficient Biosensor Development
The Limitations of One-Variable-at-a-Time (OVAT) Optimization in Biosensor Fabrication
The development of high-performance biosensors is a complex, multi-parameter challenge that requires careful optimization to achieve the sensitivity, selectivity, and reproducibility demanded by modern diagnostic and research applications. For decades, the one-variable-at-a-time (OVAT) approach has been a commonly used method for optimizing biosensor fabrication parameters. This traditional technique involves systematically varying a single experimental factor while holding all others constant, iterating this process across all parameters believed to influence biosensor performance. While intuitively simple and straightforward to implement, OVAT optimization possesses significant limitations that can impede the development of optimally performing biosensing platforms, particularly as sensor architectures grow increasingly sophisticated.
Within the context of advanced optimization methodologies, response surface methodologies (RSM) and specifically central composite design (CCD) have emerged as powerful statistical alternatives that address the fundamental shortcomings of the OVAT approach. This technical guide examines the inherent limitations of OVAT optimization in biosensor fabrication and demonstrates how central composite design provides a superior framework for navigating complex multi-parameter spaces, ultimately leading to enhanced sensor performance with greater experimental efficiency.
Fundamental Limitations of the OVAT Approach
The OVAT approach, while methodologically simple, suffers from several critical limitations that reduce its effectiveness for optimizing complex biosensor systems.
Inability to Detect Factor Interactions
The most significant limitation of OVAT is its fundamental inability to detect interactions between different experimental factors [1]. Biosensor fabrication and performance are influenced by numerous interconnected parameters whose effects are often non-additive and interdependent. For instance, the optimal concentration of a capture probe may depend on the ionic strength of the buffer, or the ideal hybridization temperature might vary with probe density. Since OVAT varies only one factor at a time while holding others constant, these critical interactions remain undetected, potentially leading researchers to suboptimal conditions [2].
Experimental Inefficiency and Resource Consumption
OVAT optimization requires an impractically large number of experiments when multiple parameters need optimization. The number of experiments grows linearly with the number of factors and their levels, quickly becoming resource-prohibitive. For example, optimizing six variables at three levels each would require 729 (3⁶) experiments – a nearly impossible task given the time and resource constraints of most research settings [1]. This inefficiency is particularly problematic in biosensor development where reagents are often expensive and preparation time-consuming.
Risk of Identifying Local Optima
By exploring the experimental space in a sequential, unidimensional manner, OVAT frequently identifies local optima rather than the global optimum for the system [2]. Without the ability to visualize the response surface across multiple dimensions, researchers can become trapped in suboptimal regions of the parameter space, ultimately limiting the performance of their final biosensor device.
Practical Evidence of OVAT Limitations
Case studies demonstrate these limitations in practical biosensor development. A paper-based electrochemical biosensor for miRNA detection initially optimized using OVAT achieved limits of detection (LOD) of 12.0 nM for miR-155 and 25.7 nM for miR-21 [1]. However, when the same system was re-optimized using design of experiments (DoE) methodology, researchers achieved a 5-fold improvement in detection limit, directly attributable to more accurate identification of optimal conditions that accounted for interacting variables [1].
Similarly, in pharmaceutical research, OVAT-optimized methods often fail to achieve the robustness and performance of those developed using multivariate approaches, particularly for complex analytical techniques requiring precision at low detection limits [3].
Central Composite Design: A Superior Framework for Biosensor Optimization
Central composite design (CCD) belongs to the family of response surface methodologies (RSM) and provides a structured, statistical framework for efficiently exploring multiple experimental factors simultaneously. As a second-order design, CCD can model curvature in the response surface, making it ideal for identifying optimal conditions in complex systems like biosensors.
Fundamental Structure of CCD
A central composite design consists of three distinct components:
- Factorial points (2ᵏ or fractional factorial): These points form the corners of the experimental cube and estimate linear effects and two-factor interactions.
- Center points: Repeated experiments at the center of the design space that estimate pure error and check for curvature.
- Axial points (star points): Points located along each factor axis at a distance ±α from the center that enable estimation of quadratic effects.
This structure provides comprehensive coverage of the experimental space while maintaining statistical efficiency, typically requiring far fewer experiments than a full OVAT approach [4].
Advantages of CCD for Biosensor Optimization
CCD addresses the specific limitations of OVAT in several crucial ways:
- Detection of Factor Interactions: By varying all factors simultaneously according to a predefined matrix, CCD can identify and quantify interactions between parameters, providing insights into the underlying system behavior [5].
- Experimental Efficiency: CCD dramatically reduces the number of experiments required for optimization. One study demonstrated optimization of six variables with only 30 experiments using a D-optimal design (a variant of CCD), compared to 486 experiments that would have been required with OVAT [1].
- Global Optima Identification: The comprehensive exploration of the experimental space enables CCD to locate true optimal conditions rather than local optima [6].
- Mathematical Modeling: CCD generates a mathematical model (typically a second-order polynomial) that describes the relationship between factors and responses, allowing prediction of biosensor performance across the entire experimental domain [3].
Comparative Analysis: OVAT versus CCD
Table 1: Quantitative Comparison of OVAT and CCD Optimization Approaches
| Characteristic | OVAT Approach | CCD Approach | Practical Implication |
|---|---|---|---|
| Experimental Efficiency | Linear increase with factors (3⁶ = 729 for 6 factors) | Quadratic increase (30-50 runs typical for 6 factors) | CCD reduces experimental burden by 80-95% [1] |
| Factor Interactions | Cannot detect | Quantifies all two-factor interactions | Prevents suboptimal conditions due to missed interactions [2] |
| Optimum Identification | Often finds local optimum | Finds global optimum | Better biosensor performance (e.g., 5x LOD improvement) [1] |
| Mathematical Model | No comprehensive model | Full quadratic model | Enables prediction and optimization across design space [4] |
| Resource Consumption | High (time, reagents, cost) | Moderate | Faster development cycle, lower cost [3] |
| Curvature Detection | Limited | Explicitly models curvature | Accurate optimization of nonlinear systems |
Table 2: Practical Applications of CCD in Biosensor Development and Related Fields
| Application Area | CCD Configuration | Factors Optimized | Performance Improvement |
|---|---|---|---|
| Electrochemical miRNA Biosensor [1] | D-optimal design (30 experiments) | 6 variables: AuNP concentration, probe density, ionic strength, hybridization time, temperature, electrochemical parameters | 5-fold improvement in detection limit compared to OVAT |
| Fluorescent Lacosamide Detection [3] | CCD (27 experiments) | pH, buffer volume, BN-GQDs concentration, incubation time | LOD of 0.033 µg/mL, meeting ICH M10 validation criteria |
| Copper-Mediated Radiofluorination [2] | CCD for response surface optimization | Temperature, reagent stoichiometry, concentration, time | >2x experimental efficiency vs OVAT, identified critical interactions |
| Pigment Production Optimization [5] | Five-level CCD | Yeast extract, K₂HPO₄, MgSO₄·7H₂O | Predicted maximum yield with 17 experiments vs ~50 with OVAT |
Implementing CCD for Biosensor Optimization: A Step-by-Step Methodology
Successful implementation of CCD for biosensor optimization follows a systematic workflow that maximizes information gain while minimizing experimental effort.
Experimental Workflow for CCD Implementation
The following diagram illustrates the comprehensive workflow for implementing CCD in biosensor optimization:
Practical Implementation Protocol
Based on successful applications in recent literature, the following protocol provides a template for implementing CCD in biosensor optimization:
Phase 1: Preliminary Screening (Weeks 1-2)
- Define critical quality attributes: Identify key biosensor performance metrics (e.g., limit of detection, signal-to-noise ratio, reproducibility, dynamic range).
- Brainstorm potential factors: Compile a comprehensive list of all material, fabrication, and operational parameters that could influence biosensor performance.
- Conduct screening design: Use a fractional factorial or Plackett-Burman design to identify the most influential factors from the broader list [1].
- Establish factor ranges: Based on screening results and practical constraints, determine appropriate low, middle, and high levels for each significant factor to be included in the CCD.
Phase 2: CCD Execution (Weeks 3-6)
- Design CCD matrix: Use statistical software (JMP, Minitab, Design-Expert, or R) to generate a CCD experimental matrix. For 4-6 factors, this typically requires 27-54 experimental runs, including center points for error estimation [3].
- Randomize run order: Execute experiments in randomized order to minimize confounding from external variables.
- Measure responses: For each experimental run, measure all predefined critical quality attributes with appropriate replication.
Phase 3: Data Analysis and Optimization (Weeks 7-8)
- Model development: Fit experimental data to a second-order polynomial model using multiple linear regression: ( Y = β0 + ΣβiXi + ΣβiiXi^2 + ΣβijXiXj + ε )
- Statistical validation: Evaluate model adequacy using ANOVA, lack-of-fit tests, and examination of residuals.
- Response surface analysis: Visualize factor-response relationships using contour and 3D surface plots.
- Identify optimal conditions: Use numerical optimization algorithms (desirability function) to identify factor settings that simultaneously optimize all critical responses.
- Experimental verification: Conduct confirmation experiments at predicted optimal conditions to validate model accuracy.
Essential Research Reagent Solutions for CCD-Optimized Biosensors
Successful implementation of CCD for biosensor optimization requires specific materials and reagents tailored to the biosensing platform. The following table summarizes key research reagent solutions used in CCD-optimized biosensor development:
Table 3: Essential Research Reagent Solutions for Biosensor Optimization
| Reagent Category | Specific Examples | Function in Biosensor Development | Optimization Considerations |
|---|---|---|---|
| Nanomaterial Transducers | Gold nanoparticles (AuNPs), graphene quantum dots (GQDs), boron-nitrogen doped GQDs (BN-GQDs) [1] [3] | Signal amplification, electron transfer enhancement, fluorescence properties | Concentration, size, functionalization, doping levels |
| Biorecognition Elements | DNA probes, antibodies, aptamers, enzymes (glucose oxidase, lactate oxidase) [1] [7] | Target recognition and binding specificity | Immobilization density, orientation, activity preservation |
| Surface Chemistry Reagents | Cross-linkers, SAM-forming thiols, PEG spacers, blocking agents [7] | Interface engineering, non-specific binding reduction, bioreceptor immobilization | Concentration, incubation time, buffer composition |
| Buffer Components | Phosphate buffers, Briton-Robinson buffer, Tris-HCl, ionic strength modifiers [1] [3] | Control of pH, ionic strength, chemical environment | pH, ionic strength, buffer capacity, compatibility |
| Electrochemical Mediators | Ferricyanide, methylene blue, organometallic complexes [1] | Facilitate electron transfer in electrochemical detection | Concentration, redox potential, stability |
| Signal Generation Reagents | Enzyme substrates, electrochemiluminescent compounds, fluorescent dyes [3] | Generate measurable signal upon target binding | Concentration, stability, compatibility with detection system |
The one-variable-at-a-time approach to biosensor optimization presents fundamental limitations in detecting factor interactions, experimental efficiency, and identification of global optima. These shortcomings become increasingly problematic as biosensor architectures grow more complex and performance requirements more stringent. Central composite design addresses these limitations through a structured, statistical framework that simultaneously varies multiple factors according to mathematically derived experimental matrices.
The implementation of CCD in biosensor development enables researchers to efficiently navigate complex multi-parameter spaces, quantify factor interactions, and identify true optimal conditions with significantly reduced experimental effort compared to OVAT. As evidenced by numerous case studies across electrochemical, optical, and fluorescent biosensing platforms, CCD-optimized biosensors consistently outperform their OVAT-optimized counterparts, achieving lower detection limits, improved reproducibility, and enhanced overall performance.
For researchers pursuing the development of next-generation biosensors with demanding performance specifications, the adoption of central composite design and related response surface methodologies represents not merely a statistical refinement, but a fundamental paradigm shift in optimization strategy—one that aligns with the complex, interdependent nature of modern biosensing systems.
Core Principles of Response Surface Methodology (RSM) and CCD
Response Surface Methodology (RSM) is a powerful collection of mathematical and statistical techniques used for empirical model building and process optimization when multiple independent variables influence one or more responses of interest [8] [9]. Originally developed by Box and Wilson in the 1950s, RSM has evolved into a fundamental methodology within the broader framework of Design of Experiments (DoE) [8] [9] [10]. The primary objective of RSM is to efficiently map a region of interest in the factor space, identify optimal factor settings that produce desired response values, and understand how changes in input variables jointly affect response variables [8] [11] [10].
In scientific and industrial contexts, RSM enables researchers to navigate complex relationships between factors and responses through a structured sequence of experiments. This approach is particularly valuable when traditional one-variable-at-a-time methods prove inadequate due to factor interactions or when the relationship between variables exhibits significant curvature [4] [11]. By employing carefully designed experiments and regression analysis, RSM develops mathematical models that approximate the true underlying relationship between factors and responses, providing a systematic framework for process improvement and optimization [8] [9].
Fundamental Concepts and Terminology
Core Components of RSM
Understanding RSM requires familiarity with its fundamental components and their relationships:
- Factors: These are the input variables (also called independent variables) that can be controlled or adjusted during experimentation. In biosensor research, factors might include material concentrations, process temperatures, incubation times, or pH levels [8] [4].
- Responses: The output variables (dependent variables) that represent the measurable outcomes of the experiment. For biosensor optimization, typical responses include sensitivity, detection limit, selectivity, and response time [12] [4].
- Experimental Domain: The multidimensional space defined by the ranges of all factors being studied. This region represents the boundaries within which the experimenter believes the optimal operating conditions are likely to be found [8] [4].
- Response Surface: The geometrical representation of the relationship between factors and response, typically visualized as contours or 3D surfaces [8] [10].
Mathematical Foundation
RSM typically employs polynomial regression models to approximate the true functional relationship between factors and responses. The general form of a second-order (quadratic) model for k factors is represented as:
[Y = \beta0 + \sum{i=1}^{k}\betaiXi + \sum{i=1}^{k}\beta{ii}Xi^2 + \sum{i=1}^{k-1}\sum{j=i+1}^{k}\beta{ij}XiXj + \varepsilon]
Where Y is the predicted response, β₀ is the constant term, βᵢ represents the linear coefficients, βᵢᵢ represents the quadratic coefficients, βᵢⱼ represents the interaction coefficients, and ε represents the random error term [8] [10].
This quadratic model is particularly valuable because it can capture curvature in the response surface, which is essential for identifying optimal conditions when the true optimum lies within the experimental region rather than at its boundaries [11].
Central Composite Design (CCD): Structure and Variants
Components of CCD
Central Composite Design is the most widely used response surface design, consisting of three distinct element types that provide comprehensive information about the factor-response relationship [13] [11]:
- Factorial Points: A full or fractional factorial design that captures linear effects and factor interactions. For k factors, this portion contains 2ᵏ points (or 2ᵏ⁻ᵖ for fractional factorial).
- Axial Points (Star Points): Additional points located along each factor axis at a distance ±α from the design center. These points enable estimation of quadratic effects and provide additional information about curvature in the response surface. For k factors, there are 2k axial points.
- Center Points: Multiple replicates at the center of the design space that provide an estimate of pure error and model stability, and help detect curvature in the response [13] [11].
The specific value of α depends on the desired design properties, with rotatability being a common criterion. A design is rotatable if the prediction variance depends only on the distance from the design center, not on direction [11].
Types of Central Composite Designs
Central Composite Designs are categorized based on the placement of the axial points relative to the factorial portion [13]:
Table 1: Comparison of Central Composite Design Types
| Design Type | Axial Point Location | Factor Levels | Key Characteristics | Application Context |
|---|---|---|---|---|
| Circumscribed (CCC) | Outside the factorial cube | 5 levels per factor | Rotatable property; extends beyond original factor range | General optimization when extreme conditions are feasible |
| Face-Centered (CCF) | On the faces of the factorial cube | 3 levels per factor | Practical constraint adherence; α = ±1 | When factor ranges cannot be exceeded |
| Inscribed (CCI) | Within the factorial cube | 5 levels per factor | Factorial points at extremes of original range | When the original range represents actual operating limits |
For biosensor optimization research, the face-centered CCD is often preferred because it operates within a clearly defined "safe operating zone" where all factor combinations are feasible and biologically relevant [11]. This is particularly important when working with sensitive biological components that may be denatured or inactivated under extreme conditions.
Comparison of Response Surface Designs
Table 2: Comparison of CCD and Box-Behnken Designs for Response Surface Methodology
| Characteristic | Central Composite Design (CCD) | Box-Behnken Design (BBD) |
|---|---|---|
| Design Structure | Factorial + axial points + center points | Balanced incomplete block design |
| Number of Levels | 3 or 5 per factor | 3 per factor |
| Sequential Usage | Excellent - can build on previous factorial designs | Not suitable - requires new design |
| Extreme Conditions | Includes all factors at high/low simultaneously | Avoids extreme combinations |
| Number of Runs (3 factors) | 15-20 depending on center points | 13-15 |
| Region of Exploration | Spherical or cuboidal | Spherical |
| Optimal For | Sequential optimization after screening | When extreme combinations are risky |
The choice between CCD and BBD depends on research constraints and objectives. CCD is preferable for sequential experimentation where researchers can build upon previous factorial results, while BBD offers greater efficiency when the number of experimental runs must be minimized and extreme factor combinations should be avoided [13] [11].
Implementation Workflow for RSM and CCD
The following diagram illustrates the systematic workflow for implementing Response Surface Methodology in optimization research:
Problem Definition and Factor Screening
The initial phase of any RSM study involves clearly defining the optimization objectives and identifying the critical response variables that measure process performance [8]. In biosensor research, this might include defining target performance metrics such as detection limit, sensitivity, dynamic range, or response time [4]. Subsequent factor screening helps identify which of the many potential input variables significantly influence the responses of interest. Effective screening prevents resource waste on insignificant factors while ensuring critical variables are not overlooked [8].
Experimental Design and Execution
After identifying critical factors, researchers must select appropriate ranges for each factor and choose a specific experimental design. For CCD implementation, this involves determining whether a circumscribed, face-centered, or inscribed design is most appropriate based on operational constraints [13]. Factors are typically coded to a -1 (low), 0 (center), +1 (high) scale to minimize multicollinearity and place all factors on a common scale for coefficient comparison [8] [13]. Experiments are then conducted in randomized order to minimize the effects of uncontrolled variables and external influences [8].
Model Development and Validation
Following data collection, regression analysis is used to fit an appropriate model (typically second-order) to the experimental data [8] [10]. The model's statistical significance and adequacy are then evaluated using Analysis of Variance (ANOVA), which partitions total variability into components attributable to the model, individual terms, and residual error [8] [14]. Key validation metrics include:
- Lack-of-fit testing: Determines whether the selected model adequately describes the observed data
- R-squared values: Measure the proportion of response variation explained by the model
- Residual analysis: Checks model assumptions by examining patterns in prediction errors [8]
Optimization and Validation
Once an adequate model is established, optimization techniques identify factor settings that produce optimal response values [8] [10]. For single responses, this may involve analytical or numerical methods to locate stationary points on the response surface. For multiple responses, desirability functions or overlaid contour plots help identify compromise conditions that simultaneously satisfy multiple objectives [11] [10]. Finally, confirmation experiments at the predicted optimal conditions validate both the model's predictive capability and the practical viability of the identified optimum [8].
Application in Biosensor Optimization Research
Case Study: Glucose Biosensor Optimization
A compelling demonstration of CCD in biosensor research comes from the optimization of an amperometric glucose biosensor [12]. Researchers applied a five-level, three-factor CCD to determine the optimal electrode surface composition, focusing on three critical variables: amounts of carboxylated multiwall carbon nanotubes (c-MWCNT), titanium dioxide nanoparticles (TiO₂NP), and glucose oxidase (GOx) [12].
The experimental design enabled researchers to not only identify optimal factor levels but also to understand interaction effects between components that would have been missed in traditional one-variable-at-a-time approaches. The resulting optimized biosensor showed a linear response to glucose from 2.0×10⁻⁵ to 1.9×10⁻³ M with a detection limit of 2.1×10⁻⁶ M and sensitivity of 168.5 μA mM⁻¹ cm⁻² [12]. Performance comparison confirmed that the CCD-optimized biosensor outperformed those developed using conventional optimization methods.
Systematic Optimization of Ultrasensitive Biosensors
Recent research emphasizes the critical importance of systematic optimization for ultrasensitive biosensors, particularly those with sub-femtomolar detection limits [4]. The complex relationships between fabrication parameters, immobilization strategies, and detection conditions in these sophisticated platforms make them ideal candidates for RSM approaches [4].
Experimental design addresses a fundamental limitation of conventional biosensor development: the tendency to optimize variables independently, which fails to account for potentially significant interaction effects [4]. By contrast, RSM approaches enable researchers to efficiently explore the multidimensional factor space while quantifying both main effects and factor interactions, ultimately leading to more robust and reliably optimized biosensing platforms [4].
Research Reagent Solutions for Biosensor Optimization
Table 3: Essential Research Reagents and Materials for Biosensor Optimization Studies
| Reagent/Material | Function in Biosensor Development | Application Example |
|---|---|---|
| Carboxylated Multiwall Carbon Nanotubes (c-MWCNT) | Electron transfer enhancement; increased electrode surface area | Glucose biosensor electrode composition [12] |
| Titanium Dioxide Nanoparticles (TiO₂NP) | Biocompatibility; photocatalytic properties; electron transfer mediation | Glucose biosensor performance enhancement [12] |
| Glucose Oxidase (GOx) | Biological recognition element; specific catalysis of glucose oxidation | Enzymatic glucose biosensors [12] |
| Spirulina platensis Biomass | Biosorbent material for contaminant removal | Environmental biosensor development [14] |
| Diphenylcarbazide | Colorimetric detection reagent for chromium(VI) | Analytical detection in biosensor validation [14] |
Advanced Considerations and Future Perspectives
Addressing Multiple Response Optimization
Many practical biosensor optimization problems involve multiple competing responses that must be simultaneously optimized [10]. For example, a researcher might need to maximize sensitivity while minimizing response time and manufacturing cost. RSM addresses this challenge through several approaches:
- Overlaid Contour Plots: Visualizing the acceptable operating region for each response and identifying regions where all responses simultaneously meet specifications [10]
- Desirability Functions: Transforming each response into an individual desirability value (0-1 scale) and combining these into an overall composite desirability to be maximized [10]
- Dual Response Surface Methodology: Modeling both the mean response and variability to find conditions that achieve target performance with minimal variation [8]
Emerging Trends and Methodological Advances
The ongoing integration of RSM with computational modeling and machine learning represents a significant frontier in biosensor research [4] [10]. Surrogate modeling techniques, such as Gaussian process regression and neural networks, can complement traditional RSM when physical experimentation is costly or time-consuming [8]. Additionally, the growing emphasis on robust parameter design ensures that optimized biosensors perform consistently despite uncontrollable environmental factors and manufacturing variations [8].
As biosensor technology advances toward increasingly complex multiplexed detection systems and point-of-care applications, the systematic optimization approaches provided by RSM and CCD will remain indispensable tools for researchers seeking to translate laboratory innovations into practical, reliable diagnostic solutions [4].
In the field of biosensor development, achieving optimal performance is a complex multivariate challenge. Parameters such as the concentration of the biorecognition element, immobilization time, pH, and temperature do not act in isolation; they interact in ways that can significantly impact the sensor's sensitivity, selectivity, and limit of detection [15]. The traditional "one-variable-at-a-time" optimization approach is not only inefficient but also fails to capture these critical interactions, often leading to suboptimal results [16].
Central Composite Design (CCD) is a powerful statistical methodology for response surface modeling and optimization that addresses these limitations. Developed by Box and Wilson, CCD provides a structured and efficient framework for designing experiments, fitting a second-order model, and locating optimal factor settings [16] [15]. For researchers developing ultrasensitive biosensors, where maximizing the signal-to-noise ratio is paramount, the systematic application of CCD has proven to be particularly valuable [15]. Its flexibility and robustness have made it a cornerstone of chemometrics, enabling the efficient development of reliable analytical methods [16].
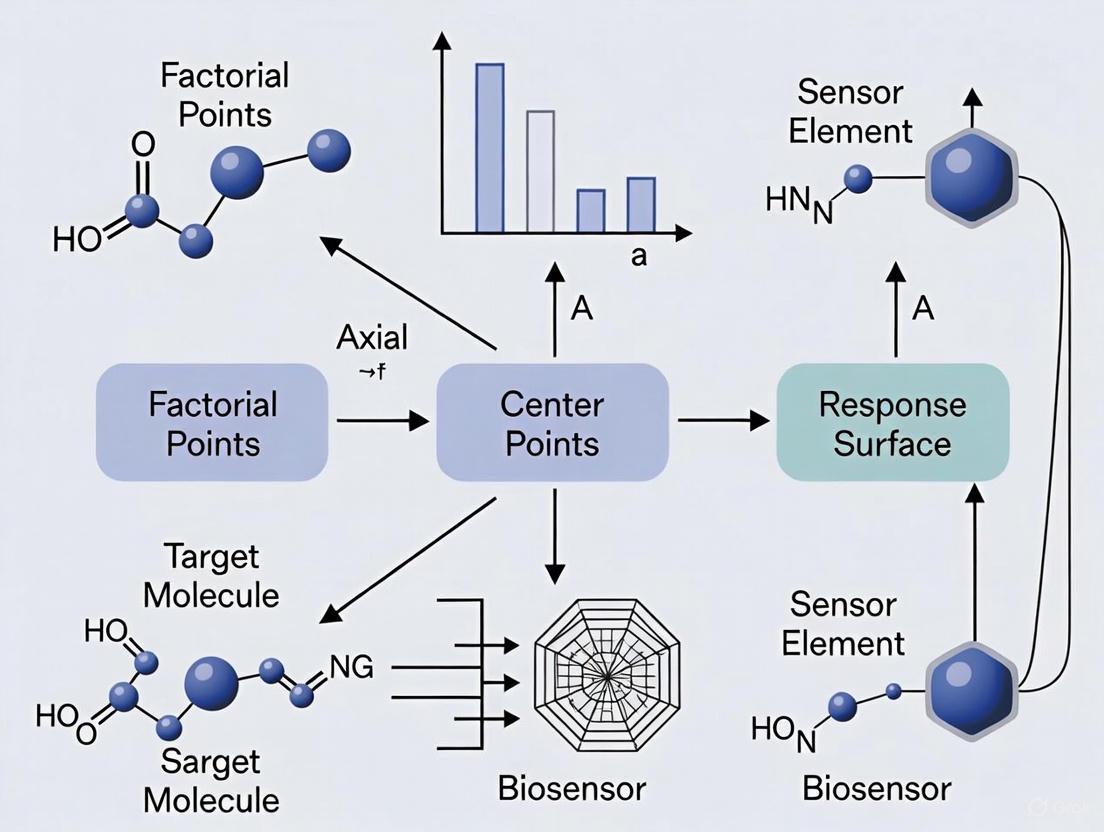
This guide decodes the core components of a CCD matrix—the factorial, axial, and center points—and provides a detailed protocol for its application in biosensor optimization research.
The Core Components of a CCD Matrix
A Central Composite Design is built from three distinct types of experimental points that work in concert to enable the estimation of a complex, curved response surface.
The Factorial Points
- Purpose: The factorial points form a two-level full or fractional factorial design that is the foundation of the CCD. These points are primarily responsible for estimating the linear effects and two-factor interaction effects of the model [17] [15].
- Encoding: In their coded form, the levels of these factors are set to -1 (low) and +1 (high), which correspond to the practical limits of the experimental region of interest [18]. For k factors, a full factorial requires (2^k) points.
- Geometric Location: These points represent the "corners" of the experimental domain. For two factors, they form a square; for three factors, a cube; and for more factors, a hypercube [15].
The Axial Points (Star Points)
- Purpose: The axial points are the defining feature of the CCD. They are specifically added to allow for the estimation of curvature in the response surface, meaning they enable the model to fit quadratic effects [17] [16].
- Encoding: Also called star points, they are located on the axes of the experimental factors at a distance of ±α from the design center, with all other factors set to zero (the center point) [17].
- Geometric Location: The axial points lie along the coordinate axes of the design space. In a two-factor design, they are located on the horizontal and vertical axes, outside the factorial square.
The Center Points
- Purpose: Center points serve multiple critical functions. They provide an independent estimate of pure experimental error, allow for the detection of curvature in the response surface, and stabilize the prediction variance across the experimental region [17] [19]. Replicating the center point is essential for obtaining a reliable estimate of variability.
- Encoding: The center point is located at the midpoint of the design space, with all factors set to their mid-level, coded as 0 [15].
- Geometric Location: This point sits at the very center of the factorial square, cube, or hypercube.
The following diagram illustrates the geometric arrangement of these three components in a standard two-factor CCD.
Diagram 1: A two-factor Central Composite Design, showing the factorial (blue), axial (yellow), and center (red) points.
Types of Central Composite Designs
The value chosen for the axial distance α and the relationship of the star points to the factorial cube determine the specific type of CCD. The choice depends on the experimental region of interest and desired properties.
Table 1: Types of Central Composite Designs
| CCD Type | Terminology | Axial Distance (α) | Factor Levels | Key Properties |
|---|---|---|---|---|
| Circumscribed | CCC | |α| > 1 [17] | 5 per factor [17] | Original form; requires the factorial points to be strictly within the operability region; rotatable [17]. |
| Inscribed | CCI | α = ±1 [17] | 5 per factor [17] | A scaled-down CCC where the star points define the region of interest; used when the design space has strict limits [17]. |
| Face-Centered | CCF | α = ±1 [17] | 3 per factor [17] | Star points are at the center of the factorial space's faces; not rotatable but easier to execute [17]. |
The diagram below visualizes the differences in the experimental domains explored by these three common CCD types.
Diagram 2: A comparison of Circumscribed (CCC), Inscribed (CCI), and Face-Centered (CCF) central composite designs.
The total number of experiments required for a CCD is a direct function of the number of factors being studied. This allows researchers to plan resources effectively.
Table 2: Total Number of Runs in a Central Composite Design
| Number of Factors (k) | Factorial Points ((2^k)) | Axial Points (2k) | Center Points (N0) | Total Runs (Example with N0=5) |
|---|---|---|---|---|
| 2 | 4 | 4 | User-defined | 13 |
| 3 | 8 | 6 | User-defined | 19 |
| 4 | 16 | 8 | User-defined | 29 |
| 5 | 32 | 10 | User-defined | 47 |
The general formula for the total number of runs is: Total runs = (2^k) + 2k + N0, where (k) is the number of input variables and (N_0) is the number of center points [17].
The value of the axial distance α can be calculated to achieve desired statistical properties. For a rotatable design, which ensures constant prediction variance at all points equidistant from the center, the value is α = (2^{k/4}) [17]. For a face-centered design (CCF), α is simply set to 1 [17] [19].
The Researcher's Toolkit for CCD
Table 3: Essential Research Reagent Solutions for a Biosensor Optimization CCD
| Reagent / Material | Function in the Experiment | Example from Literature |
|---|---|---|
| Biorecognition Element | The biological molecule (e.g., antibody, enzyme, DNA probe) that confers specificity to the biosensor by binding the target analyte. | Immobilization strategy is a key parameter for optimization via CCD [15]. |
| Immobilization Buffer | The chemical solution used to fix the biorecognition element onto the transducer surface; its pH and ionic strength are critical factors. | A key variable in optimizing the biosensor's detection interface [15]. |
| Signal Probe/Amplification Reagent | A chemical or nanomaterial (e.g., enzyme conjugate, nanoparticle) used to generate or amplify the detection signal. | Formulation of the detection interface is a primary optimization target in biosensor development [15]. |
| Blocking Buffer | A solution (e.g., BSA, casein) used to cover non-specific binding sites on the sensor surface, reducing background noise. | Contributes to maximizing the signal-to-noise ratio, a key response in ultrasensitive biosensor optimization [15]. |
| Washing Buffer | A solution used to remove unbound reagents; its stringency (e.g., salt concentration, detergent) can be a critical factor. | Optimized to minimize non-specific binding and improve the limit of detection [15]. |
| Analyte Standard | A purified preparation of the target molecule at a known concentration, used to construct the calibration curve and define the response. | The detection of specific biomarkers (proteins, peptides, genomic markers) is the central goal of the optimized method [15]. |
Experimental Protocol: A CCD for Biosensor Optimization
The following workflow provides a detailed, step-by-step protocol for implementing a CCD in a biosensor development project.
Diagram 3: A sequential workflow for conducting a Central Composite Design (CCD) study.
Step 1: Define Objective and Response Variable(s)
Clearly state the goal of the optimization (e.g., "to maximize the electrochemical signal-to-noise ratio for a target DNA sequence"). Select one or more quantifiable response variables that accurately reflect biosensor performance, such as Limit of Detection (LOD), signal intensity, or assay time [15].
Step 2: Select Critical Factors and Ranges
Identify the key input variables (factors) that most likely influence the response. In biosensor optimization, these often include:
- pH of the immobilization or detection buffer.
- Concentration of the biorecognition element (e.g., antibody, DNA probe).
- Immobilization time.
- Temperature of the reaction.
- Concentration of a signal amplification reagent [15]. Define a realistic and practical range for each factor (e.g., pH 6.0 to 8.0). The low and high levels will be coded as -1 and +1, respectively [18].
Step 3: Choose CCD Type and Generate Design Matrix
Select an appropriate CCD type based on your experimental constraints and goals.
- Use a Face-Centered (CCF) design for a simple, three-level design that is easy to implement [17] [19].
- Use a Circumscribed (CCC) design if you need a rotatable design and can operate outside the factorial cube [17]. Software tools like JMP, Design-Expert, or Statease are typically used to generate the randomized run order, which is critical for avoiding confounding from lurking variables [19].
Step 4: Execute Experiments in Randomized Order
Conduct all experiments as specified by the design matrix. It is imperative to follow the randomized run order to minimize the impact of uncontrolled variables and time-related effects. Precisely record the response(s) for each experimental run.
Step 5: Fit Second-Order Model via Regression
Using standard statistical software, perform a multiple linear regression to fit a second-order polynomial model to the data. The general form of the model for two factors (X₁, X₂) is: Y = b₀ + b₁X₁ + b₂X₂ + b₁₂X₁X₂ + b₁₁X₁² + b₂₂X₂² + e Where Y is the predicted response, b₀ is the constant, b₁ and b₂ are linear coefficients, b₁₂ is the interaction coefficient, b₁₁ and b₂₂ are quadratic coefficients, and e is the error [20]. The model's significance and lack-of-fit should be evaluated using ANOVA.
Step 6: Analyze Model and Generate Response Surfaces
Interpret the fitted model to understand the relationship between factors and the response.
- Examine the magnitude and sign of coefficients to determine the influence of each factor.
- Use Response Surface Methodology (RSM) to visualize the model by generating contour or 3D surface plots [16] [20]. These plots are invaluable for identifying the direction toward the optimum.
Step 7: Locate Optimum and Verify with Experiments
The software can use the fitted model to numerically and graphically identify the optimal factor settings (e.g., the point that maximizes the signal). Crucially, this predicted optimum must be validated by performing a small set of confirmation experiments under these recommended conditions [15]. If the results from the confirmation runs align with the predictions, the optimal conditions have been successfully identified.
The structured matrix of factorial, axial, and center points in a Central Composite Design provides a highly efficient and information-rich framework for navigating complex experimental landscapes. For researchers and scientists in drug development and biosensor technology, mastering the decoding of this matrix is a critical skill. It enables a comprehensive understanding of the system under study—capturing not just linear effects, but also the crucial interaction and quadratic effects that define a true optimum. By following the detailed protocols outlined in this guide, professionals can systematically overcome the limitations of one-variable-at-a-time experimentation, thereby accelerating the development of robust, high-performance biosensing systems.
In the behavioral, social, and biological sciences, real-world phenomena rarely operate through simple linear relationships alone. Positing structural models with nonlinear terms is often necessary to adequately account for the complexities underlying experimental processes [21]. Among various nonlinear relations, the quadratic function remains an exceptionally attractive alternative for practitioners due to its effectiveness in summarizing many experimental processes within the observed data range [21]. A quadratic effect manifests as a curvilinear relationship where the direction of the effect changes across the level of the predictor variable, creating either a concave (downward-curving) or convex (upward-curving) pattern [21]. Understanding and properly modeling these effects, particularly in conjunction with variable interactions, provides researchers with a critical advantage in optimizing experimental outcomes and extracting meaningful insights from complex data.
The methodological approaches for testing quadratic effects have evolved significantly since Kenny and Judd's seminal 1984 work, now encompassing latent variable score approaches, product indicator methods, maximum likelihood estimation, and Bayesian estimation techniques [21]. These advanced statistical methods enable researchers to move beyond traditional linear models and capture the nuanced relationships that frequently occur in experimental systems, particularly in optimization research where response surfaces often exhibit curvature that must be precisely quantified to identify optimal operating conditions.
Theoretical Foundations of Quadratic Effects
Structural Equation Modeling Framework for Nonlinear Effects
Structural equation modeling (SEM), initially conceived for modeling linear relations among latent variables, can be extended to incorporate nonlinear effects through a structured framework comprising two distinct components [21]. The measurement model defines the relationship between latent variables and their observed indicators, while the structural model delineates the effects of exogenous latent variables on the endogenous variable, including any nonlinear pathways [21].
In a prototypical nonlinear structural equation model with quadratic effects, the full specification can be represented by two primary equations. The measurement model is defined as:
Zᵢ = τ + Λfᵢ + eᵢ
where Zᵢ is a vector of observed variables for the i-th individual, τ represents a vector of intercepts, Λ is the matrix of factor loadings relating latent variables to their measured indicators, and eᵢ represents the vector of unique factors independent of fᵢ with E(eᵢ) = 0 and Var(eᵢ) = Θ, where Θ is diagonal [21].
The structural model incorporating quadratic effects is specified as:
f₁ᵢ = γ₀ + γ₁f₂ᵢ + γ₂f₂ᵢ² + dᵢ
where γ₀, γ₁, and γ₂ are the intercept and direct path estimates of the first-order latent variable f₂ᵢ and the quadratic term f₂ᵢ², respectively [21]. The sign of γ₂ indicates whether the endogenous-exogenous curvilinear relation is concave (negative, implying curving downward) or convex (positive, implying curving upward) [21]. The residuals dᵢ have E(dᵢ) = 0 and Var(dᵢ) = σd², and are independent of f₂ᵢ as well as eᵢ.
Table 1: Interpretation of Quadratic Effect Parameters
| Parameter | Symbol | Interpretation | Implied Relationship |
|---|---|---|---|
| Intercept | γ₀ | Baseline level of endogenous variable | Starting point of curve |
| Linear effect | γ₁ | Direction and steepness of initial relationship | Slope at origin |
| Quadratic effect | γ₂ | Rate of curvature | Concave (γ₂ < 0) or convex (γ₂ > 0) |
Methodological Approaches for Estimating Quadratic Effects
The current methodological landscape offers multiple approaches for estimating and testing hypotheses of quadratic effects in latent variable regression models. Based on comprehensive Monte Carlo simulations comparing these methods, distinct performance patterns emerge across different experimental conditions [21].
The latent variable scores (LVS) approach involves a two-step process where factor scores for endogenous and exogenous latent variables are first computed, then squared to form quadratic terms before submission to multiple regression analysis [21]. While computationally straightforward, this method may produce biased or inconsistent parameter estimates due to measurement error in the factor scores [21].
Product indicator approaches construct interactions between indicators of latent variables and may be implemented as fully constrained, partially constrained, or unconstrained versions [21]. Maximum likelihood estimation methods, including marginal maximum likelihood and approximate maximum likelihood estimation schemes, generally demonstrate strong performance in terms of bias, root-mean-square error, standard error ratios, power, and Type I error control [21]. Similarly, Bayesian estimation approaches have shown excellent performance characteristics in simulation studies [21].
Table 2: Comparison of Quadratic Effect Estimation Methods
| Method | Key Characteristics | Performance Advantages | Implementation Considerations |
|---|---|---|---|
| Latent Variable Scores | Two-step factor score regression | Computational simplicity | Potential measurement error bias |
| Product Indicator | Constrained/unconstrained indicator products | Handles measurement error | Complex constraint specification |
| Maximum Likelihood | Direct model fitting | Low bias, good Type I error control | Computational intensity |
| Bayesian | Prior incorporation, MCMC sampling | Excellent overall performance | Prior specification sensitivity |
Central Composite Design for Optimization
Fundamental Principles and Design Structure
In response surface methodology, central composite design (CCD) represents an efficient experimental framework for building second-order (quadratic) models without requiring a complete three-level factorial experiment [22] [23]. This design approach is particularly valuable in biosensor optimization research, where understanding curvature in response surfaces is essential for identifying optimal operational parameters.
A CCD consists of three distinct sets of experimental runs that work in concert to efficiently estimate quadratic effects [22] [23]. The factorial portion, often a two-level full or fractional factorial design, forms the foundation for estimating linear and interaction effects. The center points, experimental runs where each factor is set at the median value of the factorial levels, provide pure error estimation and detect curvature. The axial points (or "star points"), where one factor is set at extremes beyond the factorial range while other factors remain at center points, enable estimation of quadratic terms [22] [23]. The number of axial points is always twice the number of factors in the design (2k for k factors) [22].
The complete design matrix for a central composite design is formed by vertically concatenating these three components [23]. For linear regression analysis, the design matrix X includes a column of 1s (intercept), the design matrix d, and all elementwise products of pairs of columns of d along with squared terms [23].
Figure 1: Central Composite Design Component Structure
Variants and Configuration Parameters
Central composite designs exist in three primary variants, each with distinct properties and applications [22]. The circumscribed (CCC) design, the original form of CCD, positions star points at a distance α from the center that establishes new extremes for all factors, creating designs with circular, spherical, or hyperspherical symmetry that require five levels for each factor [22]. The inscribed (CCI) design uses the actual factor limits as star points and creates a factorial design within those limits, effectively scaling down a CCC design [22]. The face-centered (CCF) design positions star points at the center of each face of the factorial space (α = ±1), requiring only three levels of each factor [22].
The value of α, the distance from the center to the axial points, is a critical design parameter determined based on desired properties [22] [23]. For rotatable designs, which provide consistent prediction variance at all points equidistant from the design center, α = F¹ᐟ⁴, where F is the number of points in the factorial portion of the design [23]. For orthogonal designs, which enable independent estimation of model coefficients, α = (Q × F/4)¹ᐟ⁴, where Q = (√(F + T) - √F)² and T is the number of additional points beyond the factorial portion [23].
Table 3: Central Composite Design Variants and Properties
| Design Type | α Value | Factor Levels | Process Space | Key Properties |
|---|---|---|---|---|
| Circumscribed (CCC) | α > 1 | 5 levels | Largest | Rotatable, spherical symmetry |
| Inscribed (CCI) | α = 1 | 5 levels | Smallest | Rotatable, uses actual factor limits |
| Face-Centered (CCF) | α = 1 | 3 levels | Intermediate | Not rotatable, practical implementation |
Application in Biosensor Optimization Research
Case Study: Glucose Biosensor Fabrication
The practical implementation of central composite design for modeling quadratic effects and interactions is powerfully illustrated in biosensor optimization research. In a seminal study, researchers employed a five-level three-factorial CCD to determine the optimum electrode surface composition for an amperometric glucose biosensor [12]. This approach systematically investigated three critical variables—amounts of carboxylated multiwall carbon nanotubes (c-MWCNT), titanium dioxide nanoparticles (TiO₂NP), and glucose oxidase (GOx)—and their quadratic effects on biosensor performance.
The experimental methodology involved applying the CCD to identify optimal combinations of the three critical factors, with statistical significance of the model and factors evaluated using variance analysis (ANOVA) at a 95% confidence level [12]. The resulting optimized electrode composition yielded a biosensor with linear response to glucose from 2.0 × 10⁻⁵ to 1.9 × 10⁻³ M, a detection limit of 2.1 × 10⁻⁶ M, and sensitivity of 168.5 μA mM⁻¹ cm⁻² under optimal experimental conditions [12]. Comparative analysis demonstrated that the CCD-optimized biosensor outperformed those fabricated using conventional one-factor-at-a-time optimization and smaller 2² CCD approaches, highlighting the critical advantage of comprehensive quadratic effect modeling [12].
Figure 2: Biosensor Optimization Workflow Using CCD
Research Reagent Solutions for Biosensor Development
The effective implementation of CCD in biosensor optimization relies on specific research reagents and materials, each serving distinct functions in the experimental system. The glucose biosensor case study exemplifies how these components interact within an optimized framework [12].
Table 4: Essential Research Reagents for Biosensor Optimization
| Research Reagent | Function in Experimental System | Role in Optimization |
|---|---|---|
| Carboxylated Multiwall Carbon Nanotubes (c-MWCNT) | Enhance electron transfer, increase electrode surface area | Critical variable with potential quadratic effects on sensitivity |
| Titanium Dioxide Nanoparticles (TiO₂NP) | Improve biocompatibility, facilitate enzyme immobilization | Interactive effects with other components |
| Glucose Oxidase (GOx) | Biological recognition element, catalyzes glucose oxidation | Primary enzymatic component with potential optimal concentration |
| Electrode Substrate | Platform for biosensor construction | Fixed component with standardized properties |
| Buffer Solutions | Maintain optimal pH for enzymatic activity | Standardized experimental condition |
Methodological Implementation
Computational Approaches for Quadratic Effect Modeling
Implementing quadratic effect models requires specialized computational approaches for generating and handling the necessary mathematical terms. For continuous variables, all pairwise interactions including "pure quadratic" terms (where a variable interacts with itself) can be systematically generated using computational techniques [24].
The total number of quadratic interactions for k variables is "(k+1) choose 2," which simplifies to (k+1)×k/2 [24]. This includes k pure quadratic terms and "k choose 2" cross-variable interaction terms. In statistical computing environments like SAS, the EFFECT statement with POLYNOMIAL option in PROC GLMSELECT can automatically generate these degree-2 effects, while the horizontal direct product (HDIR) function provides a flexible matrix-based approach for calculating interaction terms [24].
A robust algorithmic implementation for generating quadratic interactions involves computing HDIR(X[,i], X[,i:k]) for i=1,2,...,k, where X is the design matrix [24]. This systematic approach ensures all unique pairs are generated without redundancy. For researchers implementing these models, proper handling of missing values is essential, as missing data in main effects will propagate through to interaction terms, potentially requiring complete case analysis or sophisticated missing data techniques [24].
Statistical Testing and Interpretation Framework
When modeling complex quadratic and interaction effects, a comprehensive approach to statistical testing and interpretation is essential. Rather than employing stepwise term selection, which introduces co-linearity issues and distorts degrees of freedom, researchers should fit full models with all relevant product and main effect terms, then interpret results using comprehensive "chunk tests" (multiple degree-of-freedom tests of related terms) and visualizations [25].
This approach includes testing for overall interaction effects, nonlinear interactions, and combined tests for overall effects including both main effects and interactions [25]. For models incorporating spline terms (often superior to ordinary polynomials for flexibility), the ANOVA output provides meaningful hypothesis tests for all components, including tests for combined interaction influences when predictors interact with multiple variables [25].
Visualization techniques play a crucial role in interpreting complex quadratic and interaction effects. Surface plots display joint effects of two predictors on the response, while conditional effect plots show how the relationship between one predictor and the response changes at different levels of a modifying variable [25]. These graphical representations are particularly valuable for communicating complex nonlinear relationships to interdisciplinary research teams.
The strategic implementation of quadratic effect modeling and central composite design represents a critical methodological advantage in biosensor optimization and broader scientific research. By formally accounting for curvature and interaction effects in experimental systems, researchers can more accurately identify optimal operating conditions and understand complex biological relationships. The structured approach of CCD provides an efficient framework for building these second-order models while minimizing experimental burden. As computational methods continue to advance, incorporating robust statistical techniques for estimating and testing quadratic effects becomes increasingly accessible across scientific disciplines, promising enhanced optimization capabilities in pharmaceutical development, diagnostic biosensor fabrication, and beyond.
Central Composite Design (CCD) is a cornerstone of Response Surface Methodology (RSM), providing an efficient experimental framework for optimizing complex processes in biosensor development. As a branch of chemometrics, CCD belongs to the broader field of Design of Experiments (DOE), which employs structured experimental matrices to study and optimize systems, procedures, and processes to enhance their performance [16]. CCD has emerged as a particularly powerful tool due to its flexibility and robustness in modeling quadratic response surfaces, which are essential for understanding and optimizing the multifaceted parameters influencing biosensor performance.
In biosensor research and drug development, optimization challenges frequently involve numerous interacting variables—including biological recognition elements, transducer materials, surface chemistry, and detection conditions. Traditional One-Factor-at-a-Time (OFAT) optimization approaches prove inadequate for these complex systems because they cannot detect interaction effects between variables and may miss true optimal conditions [26]. CCD overcomes these limitations by systematically exploring multiple factors simultaneously while requiring fewer experimental runs than full factorial designs. This efficiency is particularly valuable in biosensor optimization, where reagents and biological materials are often costly and experimental procedures are time-intensive.
CCD achieves its power through a structured combination of three distinct types of experimental points: factorial points that map the primary experimental space, axial (star) points that estimate curvature, and center points that assess experimental error and system stability [16] [17]. The arrangement and scaling of these points give rise to different CCD modalities—Circumscribed (CCC), Inscribed (CCI), and Face-Centered (CCF)—each with specific characteristics, advantages, and limitations suited to particular experimental scenarios in biosensor development.
Fundamental Principles of CCD Modalities
Structural Components and Mathematical Basis
All CCD modalities share a common structural foundation, combining three types of experimental points to efficiently model second-order (quadratic) response surfaces. The factorial component consists of a full or fractional two-level factorial design, typically coded as -1 (low level) and +1 (high level) for each factor. This component estimates main effects and two-factor interactions. The axial (or star) component adds points along each factor axis at a distance ±α from the design center, enabling estimation of quadratic effects. The center points, typically replicated multiple times at the midpoint (0,0) of all factors, provide an estimate of pure error and model stability [17] [18].
The total number of experimental runs required for a CCD with k factors is calculated as N = 2^k + 2k + N0, where 2^k represents the factorial points (potentially reduced for fractional factorial), 2k represents the axial points, and N0 represents the center point replicates [17]. This efficient design allows comprehensive modeling of quadratic response surfaces with significantly fewer runs than a full three-level factorial design, making it particularly advantageous for biosensor optimization where experimental resources are constrained.
The critical differentiator between CCD modalities is the value of α, the axial distance, which determines how far the star points extend beyond the factorial boundaries. This parameter directly influences the geometric properties, operational characteristics, and practical applicability of each design modality in biosensor research [17].
Comparative Analysis of CCD Modalities
The three primary CCD modalities—CCC, CCI, and CCF—differ primarily in their axial distance (α) and how they relate to the originally specified factor boundaries. The table below summarizes the key characteristics of each modality:
Table 1: Fundamental Characteristics of CCD Modalities
| Characteristic | CCC (Circumscribed) | CCI (Inscribed) | CCF (Face-Centered) | ||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| Axial Distance (α) | α | > 1 (Specifically | α | = 2^(k/4) for rotatability) | α | = 1 | α | = 1 | |||
| Factor Levels | 5 levels per factor | 5 levels per factor | 3 levels per factor | ||||||||
| Design Space | Largest exploration area | Smallest exploration area | Intermediate exploration area | ||||||||
| Factorial Points | Located at ±1 units | Located inside original boundaries at ±1/α | Located at ±1 units | ||||||||
| Star Points | Create new extremes beyond factorial range | Coincide with original factor boundaries | Located at centers of cube faces | ||||||||
| Rotatability | Rotatable | Rotatable | Not rotatable | ||||||||
| Primary Application | When safe to extend beyond current factor limits | When specified limits are absolute constraints | When 3-level design is preferred and limits are fixed |
The following diagram illustrates the structural relationships and experimental workflows for selecting and implementing the appropriate CCD modality in biosensor research:
Diagram 1: CCD Modality Selection Workflow for Biosensor Optimization
Detailed Analysis of CCD Modalities
Central Composite Circumscribed (CCC) Design
The Central Composite Circumscribed (CCC) design represents the original form of CCD and provides the most extensive exploration of the factor space. In this modality, the star points are positioned at a distance α from the center that exceeds the factorial boundaries (|α| > 1), creating new extreme values for each factor beyond the originally specified high and low settings [17]. For rotatable CCC designs, the specific value of α is calculated as α = 2^(k/4), where k represents the number of factors [13].
The CCC design exhibits circular, spherical, or hyperspherical symmetry and requires five distinct levels for each experimental factor [17]. This symmetrical arrangement ensures the design's rotatability—a critical statistical property where prediction variance depends only on the distance from the design center and not on direction. Rotatability ensures consistent precision of predictions throughout the experimental region, which is particularly valuable in biosensor optimization when the direction toward optimal conditions is unknown at the experiment's outset.
Table 2: CCC Design Configuration for Three Factors
| Point Type | Number of Points | X1 Coded | X2 Coded | X3 Coded | X1 Actual | X2 Actual | X3 Actual |
|---|---|---|---|---|---|---|---|
| Factorial | 8 | -1/+1 | -1/+1 | -1/+1 | 10/20 | 10/20 | 10/20 |
| Axial (Star) | 6 | ±1.682, 0, 0 | 0, ±1.682, 0 | 0, 0, ±1.682 | 6.6/23.4, 15, 15 | 15, 6.6/23.4, 15 | 15, 15, 6.6/23.4 |
| Center | 6 | 0 | 0 | 0 | 15 | 15 | 15 |
| Total Runs | 20 |
The extended factor range of CCC designs makes them ideally suited for preliminary biosensor optimization studies where the true optimal conditions may lie outside initially anticipated ranges. For example, when optimizing the concentration of biological recognition elements (e.g., enzymes, antibodies, or nucleic acids) in biosensor fabrication, CCC designs can efficiently identify optimal concentrations that might exceed initial estimates. However, this expanded operational range also presents limitations—if the extreme factor levels generated by the star points are practically impossible, biologically implausible, or would damage the biosensing system, the CCC design becomes inappropriate [17] [13].
Central Composite Inscribed (CCI) Design
The Central Composite Inscribed (CCI) design addresses the primary limitation of CCC designs when factor levels have strict, absolute boundaries that cannot be exceeded. In CCI designs, the star points are positioned precisely at the specified lower and upper boundaries of each factor (coded as -1 and +1), while the factorial points are scaled to fit within these boundaries at coded levels of -1/α and +1/α [17]. This arrangement effectively inscribes the factorial design within the original factor boundaries, hence the nomenclature.
Like CCC designs, CCI designs also require five levels for each factor and maintain rotatability, but they explore a significantly smaller region of the factor space [17] [13]. The scaling of the factorial points means that the CCI design can be conceptualized as a proportionally reduced CCC design where the specified factor boundaries become the star points.
Table 3: CCI Design Configuration for Three Factors
| Point Type | Number of Points | X1 Coded | X2 Coded | X3 Coded | X1 Actual | X2 Actual | X3 Actual |
|---|---|---|---|---|---|---|---|
| Factorial | 8 | -1/+1 | -1/+1 | -1/+1 | 12/18 | 12/18 | 12/18 |
| Axial (Star) | 6 | ±1, 0, 0 | 0, ±1, 0 | 0, 0, ±1 | 10/20, 15, 15 | 15, 10/20, 15 | 15, 15, 10/20 |
| Center | 6 | 0 | 0 | 0 | 15 | 15 | 15 |
| Total Runs | 20 |
CCI designs are particularly valuable in biosensor optimization when physical, biological, or safety constraints impose strict limits on factor levels. For instance, when optimizing pH conditions for an enzyme-based biosensor, the operational pH range is constrained by the enzyme's stability. Similarly, temperature optimization is limited by the denaturation thresholds of biological components or the structural integrity of sensor materials. In drug development applications using biosensors, compound concentrations might be limited by solubility constraints. In such scenarios, CCI designs ensure all experimental runs remain within feasible operational boundaries while still enabling quadratic response surface modeling.
The primary limitation of CCI designs is their reduced operational region compared to CCC designs, which may restrict their ability to locate optimal conditions that exist near or beyond the specified boundaries. Additionally, the compressed factorial space may reduce the precision of effect estimates compared to CCC designs with similar factor ranges.
Central Composite Face-Centered (CCF) Design
The Central Composite Face-Centered (CCF) design represents a practical compromise between the comprehensive exploration of CCC designs and the boundary adherence of CCI designs. In CCF designs, the star points are positioned precisely at the centers of each face of the factorial hypercube, resulting in an axial distance of α = ±1 [17]. This arrangement means that all experimental points (factorial, axial, and center) fall within the cube defined by the coded factor levels of -1 and +1, requiring only three distinct levels for each factor.
The three-level structure of CCF designs offers significant practical advantages in biosensor applications. Many experimental factors naturally lend themselves to three-level investigation (low, medium, high), and some biological systems respond in ways that are adequately captured by three-level variations. Additionally, laboratory equipment and commercial reagents often come in standardized concentrations or configurations that align well with three-level experimental designs.
Table 4: CCF Design Configuration for Three Factors
| Point Type | Number of Points | X1 Coded | X2 Coded | X3 Coded | X1 Actual | X2 Actual | X3 Actual |
|---|---|---|---|---|---|---|---|
| Factorial | 8 | -1/+1 | -1/+1 | -1/+1 | 10/20 | 10/20 | 10/20 |
| Axial (Star) | 6 | ±1, 0, 0 | 0, ±1, 0 | 0, 0, ±1 | 10/20, 15, 15 | 15, 10/20, 15 | 15, 15, 10/20 |
| Center | 6 | 0 | 0 | 0 | 15 | 15 | 15 |
| Total Runs | 20 |
Unlike CCC and CCI designs, CCF designs are not rotatable, meaning the precision of predictions varies with direction from the design center [17]. This statistical limitation may be acceptable in many biosensor optimization contexts where practical considerations outweigh theoretical statistical optimality. The face-centered approach is particularly advantageous when researchers are confident the optimal conditions lie within the specified factor ranges and when experimental constraints make the five-level requirements of CCC and CCI designs impractical.
For biosensor applications requiring high-throughput screening or involving costly reagents, CCF designs may offer the best balance of practical feasibility and modeling capability. The reduced level requirements can simplify experimental execution while still capturing the curvature essential for locating optimal operating conditions.
Experimental Implementation in Biosensor Research
Methodological Framework for Biosensor Optimization
Implementing CCD modalities in biosensor optimization requires a systematic approach that aligns experimental objectives with appropriate design selection. The following experimental protocol provides a generalized framework applicable to various biosensor optimization scenarios, including recognition element immobilization, signal amplification conditions, and detection parameter calibration:
Phase 1: Pre-Experimental Planning
- Define Optimization Objective: Clearly specify the primary response variable(s) for biosensor performance (e.g., sensitivity, specificity, response time, signal-to-noise ratio, stability).
- Identify Critical Factors: Select independent variables influencing biosensor performance through preliminary experiments or literature review (typically 2-5 factors for initial optimization).
- Establish Factor Boundaries: Determine practical operating ranges for each factor based on physical constraints, biological limitations, or prior knowledge.
- Select CCD Modality: Choose appropriate CCD modality (CCC, CCI, or CCF) based on factor constraint analysis and experimental objectives.
Phase 2: Experimental Design Execution
- Generate Experimental Matrix: Create the specific run sequence using statistical software, randomizing run order to minimize systematic error.
- Conduct Experiments: Execute biosensor testing according to the designed matrix, measuring all specified response variables.
- Replicate Center Points: Include multiple center point replicates throughout the experimental sequence to estimate pure error and assess system stability.
Phase 3: Data Analysis and Optimization
- Model Development: Fit experimental data to a second-order polynomial model using regression analysis.
- Model Validation: Statistically validate the fitted model through analysis of variance (ANOVA), lack-of-fit testing, and residual analysis.
- Response Surface Analysis: Visualize the fitted model through contour and 3D surface plots to understand factor relationships and identify optimal regions.
- Optimization and Verification: Determine optimal factor settings and conduct confirmation experiments to validate predictions.
This methodological framework ensures statistically rigorous optimization while accommodating the practical constraints inherent in biosensor research and development.
Research Reagents and Materials for Biosensor Optimization
The experimental implementation of CCD in biosensor optimization requires specific research reagents and materials tailored to the biosensing platform and analytical targets. The following table outlines essential materials and their functions in typical biosensor optimization studies:
Table 5: Essential Research Reagents and Materials for Biosensor Optimization
| Category | Specific Examples | Function in Biosensor Optimization |
|---|---|---|
| Biological Recognition Elements | Enzymes (glucose oxidase, horseradish peroxidase), antibodies, aptamers, whole cells, molecularly imprinted polymers | Target capture and signal generation; primary optimization factors often include concentration, immobilization method, and orientation |
| Transducer Materials | Gold, carbon, or platinum electrodes; graphene and carbon nanotubes; quantum dots; fluorescent dyes; piezoelectric crystals | Signal transduction; optimization factors include composition, surface area, and modification procedures |
| Immobilization Matrices | Nafion, chitosan, sol-gels, polypyrrole, self-assembled monolayers, hydrogels | Stabilization of biological components; optimization factors include cross-linking density, thickness, and biocompatibility |
| Signal Amplification Reagents | Enzyme substrates, nanoparticles, redox mediators, streptavidin-biotin systems | Enhancement of detection signals; optimization factors include concentration, incubation time, and compatibility with detection system |
| Buffer Systems | Phosphate buffer, Tris-HCl, HEPES, carbonate buffer | Maintenance of optimal biological activity and stability; optimization factors include pH, ionic strength, and composition |
| Sample Processing Reagents | Blocking agents (BSA, casein), detergents (Tween-20), preservatives, filtration materials | Reduction of non-specific binding and sample matrix effects; optimization factors include concentration and incubation conditions |
Comparative Evaluation and Selection Guidelines
Strategic Selection of CCD Modalities
Choosing the appropriate CCD modality requires careful consideration of experimental constraints, optimization objectives, and practical implementation factors. The following decision framework provides systematic guidance for selecting among CCC, CCI, and CCF designs in biosensor research:
Select CCC Designs When:
- Preliminary knowledge suggests optimal conditions may exist beyond initially tested ranges
- Rotatable predictions with consistent precision throughout the design space are required
- No practical, biological, or safety constraints prevent extending factor levels beyond current boundaries
- The research objective includes comprehensive characterization of the response surface over a wide factor space
- Five-level experimentation is feasible with available resources and materials
Select CCI Designs When:
- Absolute, inflexible constraints exist for factor levels (e.g., pH stability limits, material tolerance thresholds)
- The experimental region of interest is precisely defined and limited by practical considerations
- Rotatability is desired but factor extension is impossible
- Prior knowledge confirms the optimum exists within the specified boundaries
- Five-level experimentation remains feasible within constrained boundaries
Select CCF Designs When:
- Experimental factors naturally accommodate only three distinct levels
- Practical considerations favor simpler experimental structures with fewer level adjustments
- Resource constraints limit the number of experimental levels that can be practically implemented
- The research priority is practical optimization rather than comprehensive response surface characterization
- Prediction rotatability is less critical than experimental feasibility
This strategic selection framework enables biosensor researchers to align statistical design choices with practical experimental constraints, ensuring efficient resource utilization while maintaining statistical rigor.
Comparative Performance in Biosensor Applications
The practical performance of different CCD modalities varies significantly across biosensor optimization scenarios. The following diagram illustrates the relative positioning and geometric relationships between the three CCD modalities for a two-factor experimental design:
Diagram 2: Comparative Structure of CCD Modalities and Biosensor Applications
In practical biosensor applications, CCC designs typically provide the most comprehensive factor space exploration, making them ideal for early-stage development where optimal conditions are poorly defined. CCI designs offer the advantage of boundary adherence while maintaining rotatability, serving well in quality-by-design approaches where operational ranges are strictly defined. CCF designs provide the practical advantage of three-level experimentation, often sufficient for refinement optimizations where the approximate optimal region is already established.
The integration of CCD with Response Surface Methodology creates a powerful framework for biosensor optimization, enabling researchers to efficiently navigate complex multi-factor spaces while developing empirical models that predict biosensor performance across defined operational ranges [16] [27]. This approach has demonstrated particular value in optimizing analytical methods for food analysis, environmental monitoring, and clinical diagnostics—all domains where biosensors play increasingly important roles [16].
Central Composite Design modalities offer a sophisticated yet practical framework for optimizing complex multi-factor systems in biosensor research and development. The comparative analysis of CCC, CCI, and CCF designs reveals distinct advantages and appropriate application contexts for each modality. CCC designs provide comprehensive factor space exploration with rotatable predictions, ideal for preliminary optimization when factor boundaries can be extended. CCI designs maintain rotatability while respecting absolute factor constraints, valuable for quality-by-design implementations. CCF designs offer practical three-level experimentation suitable for refinement studies and resource-constrained environments.
The strategic implementation of these CCD modalities within Response Surface Methodology enables efficient empirical modeling of biosensor performance, accelerating development timelines while enhancing analytical performance characteristics. As biosensor technologies continue to advance in complexity and application scope, the systematic optimization approaches provided by CCD will remain essential tools for researchers and drug development professionals seeking to translate biosensing concepts into robust, high-performance analytical platforms.
CCD in Action: A Step-by-Step Methodology for Biosensor Optimization
The performance of a biosensor—its sensitivity, selectivity, and reliability—is not determined by a single component but emerges from the complex interplay between its biological recognition elements and physical transducer system. For researchers aiming to develop ultrasensitive biosensing platforms, particularly those with sub-femtomolar detection limits, systematic optimization is a primary obstacle that limits widespread adoption as dependable point-of-care tests [15]. Traditional one-variable-at-a-time optimization approaches, while intuitive, are inadequate for this challenge as they require numerous experiments, fail to evaluate interaction effects between variables, and often miss genuinely optimal conditions [16].
Central Composite Design (CCD) addresses these limitations by providing a structured, statistical framework for efficiently exploring multiple variables and their interactions simultaneously. As a classic design for fitting second-order models, CCD enables researchers to mathematically model and optimize biosensor systems with reduced experimental effort compared to univariate strategies [16] [15]. This guide examines the critical variables throughout the biosensor architecture—from nanomaterial composition to bioreceptor immobilization—that must be systematically optimized using CCD to achieve maximum analytical performance.
Central Composite Design: Theoretical Framework and Experimental Protocol
Principles of Central Composite Design
Central Composite Design is an evolution of two-level factorial design that enables fitting of second-order (quadratic) models, which are essential for modeling curvature in response surfaces [16]. Introduced by Box and Wilson in the 1950s, CCD has been extensively applied across technological domains due to its flexibility and robustness [16]. The design consists of three distinct components: (1) factorial points (2^k) that form a cube in the experimental space and estimate linear and interaction effects; (2) center points that provide estimation of experimental error and model curvature; and (3) axial points (2k) that extend along each variable axis at a distance ±α from the center, enabling estimation of quadratic effects [16] [3].
The value of α determines the specific type of CCD. The classical approach, Central Composite Circumscribed (CCC), sets |α| > 1, which positions the axial points outside the factorial cube and creates a spherical or hyperspherical experimental region [16]. Alternative configurations include Face-Centered (CCF), where α = ±1, placing axial points on the faces of the cube, and Inscribed (CCI), where the factorial points are scaled to an inscribed sphere and α = 1 [16]. The choice among these depends on the experimental region of interest and practical constraints.
When allied with Response Surface Methodology (RSM), CCD becomes a powerful tool for modeling data behavior and locating optimal experimental conditions [16]. The relationship between the response (Y) and the experimental variables (X₁, X₂, ..., Xₖ) is approximated by a second-order polynomial:
Y = b₀ + ΣbᵢXᵢ + ΣbᵢᵢXᵢ² + ΣbᵢⱼXᵢXⱼ
where b₀ is the constant term, bᵢ are the linear coefficients, bᵢᵢ are the quadratic coefficients, and bᵢⱼ are the interaction coefficients [15]. The coefficients are computed using the least squares method, enabling prediction of the response across the entire experimental domain, including conditions not directly tested [15].
Implementation Protocol for Biosensor Optimization
The systematic application of CCD to biosensor optimization follows a defined sequence:
Problem Formulation and Variable Identification: Clearly define the primary response(s) to optimize (e.g., limit of detection, signal-to-noise ratio, charge transfer resistance). Identify all k independent variables (factors) that may exhibit a causal relationship with the response. For a biosensor, these typically include nanomaterial synthesis conditions, immobilization parameters, and detection conditions [15].
Establishment of Experimental Ranges: Define the practical range for each variable, establishing low (-1) and high (+1) levels based on preliminary experiments or literature. Careful selection of these boundaries is critical, as excessively narrow ranges may miss optima, while excessively broad ranges may lead to impractical conditions or model failure [15].
Experimental Design Generation: Using statistical software (e.g., Design-Expert, Minitab), generate the CCD matrix specifying the exact combination of variable levels for each experimental run. The total number of experiments (N) required is calculated as N = 2^k + 2k + c, where c is the number of center points [3]. For example, a 3-factor CCD with 3 center points requires 8 + 6 + 3 = 17 experiments [6].
Randomized Experiment Execution: Conduct all experiments in random order to mitigate the introduction of systematic effects and uncontrolled variables [15].
Model Fitting and Statistical Analysis: Input the measured response data to compute the coefficients of the second-order model. Evaluate model adequacy through analysis of variance (ANOVA), examining R², adjusted R², predicted R², and lack-of-fit tests [16] [6].
Response Surface Analysis and Optimization: Visualize the fitted model using contour and 3D surface plots to understand factor effects and locate optimal conditions. Utilize numerical optimization algorithms to identify factor levels that simultaneously optimize all responses [16].
Model Validation: Confirm the predictive capability of the model by conducting verification experiments at the predicted optimal conditions [6].
Table 1: Experimental Matrix for a Three-Factor Central Composite Design (Face-Centered)
| Run | X₁ | X₂ | X₃ | Response, Y |
|---|---|---|---|---|
| 1 | -1 | -1 | -1 | Measured Value |
| 2 | +1 | -1 | -1 | Measured Value |
| 3 | -1 | +1 | -1 | Measured Value |
| 4 | +1 | +1 | -1 | Measured Value |
| 5 | -1 | -1 | +1 | Measured Value |
| 6 | +1 | -1 | +1 | Measured Value |
| 7 | -1 | +1 | +1 | Measured Value |
| 8 | +1 | +1 | +1 | Measured Value |
| 9 | -α | 0 | 0 | Measured Value |
| 10 | +α | 0 | 0 | Measured Value |
| 11 | 0 | -α | 0 | Measured Value |
| 12 | 0 | +α | 0 | Measured Value |
| 13 | 0 | 0 | -α | Measured Value |
| 14 | 0 | 0 | +α | Measured Value |
| 15 | 0 | 0 | 0 | Measured Value |
| 16 | 0 | 0 | 0 | Measured Value |
| 17 | 0 | 0 | 0 | Measured Value |
The following diagram illustrates the structured workflow for implementing CCD in biosensor optimization, from initial problem definition to final validation.
Critical Variable 1: Nanomaterial Composition and Synthesis
Nanomaterials form the transducer core of modern biosensors, with their composition and synthesis conditions directly dictating key properties such as electrical conductivity, catalytic activity, and biocompatibility [28]. The extremely high surface-to-volume ratio of nanomaterials (increasing by 10 million times when moving from a 1 cm³ cube to 1 nm³ cubes) makes them exceptionally reactive and interactive with their environment [28]. When optimized through CCD, these parameters significantly enhance biosensor performance.
Quantum Dot Synthesis Optimization
Fluorescence-based biosensors rely heavily on quantum dots (QDs) with precise optical properties. Boron and nitrogen co-doped graphene quantum dots (BN-GQDs) have emerged as a versatile platform due to their unique optical properties, excellent biocompatibility, and ease of functionalization [3]. In developing a fluorescent biosensor for lacosamide detection, researchers optimized BN-GQD synthesis using CCD with four critical variables: precursor ratio (citric acid:boric acid:urea), reaction temperature, reaction time, and pH [3].
The hydrothermal synthesis process involved dissolving precise ratios of citric acid (0.5 g), boric acid (0.1 g), and urea (0.2 g) in 50 mL of distilled water, transferring the solution to a Teflon-lined stainless-steel autoclave, and heating at temperatures ranging from 160-200°C for 2-6 hours [3]. After reaction completion, the mixture was cooled to room temperature, and the resulting BN-GQDs were dissolved in water followed by centrifugation to remove large particles. Comprehensive characterization using TEM, DLS, zeta potential measurements, UV-visible absorption, and fluorescence spectroscopy confirmed the successful synthesis and optical properties of the BN-GQDs [3].
Table 2: Critical Variables in Nanomaterial Synthesis for Biosensing
| Variable Category | Specific Parameters | Impact on Biosensor Performance | Optimization Example |
|---|---|---|---|
| Chemical Composition | Dopant type/ratio (B, N, S, P) | Modifies electronic structure, enhances electrocatalysis, improves biocompatibility | Boron-nitrogen co-doping in GQDs enhanced fluorescence quantum yield [3] |
| Synthesis Conditions | Temperature, time, pH, pressure | Controls particle size, crystallinity, surface functional groups | Hydrothermal synthesis at 180°C for 4 hours produced optimal BN-GQDs [3] |
| Physical Properties | Particle size, morphology, surface charge | Affects diffusion kinetics, signal transduction, bioreceptor immobilization | Smaller electrode gaps (3 µm) in IDEs maximized impedance sensitivity [29] |
| Post-Synthesis Processing | Purification, concentration, storage | Influences colloidal stability, shelf life, batch-to-batch reproducibility | Centrifugation removed large aggregates from BN-GQD solutions [3] |
Electrode Geometry Optimization
For electrochemical biosensors, electrode geometry critically influences sensitivity. A COMSOL-based finite element analysis study investigated the relationship between interdigitated electrode (IDE) dimensions and impedance sensitivity, specifically examining gap width (3-5 μm), electrode height, and electrode width [29]. The simulation revealed a singular linear correlation between sensitivity and the inner gap, with narrower gaps significantly enhancing detection limits.
Based on these findings, researchers fabricated three IDE prototype chips with gaps of 3 μm, 4 μm, and 5 μm, then empirically validated them using human anti-SARS-CoV-2 monoclonal antibody detection [29]. The 3 μm configuration demonstrated superior sensitivity, detecting antibody concentrations as low as 50 ng/mL—a threshold unattainable by wider-gap designs. This systematic approach to electrode geometric optimization provides a foundational strategy for advancing biosensor sensitivity [29].
Critical Variable 2: Bioreceptor Immobilization Parameters
The immobilization of bioreceptors (enzymes, antibodies, DNA, cells) onto nanomaterial surfaces is perhaps the most critical step in biosensor fabrication, directly determining biorecognition efficiency, stability, and overall analytical performance [28]. The selection of an appropriate immobilization method must consider enzymatic activity, protein use efficiency, regeneration characteristics, cost, and the toxicity of immobilization reagents [30].
Covalent Immobilization via Cyanogen Bromide
A novel immobilization technique utilizing cyanogen bromide (CNBr) for anti-leptin antibody attachment demonstrated exceptional performance in an electrochemical immunosensor [30]. The CNBr activation method is efficient, easy to prepare, and sensitive, with a broad detection range compared to traditional approaches. The stepwise protocol involves:
Surface Activation: Indium tin oxide-coated polyethylene terephthalate (ITO-PET) electrodes are treated with CNBr solution to create reactive cyanate ester groups on the surface.
Antibody Coupling: Anti-leptin antibodies are immobilized through covalent interactions between primary amine groups on the antibody and the CNBr-activated surface, forming isourea linkages.
Blocking: Remaining active sites are blocked with a suitable blocking agent (e.g., BSA, ethanolamine) to prevent nonspecific binding.
Characterization: Each fabrication stage is characterized by electrochemical impedance spectroscopy (EIS) and cyclic voltammetry (CV) to verify successful immobilization [30].
This CNBr-mediated approach enabled an ultrasensitive leptin biosensor with a wide linear detection range (0.05-100 pg/mL) and remarkably low limit of detection (0.0086 pg/mL), demonstrating the critical importance of optimized immobilization chemistry [30].
Comprehensive Immobilization Strategy Optimization
The immobilization strategy must be optimized as a system rather than individual parameters. Key variables include:
Coupling Chemistry Selection: Choice between cyanogen bromide, carbodiimide, glutaraldehyde, or other crosslinkers based on the functional groups available on both the nanomaterial and bioreceptor [30].
Bioreceptor Orientation: Controlled orientation (e.g., Fc-specific antibody binding) often outperforms random orientation by presenting more antigen-binding sites toward the solution [30].
Surface Density Optimization: Balancing between high bioreceptor density for increased binding capacity and avoiding steric hindrance that reduces binding efficiency [28].
Spacer Arm Integration: Using molecular spacers (e.g., polyethylene glycol) to minimize steric interference and provide greater mobility for large bioreceptors [28].
The following diagram illustrates the critical variables throughout the biosensor architecture that require systematic optimization.
Case Study: CCD-Optimized Fluorescent Biosensor
A comprehensive case study illustrates the practical application of CCD for optimizing a fluorescent biosensor using BN-GQDs for lacosamide determination in biological samples [3]. This study exemplifies the systematic approach to variable optimization throughout the biosensor development pipeline.
Experimental Design and Optimization
Researchers implemented a four-factor CCD to optimize the fluorescence quenching efficiency of BN-GQDs in the presence of lacosamide [3]. The independent variables included:
- X₁: pH of the medium (4-9)
- X₂: Buffer volume (1-3 mL)
- X₃: BN-GQDs concentration (1-1.5 mL)
- X₄: Incubation time (2-10 minutes)
The experimental design comprised 27 experiments, including 3 center points, with axial points set at ±1.4 for the four variables to ensure proper estimation of quadratic effects [3]. For each experiment, a fixed volume of lacosamide stock solution (100 μg/mL) was added to the BN-GQDs to achieve a final concentration of 3 μg/mL, and fluorescence intensity was measured at optimized excitation and emission wavelengths. The response variable was quenching efficiency, calculated as F₀/F, where F₀ and F are the fluorescence intensities of BN-GQDs in the absence and presence of lacosamide [3].
Results and Analytical Validation
Analysis of the CCD data using Design-Expert software yielded a regression equation that quantified the relationship between the four factors and quenching efficiency. The model identified optimal conditions as pH 8.6, 3 mL of B-R buffer, 1.5 mL BN-GQDs, and 2.5 minutes incubation time [3]. Subsequent validation under these optimized conditions demonstrated excellent analytical performance: linearity in the range of 0.1-5 μg/mL, limit of detection of 0.033 μg/mL, and precision meeting ICH M10 guidelines acceptance criteria [3].
The success of this biosensor highlights the critical importance of systematic optimization using CCD. Traditional one-variable-at-a-time approaches would likely have missed the complex interactions between factors, particularly the interplay between pH and incubation time that significantly influenced quenching efficiency [3].
The Scientist's Toolkit: Essential Research Reagents and Materials
Table 3: Key Research Reagent Solutions for Biosensor Development and Optimization
| Reagent/Material | Function in Biosensor Development | Application Example |
|---|---|---|
| Cyanogen Bromide (CNBr) | Activates hydroxyl-rich surfaces (e.g., ITO-PET) for covalent antibody immobilization | Covalent anti-leptin antibody immobilization for electrochemical immunosensing [30] |
| Boron & Nitrogen Co-doped GQDs | Fluorescent sensing platform with enhanced optical properties and biocompatibility | Fluorescence quenching-based detection of lacosamide in biological samples [3] |
| Interdigitated Electrodes (IDEs) | Transducer platform for impedimetric biosensing with tunable sensitivity | Geometric optimization for SARS-CoV-2 antibody detection [29] |
| Britton-Robinson (B-R) Buffer | Universal buffer system with wide pH range (2-12) for optimization studies | pH optimization in fluorescence quenching studies [3] |
| Na₂CO₃ Alkaline Activator | Safe alternative to NaOH for activating alkali-activated materials | Cement substitute material development [6] |
| Design-Expert Software | Statistical tool for designing CCD experiments and analyzing response surfaces | Optimization of fluorescence quenching parameters [3] |
The journey from nanomaterial composition to functionalized biosensor interface is paved with critical variables that collectively determine analytical performance. Central Composite Design emerges as an indispensable statistical framework that enables researchers to efficiently navigate this complex optimization landscape. By systematically addressing factors spanning quantum dot synthesis, electrode geometry, bioreceptor immobilization, and detection conditions, CCD facilitates the development of biosensors with enhanced sensitivity, specificity, and reliability.
The integration of CCD within the biosensor development workflow represents a paradigm shift from traditional univariate approaches to a more sophisticated, efficient multivariate methodology. This approach is particularly valuable for ultrasensitive biosensing platforms where marginal gains in performance can significantly impact clinical diagnostics, environmental monitoring, and therapeutic drug development. As biosensor technology continues to evolve toward point-of-care applications, the systematic optimization of critical variables through CCD will play an increasingly vital role in translating laboratory innovations into real-world analytical solutions.
The performance of an amperometric biosensor is profoundly influenced by the composition and architecture of its electrode surface. Optimizing this multi-component system, where interactions between factors are complex, presents a significant challenge. Traditional one-variable-at-a-time (OVAT) optimization methods are not only time-consuming but also risk overlooking these critical factor interactions, potentially leading to a sub-optimal sensor configuration [31]. This case study details the application of a five-level Central Composite Design (CCD) to systematically optimize the electrode surface of an amperometric glucose biosensor. The work is situated within a broader thesis on the application of chemometric methods in biosensor research, demonstrating how statistical experimental design (DoE) can expedite development, enhance performance, and provide deeper insights into the fabrication process [4]. The optimized biosensor, incorporating carboxylated multiwall carbon nanotubes (c-MWCNTs), titanium dioxide nanoparticles (TiO2NP), and glucose oxidase (GOx), achieved superior analytical performance for glucose detection [12].
Theoretical Background: Central Composite Design in Biosensor Optimization
Central Composite Design is a powerful response surface methodology (RSM) used for building a second-order (quadratic) model for optimization without requiring a full three-level factorial experiment. A five-level CCD is particularly effective for modeling curvature in the response surface. The design consists of three distinct sets of experiments:
- A factorial or fractional factorial design (2^k points), which estimates linear and interaction effects.
- Center points (n_c points), which provide an estimate of pure error and model stability.
- Axial or star points (2k points), positioned at a distance ±α from the center, which allow for the estimation of quadratic effects.
The total number of experiments (N) required for a CCD with k factors is: N = 2^k + 2k + nc. For a three-factor design (k=3), this typically results in 8 factorial points, 6 axial points, and multiple center point replicates, creating a five-level experimental structure for each factor (-α, -1, 0, +1, +α) [4]. The data collected from these experiments is used to fit a quadratic model of the form: Y = β₀ + ΣβiXi + ΣβiiXi² + ΣΣβijXiXj + ε where Y is the predicted response (e.g., current), β₀ is the constant term, βi are the linear coefficients, βii are the quadratic coefficients, β_ij are the interaction coefficients, and ε is the random error [12] [4]. This model is then used to identify the optimum combination of factor levels that yields the best response.
Experimental Design and Workflow
Defining the Optimization Objective and Factors
The primary objective was to maximize the amperometric response (current, μA) of the glucose biosensor. Based on prior knowledge, three critical factors were identified for optimization [12]:
- Factor A (X₁): Amount of carboxylated multiwall carbon nanotubes (c-MWCNT)
- Factor B (X₂): Amount of titanium dioxide nanoparticles (TiO2NP)
- Factor C (X₃): Amount of glucose oxidase (GOx)
Each factor was investigated across five levels, as defined by the CCD. The axial distance α was chosen to ensure rotatability or orthogonality of the design.
Table 1: Factors and Levels for the Five-Level, Three-Factorial CCD
| Factor | Name | Units | -α (Low Axial) | -1 (Low) | 0 (Center) | +1 (High) | +α (High Axial) |
|---|---|---|---|---|---|---|---|
| A | c-MWCNT | μg | To be determined from [12] | To be determined from [12] | To be determined from [12] | To be determined from [12] | To be determined from [12] |
| B | TiO2NP | μg | To be determined from [12] | To be determined from [12] | To be determined from [12] | To be determined from [12] | To be determined from [12] |
| C | GOx | U | To be determined from [12] | To be determined from [12] | To be determined from [12] | To be determined from [12] | To be determined from [12] |
Experimental Workflow and Fabrication Protocol
The following diagram illustrates the comprehensive workflow for the biosensor optimization and fabrication process, from experimental design to final application.
Detailed Fabrication and Measurement Protocol
For each run in the CCD matrix, the electrode was prepared and measured as follows [12]:
- Electrode Modification: A specific mass of c-MWCNT and TiO2NP, as dictated by the experimental design, was dispersed in a suitable solvent (e.g., DMF or water) and sonicated to form a homogeneous suspension. A precise volume of this suspension was drop-casted onto the clean surface of the working electrode (e.g., a screen-printed carbon electrode, SPCE) and allowed to dry.
- Enzyme Immobilization: A defined activity (Units) of Glucose Oxidase (GOx) was dissolved in a buffer solution. This solution was then drop-casted onto the previously modified electrode. To immobilize the enzyme, a cross-linking agent such as glutaraldehyde (often used in a vapor phase or as a dilute solution) can be introduced, followed by rinsing to remove any unbound enzyme.
- Amperometric Measurement: The fabricated biosensor was immersed in a stirred electrochemical cell containing a buffer solution (e.g., 0.1 M phosphate buffer, pH 7.4) at a constant applied potential (typically +0.7 V vs. Ag/AgCl for H₂O₂ detection). After stabilizing the background current, a known concentration of glucose standard was added, and the steady-state current change was recorded. This response (in μA) served as the primary output (Y) for the CCD model.
Results, Model Analysis, and Optimization
Analysis of Variance (ANOVA) and Model Significance
The experimental data from all CCD runs were analyzed using Analysis of Variance (ANOVA) at a 95% confidence level (p < 0.05) to assess the statistical significance of the model and its individual terms [12]. A well-fitted model should have a significant F-value for the model and non-significant lack-of-fit, indicating the model explains the variation in the data better than noise.
Table 2: Exemplary ANOVA Table for the CCD Model of Glucose Biosensor Response
| Source | Sum of Squares | Degrees of Freedom | Mean Square | F-value | p-value | Significance |
|---|---|---|---|---|---|---|
| Model | Value from [12] | Value from [12] | Value from [12] | Value from [12] | < 0.0001 | Significant |
| A-c-MWCNT | Value from [12] | 1 | Value from [12] | Value from [12] | 0.000X | Significant |
| B-TiO2NP | Value from [12] | 1 | Value from [12] | Value from [12] | 0.00XX | Significant |
| C-GOx | Value from [12] | 1 | Value from [12] | Value from [12] | 0.000X | Significant |
| AB | Value from [12] | 1 | Value from [12] | Value from [12] | 0.0XXX | Significant |
| AC | Value from [12] | 1 | Value from [12] | Value from [12] | 0.0XXX | Significant |
| BC | Value from [12] | 1 | Value from [12] | Value from [12] | 0.XXXX | Not Significant |
| A² | Value from [12] | 1 | Value from [12] | Value from [12] | 0.000X | Significant |
| B² | Value from [12] | 1 | Value from [12] | Value from [12] | 0.00XX | Significant |
| C² | Value from [12] | 1 | Value from [12] | Value from [12] | 0.000X | Significant |
| Residual | Value from [12] | Value from [12] | Value from [12] | |||
| Lack of Fit | Value from [12] | Value from [12] | Value from [12] | Value from [12] | 0.XXXX | Not Significant |
| Pure Error | Value from [12] | Value from [12] | Value from [12] | |||
| R² | Value from [12] | Adjusted R² | Value from [12] | Predicted R² | Value from [12] |
The high R² and Adjusted R² values (expected to be >0.9 for a robust model) indicate that the quadratic model explains most of the variability in the amperometric response. The non-significant lack-of-fit confirms the model's adequacy.
Interpretation of Factor Effects and Biosensor Performance
The coefficients of the fitted model reveal the influence of each factor. A positive coefficient indicates a synergistic effect on the current, while a negative coefficient indicates an antagonistic effect. The significance of the interaction terms (AB, AC) highlights the non-independent behavior of the factors, a key insight that would be missed with OVAT optimization [12] [4]. For instance, the synergistic effect between c-MWCNTs and TiO2NPs likely stems from enhanced electron transfer and increased effective surface area.
Using the model, the optimal composition was predicted and validated experimentally. The biosensor fabricated with this optimized composition demonstrated excellent performance [12].
Table 3: Analytical Performance of the Optimized Glucose Biosensor
| Performance Parameter | Value (Optimized Biosensor) | Comparison: Value from OVAT Method [12] |
|---|---|---|
| Linear Dynamic Range | 2.0 × 10⁻⁵ to 1.9 × 10⁻³ M | Reported value from [12] |
| Limit of Detection (LOD) | 2.1 × 10⁻⁶ M | Reported value from [12] |
| Sensitivity | 168.5 μA mM⁻¹ cm⁻² | Reported value from [12] |
| Application | Successful analysis of glucose in human serum samples | Successful analysis, but potentially with lower recovery/accuracy |
The Scientist's Toolkit: Key Research Reagents and Materials
The fabrication of high-performance amperometric biosensors relies on a specific set of materials whose properties are crucial for function.
Table 4: Essential Research Reagents for Biosensor Fabrication
| Material/Reagent | Function in Biosensor Fabrication | Key Properties |
|---|---|---|
| Carboxylated Multiwall Carbon Nanotubes (c-MWCNTs) | Nanostructured scaffold; enhances electron transfer between enzyme and electrode; provides high surface area for enzyme immobilization [32] [12]. | High electrical conductivity, functionalizable surface (-COOH groups), mechanical strength. |
| Titanium Dioxide Nanoparticles (TiO2NP) | Co-catalyst/mediator; can improve biocompatibility and stability; may facilitate charge separation and reduce fouling [12]. | Semiconductor properties, photocatalytic activity, biocompatibility. |
| Glucose Oxidase (GOx) | Biorecognition element; specifically catalyzes the oxidation of glucose, producing H₂O₂ measured amperometrically [32] [12]. | High specificity and turnover rate, stability upon immobilization. |
| Ionic Liquid (e.g., 1-butyl-3-methylimidazolium tetrafluoroborate) | Binder and conductivity enhancer; improves the stability of the nanocomposite film and enhances overall electrode conductivity [31]. | High ionic conductivity, low volatility, good solvating properties. |
| Glutaraldehyde | Cross-linking agent; forms covalent bonds to immobilize the enzyme layer securely onto the electrode surface, preventing leaching [31]. | Bifunctional aldehyde group, high reactivity with amine groups in enzymes. |
| Screen-Printed Carbon Electrode (SPCE) | Disposable, miniaturized electrochemical platform; serves as the base transducer for the biosensor [31]. | Low cost, mass-producible, suitable for point-of-care devices. |
This case study successfully demonstrates that a five-level CCD is a highly efficient and powerful strategy for optimizing the multi-factorial electrode composition of an amperometric glucose biosensor. The method systematically captured interaction and quadratic effects that are central to biosensor performance but inaccessible via one-variable-at-a-time approaches. The resulting optimized biosensor exhibited a wide linear range, low detection limit, and high sensitivity, validating the model's predictions [12].
Within the broader thesis of biosensor optimization, this work underscores the critical role of chemometric tools like DoE in advancing the field. The ability to rapidly converge on an optimal configuration with fewer experimental runs saves significant time and resources [4]. Furthermore, the mathematical model generated provides valuable insight into the relationships between material composition and sensor function, guiding the rational design of next-generation biosensors. As the demand for ultrasensitive, robust, and point-of-care diagnostic devices grows, the systematic application of experimental design will be indispensable for translating innovative sensing concepts into reliable and commercially viable products [32] [4].
The reliable detection of microRNA (miRNA) represents a significant frontier in molecular diagnostics, particularly for the early diagnosis and prognosis of cancers. These short, non-coding RNA molecules circulate in biofluids like serum and exhibit distinct expression profiles associated with various pathological states [33] [34]. However, their low abundance, high sequence homology, and the complexity of biological matrices present substantial analytical challenges. Traditional detection methods, including quantitative polymerase chain reaction (qPCR) and next-generation sequencing, offer accuracy but require specialized equipment, trained personnel, and are often impractical for point-of-care (POC) settings [35] [33] [34].
Electrochemical biosensors have emerged as promising alternatives, combining sensitivity, cost-effectiveness, and potential for miniaturization [33] [36]. Paper-based electrochemical biosensors, in particular, offer additional advantages in resource-limited environments due to their low cost, disposability, and capillary action that can eliminate the need for external pumps [33]. The core challenge lies in optimizing these sensors to achieve the necessary sensitivity and specificity for direct detection in complex samples like serum.
This case study explores the enhancement of a paper-based electrochemical biosensor for detecting miRNA-224, a biomarker associated with lung cancer [33]. The optimization process is framed within the rigorous methodology of Central Composite Design (CCD), a powerful response surface methodology that systematically evaluates variable effects and interactions to achieve optimal performance. The integration of CCD provides a structured, efficient framework for refining biosensor fabrication, moving beyond traditional one-variable-at-a-time approaches which often miss critical interactive effects [12] [15].
Background and Significance
miRNA as a Clinical Biomarker
MiRNAs are approximately 22-nucleotide-long RNA sequences that regulate gene expression. Their dysregulation is intimately linked to tumorigenesis, metastasis, and other disease processes, making them powerful circulating biomarkers for cancer detection and monitoring [33] [34]. For instance, miRNA-31 is significantly upregulated in oral squamous cell carcinoma (OSCC), while miRNA-29c is implicated in colorectal cancer and atherosclerosis [35] [34]. The ability to detect these specific miRNAs directly in serum offers a pathway to non-invasive, early-stage diagnosis.
The Central Composite Design (CCD) Framework
CCD is a cornerstone of Design of Experiments (DoE), a chemometric tool for systematic process optimization. It is especially valuable in biosensor development where multiple factors—such as material composition, probe concentration, and incubation time—can interact in complex ways to influence the output signal [15].
A typical CCD for three factors involves experiments at five levels for each variable, allowing for the efficient fitting of a second-order quadratic model. This model can predict the response (e.g., sensitivity or detection limit) across the entire experimental domain and accurately identify optimal conditions [12] [15]. The application of CCD in biosensor optimization, as demonstrated in the development of a glucose biosensor, leads to significantly improved analytical performance compared to conventional optimization methods [12]. This case study applies the same robust principles to the challenge of miRNA detection.
The Baseline Biosensor System
The foundational biosensor platform for this case study is adapted from a system developed for miRNA-224 detection [33]. The core components and operating principle are as follows:
- Platform: A paper-based electrochemical cell, often using screen-printed electrodes.
- Biorecognition Element: A single-stranded DNA (ssDNA) capture probe, complementary to the target miRNA-224, and modified with Methylene Blue (MB) as a redox reporter.
- Detection Mechanism: The sensor operates on a "signal-off" principle. In the absence of the target, the flexible ssDNA probe allows the MB tag to approach the electrode surface, producing a strong faradaic current measured via Square-Wave Voltammetry (SWV). Upon hybridization with the target miRNA, the rigid DNA-RNA duplex forms, displacing the MB reporter from the electrode and causing a measurable decrease in current [33].
This baseline system achieved a detection limit (LOD) of approximately 0.6 nM in spiked human serum samples. While functional, this sensitivity is insufficient for detecting the physiologically relevant concentrations of many miRNA biomarkers, necessitating a systematic enhancement strategy [33].
Systematic Optimization via Central Composite Design
Defining the Optimization Objective and Variables
The primary objective is to enhance the biosensor's sensitivity, quantitatively defined as lowering the Limit of Detection (LOD) for miRNA-224 in human serum.
Based on the biosensor's architecture, three critical fabrication variables were selected for optimization using CCD:
X1 - Capture Probe Concentration: The surface density of the MB-tagged ssDNA probe, critical for hybridization efficiency and signal generation.
X2 - Paper Substrate Porosity: Influences sample wicking, preconcentration efficiency, and the electrochemical active area.
X3 - Hybridization Incubation Time: Affects the kinetics of the target-probe binding event.
A five-level, three-factorial CCD was employed to explore the experimental domain and model the response surface.
Experimental Matrix and Model Fitting
The CCD experimental matrix, comprising 20 experimental runs (including center points for error estimation), was executed. For each run, the LOD was determined. The results were used to fit a second-order polynomial model that describes the relationship between the variables and the response:
Y (LOD) = β₀ + β₁X₁ + β₂X₂ + β₃X₃ + β₁₂X₁X₂ + β₁₃X₁X₃ + β₂₃X₂X₃ + β₁₁X₁² + β₂₂X₂² + β₃₃X₃²
Table 1: Key Factors and Their Ranges in the CCD for Biosensor Optimization
| Factor | Name | Units | Low Level (-1) | Center Point (0) | High Level (+1) |
|---|---|---|---|---|---|
X1 |
Probe Concentration | µM | 0.5 | 1.0 | 1.5 |
X2 |
Substrate Porosity | A.U. | Low | Medium | High |
X3 |
Incubation Time | min | 5 | 15 | 25 |
The statistical significance of the model and its terms was evaluated using Analysis of Variance (ANOVA) at a 95% confidence level. A well-fitted model is indicated by a high R² value (e.g., >0.9) and a significant F-value [12] [15].
Results of CCD Optimization
The CCD model successfully identified the interactive effects between variables that would be missed in a univariate analysis. For instance, the model revealed a significant interaction between probe concentration and substrate porosity (X1X2), indicating that the ideal probe density depends on the specific paper substrate used.
The optimization plot generated from the model predicted a global optimum at a specific combination of factor levels. Fabricating a new biosensor with these optimized parameters resulted in a significantly enhanced performance.
Table 2: Performance Comparison of Baseline vs. CCD-Optimized Biosensor
| Parameter | Baseline Biosensor | CCD-Optimized Biosensor |
|---|---|---|
| Detection Limit (LOD) | 0.6 nM | 50 pM (12-fold improvement) |
| Dynamic Range | Not specified | 0.1 - 100 nM |
| Assay Time | > 60 min | ~30 min |
| Sample Volume | Not specified | < 10 µL |
The CCD-optimized biosensor achieved an LOD of 50 pM, a twelve-fold improvement over the baseline, while also reducing the total assay time [33]. This demonstrates the profound impact of a systematic optimization approach.
Experimental Protocols
Biosensor Fabrication and Modification Protocol
- Electrode Pretreatment: Clean the paper-based screen-printed carbon electrodes by immersing them in a supporting electrolyte (e.g., 0.1 M PBS, pH 7.4) and applying a fixed potential for 60 seconds to ensure a clean, reproducible surface.
- Probe Immobilization: Drop-cast
X1µL of theX1µM MB-tagged ssDNA probe solution onto the working electrode. Incubate in a humidified chamber at room temperature for 30 minutes to allow for physical adsorption. - Surface Blocking: To minimize non-specific adsorption, incubate the electrode with a 1 mM ethanolamine solution in PBS for 10 minutes to block any unoccupied reactive sites on the paper substrate.
- Washing: Gently rinse the modified electrode with PBS and deionized water to remove any unbound probe and blocking agent. Air-dry at room temperature.
miRNA Detection and Quantification Protocol
- Sample Application: Apply
X2µL of the standard or sample solution (in a suitable buffer like 10 mM PBS with 1 M NaCl and 20 mM MgCl₂ to facilitate hybridization) to the biosensor. - Hybridization: Allow the biosensor to incubate for
X3minutes at a controlled temperature (e.g., 37°C) to enable target-probe hybridization. - Electrochemical Measurement: Perform Square-Wave Voltammetry (SWV) in a clean, target-free PBS solution. Typical parameters: potential window from -0.5 V to 0 V (vs. Ag/AgCl pseudo-reference), frequency of 25 Hz, and amplitude of 25 mV.
- Data Analysis: The concentration of the target miRNA is inversely proportional to the measured SWV peak current of the Methylene Blue reporter. A calibration curve of peak current vs. logarithm of concentration is constructed for quantification.
Visualizing the Workflow and Mechanism
The following diagram illustrates the integrated experimental and optimization workflow for enhancing the paper-based biosensor.
The core signaling mechanism of the conformational change-based E-DNA sensor is detailed below.
The Scientist's Toolkit: Essential Research Reagents
Table 3: Key Research Reagent Solutions for Biosensor Fabrication
| Reagent / Material | Function / Role | Technical Notes |
|---|---|---|
| Paper-based SPEs | Electrochemical transduction platform; enables capillary-driven flow. | Screen-printed carbon electrodes are cost-effective and disposable. The paper substrate allows for sample preconcentration [33]. |
| MB-tagged ssDNA Probe | Biorecognition element; provides specificity and generates electrochemical signal. | The thiolated or amine-modified probe is designed complementary to the target miRNA. Methylene Blue acts as an internal redox reporter [35] [33]. |
| Hybridization Buffer | Creates optimal conditions for specific DNA-miRNA hybridization. | Typically contains high salt concentration (e.g., 1 M NaCl) and Mg²⁺ (e.g., 20 mM) to stabilize the DNA-RNA duplex [34]. |
| Ethanolamine | Surface blocking agent. | Passivates unoccupied sites on the electrode surface to reduce non-specific adsorption and background signal [34]. |
| Synthetic miRNA Targets | Used for calibration, optimization, and validation of the biosensor. | Purified synthetic miRNA sequences are essential for establishing the standard curve and assessing selectivity against mismatched sequences [35] [33]. |
This case study demonstrates a successful framework for enhancing the analytical performance of a paper-based electrochemical biosensor for miRNA detection. By integrating a sample preconcentration step with a systematic optimization strategy using Central Composite Design, a twelve-fold improvement in the detection limit was achieved, reaching a clinically relevant 50 pM for miRNA-224 in serum [33].
The CCD approach proved invaluable for efficiently navigating a multi-variable parameter space, identifying significant interactive effects, and establishing a robust predictive model for the biosensor's performance. This methodology transcends the specific example presented, providing a generalizable blueprint for optimizing a wide range of biosensing platforms. The resulting biosensor architecture, combining the cost-effectiveness of paper substrates with the sensitivity of conformational change-based electrochemical detection, holds significant promise for the development of future point-of-care diagnostics for cancer and other diseases. Future work will focus on translating this optimized assay into a fully integrated, user-friendly device and validating its performance with clinical patient samples.
Integrating CCD with Machine Learning for Ultra-Sensitive Platforms
The pursuit of ultra-sensitive biosensing platforms necessitates rigorous optimization strategies to maximize performance metrics such as sensitivity, selectivity, and reproducibility. Central Composite Design (CCD) is a powerful response surface methodology (RSM) that systematically explores the relationship between multiple experimental variables and their responses [37]. When integrated with machine learning (ML) algorithms, CCD transforms from a traditional optimization tool into an intelligent framework capable of modeling complex, non-linear interactions in biosensor systems [4] [38]. This integration is particularly crucial for biosensors with sub-femtomolar detection limits, where challenges like enhancing the signal-to-noise ratio and improving reproducibility are most pronounced [4].
CCD operates by augmenting a two-level factorial or fractional factorial design with center points and axial ("star") points, allowing efficient estimation of second-order (quadratic) effects and curvature in the response surface [22]. This structured approach enables researchers to optimize multiple factors simultaneously while accounting for interaction effects that traditional one-variable-at-a-time approaches would miss [4]. The fusion of this statistically rigorous experimental framework with ML's predictive capabilities creates a powerful paradigm for developing next-generation biosensing platforms with unprecedented sensitivity and reliability for pharmaceutical and clinical applications [38].
Theoretical Foundations of Central Composite Design
Core Architecture and Mathematical Formulation
A Box-Wilson Central Composite Design contains an embedded factorial or fractional factorial design with center points that is augmented with a group of 'star points' to enable estimation of curvature [22]. The total number of experiments (N) required in a CCD is determined by the equation: N = 2^k + 2k + C, where k is the number of factors, 2^k represents the factorial points, 2k represents the star points, and C is the number of center points [22] [37]. This efficient experimental structure allows for the fitting of a second-order polynomial model without requiring a complete three-level factorial experiment, which would be prohibitively large in terms of experimental runs [37].
The second-order model fitted using CCD data takes the form: Y = β₀ + ΣβᵢXᵢ + ΣβᵢᵢXᵢ² + ΣΣβᵢⱼXᵢXⱼ + ε Where Y is the predicted response, β₀ is the constant coefficient, βᵢ are the linear coefficients, βᵢᵢ are the quadratic coefficients, βᵢⱼ are the interaction coefficients, Xᵢ and Xⱼ are the independent variables, and ε represents the error term [37]. This model effectively captures linear, quadratic, and interaction effects between factors, providing a comprehensive understanding of the factor-response relationships.
Types of Central Composite Designs
CCD can be implemented in three primary variations, each with distinct properties and applications:
Circumscribed CCD (CCC): This is the original CCD form where star points are positioned at a distance α from the center based on desired design properties, establishing new extremes for each factor [22]. This design requires five levels for each factor and exhibits rotatable properties, meaning the prediction variance depends only on the distance from the design center [22] [37].
Inscribed CCD (CCI): In this design, the star points are positioned at the factorial boundaries of the design space, effectively scaling down a CCC design so that the star points become the factorial points [22]. This approach is suitable when the experimental region is constrained by practical limitations, and it also produces a rotatable design [37].
Face-Centered CCD (CCF): This variation positions star points at the center of each face of the factorial space, resulting in α = ±1 [22]. This design requires only three levels for each factor but is not rotatable, though it often provides practical advantages in experimental setups where only three factor levels are feasible [22] [37].
The value of α is crucial for design properties and is calculated differently depending on the desired characteristics. For rotatability, the value is determined by α = (2^k)^(1/4) for a full factorial design, where k is the number of factors [22]. The selection of the appropriate CCD type depends on the experimental region of interest, the number of feasible factor levels, and the desired statistical properties.
Integration Frameworks: Coupling CCD with Machine Learning
Sequential Optimization and Predictive Modeling
The most straightforward integration approach involves using CCD to generate optimized experimental parameters, which then serve as training data for machine learning models. In this framework, CCD first identifies the critical factors and their optimal ranges through response surface methodology [37]. The data obtained from CCD experiments, including main effects, interaction terms, and quadratic responses, then train ML algorithms to build predictive models that can interpolate and extrapolate beyond the experimental conditions tested [38].
This sequential approach was demonstrated in the development of a visual biosensor for sertraline detection using core-shell AuNPs@CDs nanoprobes, where CCD was first employed to optimize key variables affecting the sensing process before analysis [39]. The researchers used CCD to simulate assay conditions and identify optimal parameter combinations, which could subsequently be used to train ML models for predicting sensor performance under new conditions. This methodology combines the rigorous experimental design of CCD with the predictive power of ML, creating a powerful tool for biosensor optimization [39] [38].
AI-Enhanced Experimental Design and Response Surface Analysis
Machine learning algorithms can enhance CCD implementation by analyzing complex, non-linear responses that may not perfectly fit traditional polynomial models. While CCD effectively captures quadratic responses, ML models like artificial neural networks (ANNs), support vector machines (SVMs), and random forests can model more complex response surfaces with higher accuracy [38] [40].
Deep learning models, including convolutional neural networks (CNNs) and transformer-based architectures, have shown particular promise in processing high-dimensional data from biosensors, such as spectral information or complex signal patterns [4] [40]. These models can identify subtle patterns and relationships in CCD-generated data that might be missed by traditional analysis methods. For instance, in portable chemiluminescence sensing platforms, deep learning models like InceptionV3, VGG16, and ResNet-50 have been successfully applied to enhance the analysis of complex signal outputs, demonstrating the potential for similar applications in CCD-optimized biosensing systems [40].
Table 1: Machine Learning Models for Enhanced CCD Data Analysis
| ML Model | Application in CCD Integration | Advantages | Limitations |
|---|---|---|---|
| Artificial Neural Networks (ANNs) | Modeling complex non-linear response surfaces | High predictive accuracy for complex systems | Requires large datasets, risk of overfitting |
| Random Forest | Feature importance analysis from CCD factors | Handles non-linear relationships, robust to outliers | Limited extrapolation capability |
| Support Vector Machines (SVM) | Classification of optimal vs. suboptimal regions | Effective in high-dimensional spaces | Memory-intensive with large datasets |
| Convolutional Neural Networks (CNNs) | Analysis of spectral or image-based biosensor outputs | Automatic feature extraction from complex data | Computationally intensive, requires specialized architecture |
| Genetic Algorithms | Optimization of CCD factor levels | Global optimization, avoids local minima | Computationally expensive, parameter tuning sensitive |
Real-Time Adaptive Optimization
The most advanced integration framework combines CCD with ML for real-time, adaptive optimization of biosensing platforms. In this approach, ML models continuously analyze incoming sensor data and dynamically adjust experimental parameters originally identified through CCD [38]. This creates a closed-loop optimization system where the biosensor platform self-optimizes based on real-time performance feedback.
This adaptive learning capability is particularly valuable for biosensors deployed in fluctuating environments or for detecting multiple analytes simultaneously [38]. ML algorithms can continuously learn from new data, improving the resilience and reliability of biosensor readings over time and adapting to changing environmental conditions and sample matrices [38]. While this represents a more complex implementation, it offers the highest level of automation and performance optimization for ultra-sensitive detection platforms.
Experimental Protocols and Case Studies
Case Study: CCD-Optimized Sertraline Detection Using AuNPs@CDs Nanoprobes
A recent study demonstrates the effective application of CCD in optimizing a visual and non-invasive biosensor for detecting the antidepressant sertraline (SRT) [39]. The research employed core-shell gold nanoparticles and carbon dots (AuNPs@CDs) synthesized eco-friendly from sweet lemon waste as nanoprobes. The CCD approach was utilized to simulate assay conditions and optimize key variables affecting the sensing process before analysis [39].
Experimental Protocol:
- Nanoprobe Synthesis: Carbon dots (CDs) were synthesized from sweet lemon waste via a green synthesis approach, which were then used to reduce Au³⁺ ions to Au⁰ without external photoirradiation or additional reductants, forming core-shell AuNPs@CDs structures [39].
CCD Experimental Design: Key factors influencing sertraline detection were identified, including:
- pH of the medium
- Incubation time
- Nanoprobe concentration
- Temperature A face-centered CCD (CCF) with these factors was implemented, requiring approximately 30 experimental runs (including center points) to model the quadratic response surface [39].
Response Measurement: The colorimetric response was measured via absorbance at 560 nm, with visual color changes from pink to purple to violet indicating sertraline concentration [39].
Model Validation: The optimized conditions derived from CCD were validated experimentally, demonstrating a linear detection range of 1-35 μM with a limit of detection (LOD) of 100 nM for sertraline in biofluids [39].
Table 2: Research Reagent Solutions for CCD-Optimized Biosensing
| Reagent/Material | Function in Biosensing Platform | Specifications/Alternatives |
|---|---|---|
| Sweet lemon waste-derived CDs | Green synthesis of carbon dots as reducing and stabilizing agents | Alternative: Other fruit peel wastes (watermelon, pomelo) or chemical precursors |
| Tetrachloroauric acid (HAuCl₄·3H₂O) | Gold precursor for AuNPs formation | Concentration typically 1-10 mM in final solution |
| Sertraline standard | Target analyte for method development and validation | Pharmaceutical grade, 50 mg tablets for real sample analysis |
| Buffer solutions (various pH) | Maintaining optimal pH for detection | pH range 3.0-9.0 for investigating pH influence |
| Biological fluids | Real sample matrix for validation | Serum, urine, or saliva samples |
Protocol for General CCD-ML Integration in Biosensor Development
For researchers implementing CCD-ML integration for novel biosensing platforms, the following generalized protocol provides a methodological framework:
Phase 1: Preliminary Factor Screening
- Identify potential factors influencing biosensor performance through literature review and preliminary experiments.
- Conduct fractional factorial or Plackett-Burman designs to screen for statistically significant factors.
- Select 3-5 most influential factors for detailed CCD optimization.
Phase 2: Central Composite Design Implementation
- Define factor levels based on screening results, ensuring levels cover the anticipated optimal region.
- Choose appropriate CCD type (CCC, CCI, or CCF) based on experimental constraints and desired statistical properties.
- Execute the designed experiments in randomized order to minimize systematic error.
- Measure multiple response variables relevant to biosensor performance (sensitivity, selectivity, response time, etc.).
Phase 3: Machine Learning Integration
- Preprocess CCD data, including normalization and feature scaling as required by ML algorithms.
- Partition data into training (80%) and testing (20%) sets, maintaining representation of all experimental regions.
- Train multiple ML models (ANN, Random Forest, SVM, etc.) on the CCD data.
- Validate model performance using test set data and cross-validation techniques.
- Select the best-performing model for predictive optimization and response surface analysis.
Phase 4: Model Validation and Verification
- Conduct confirmation experiments at predicted optimal conditions.
- Compare predicted versus actual biosensor performance.
- If discrepancy exceeds acceptable limits (typically >10%), iteratively refine the model with additional data points.
Data Analysis and Interpretation Strategies
Response Surface Methodology and Statistical Analysis
The analysis of CCD experiments focuses on interpreting the second-order polynomial model to understand factor effects and identify optimal conditions. Key analytical approaches include:
Analysis of Variance (ANOVA): Determines the statistical significance of the model and individual terms. A high F-value and low p-value (<0.05) indicate model significance, while lack-of-fit tests assess model adequacy [37].
Response Surface Plots: Three-dimensional representations of the relationship between two factors and the response, while holding other factors constant. These visualizations help identify optimal regions, interaction effects, and response curvature [37].
Contour Plots: Two-dimensional projections of response surfaces that facilitate identification of factor level combinations yielding desired responses.
Optimization Functions: Utilization of desirability functions to simultaneously optimize multiple responses, balancing potentially competing objectives (e.g., maximizing sensitivity while minimizing cost).
Table 3: Key Parameters in CCD Implementation for Biosensor Optimization
| Parameter | Role in CCD | Typical Values/Ranges | Considerations for Biosensors |
|---|---|---|---|
| Number of Factors (k) | Determines experimental size and complexity | 3-5 factors typical | Balance comprehensiveness with practical feasibility |
| Alpha (α) Value | Determines star point placement | 1.0 (CCF) to 2.378 (k=5, CCC) | Choice affects rotatability and number of factor levels |
| Center Points (C) | Estimates pure error and curvature | 3-6 replicates | More center points improve error estimation |
| Model R² | Measures model fit | >0.80 acceptable, >0.90 desirable | High R² ensures good predictive capability |
| Adjusted R² | R² adjusted for number of terms | Close to R² indicates appropriate model | Prevents overfitting with unnecessary terms |
Machine Learning-Enhanced Data Interpretation
Machine learning approaches complement traditional RSM by identifying complex patterns in CCD data that might not be captured by polynomial models. Specific ML applications include:
Feature Importance Analysis: Random Forest and similar algorithms quantify the relative importance of each factor, helping prioritize optimization efforts [38].
Non-Linear Pattern Recognition: Neural networks detect and model complex non-linear relationships and higher-order interactions between factors [38] [40].
Cluster Analysis: Unsupervised learning techniques identify natural groupings in the response data, potentially revealing distinct operational regimes for the biosensor.
Dimensionality Reduction: Techniques like Principal Component Analysis (PCA) visualize high-dimensional CCD data in two or three dimensions, facilitating interpretation of complex factor-response relationships.
The integration of ML interpretation with traditional RSM creates a more comprehensive understanding of the biosensor system, leading to more robust optimization and better performance in real-world applications.
Future Perspectives and Challenges
The integration of CCD with machine learning for ultra-sensitive biosensing platforms represents a cutting-edge approach with significant potential, but several challenges must be addressed for widespread adoption. Future developments will likely focus on:
Automated Experimental Platforms: The development of fully automated systems that combine CCD-designed experiments with robotic liquid handling and real-time ML analysis will dramatically accelerate optimization cycles [38]. These systems could autonomously refine biosensor parameters based on continuous performance feedback, reducing human intervention and potential errors.
Explainable AI for Biosensor Optimization: As ML models become more complex, ensuring interpretability of predictions becomes crucial for scientific acceptance and regulatory approval [41]. Research into explainable AI techniques that provide transparent reasoning for optimization recommendations will be essential for adoption in regulated fields like pharmaceutical development and clinical diagnostics [41].
Multi-Objective Optimization Frameworks: Advanced optimization approaches that simultaneously consider multiple, potentially competing objectives (sensitivity, cost, speed, stability) will be enhanced through ML techniques that can navigate complex trade-off spaces more effectively than traditional desirability functions [4].
Standardization and Validation Protocols: The field requires standardized protocols for validating CCD-ML integrated models, particularly for regulatory submission in healthcare applications [41]. This includes establishing guidelines for dataset quality, model transparency, and independent validation procedures to ensure reliability and reproducibility [38] [41].
Despite these promising directions, challenges remain in computational resource requirements, data quality and quantity needs, interdisciplinary knowledge barriers, and regulatory acceptance of ML-driven optimization in diagnostic devices. Addressing these challenges through collaborative research between statisticians, computer scientists, and biosensor developers will unlock the full potential of CCD-ML integration for creating next-generation ultra-sensitive biosensing platforms.
Biosensors are analytical devices that convert biological, chemical, or biochemical signals into measurable electrical signals through a system containing a biological detection material combined with a chemical or physical transducer [42]. These devices utilize the specificity of a biological molecule to detect specific analytes, providing detailed insight into physiology and pathophysiology [43]. The fundamental components of an ideal biosensor include a biological recognition element (such as enzymes, antibodies, nucleic acids, or whole cells) that specifically interacts with the target analyte, a transducer that converts the biological response into a quantifiable signal, and a signal processing system that amplifies and displays the results in a user-readable format [42] [43].
The application spectrum of biosensors has expanded dramatically, encompassing diverse fields from food safety to clinical diagnostics. In recent years, substantial efforts have been directed toward advancing rapid, dependable, highly sensitive, and selective biosensing technologies aimed at identifying specific biomarkers with limits of detection (LOD) lower than femtomolar, which is increasingly regarded as essential for facilitating early diagnosis of progressive, life-threatening diseases [15]. The progression of biomedical research and clinical practices hinges largely on the development of robust methodologies for accurately and sensitively detecting biomolecules, providing clinicians with crucial tools for combating diseases through early interventions that significantly improve treatment outcomes [15].
Biosensors in Food Safety Pathogen Detection
The Burden of Foodborne Pathogens
Foodborne illnesses represent an enormous challenge to worldwide healthcare systems, with the Centers for Disease Control and Prevention (CDC) estimating that foodborne or waterborne pathogens cause approximately 76 million illnesses annually in the United States alone [42]. The economic impact is substantial, with approximately $15.6 billion in economic losses occurring each year due to foodborne illnesses [42]. Bacterial pathogens constitute the most well-known foodborne pathogens, causing the greatest number of illnesses, including the most hospitalizations (63.9%) and deaths (63.7%) [42]. These pathogens can contaminate various food matrices, including raw meat and poultry, vegetables, fruits, eggs, dairy products, and cooked seafood, leading to serious health consequences when consumed [42].
Conventional vs. Biosensor-Based Detection Methods
Traditional methods for detecting microbial contaminants in foods include viable cell counting, staining, carbohydrate fermentation assays, enzyme-linked immunosorbent assays (ELISA), polymerase chain reaction (PCR), ultraviolet detection, and fluorescence techniques [42]. Despite their widespread use, these conventional approaches present several limitations, including requirements for large sample volumes, high skill levels, extended processing time, and significant costs [42]. Furthermore, most traditional methods require prolonged incubation periods to obtain accurate microbiological results, delaying critical decisions in food safety management [42].
Biosensors represent an innovative approach that overcomes these limitations by providing rapid, real-time information superior to traditional microbiological approaches [42]. Among the most effective biosensors for food safety applications is adenosine triphosphate (ATP) bioluminescence, which can be used for monitoring food processing manufacturing within Hazard Analysis and Critical Control Points (HACCP) frameworks [42]. ATP bioluminescence operates on the principle that ATP serves as a significant biological source of energy found in various microbes, thus indicating the presence of living microorganisms [42].
Table 1: Major Foodborne Pathogens and Associated Food Matrices
| Pathogenic Sources | Food Matrix | Symptoms and Illnesses |
|---|---|---|
| Staphylococcus aureus | Unpasteurized Milk and Cheese Products | Food Poisoning |
| Bacillus cereus | Dairy Products, Dry Foods, Rice, Egg Products | Diarrhea, Vomiting |
| E. coli O157:H7 | Meat Products and Milk | Diarrheal Diseases and Producing of Shiga Toxins |
| Vibrio parahaemolyticus | Seafood | Diarrhea |
| Salmonella enteritidis | Meats, Eggs, Fruits, Vegetables | Vomiting, Diarrhea, Cramps, Fever |
| Campylobacter jejuni | Meat, Poultry | Postinfectious Reactive Arthritis |
| Listeria monocytogenes | Lentil Salad | Gastroenteritis and Invasive Infection |
| Clostridium botulinum | Uncooked Food, Canned Foods | Botulism |
Table 2: Comparison of Microbiological Analysis Approaches
| Microbiological Approaches | Detection Limit (Log CFU/mL) | Time Consumed |
|---|---|---|
| Viable Cell Counting | Unlimited | days |
| Microscopy | Unlimited | minutes |
| Absorbance | 8–9 | Immediate |
| Enzyme Linked Immunosorbence | 2.83–3 | 3 hours |
Biosensors in Clinical Cancer Biomarker Detection
The Cancer Diagnostic Imperative
Cancer remains one of the leading causes of death worldwide, with nearly 570,000 people expected to die from cancer in a single year in the United States alone [44]. The earlier cancer can be detected, the better the chance of a cure; however, many cancers are diagnosed only after they have metastasized throughout the body [44]. The five-year survival rate for certain cancers such as liver, pancreatic, and lung remains very low (6%–16%), highlighting the critical need for early detection technologies [44]. Effective, accurate methods of cancer detection and clinical diagnosis are urgently needed to improve patient outcomes and reduce mortality rates [44] [45].
Cancer is defined as abnormal and uncontrolled cell growth due to an accumulation of specific genetic and epigenetic defects, both environmental and hereditary in origin [44]. The two major tumorigenesis mechanisms are activation of oncogenes and inactivation of tumor suppressor genes (TSGs) [44]. Oncogene activation occurs through mutation or duplication of a normal gene (a proto-oncogene) involved in regulating cell growth, proliferation, and/or differentiation, leading to constitutive activation or excess levels of a normal gene product [44]. Notable examples include the human epidermal growth factor receptor Her-2, which is amplified in approximately 33% of all breast cancers [44].
Cancer Biomarkers and Their Clinical Significance
The National Cancer Institute (NCI) defines a biomarker as "a biological molecule found in blood, other body fluids, or tissues that is a sign of a normal or abnormal process or of a condition or disease" [44]. Cancer biomarkers can be of various molecular origins, including DNA (specific mutation, translocation, amplification, and loss of heterozygosity), RNA, or protein (hormone, antibody, oncogene, or tumor suppressor) [44]. These biomarkers are potentially among the most valuable tools for early cancer detection, accurate pretreatment staging, determining response to chemotherapy treatment, and monitoring disease progression [44].
Table 3: Key Cancer Biomarkers and Their Diagnostic Applications
| Type of Cancer | Biomarker | Clinical Utility |
|---|---|---|
| Prostate | Prostate-Specific Antigen (PSA) | Screening and diagnosis; levels >4.0 ng/mL correlate with prostate cancer |
| Ovarian | Cancer Antigen 125 (CA 125) | Elevated in 90% of advanced ovarian cancer cases; monitoring treatment response |
| Breast | BRCA1, BRCA2, HER2/NEU | BRCA mutations account for ~50% of hereditary breast cancers; HER2 status guides targeted therapy |
| Multiple Cancers | p53 | Mutations found in brain, breast, colon, lung, hepatocellular carcinomas, and leukemia |
| Colon | Carcinoembryonic Antigen (CEA) | Monitoring disease progression and treatment response |
Prostate-specific antigen (PSA) represents one of the first tumor biomarkers identified and put into routine clinical use for prostate cancer screening and diagnosis [44]. Studies have shown that above-normal PSA levels (typically >4.0 ng/mL) correlate directly with prostate cancer, with approximately 30% of men with PSA levels between 4.1 and 9.9 ng/mL having prostate cancer [44]. Similarly, elevated cancer antigen (CA) 125 is most commonly associated with ovarian cancer and is elevated in 90% of women with advanced ovarian cancer [44]. These biomarkers, along with many others, provide critical information for cancer diagnosis, treatment selection, and monitoring.
Central Composite Design in Biosensor Optimization
The Need for Systematic Optimization in Biosensor Development
The systematic optimization of ultrasensitive biosensors remains a primary obstacle limiting their widespread adoption as dependable point-of-care tests [15]. Biosensor performance depends on multiple interconnected parameters, including the formulation of the detection interface, the immobilization strategy of the biorecognition elements, and the detection conditions [15]. Traditionally, many studies have optimized individual variables independently, a straightforward yet problematic approach particularly when dealing with interacting variables [15]. This univariate approach may fail to identify true optimal conditions, as the conditions established for sensor preparation and operation may not represent the genuine optimum when variable interactions are present [15].
Experimental design, or design of experiment (DoE), provides a powerful chemometric tool that facilitates the systematic and statistically reliable optimization of parameters [15]. The DoE approach follows a model-based optimization, resulting in the development of a data-driven model that connects variations in input variables (such as material properties engaged in biosensor development and production parameters) to sensor outputs [15]. This methodology offers significant advantages over traditional one-variable-at-a-time approaches, including reduced experimental effort, enhanced information quality, and the ability to detect and quantify interactions between variables [15].
Fundamental DoE Methodologies
Several experimental design strategies are employed in biosensor optimization, with factorial designs and central composite designs being among the most prominent:
Factorial Designs: The 2^k factorial designs are first-order orthogonal designs requiring 2^k experiments, where k represents the number of variables being studied [15]. In these models, each factor is assigned two levels coded as -1 and +1, corresponding to the variable's range selected based on the specific application [15]. These designs are effective for fitting first-order approximating models and identifying interactions between variables, though they may fail to account for curvature in certain responses [15].
Central Composite Design (CCD): Central composite designs represent one of the most widely used classes of second-order experimental designs for optimization studies [6]. CCD can be conceptualized as a factorial or fractional factorial design augmented with additional points (center points and axial points) that enable estimation of curvature in the response surface [15] [6]. This design is particularly valuable for building second-order quadratic models, which are essential when the response follows a quadratic function with respect to the experimental variables [15].
CCD Optimization Workflow
Practical Application of CCD in Biosensor Development
A practical example of CCD application can be found in the optimization of alkali-activated materials, where researchers used central composite design to investigate the effects of silica content, particle size, and replacement ratio of rice husk ash (RHA) on compressive strength [6]. The experimental design incorporated 8 cube points, 1 center point in the cube, 6 axis points, and 2 center points in axial, totaling 17 experiments [6]. The results demonstrated that the replacement ratio of RHA most significantly affected compressive strength (p-values < 0.002) compared with particle size (p-values < 0.450) and silica content (p-values < 0.017) [6]. The optimum values identified through this systematic approach were 50 µm particle size, 90% silica content, and 15 wt.% replacement ratio [6].
In biosensor optimization, CCD can be similarly applied to critical parameters such as bioreceptor density, incubation time, temperature, pH, and signal amplification conditions. The methodology enables researchers to not only identify optimal conditions but also to understand the relationship and interactions between different variables affecting biosensor performance [15]. This approach is particularly crucial for ultrasensitive platforms with sub-femtomolar detection limits, where challenges like enhancing the signal-to-noise ratio, improving selectivity, and ensuring reproducibility are particularly pronounced [15].
Experimental Protocols and Methodologies
Biosensor Fabrication and Characterization Protocol
The development of high-performance biosensors requires meticulous attention to fabrication protocols and characterization methods. A generalized protocol for biosensor development includes the following key steps:
Substrate Preparation and Functionalization: Clean and functionalize the transducer surface (e.g., gold, carbon, or silicon-based substrates) to enable efficient immobilization of biorecognition elements. Surface characterization techniques such as atomic force microscopy (AFM), scanning electron microscopy (SEM), or contact angle measurements verify successful functionalization.
Bioreceptor Immobilization: Immobilize biological recognition elements (antibodies, aptamers, enzymes, or nucleic acids) onto the functionalized surface using appropriate coupling chemistry (e.g., EDC-NHS for carboxyl-amine coupling, avidin-biotin interaction, or thiol-gold chemistry). Optimization of immobilization conditions (pH, concentration, time, temperature) is critical for maintaining bioreceptor activity and orientation.
Blocking and Surface Passivation: Apply blocking agents (e.g., bovine serum albumin, casein, or ethanolamine) to cover any remaining reactive sites on the sensor surface, minimizing non-specific binding that contributes to background noise.
Assay Optimization and Characterization: Systematically optimize assay conditions including incubation time, temperature, pH, and ionic strength using design of experiments (DoE) approaches. Characterize biosensor performance through calibration curves, limit of detection (LOD) determination, specificity testing against interferents, and reproducibility assessments.
Central Composite Design Implementation Protocol
Implementing central composite design for biosensor optimization follows a structured methodology:
Critical Parameter Identification: Conduct preliminary studies to identify factors significantly influencing biosensor performance. Typical factors include bioreceptor concentration, incubation time, temperature, pH, and signal amplification reagent concentrations.
Experimental Domain Definition: Establish appropriate ranges for each factor based on preliminary experiments or literature data. The experimental domain should be sufficiently wide to detect potential curvature in the response while remaining within practically feasible ranges.
Experimental Matrix Construction: Generate the CCD experimental matrix using statistical software (e.g., Minitab, Design-Expert, or R). A typical CCD for three factors requires 20 experiments (8 factorial points, 6 axial points, and 6 center points).
Randomized Experiment Execution: Conduct experiments in randomized order to minimize the effects of uncontrolled variables and systematic errors. Replicate center points to estimate pure error and model adequacy.
Response Surface Modeling: Fit experimental data to a second-order polynomial model using regression analysis. The general form of the model for three factors is: Y = β₀ + β₁X₁ + β₂X₂ + β₃X₃ + β₁₂X₁X₂ + β₁₃X₁X₃ + β₂₃X₂X₃ + β₁₁X₁² + β₂₂X₂² + β₃₃X₃²
Model Validation and Optimization: Validate the fitted model through statistical significance testing (ANOVA), lack-of-fit tests, and residual analysis. Once validated, use the model to identify optimal factor settings that maximize or minimize the response as desired.
Table 4: Research Reagent Solutions for Biosensor Development
| Reagent/Material | Function | Application Examples |
|---|---|---|
| Gold Nanoparticles | Signal amplification, electron transfer enhancement, bioreceptor immobilization platform | Electrochemical and optical biosensors for pathogen and biomarker detection |
| Graphene Oxide | High surface area substrate, excellent electrical conductivity, fluorescence quenching | Field-effect transistor biosensors, electrochemical aptasensors |
| EDC/NHS Chemistry | Carboxyl group activation for amine coupling to form stable amide bonds | Antibody and enzyme immobilization on sensor surfaces |
| Avidin-Biotin System | High-affinity interaction for oriented immobilization of biotinylated bioreceptors | DNA biosensors, immunosensors with controlled receptor orientation |
| Polycarboxylate-based Polymers | Signal enhancement, noise reduction, stabilization of bioreceptors | Electrochemical biosensors, point-of-care diagnostic devices |
| Enzyme Labels (HRP, ALP) | Catalytic signal amplification through substrate conversion | Enzyme-linked immunosorbent assays (ELISA), electrochemical immunosensors |
Emerging Trends and Future Perspectives
The field of biosensing continues to evolve rapidly, with several emerging trends shaping future developments in both food safety and clinical diagnostics. The integration of artificial intelligence (AI) with biosensor technologies represents a particularly promising direction, enabling more accurate data interpretation, pattern recognition, and predictive analytics [45]. AI algorithms can enhance biosensor performance by compensating for environmental variations, improving signal-to-noise ratios, and enabling multiplexed detection through advanced data deconvolution techniques [45].
Point-of-care testing (POCT) represents another significant trend, driven by demands for rapid, decentralized diagnostics in both food safety monitoring and clinical settings [15]. The development of portable, user-friendly biosensing platforms with minimal sample preparation requirements will dramatically expand the application spectrum of biosensors beyond traditional laboratory settings [15]. Smartphone-based biosensors, paper-based microfluidic devices, and wearable continuous monitoring systems represent exciting developments in this domain [43].
Multiplexed detection capabilities are increasingly becoming a standard requirement for both food safety and clinical applications. The ability to simultaneously detect multiple pathogens or biomarkers in a single assay provides comprehensive diagnostic information while reducing analysis time and cost [42] [44]. Advanced biosensor platforms incorporating microarray technologies, multiplexed electrochemical detection, or spectral encoding strategies are addressing this need.
Synthetic biology approaches are also revolutionizing biosensor design through the engineering of novel biological recognition elements with enhanced specificity, stability, and signal generation capabilities [43]. Designed aptamers, engineered enzymes, and synthetic genetic circuits expand the toolbox available for biosensor development, enabling detection of analytes that were previously challenging to monitor with conventional biological elements.
Biosensor Application Spectrum
Biosensor technologies have demonstrated remarkable versatility across diverse application domains, from ensuring food safety through pathogen detection to enabling early cancer diagnosis through biomarker monitoring. The effectiveness of biosensors in these critical areas stems from their ability to provide rapid, sensitive, and specific detection of target analytes, often in real-time and with minimal sample preparation. The continuing evolution of biosensing platforms, incorporating advances in nanotechnology, materials science, and microengineering, promises even greater performance capabilities in the future.
Central composite design and other systematic optimization methodologies play an indispensable role in advancing biosensor technology toward its full potential. By enabling efficient, statistically rigorous optimization of multiple interdependent parameters, these experimental design strategies accelerate biosensor development while ensuring robust performance characteristics. As biosensors continue to transition from laboratory prototypes to commercially viable products for food safety monitoring and clinical diagnostics, systematic optimization approaches will remain essential for achieving the reliability, reproducibility, and sensitivity required for real-world applications.
The convergence of biosensor technology with artificial intelligence, point-of-care testing platforms, and synthetic biology approaches heralds an exciting future where rapid, accurate diagnostic information becomes increasingly accessible across healthcare, food safety, and environmental monitoring domains. Through continued interdisciplinary collaboration and the application of systematic optimization frameworks like central composite design, biosensor technologies will undoubtedly expand their application spectrum even further, addressing emerging challenges in public health and personalized medicine.
Troubleshooting and Advanced Optimization Strategies for Robust Biosensor Performance
Identifying and Resolving Common Pitfalls in Experimental Domain Selection
In biosensor optimization research, the selection of an appropriate experimental domain represents a critical foundation upon which successful experimental design is built. The experimental domain—defined as the multidimensional space bounded by the upper and lower limits assigned to each factor under investigation—directly controls the efficiency of resource utilization, the validity of mathematical models, and the reliability of optimized conditions. Within the context of central composite design (CCD), a widely employed response surface methodology (RSM) for biosensor development, improper domain selection can compromise the entire optimization endeavor, leading to wasted resources, inconclusive results, and failed validation [16] [15].
CCD has established itself as a powerful optimization tool in biosensor research due to its flexibility and robustness in building second-order models, which are essential for capturing the complex, often nonlinear, relationships between biosensor fabrication parameters and performance metrics [16] [46]. However, its effectiveness is predicated on a correctly defined experimental space. This technical guide examines the prevalent pitfalls encountered during experimental domain selection in CCD studies, provides methodologies for their identification and resolution, and presents a structured framework to enhance the robustness of biosensor optimization.
Central Composite Design: A Primer for Biosensor Applications
Central composite design is a cornerstone of response surface methodology, specifically engineered to efficiently fit quadratic models. A standard CCD comprises three distinct sets of design points: a two-level factorial design (or fractional factorial), axial (star) points positioned at a distance α from the center, and replicated center points [47] [48]. The value of α determines the design type: a circumscribed CCD (CCCD) with |α| > 1, a face-centered CCD (FCCD) with |α| = 1, or an inscribed CCD (ICCD) [16] [47].
The structure of a CCD for two factors is visually summarized in the diagram below, illustrating the arrangement of factorial, axial, and center points.
In biosensor optimization, factors may include the concentration of biorecognition elements (e.g., enzymes, antibodies), immobilization time, pH of the sensing environment, or nanomaterial loading, while responses often include critical performance metrics such as sensitivity, dynamic range, limit of detection (LOD), and response time [49] [15] [46]. For instance, a study optimizing a CRISPR/Cas12a-based smartphone biosensor for nucleic acid detection achieved a LOD of 40 femtograms by carefully optimizing factors like reagent concentration and incubation time [50]. The primary advantage of CCD over one-variable-at-a-time (OVAT) approaches is its ability to quantify factor interactions and nonlinear effects with a minimized number of experimental runs, thereby accelerating the development cycle for sophisticated biosensing platforms [16] [51].
Common Pitfalls in Experimental Domain Selection
Pitfall 1: Incorrect Factor Ranges Leading to Model Inaccuracy
One of the most frequent and critical errors is selecting factor ranges that are either too narrow or too wide. Excessively narrow ranges may fail to capture the true curvature of the response surface, preventing the identification of a global optimum. Conversely, excessively wide ranges can lead to model failure if the true response within the domain is too complex to be described by a second-order polynomial, or if experiments conducted at extreme factor levels yield physically impossible or irrelevant results [16] [47].
In biosensor development, this is particularly pertinent when working with biological elements. For example, if the pH range under investigation is too broad, it might denature the enzyme used for recognition, rendering the biosensor non-functional and the data from those experimental points useless [46]. A study optimizing an amperometric biosensor for heavy metals using a CCD highlighted the importance of selecting viable ranges for factors like HRP enzyme concentration and applied potential to ensure the biological activity of the sensing interface [16].
Pitfall 2: Overlooking Factor-Factor Interactions and Constraints
Biosensor systems are inherently complex, with factors that often interact and are subject to practical constraints. A classic OVAT mindset can lead experimenters to define domains that ignore these interactions and constraints. In CCD, if the experimental domain includes combinations of factor levels that are practically infeasible (e.g., a very high bioreceptor concentration paired with a very short immobilization time, preventing adequate binding), the resulting model's predictions in those regions will be invalid [15] [47].
Furthermore, some factors are inherently linked. For instance, in the formulation of a detection interface, the components of a mixture (e.g., different polymers or solvents) must sum to 100%. Using a standard CCD in such a scenario is inappropriate, as it would violate this fundamental constraint. In these cases, a mixture design is the correct methodological choice [47].
Pitfall 3: Inadequate Consideration of the Alpha Value and Design Choice
The choice of α value and the corresponding type of CCD (circumscribed, face-centered, or inscribed) is a strategic decision that is often made without sufficient justification. A face-centered CCD (FCCD), where α=1, is computationally simple as it requires only three levels for each factor (-1, 0, +1). However, it lacks rotatability, a property where the prediction variance depends only on the distance from the design center, providing uniform precision across the experimental domain [47] [48] [51].
For a rotatable circumscribed CCD (CCCD), α is calculated as α = (2^k)^(1/4), where k is the number of factors. This design provides excellent predictive capability across the domain but requires five levels for each factor and may place axial points at extreme, sometimes impractical, settings [51]. Selecting an FCCD for convenience when a CCCD is warranted can reduce the model's predictive power near the domain boundaries.
Pitfall 4: Insufficient Replication at the Center Point
Center points are crucial in CCD as they provide an independent estimate of pure experimental error, allow for checking the adequacy of the first-order model, and ensure stability of the design [47] [48]. A common mistake is to include only one or two center points. Insufficient replication can lead to an unreliable estimate of error, which in turn compromises the validity of statistical significance tests (e.g., ANOVA) used to evaluate the model and its terms [16] [51]. It is generally recommended to include between 4 and 6 center point replicates to obtain a robust estimate of the experimental noise [47] [48].
Methodologies for Identification and Resolution
Preliminary Research and Sequential Experimentation
A robust domain selection process begins long before the CCD is executed. Preliminary research, including literature review and prior knowledge of the biosensor system, is essential for establishing plausible initial factor boundaries [15].
A highly effective strategy is sequential experimentation. This involves starting with a broad screening design, such as a 2^k factorial design, to identify significant factors and their approximate functional relationships. The data from this initial design can then be used to refine the experimental domain for a subsequent, more detailed CCD. As noted in one review, "it is often necessary to conduct multiple DoE iterations" and "it is advisable not to allocate more than 40% of the available resources to the initial set of experiments" [15]. This iterative approach mitigates the risk of committing to an incorrect domain from the outset. The workflow for this process is illustrated below.
Leveraging Asymmetrical Designs for Constrained Problems
When the region of operability for the biosensor is asymmetrical, forcing a symmetrical CCD is a suboptimal practice. In such cases, asymmetrical response surface designs should be employed. The most common of these are D-optimal designs [47].
A D-optimal design is constructed by selecting a pre-defined number of experiments from a grid of all potential candidate points within the irregular experimental domain. The selection criterion is to maximize the determinant of the X'X matrix (the information matrix), which minimizes the variance of the model coefficients. This results in a design that is optimally tailored to the specific, often constrained, experimental domain of complex biosensor systems, ensuring the best possible parameter estimates from the available resources [47].
Computational and Statistical Verification Techniques
Before finalizing a CCD, several statistical checks can be performed to pre-emptively identify domain-related issues. Power analysis can help determine if the proposed number of experiments, including center points, is sufficient to detect effects of a desired size. Model adequacy checking is performed after data collection; a key diagnostic tool is the analysis of residuals—the differences between observed and predicted values. If residuals show a clear pattern (e.g., a curve) when plotted against predicted values, it is a strong indicator that the model is inadequate, potentially due to an incorrect domain that misses critical curvature or contains a more complex response than a quadratic model can capture [15].
The Scientist's Toolkit: Research Reagent Solutions
The successful application of CCD in biosensor optimization relies on a suite of essential materials and reagents. The table below details key components, their functions, and optimization considerations.
Table 1: Essential Research Reagents and Materials for Biosensor Optimization via CCD
| Reagent/Material | Function in Biosensor Development | Key Optimization Parameters (Examples) |
|---|---|---|
| Biorecognition Elements (Enzymes, Antibodies, Nucleic Acids, Transcription Factors [49] [46]) | Provides selective binding to the target analyte, forming the core of sensor specificity. | Concentration, Immobilization time/temperature, Activity/affinity, Stability. |
| Signal Transduction Materials (Gold Nanoparticles [50], Graphene [50] [46], Metal-Organic Frameworks (MOFs) [50]) | Converts the biological binding event into a measurable physical signal (optical, electrochemical). | Nanomaterial loading, Thickness of deposited layer, Electrical conductivity, Fluorescence quenching efficiency. |
| Immobilization Matrix/Crosslinkers (Polymers, Sol-Gels, Glutaraldehyde [20]) | Stabilizes and confines the biorecognition element to the transducer surface. | Matrix composition/concentration, Crosslinker density, Porosity, Biocompatibility (to prevent denaturation). |
| Buffer Components (Salts, pH Modifiers) | Maintains the optimal chemical environment for the biological element's activity and stability. | Ionic strength, pH, Buffer capacity. |
| Microfluidic Substrates (Polydimethylsiloxane (PDMS), Paper-based substrates [50]) | Automates and miniaturizes sample handling, reducing reagent volumes and integrating sample preparation. | Channel geometry/surface chemistry, Hydrophilicity/hydrophobicity, Flow rate. |
The strategic selection of the experimental domain is not a mere preliminary step but a decisive factor in the success of central composite design for biosensor optimization. By understanding and avoiding common pitfalls—such as incorrect factor ranges, overlooked constraints, and poor design choice—researchers can significantly enhance the efficiency and output of their development workflows. The adoption of a structured framework, incorporating preliminary screening, sequential experimentation, and the use of asymmetrical designs when necessary, provides a robust pathway to reliable optimization. As biosensors continue to evolve in complexity and application, from intelligent bio-manufacturing to point-of-care diagnostics, the rigorous application of these principles in experimental domain selection will be paramount in translating innovative concepts into high-performing, commercially viable, and clinically reliable devices [15] [50] [46].
Analyzing Variance (ANOVA) for Model and Factor Significance Validation
In biosensor development, achieving optimal performance requires precise tuning of multiple fabrication and operational parameters. Central Composite Design (CCD), a cornerstone of Response Surface Methodology (RSM), provides an efficient framework for this multivariate optimization by fitting a second-order polynomial model to experimental data [52] [51]. The model's form is expressed as:
Y = β₀ + ∑βᵢxᵢ + ∑βᵢᵢxᵢ² + ∑βᵢⱼxᵢxⱼ + ε
Here, Y represents the predicted response (e.g., biosensor sensitivity), β₀ is the constant coefficient, βᵢ are linear coefficients, βᵢᵢ are quadratic coefficients, βᵢⱼ are interaction coefficients, xᵢ and xⱼ are the coded independent variables, and ε is the residual error [51]. Once this model is developed, Analysis of Variance (ANOVA) serves as the critical statistical tool for validating the significance and reliability of the model and its individual terms, ensuring that the predicted optimum conditions are statistically sound and reproducible [52] [51].
Theoretical Framework: The Role of ANOVA in CCD
Within the CCD workflow, ANOVA fulfills several vital functions. It partitions the total variability in the observed response data into attributable components: the model's explained variability and the unexplained residual error. This partition allows researchers to test the following key hypotheses:
- Overall Model Significance: Whether the regression model as a whole explains a significant portion of the variability in the data compared to the residual noise.
- Factor Significance: Whether individual linear, quadratic, and interaction terms in the model contribute significantly to the prediction.
- Model Lack-of-Fit: Whether the chosen quadratic model is adequate to describe the data or if a more complex model is needed.
The validation process relies on several key statistical metrics derived from the ANOVA table, which are essential for interpreting the results robustly [52] [51].
Table 1: Key Statistical Metrics in ANOVA Interpretation
| Metric | Description | Interpretation in Biosensor Optimization |
|---|---|---|
| F-value | Ratio of the mean square of the model/term to the mean square of the residual. | A large F-value for the model indicates that the explained variance is significantly greater than the unexplained variance. For a factor, a large F-value suggests a strong effect on the biosensor response [52]. |
| p-value | Probability of obtaining an F-value at least as extreme as the one calculated, assuming the null hypothesis (no effect) is true. | A p-value below a significance threshold (e.g., 0.05) provides evidence to reject the null hypothesis, confirming the significance of the model or a specific factor [52] [53]. |
| R² (Coefficient of Determination) | Proportion of variance in the dependent variable that is predictable from the independent variables. | Indicates the model's goodness-of-fit. For instance, a study optimizing a glucose biosensor using CCD reported an R² value of 0.97, indicating the model explains 97% of the variability in the response [51]. |
| Lack-of-Fit Test | Compares the residual error to the pure error from replicated design points (e.g., center points). | A non-significant lack-of-fit (p-value > 0.05) is desirable, indicating the model is sufficiently complex for the data [53]. |
The following workflow outlines the integral role of ANOVA within the broader context of optimizing a biosensor using a Central Composite Design.
Practical Application: ANOVA in Biosensor Development
Case Study: Optimization of a Glucose Biosensor
The application of CCD and ANOVA is powerfully illustrated in the development of an amperometric glucose biosensor. In one study, researchers used a five-level, three-factorial CCD to optimize the electrode surface composition, with the critical variables being the amounts of carboxylated multiwall carbon nanotubes (c-MWCNT), titanium dioxide nanoparticles (TiO₂NP), and glucose oxidase (GOx) [12]. The ANOVA performed on the experimental data at a 95% confidence level confirmed the statistical significance of both the model and the key factors. This rigorous statistical validation ensured that the optimized electrode composition was reliable. The resulting biosensor demonstrated excellent performance, with a high sensitivity of 168.5 μA mM⁻¹ cm⁻² and a low detection limit of 2.1 × 10⁻⁶ M, successfully applied to analyze glucose in serum samples [12].
Case Study: Optimization of a Peptide-Based SARS-CoV-2 Antibody Biosensor
In cutting-edge diagnostic research, ANOVA was pivotal in optimizing biosensors for variant-specific detection of SARS-CoV-2 antibodies. Biosensors were fabricated using gold nanoparticles functionalized with immunodominant wild-type and mutated peptides [54]. The data obtained from these platforms, including Surface-Enhanced Raman Spectroscopy (SERS) and Electrochemical Impedance Spectroscopy (EIS), were analyzed using sophisticated methods like partial least squares discriminant analysis (PLS-DA). The performance of these biosensors, validated through statistical measures, achieved remarkable results, including 100% sensitivity and a detection limit as low as 0.43 ng mL⁻¹ for the wild-type peptide [54]. This underscores how ANOVA and related statistical models underpin the development of highly sensitive and specific diagnostic tools.
Table 2: Key Reagent Solutions in CCD-Optimized Biosensor Research
| Research Reagent | Function in Biosensor Development & Optimization |
|---|---|
| Carboxylated Multiwall Carbon Nanotubes (c-MWCNT) | Used in electrode composites to enhance electrical conductivity and surface area, with optimal loading determined by CCD/ANOVA [12]. |
| Titanium Dioxide Nanoparticles (TiO₂NP) | Acts as a biocompatible matrix to immobilize enzymes; its concentration is optimized via CCD to maximize electron transfer efficiency [12]. |
| Glucose Oxidase (GOx) | The primary recognition enzyme in glucose biosensors; its loading is a critical factor optimized using RSM to achieve maximum sensitivity and stability [12]. |
| Synthetic Peptides (e.g., P44) | Serve as selective biorecognition elements for antibodies; their sequence and immobilization density are optimized for variant-specific detection in immunosensors [54]. |
| Gold Nanoparticles (AuNPs) | Provide a high-surface-area platform for biomolecule immobilization and enhance signal transduction in optical and electrochemical biosensors [54]. |
| 4-Mercaptobenzoic Acid (MBA) | Functions as a Raman reporter molecule in SERS-based biosensors, forming a self-assembled monolayer on AuNPs for signal generation [54]. |
Experimental Protocol for ANOVA in CCD
This section provides a detailed methodology for conducting and interpreting ANOVA as part of a biosensor optimization study using CCD.
Step-by-Step Statistical Analysis Protocol
- Model Fitting: After conducting the experiments as per the CCD matrix, use statistical software (e.g., Design-Expert, Minitab, R) to perform multiple regression analysis and fit the second-order polynomial model to the response data [53].
- Generate the ANOVA Table: The software will automatically generate an ANOVA table, decomposing the total sum of squares into contributions from the model and the residual error.
- Assess Model Significance:
- Examine the F-value and p-value for the overall model. A p-value less than 0.05 typically indicates the model is statistically significant [52] [53].
- Check the Lack-of-Fit test. A p-value greater than 0.05 indicates the model is adequate and there is no evidence to suggest a more complex model is needed [53].
- Review the R² and Adjusted R² values to evaluate the model's goodness-of-fit.
- Evaluate Factor Significance: Scrutinize the p-values for each model term (linear, quadratic, interaction). Terms with p-values above 0.05 are generally considered non-significant and can be removed from the model to improve model robustness [53]. This process is known as model reduction or backward elimination.
- Refine the Model: Remove the non-significant terms one at a time (starting with the least significant) and re-run the ANOVA. This iterative process continues until a model containing only significant terms is achieved, known as a hierarchical model.
- Final Model Validation: The final, refined model should be validated by comparing its predictions with additional experimental data collected at the predicted optimum conditions.
The following diagram illustrates the logical decision process for interpreting an ANOVA table and refining the statistical model.
Worked Example: Interpretation of an ANOVA Table
The following table presents a simplified ANOVA output from a hypothetical CCD study on a biosensor, focusing on critical parameters like electron transfer resistance or sensitivity.
Table 3: Example ANOVA Table for a CCD-Optimized Biosensor Response
| Source | Sum of Squares | Degrees of Freedom | Mean Square | F-value | p-value | Significance |
|---|---|---|---|---|---|---|
| Model | 2115.32 | 3 | 705.11 | 133.12 | < 0.001 | Significant |
| A-Temperature | 438.77 | 1 | 438.77 | 27.61 | < 0.001 | Significant |
| B-pH | 972.11 | 1 | 972.11 | 61.18 | < 0.001 | Significant |
| AB (Interaction) | 1314.06 | 1 | 1314.06 | 82.69 | < 0.001 | Significant |
| A² (Quadratic) | 75.19 | 1 | 75.19 | 4.73 | 0.042 | Significant |
| Residual | 317.81 | 20 | 15.89 | |||
| Lack of Fit | 301.25 | 15 | 20.08 | 4.12 | 0.07 | Not Significant |
| Pure Error | 16.56 | 5 | 4.87 | |||
| Cor Total | 2433.13 | 23 |
Interpretation:
- The model is highly significant (p < 0.001), indicating it successfully captures the relationship between the factors and the response.
- All linear (A, B), interaction (AB), and quadratic (A²) terms are significant (p < 0.05), justifying the use of a second-order model.
- The non-significant lack-of-fit (p = 0.07 > 0.05) suggests the model is adequate, and there is no evidence of missing higher-order terms. This model can be confidently used for navigation of the design space and prediction of the optimum biosensor performance.
In the rigorous field of biosensor optimization, Central Composite Design coupled with ANOVA provides an indispensable methodology for moving beyond empirical guesswork. ANOVA delivers objective, statistical validation of the predictive models derived from CCD, ensuring that identified optimal conditions for parameters such as electrode composition, immobilization density, and pH are robust, significant, and scientifically defensible. By systematically applying the protocols outlined in this guide, researchers and drug development professionals can enhance the efficiency, sensitivity, and reliability of their biosensors, thereby accelerating advancements in diagnostics and therapeutic monitoring.
In the realm of biosensor optimization, Central Composite Design (CCD) stands as a powerful statistical methodology for modeling complex, non-linear relationships between critical process parameters and key performance responses. However, a single application of CCD rarely yields a fully optimized system. Iterative refinement—the systematic process of redefining the experimental domain based on initial model findings—is paramount for achieving truly optimal biosensor performance. This technical guide provides researchers and drug development professionals with a structured framework for determining when and how to successively refine experimental domains, integrating both statistical principles and practical case studies from contemporary biosensor research. By mastering this iterative approach, scientists can efficiently navigate multidimensional variable spaces to enhance biosensor sensitivity, specificity, and reproducibility for point-of-care diagnostics and bioprocessing applications.
Central Composite Design serves as a cornerstone of Response Surface Methodology (RSM), enabling researchers to efficiently explore quadratic response surfaces and identify optimal factor settings. In biosensor development, these factors may include fabrication parameters, immobilization conditions, and detection settings, while responses often encompass critical performance metrics such as limit of detection (LOD), dynamic range, and signal-to-noise ratio [15] [4].
The fundamental premise of iterative refinement acknowledges that the initial experimental domain—the multi-dimensional space defined by the ranges of each factor under investigation—is often based on preliminary knowledge or theoretical considerations that may not encompass the true optimum. Consequently, the process of optimization is inherently sequential, requiring researchers to successively reposition and potentially rescale the experimental domain based on empirical findings from preceding experimental cycles [15]. This approach stands in stark contrast to traditional one-variable-at-a-time (OVAT) methodologies, which not only fail to capture factor interactions but also lack a systematic framework for domain redefinition.
Statistical guidance suggests allocating no more than 40% of available resources to initial experimental cycles, reserving the majority for subsequent iterative refinement based on insights gained from preliminary data [15] [4]. This resource allocation strategy underscores the anticipated need for multiple optimization cycles and reflects the progressive nature of empirical optimization in complex biological and chemical systems.
When to Redefine Your Experimental Domain
Recognizing the appropriate triggers for domain redefinition is crucial for efficient resource allocation and timely optimization. Several key indicators, derived from statistical analysis and model diagnostics, should prompt consideration of a new experimental domain.
Statistical and Empirical Indicators
Significant Lack of Fit: When the regression model demonstrates a statistically significant lack of fit, indicating systematic variation unexplained by the current model, it suggests the true response surface may possess curvature or complexity not captured within the existing domain [15]. This warrants investigation through domain expansion or translation.
Optimum at Domain Boundary: If the estimated optimum falls on or near the boundary of the current experimental domain, the true optimum likely lies outside the explored region. This necessitates shifting the domain center toward the predicted optimum and potentially expanding factor ranges to capture the complete response surface [15] [55].
Insufficient Model Resolution: A first-order model exhibiting poor fit (low R² value) or a second-order model with insignificant quadratic terms may indicate that the experimental domain is either too large, encompassing largely linear responses, or positioned in a region of minimal factor effects. Repositioning or rescaling the domain can enhance model resolution [15].
Identification of Inactive Factors: Through statistical analysis of factor effects, if certain variables demonstrate negligible impact on the response, the experimental domain for subsequent iterations can be redefined to exclude these factors, thereby reducing dimensionality and focusing resources on critical parameters [15].
Emergence of New Constraints or Objectives: Practical considerations often emerge during optimization, such as newly identified material incompatibilities, cost constraints, or additional performance criteria. These developments frequently necessitate redefining the experimental domain to incorporate these real-world limitations [56].
Table 1: Decision Matrix for Experimental Domain Redefinition
| Indicator | Statistical Evidence | Recommended Action |
|---|---|---|
| Significant Lack of Fit | p-value for lack-of-fit < 0.05 | Expand domain or increase model order |
| Optimum at Boundary | Optimal factor level at ±1 coded value | Shift center toward optimum; consider expansion |
| Insignificant Quadratic Terms | p-value > 0.05 for quadratic terms | Reposition domain to more sensitive region |
| High Proportion of Inactive Factors | >50% factors with p-value > 0.1 | Reduce dimensionality; eliminate inactive factors |
| Model Inadequacy | R² < 0.7 or poor residual distribution | Rescale domain or transform factors/responses |
How to Systematically Redefine the Experimental Domain
The process of domain redefinition follows a structured methodology that integrates statistical inference with scientific reasoning. The following workflow provides a systematic approach for planning successive experimental cycles.
Domain Translation: Shifting the Center Point
When analysis indicates the optimum lies near or beyond the boundary of the current domain, the most appropriate action is to translate or shift the entire domain toward the predicted optimum.
Protocol for Domain Translation:
- Identify Direction Vector: From the current model, determine the path of steepest ascent (for maximization) or descent (for minimization). This vector indicates the direction in which the response improves most rapidly.
- Calculate New Center Point: The coordinates of the predicted optimum become the new center point for the subsequent CCD. For a second-order model, this corresponds to the stationary point of the fitted response surface.
- Maintain or Slightly Adjust Ranges: Initially, maintain similar factor ranges unless evidence suggests the need for expansion to capture a broader region around the new optimum.
- Execute New CCD: Implement a new central composite design centered on the translated coordinates.
Case Study Application: In the optimization of a flux cored arc welding process using CCD, researchers initially identified optimal factor settings at the boundary of their original domain. By systematically shifting the domain center toward this boundary optimum and executing a subsequent CCD iteration, they achieved a tensile strength of 7,717 kgf, significantly exceeding initial performance metrics [55].
Domain Expansion: Capturing Broader Response Surfaces
Domain expansion becomes necessary when the optimum is confirmed to lie outside the current boundaries, or when the initial domain is too narrow to capture the curvature of the response surface.
Protocol for Domain Expansion:
- Establish Expansion Rationale: Base the decision on statistical evidence, such as a significant lack-of-fit test or a ridge analysis indicating continued improvement beyond current boundaries.
- Determine Expansion Factor: Increase the range of factors, typically by 1.5 to 2 times, focusing particularly on factors for which the optimum was at the boundary. The axial distance (α) in the CCD may require adjustment to maintain rotatability.
- Reposition Center Point: The center point may remain unchanged if symmetrical expansion is warranted, or may be adjusted if evidence suggests the domain should expand preferentially in one direction.
- Validate Model Extrapolation: Execute new experimental points at the expanded boundaries to validate the predicted response trends from the previous model.
Domain Contraction and Factor Reduction: Focusing the Investigation
When the optimal region is identified within the current domain but requires more precise resolution, or when certain factors demonstrate negligible effects, domain contraction and factor reduction improve experimental efficiency.
Protocol for Domain Refinement:
- Identify Significant Factors: Through ANOVA and effect testing, eliminate factors with p-values exceeding significance thresholds (typically α=0.05 or 0.10).
- Narrow Factor Ranges: Reduce the experimental range around the predicted optimum for greater resolution of the optimal region. This is particularly valuable for final tuning of biosensor performance.
- Increase Center Point Replication: Enhance estimation of pure error and model adequacy by replicating the center point in the contracted domain.
- Execute Focused CCD: Implement a new CCD with reduced dimensionality and narrower ranges to precisely characterize the optimal operating conditions.
Case Study Application: In the development of whole-cell biosensors for lignin-derived molecules, researchers applied a Definitive Screening Design (DSD) to efficiently identify significant genetic factors (promoters, RBS sequences) affecting biosensor performance. Subsequent iterative cycles focused exclusively on these significant factors, dramatically enhancing dynamic range by >500-fold and sensitivity by >1500-fold compared to initial constructs [57].
Case Study: Iterative Optimization of an SPR Biosensor
A recent study on Surface Plasmon Resonance (SPR) biosensors exemplifies the power of iterative domain refinement. Researchers employed a multi-objective Particle Swarm Optimization (PSO) algorithm to systematically optimize three critical design parameters—incident angle, adhesive layer thickness, and metal layer thickness—against multiple performance metrics: sensitivity (S), figure of merit (FOM), and depth-of-resonance-based FOM (DFOM) [58].
Table 2: SPR Biosensor Performance Through Iterative Optimization
| Optimization Cycle | Key Domain Adjustments | Sensitivity Improvement | FOM Improvement | Key Findings |
|---|---|---|---|---|
| Initial Characterization | Baseline measurement | Reference | Reference | Identified weak spectral intensity and broadening |
| Single-Objective PSO | Focus on individual parameters | +187% | +92% | Revealed parameter interactions and conflicting objectives |
| Multi-Objective PSO | Balanced parameter space exploration | +230% | +111% | Achieved optimal compromise between competing metrics |
| Validation & Robustness | k-means clustering for error mitigation | Final performance confirmed | Final performance confirmed | Achieved LOD of 54 ag/mL (0.36 aM) for mouse IgG |
The iterative process revealed that optimizing for a single parameter (e.g., sensitivity) often degraded other critical performance metrics, necessitating a redefinition of the optimization domain to simultaneously accommodate multiple objectives. The final, iteratively optimized SPR biosensor demonstrated a 230% increase in bulk refractive index sensitivity and a detection limit of 54 ag/mL (0.36 aM) for mouse IgG, enabling single-molecule detection capabilities [58].
The Scientist's Toolkit: Essential Reagents and Materials
Successful iterative optimization requires both statistical expertise and appropriate experimental tools. The following table catalogues essential research reagents and computational solutions frequently employed in biosensor optimization studies.
Table 3: Research Reagent Solutions for Biosensor Optimization
| Category | Specific Examples | Function in Optimization | Application Context |
|---|---|---|---|
| Biorecognition Elements | PYR1 plant hormone receptor scaffolds [59]; CysB transcription factor [56]; Allosteric transcription factors (aTFs) [57] | Malleable binding pockets for sensor reprogramming; Target recognition specificity | Whole-cell biosensors; In vitro detection systems |
| Signal Transduction Components | Gold/chromium layers [58]; Graphene and transition metal dichalcogenides (MoS₂, WS₂) [58]; Fluorescent reporters (eGFP, mCherry) [57] [56] | Signal generation and amplification; Enhanced sensitivity via plasmonics or fluorescence | Optical/SPR biosensors; Fluorescence-based reporters |
| Immobilization Matrices | Streptavidin-coated magnetic beads [60]; Ni-NTA functionalized surfaces [60] | Controlled orientation of biorecognition elements; High-density surface attachment | SELEX procedures; Biosensor surface functionalization |
| Screening & Characterization Tools | Bio-layer interferometry (BLI) [61]; Surface Plasmon Resonance (SPR) [58] [61]; Flow cytometers/FACS [57] [61] | High-throughput binding kinetics measurement; Single-cell resolution screening | Lead candidate validation; Library screening |
| Computational Resources | Particle Swarm Optimization (PSO) algorithms [58]; Rosetta protein design software [59]; DOE software platforms (Weibull++, JMP, Design-Expert) [55] | Multi-parameter optimization; Protein stability prediction; Experimental design generation | In silico modeling and prediction; Statistical design |
Iterative refinement represents both a philosophy and a practical methodology in the optimization of biosensors through Central Composite Design. By recognizing the triggers for domain redefinition and implementing systematic protocols for domain translation, expansion, and contraction, researchers can efficiently navigate complex experimental landscapes toward global optima. The integration of statistical rigor with domain knowledge enables the transformation of initial screening results into robust, high-performance biosensing systems capable of meeting the demanding requirements of modern diagnostics and bioprocessing. As demonstrated through case studies in whole-cell biosensors and SPR platforms, this iterative approach consistently delivers order-of-magnitude improvements in critical performance metrics, accelerating the development of next-generation biosensing technologies.
Leveraging Residual Analysis to Assess Model Adequacy and Fit
In the development of biosensors and the optimization of pharmaceutical formulations, empirical models are crucial for understanding complex relationships between process parameters and final product qualities. These models, often developed using design of experiments (DoE) methodologies like Central Composite Design (CCD), help researchers identify optimal conditions for maximizing performance metrics. However, the mere construction of such models does not guarantee their reliability or predictive capability. Residual analysis provides a powerful, accessible suite of tools for diagnosing potential problems with fitted models, checking modeling assumptions, and suggesting improvements. Unlike summary statistics like R-squared, which can be misleading, residual analysis offers detailed insights into where and how a model may be failing to capture the underlying structure of the data [62].
Within the specific context of biosensor optimization research, where response surface methodology (RSM) and CCD are frequently employed, residual analysis becomes indispensable. The process of optimizing biosensor fabrication involves numerous variables—including material compositions, fabrication parameters, and detection conditions—whose interactions create complex response surfaces. Second-order models are often necessary to capture this complexity, as demonstrated in pharmaceutical optimization where first-order polynomials showed poor model adequacy, while second-order models provided superior fitness, sensitivity to variability, and prediction consistency [63]. Residual analysis serves as the critical bridge between merely fitting a model and ensuring it adequately represents the true underlying process, thereby enabling researchers to develop more reliable, sensitive, and robust biosensing platforms.
Fundamental Principles of Residuals
Definition and Calculation
In the context of regression analysis, a residual is defined as the difference between an observed value and the value predicted by a model. For the ith observation in a dataset, the residual (e_i) is calculated as: ei = yi - ŷi where *yi* represents the ith observed response and ŷ_i represents the corresponding predicted value from the regression function [62]. Geometrically, residuals represent the vertical distances between data points and the corresponding points on the regression curve or surface. A positive residual indicates that the model underpredicted the actual response, while a negative residual indicates overprediction. A residual of zero would represent a perfect prediction for that particular observation.
The pattern and distribution of these residuals across all observations provide critical information about the model's adequacy. When a model perfectly fits the data and all statistical assumptions are met, the residuals should appear as random, unpredictable fluctuations around zero. Systematic patterns in the residuals, however, indicate that the model is missing some systematic component of the data-generating process. In biosensor optimization, where measurements often involve complex biochemical interactions and instrumentation, understanding these residuals is key to refining models that accurately describe how factors like nanomaterial composition, enzyme immobilization parameters, and detection conditions affect sensor performance [15].
Connection to Model Assumptions
Residual analysis is fundamentally linked to verifying the core assumptions of linear regression models, which are often employed in the analysis of data from designed experiments like CCD. These assumptions include: linearity of the relationship between predictors and response, independence of errors, constant variance of errors (homoscedasticity), and normality of error distribution. Violations of these assumptions can lead to biased parameter estimates, incorrect standard errors, and invalid statistical inferences [62].
In the context of biosensor development, where calibration curves are essential for quantitative analysis, the assumption of constant variance is frequently violated. As noted in studies of chemical instrument calibration, the variance of measurements often increases with the concentration of the analyte, creating a heteroscedastic data distribution [64]. Residual plots effectively reveal such patterns, enabling researchers to apply corrective measures such as weighted regression or data transformation. Furthermore, in the optimization of alkali-activated materials using CCD, residual analysis helped confirm model adequacy, revealing that the replacement ratio of rice husk ash significantly affected compressive strength, while particle size had a lesser impact [6]. This insight is directly applicable to biosensor research, where identifying the most influential factors is paramount.
Practical Implementation of Residual Analysis
Graphical Residual Analysis Techniques
Graphical analysis of residuals is the most informative method for assessing model adequacy. Different types of residual plots can diagnose specific model deficiencies, and their interpretation is a critical skill for researchers.
Residuals vs. Fitted Values Plot: This is the primary plot for checking the assumptions of constant variance and linearity. It displays the residuals on the y-axis against the model's predicted (fitted) values on the x-axis. An adequate model will show residuals randomly scattered around zero, forming a horizontal band with no discernible structure as shown in Figure 1. A funnel-shaped pattern (increasing or decreasing spread with fitted values) indicates non-constant variance. A curved pattern, such as a U-shape, suggests that a higher-order term (e.g., a quadratic effect) may be missing from the model [62]. This is particularly relevant in biosensor optimization, where response surfaces often exhibit curvature that requires second-order models for adequate description [63].
Residuals vs. Run Order Plot: This plot checks for independence and the presence of time-related effects, such as instrument drift or operator fatigue. The residuals are plotted against the order in which the experiments were conducted. Randomly scattered points suggest independent errors, while a distinct trend (e.g., consecutive residuals with the same sign) indicates possible correlation over time [62]. For biosensor research, where experiments may be conducted over multiple days or by different technicians, this plot is vital for ensuring data quality.
Normal Probability Plot: This plot assesses the normality assumption of the residuals. If the residuals are perfectly normally distributed, they will fall approximately along a straight line in this plot. Significant deviations from the line, especially in the tails, indicate a departure from normality. While regression analysis is somewhat robust to minor violations of normality, severe deviations can affect hypothesis tests and confidence intervals [62].
Table 1: Interpretation of Common Residual Plots
| Plot Type | Pattern Observed | Potential Issue | Corrective Action |
|---|---|---|---|
| Residuals vs. Fitted | Curved, U-shaped pattern | Missing higher-order term | Add quadratic or interaction terms |
| Residuals vs. Fitted | Funnel shape (spread increases with fitted values) | Non-constant variance | Apply transformation (e.g., log) or use weighted regression |
| Residuals vs. Run Order | Clustering or drift over time | Non-independent errors, process drift | Investigate experimental procedure, randomize run order |
| Normal Q-Q Plot | Points deviate from diagonal line | Non-normal error distribution | Check for outliers, consider transformation |
Figure 1: A workflow for graphical residual analysis. This diagram outlines the iterative process of using residual plots to diagnose and correct model inadequacies.
Quantitative Methods and Statistical Tests
While graphical analysis is indispensable, quantitative tests provide objective measures to support visual interpretations. These are especially useful in borderline cases or when a more formal assessment is required.
Lack-of-Fit Testing: This is a powerful statistical test that compares the variability of the residuals from the model to the variability of pure error, which is estimated from replicated experimental points. In a well-designed CCD, center points provide a direct estimate of pure error. The test statistic for lack-of-fit is calculated as: L = (MSlack-of-fit) / (MSpure_error) where MS represents mean square. A significant lack-of-fit (typically when the p-value is less than 0.05) indicates that the model is missing important terms or that the functional form is incorrect [62]. This test is particularly relevant in the context of RSM, where ensuring the model has no lack-of-fit is a prerequisite for reliable optimization [63] [15].
Analysis of Standardized Residuals: Standardizing residuals (dividing each residual by an estimate of its standard deviation) makes it easier to identify outliers. A common rule of thumb is to flag observations with standardized residuals exceeding ±3 as potential outliers. In a study on calibration equations, the removal of correctly identified outliers significantly improved both the fit and predictive ability of the models [64]. Outliers in biosensor data could result from experimental artifacts, contamination, or instrumentation errors and warrant careful investigation.
Statistical Measures of Fit: Although the R-squared statistic is often overemphasized, it should not be the sole criterion for model adequacy. The standard error of the estimate (s) is a more informative criterion as it is in the units of the response variable, providing a measure of the typical distance between the data points and the regression line [64] [62]. The Prediction Sum of Squares (PRESS) statistic is particularly valuable as it offers a measure of a model's predictive capability by systematically excluding each observation and predicting it from the remaining data. A low PRESS value indicates a model with good predictive performance [64].
Table 2: Key Quantitative Measures for Residual Analysis
| Metric/Test | Purpose | Interpretation |
|---|---|---|
| Lack-of-Fit Test | Tests if the model form is adequate | A significant p-value (<0.05) suggests missing terms in the model |
| Standard Error of Estimate (s) | Measures the average distance data points fall from the regression line | A smaller 's' indicates a tighter fit. Reported in the units of the response variable |
| PRESS Statistic | Assesses the model's predictive ability | A smaller PRESS indicates better predictive performance |
| Standardized Residuals | Identifies potential outliers | Values beyond ±3 standard deviations are considered extreme |
Application in Central Composite Design for Biosensor Optimization
The principles of residual analysis find a critical application in the development and optimization of biosensors using Central Composite Design. CCD is a cornerstone of Response Surface Methodology, used to build second-order models when first-order models are insufficient due to curvature in the response surface [63] [15]. The iterative process of model building and validation in this context is illustrated in Figure 2.
Figure 2: Model development and validation loop in CCD. This iterative process ensures the empirical model derived from a Central Composite Design accurately represents the underlying biosensor system before optimization.
Case Study: Residual Analysis in a Pharmaceutical Optimization Study
A compelling example comes from the optimization of a Bedaquiline Solid Lipid Nanoparticle (SLN) formulation. Researchers used a three-step CCD to model the impact of four independent variables (drug, surfactant, polymer, and lipid) on critical quality attributes like particle size and zeta potential. Their initial analysis with a first-order polynomial showed poor model adequacy, lacking explanatory power and significant predictors. However, upon refining the model, the second-order models provided superior fitness, sensitivity to variability, and prediction consistency. The optimized formulation achieved a near-perfect desirability value of 0.9998 [63]. While not explicitly detailed in the source, this process of model refinement from an inadequate first-order to an adequate second-order model inherently relies on diagnostic tools like residual analysis to detect the missing curvature and interaction terms.
Protocol: Implementing Residual Analysis for a Biosensor Calibration Model
The following protocol outlines the steps for leveraging residual analysis when developing a calibration model for a biosensor, such as one designed for detecting organophosphate pesticides [65] or monitoring glucose [66].
Experimental Design and Data Collection: Conduct experiments according to a predefined CCD or factorial design. For a calibration model, this involves preparing standard solutions of the analyte at different concentrations and measuring the biosensor's response (e.g., change in absorbance, current, or voltage) [64]. Ensure replicates, especially at the center point, to estimate pure error.
Model Fitting: Fit a preliminary calibration model using regression analysis. Begin with a simple linear model (Response = β₀ + β₁ × Concentration). If residual analysis suggests inadequacy, progress to a second-order polynomial (Response = β₀ + β₁ × Concentration + β₂ × Concentration²) [64].
Generate and Analyze Residual Plots:
- Create a residuals vs. fitted values plot. Look for a random scatter. A systematic curve indicates the need for a higher-order term. A funnel shape suggests the need for a weighted regression or a transformation (e.g., logarithmic) of the response data to stabilize variance [64] [62].
- Create a normal probability plot of the residuals. Significant deviations from the diagonal line challenge the normality assumption.
- If the run order is recorded, create a residuals vs. run order plot to check for temporal dependencies.
Conduct Quantitative Tests:
- Perform a lack-of-fit test using the replicate measurements. A significant p-value (p < 0.05) indicates the model is not capturing the true relationship.
- Calculate the standard error of the estimate (s). This value represents the average error in predicting the biosensor's response and should be minimized.
- Examine standardized residuals to identify and investigate any potential outliers that may be unduly influencing the model.
Model Refinement and Validation: Based on the residual analysis, refine the model by adding terms, applying transformations, or removing outliers. Once an adequate model is found, validate its predictive performance using a new set of validation data or through cross-validation techniques like the PRESS statistic [64].
Essential Research Reagent Solutions for Biosensor Development
The following table details key materials used in the development and validation of enzymatic biosensors, as referenced in the studies.
Table 3: Key Reagents in Biosensor Development and Optimization
| Reagent/Material | Function in Research | Example from Literature |
|---|---|---|
| Acetylcholinesterase (AChE) | Enzyme used as biological recognition element for detecting organophosphate pesticides; its inhibition is measured. | Used in a nanocellulose-AgNP biosensor for chlorpyrifos and malathion detection [65]. |
| Dialdehyde Nanocellulose (DANC) | A functionalized, biocompatible nanomaterial that acts as a stabilizing and immobilizing matrix for enzymes and nanoparticles. | Served as a matrix for silver nanoparticles and AChE in a pesticide biosensor [65]. |
| Silver Nanoparticles (AgNPs) | Nanoparticles used for signal amplification and transduction due to their unique optical and electrochemical properties. | Capped with DANC; aggregation caused by enzymatic product led to a detectable signal change [65]. |
| Acetylthiocholine Chloride (ATChCl) | Enzyme substrate; its hydrolysis by AChE produces thiocholine, which interacts with the transducer to generate a signal. | Substrate for AChE in the inhibition-based biosensor for pesticides [65]. |
| Nafion | A perfluorosulfonated ionomer used to coat electrodes, improving selectivity and stability by preventing fouling. | Mentioned as part of a hybrid film in an electrochemical acetylcholinesterase biosensor [65]. |
Residual analysis is not merely a supplementary step in model checking but a fundamental practice for ensuring the validity and reliability of empirical models derived from Central Composite Design and other statistical frameworks. In the high-stakes field of biosensor optimization and pharmaceutical development, where model predictions directly influence experimental direction and product quality, a model that fits the data poorly can lead to wasted resources and erroneous conclusions. By systematically employing both graphical and quantitative residual analysis, researchers can diagnose and correct model deficiencies, validate assumptions, and ultimately build robust models that truly capture the underlying phenomena. This rigorous approach to model assessment, as demonstrated in the transition from inadequate first-order to superior second-order models, is essential for advancing the development of sensitive, reliable, and optimally performing biosensors.
The development of high-performance biosensors represents a critical frontier in analytical chemistry, clinical diagnostics, and food safety monitoring. Achieving optimal performance requires the simultaneous optimization of multiple, often competing response variables: sensitivity (limit of detection), stability (operational lifespan and reproducibility), and cost (manufacturing and material expenses). Traditional one-factor-at-a-time (OFAT) optimization approaches prove inadequate for this challenge, as they ignore interactive effects between factors and only provide localized understanding of the experimental domain [67]. Within the broader thesis of central composite design (CCD) in biosensor optimization research, this technical guide establishes how Design of Experiments (DoE), particularly CCD, provides a structured, statistical framework for effectively balancing these multiple responses.
Biosensors are versatile in many applications, but their widespread adoption as reliable point-of-care tests is often hindered by challenges in systematic optimization [15]. The fundamental obstacle lies in the complex, multi-factorial nature of biosensor fabrication and operation. Parameters such as electrode surface composition, biorecognition element immobilization strategy, and detection conditions interact in nonlinear ways, simultaneously affecting all critical responses [4]. For instance, increasing the amount of an expensive nanomaterial like carboxylated multiwalled carbon nanotubes might enhance sensitivity but adversely impact cost and potentially even stability. Experimental design, a powerful chemometric tool, offers a solution by effectively guiding the development and optimization of ultrasensitive biosensors while explicitly accounting for these trade-offs [15] [4].
This guide details the application of Central Composite Design as a response surface methodology (RSM) for global optimization of biosensor systems. CCD is especially valuable for optimizing ultrasensitive platforms with sub-femtomolar detection limits, where challenges like enhancing the signal-to-noise ratio, improving selectivity, and ensuring reproducibility are particularly pronounced [4]. By employing CCD, researchers can develop empirical models that predict biosensor performance based on multiple input variables, thereby identifying the sweet spot that balances sensitivity, stability, and cost-effectiveness.
Theoretical Foundations of Central Composite Design
Central Composite Design belongs to the class of second-order experimental designs used in Response Surface Methodology to build quadratic models for process optimization. A standard CCD comprises three distinct element types: (1) a factorial or fractional factorial design (2^k points) that estimates linear and interaction effects; (2) axial (star) points (±α) that allow estimation of curvature; and (3) center points (multiple replicates at the mid-level of all factors) that estimate pure error and lack of fit [12] [4]. This composite structure enables efficient estimation of a full quadratic model with a reasonable number of experimental runs, making it particularly suitable for biosensor optimization where experimental resources may be limited.
The mathematical model for a CCD with k factors takes the form of a second-order polynomial: Y = β₀ + ΣβᵢXᵢ + ΣβᵢᵢXᵢ² + ΣΣβᵢⱼXᵢXⱼ + ε Where Y is the predicted response, β₀ is the constant term, βᵢ are the linear coefficients, βᵢᵢ are the quadratic coefficients, βᵢⱼ are the interaction coefficients, and ε represents the error term [4]. This model can accurately capture nonlinear relationships common in biosensor systems, such as the saturation effects in enzyme kinetics or optimal concentration ranges for nanomaterials.
The value of α, the axial distance, determines whether the CCD is rotatable (a desirable property ensuring constant prediction variance at all points equidistant from the center). For a full factorial design with k factors, rotatability is achieved when α = (2^k)^(1/4). Face-centered CCDs (α = ±1) represent a common alternative when practical constraints limit the experimental region [4]. The number of experimental runs required for a CCD with k factors is 2^k + 2k + nc, where nc is the number of center points. This efficiency makes CCD particularly valuable for biosensor optimization, where experimental runs may be time-consuming and resource-intensive.
Table 1: Experimental Matrix for a Three-Factor Central Composite Design
| Run Type | Number of Runs | Factor X₁ | Factor X₂ | Factor X₃ |
|---|---|---|---|---|
| Factorial | 8 | ±1 | ±1 | ±1 |
| Axial | 6 | ±α, 0, 0 | 0, ±α, 0 | 0, 0, ±α |
| Center | 3-5 | 0 | 0 | 0 |
Implementing CCD for Biosensor Optimization: A Step-by-Step Protocol
Phase I: Pre-Experimental Planning and Factor Selection
Step 1: Define Clear Optimization Objectives Quantitatively define the target responses. For biosensors, these typically include:
- Sensitivity: Expressed as limit of detection (LOD), slope of calibration curve, or signal-to-noise ratio.
- Stability: Operational stability (e.g., % signal retention over time or number of cycles), shelf life, or reproducibility (%RSD).
- Cost: Material costs, manufacturing time, or cost per test [68] [69].
Step 2: Identify Critical Factors and Their Ranges Select input variables (factors) likely to influence the responses through prior knowledge or screening designs. Common factors in biosensor development include:
- Nanomaterial concentration (e.g., mg/mL of graphene oxide, carbon nanotubes)
- Bioreceptor concentration (e.g., mg/mL of enzyme, antibody)
- Immobilization time and temperature
- pH of detection medium
- Incubation time [12] [3].
Establish practical ranges for each factor based on preliminary experiments or literature. For example, in optimizing a glucose biosensor, Okman Koçoğlu et al. investigated carboxylated multiwalled carbon nanotubes (0.5-2.0 mg/mL), titanium dioxide nanoparticles (0.25-1.5 mg/mL), and glucose oxidase (5-20 U) as critical factors [12].
Step 3: Select Appropriate CCD Variant Choose the specific CCD configuration based on the number of factors, resource availability, and need for rotatability. For 2-4 factors, a full CCD with 4-5 center points typically provides sufficient data for model development without excessive experimental burden [4].
Phase II: Experimental Execution and Data Collection
Step 4: Randomize and Execute Experimental Runs Conduct all experiments in randomized order to minimize confounding from systematic errors. For biosensor fabrication, this typically involves preparing multiple electrode modifications according to the CCD matrix in random sequence, then measuring all response variables for each fabrication condition [12].
Step 5: Measure All Response Variables For each experimental run, quantitatively measure all predefined responses. For example:
- Sensitivity: Record calibration curves and calculate LOD using standard formulas (e.g., 3σ/slope).
- Stability: Monitor signal response over repeated measurements or time.
- Cost: Calculate based on material consumption and processing time [69].
Phase III: Data Analysis and Multi-Response Optimization
Step 6: Develop Empirical Models for Each Response Use multiple linear regression to fit the second-order polynomial models for each response. Statistically validate model adequacy using ANOVA (Analysis of Variance), Lack-of-Fit tests, and R² values. Nonsignificant terms (p > 0.05) may be removed to simplify models [12] [4].
Step 7: Identify Optimal Conditions Using Desirability Functions The desirability function approach is the most widely used method for multi-response optimization. This technique converts each predicted response (Ŷᵢ) into an individual desirability value (dᵢ) ranging from 0 (undesirable) to 1 (fully desirable), then combines these into an overall desirability (D) as the geometric mean: D = (d₁ × d₂ × ... × dₙ)^(1/n) [69].
Individual desirability functions are defined based on optimization goal:
- Maximization: d = 0 if Y < Yₘᵢₙ; d = 1 if Y > Yₘₐₓ; linear or nonlinear transition between.
- Minimization: d = 1 if Y < Yₘᵢₙ; d = 0 if Y > Yₘₐₓ; linear or nonlinear transition between.
- Target: d = 1 if Y = target; d = 0 if Y outside acceptable range.
Step 8: Validation of Optimized Conditions Confirm model predictions by conducting verification experiments at the identified optimum conditions. Compare predicted and observed response values to validate model adequacy [12].
Diagram 1: CCD Multi-Response Optimization Workflow (76 characters)
Case Studies in Biosensor Optimization Using CCD
Case Study 1: Optimization of Glucose Biosensor Electrode Composition
Okman Koçoğlu et al. demonstrated the application of a five-level, three-factor CCD to optimize the electrode surface composition of an amperometric glucose biosensor [12]. The researchers investigated three critical factors: amounts of carboxylated multiwalled carbon nanotubes (c-MWCNT), titanium dioxide nanoparticles (TiO₂NP), and glucose oxidase (GOx). The central composite design enabled them to systematically explore the factor space and model the relationships between these fabrication parameters and biosensor performance.
Through response surface analysis and multi-objective optimization, the study identified the optimal electrode composition that balanced sensitivity, stability, and cost-effectiveness. The resulting optimized glucose biosensor showed a linear response from 2.0×10⁻⁵ to 1.9×10⁻³ M with a detection limit of 2.1×10⁻⁶ M and sensitivity of 168.5 μA mM⁻¹ cm⁻². The authors compared these performance parameters with those obtained using conventional one-factor-at-a-time optimization and found significant improvements in both sensitivity and stability, demonstrating the superiority of the CCD approach for balancing multiple responses [12].
Table 2: CCD Optimization Parameters for Glucose Biosensor [12]
| Factor | Symbol | Low Level (-1) | High Level (+1) | Optimal Value |
|---|---|---|---|---|
| Carboxylated MWCNT (mg/mL) | X₁ | 0.5 | 2.0 | 1.4 |
| TiO₂ Nanoparticles (mg/mL) | X₂ | 0.25 | 1.5 | 0.8 |
| Glucose Oxidase (U) | X₃ | 5 | 20 | 14 |
Case Study 2: Optimization of Fluorescence Quenching for Lacosamide Detection
A recent study applied CCD to optimize a fluorescence-based biosensing platform for determination of the antiepileptic drug lacosamide in biological samples [3]. The researchers employed boron and nitrogen co-doped graphene quantum dots (BN-GQDs) as the sensing platform and used a four-factor CCD to optimize the fluorescence quenching conditions.
The experimental design included 27 runs with four independent variables: pH of the medium (4-9), buffer volume (1-3 mL), BN-GQDs concentration (1-1.5 mL), and incubation time (2-10 minutes). The response variable was quenching efficiency (F₀/F), where F₀ and F are the fluorescence intensities in the absence and presence of lacosamide, respectively. Through response surface analysis, the study identified optimal conditions of pH 8.6, 3 mL of B-R buffer, 1.5 mL BN-GQDs, and 2.5 minutes incubation time, which maximized sensitivity while maintaining operational stability [3].
This optimization resulted in a highly sensitive method with a linear range of 0.1-5 μg/mL and a detection limit of 0.033 μg/mL, successfully applied to quantify lacosamide in pharmaceutical formulations and pharmacokinetic studies. The study highlights how CCD can effectively balance multiple performance characteristics, including sensitivity, analysis time, and reagent consumption.
Table 3: Analytical Performance Comparison of CCD-Optimized Biosensors
| Biosensor Type | Response Variables Optimized | Performance Before CCD | Performance After CCD |
|---|---|---|---|
| Glucose Biosensor [12] | Sensitivity, Stability, Cost | Suboptimal with OFAT | LOD: 2.1×10⁻⁶ M, Sensitivity: 168.5 μA mM⁻¹ cm⁻² |
| Lacosamide Fluorescence Sensor [3] | Sensitivity, Analysis Time, Reagent Use | Not reported | LOD: 0.033 μg/mL, Linear range: 0.1-5 μg/mL |
| GDH-Based Biosensor [69] | Apparent Michaelis Constant, Output Current, Enzyme Amount | Conflicting objectives | Balanced performance through multi-objective optimization |
The Scientist's Toolkit: Essential Research Reagents and Materials
Table 4: Key Research Reagent Solutions for CCD-Optimized Biosensor Development
| Reagent/Material | Function in Biosensor Development | Application Example |
|---|---|---|
| Carboxylated Multiwalled Carbon Nanotubes (c-MWCNT) | Enhance electron transfer, increase surface area for bioreceptor immobilization | Electrode modification in electrochemical biosensors [12] |
| Titanium Dioxide Nanoparticles (TiO₂NP) | Improve biocompatibility, provide functional groups for immobilization | Electron transfer mediator in enzyme-based biosensors [12] |
| Graphene Quantum Dots (GQDs) | Fluorescent sensing platform with tunable optical properties | Fluorescence quenching-based detection of pharmaceuticals [3] |
| Boron and Nitrogen Co-dopants | Modify electronic properties, enhance quantum yield of GQDs | Improving sensitivity of fluorescence-based sensors [3] |
| Glucose Dehydrogenase (GDH) | Biorecognition element for glucose detection | Model enzyme system for biosensor optimization studies [69] |
| Screen-Printed Electrodes | Low-cost, disposable sensor platforms | Point-of-care biosensor applications [67] |
| Redox Mediators (e.g., Ferrocene derivatives) | Shuttle electrons between bioreceptor and electrode | Second-generation electrochemical biosensors [67] |
Advanced Multi-Objective Optimization Strategies
For complex biosensor systems with strongly conflicting objectives, more sophisticated multi-objective optimization approaches may be required. Žilinskas et al. demonstrated the application of multi-objective optimization with Pareto optimality for designing amperometric biosensors [69]. In this approach, multiple objectives are optimized simultaneously without artificially combining them into a single function, generating a set of non-dominated solutions known as the Pareto front.
In their study of a glucose dehydrogenase-based biosensor, three conflicting objectives were considered: the apparent Michaelis constant (Kₘᵃᵖᵖ), the maximal output current (Iₘₐₓ), and the enzyme amount (AₑE₀). The Pareto front visualization enabled researchers to understand the trade-offs between these objectives and select the most appropriate compromise solution based on specific application requirements [69].
Diagram 2: Multi-Objective Optimization Concept (52 characters)
Machine learning approaches are increasingly being integrated with traditional DoE for enhanced biosensor optimization. Recent research demonstrates how machine learning algorithms can optimize structural parameters of graphene-based biosensors, systematically refining detection accuracy and reproducibility beyond what traditional methods achieve [70]. These approaches are particularly valuable for highly complex biosensor systems with numerous interacting factors where traditional RSM may be insufficient.
Central Composite Design provides a powerful, statistically rigorous framework for balancing the multiple competing responses inherent in biosensor optimization. By enabling simultaneous optimization of sensitivity, stability, and cost, CCD addresses a critical challenge in biosensor development and facilitates the creation of high-performance analytical devices suitable for real-world applications. The methodology's ability to model complex nonlinear relationships and factor interactions while requiring a reasonable number of experimental runs makes it particularly valuable for resource-conscious research environments.
Future developments in this field will likely involve increased integration of machine learning with traditional DoE, hybrid approaches combining physical models with empirical data, and adaptive design strategies that optimize the optimization process itself [70]. As biosensor technology continues to advance toward increasingly complex multienzyme systems and nano-engineered interfaces [68], the role of systematic optimization approaches like CCD will become increasingly critical for translating laboratory innovations into practical diagnostic tools that successfully balance performance, reliability, and cost-effectiveness.
Validation and Comparative Analysis: Demonstrating the Superiority of CCD-Optimized Biosensors
In the rigorous field of biosensor development, the transition from a predicted optimum to a validated, high-performing device is critical. This guide details the experimental protocols and quantitative assessments required to confirm that conditions identified through Central Composite Design (CCD) yield a biosensor with enhanced analytical performance. Framed within biosensor optimization research, this whitepaper provides drug development professionals and researchers with a technical roadmap for experimental validation, from fabricating the optimized sensor configuration to statistically verifying its response against a reference method.
Central Composite Design (CCD) is a powerful response surface methodology used to build a second-order (quadratic) model for a response variable without a full three-level factorial experiment [23]. In biosensor optimization, a CCD model generates a predicted optimum—a specific combination of input factors (e.g., material concentrations, process parameters) expected to deliver the best possible sensor response (e.g., sensitivity, detection limit) [15] [37]. However, this prediction is merely a statistical assertion. Experimental validation is the crucial final step that quantifies success by demonstrating that the biosensor fabricated at the CCD-predicted conditions actually achieves the anticipated performance in practice. This process bridges statistical modeling with tangible, reliable biosensor performance, providing the empirical evidence required for adopting these devices in critical applications like point-of-care diagnostics and drug development [15].
A Central Composite Design is constructed from three distinct sets of experimental runs, which, when combined, allow for the efficient fitting of a quadratic model [23] [22]:
- A Factorial Design: A two-level full or fractional factorial design (coded as -1 and +1) that estimates linear and interaction effects.
- Axial Points (Star Points): A set of points where all factors are at their center levels except one, which is set to a distance ±α from the center. This allows for the estimation of curvature. The value of α is chosen based on desired properties like rotatability [23] [22].
- Center Points: Multiple replicates at the center of the design space (coded as 0) to estimate pure error and model stability [23].
The total number of experiments (N) in a CCD with k factors is given by: N = 2^(k) + 2k + n₀, where n₀ is the number of center points [37]. The resulting data is used to fit a quadratic model of the form: Y = b₀ + ΣbᵢXᵢ + ΣbᵢᵢXᵢ² + ΣbᵢⱼXᵢXⱼ [37] where Y is the predicted response, b₀ is the constant term, bᵢ are the linear coefficients, bᵢᵢ are the quadratic coefficients, and bᵢⱼ are the interaction coefficients.
The workflow below illustrates the iterative process of optimization using CCD, culminating in the experimental validation that is the focus of this guide.
Core Experimental Protocol for Validation
The validation process is a multi-stage investigation designed to comprehensively evaluate the biosensor fabricated at the CCD-predicted optimum.
Fabrication of the Optimized Biosensor
Based on a case study optimizing a glucose biosensor, the fabrication protocol for the validated configuration is as follows [12]:
- Objective: To fabricate a biosensor electrode using the precise amounts of critical components (e.g., carboxylated multiwall carbon nanotubes (c-MWCNT), titanium dioxide nanoparticles (TiO₂NP), and glucose oxidase (GOx)) as determined by the CCD model.
- Materials:
- Electrode base (e.g., glassy carbon electrode)
- Dispersion of c-MWCNT, TiO₂NP, and GOx in a suitable buffer or solvent, prepared at the optimized concentrations.
- Binding agents (if required, e.g., Nafion).
- Methodology:
- Electrode Preparation: Clean and polish the base electrode surface to a mirror finish according to standard electrochemical protocols.
- Modification with Optimized Composite: Prepare a homogeneous suspension containing the optimized quantities of c-MWCNT, TiO₂NP, and GOx. The exact volumes and concentrations are defined by the CCD model.
- Immobilization: Deposit a precise volume of the optimized suspension onto the pre-treated electrode surface and allow it to dry under controlled conditions (e.g., temperature, humidity). A stabilizing layer (e.g., Nafion) may be applied over the modified surface to entrap the enzymatic component and enhance stability.
- Curing: The fabricated biosensor is typically cured or stored at a specified temperature to ensure stability before testing.
Analytical Performance Assessment
The fabricated biosensor is then subjected to a series of tests to quantify its key performance metrics (responses) which were originally optimized in the CCD model.
- Objective: To measure the biosensor's detection limit, linear range, sensitivity, and selectivity under optimal operational conditions.
- Methodology:
- Calibration Curve: Prepare a series of standard solutions of the analyte (e.g., glucose) across a concentration range that encompasses the predicted linear range. Measure the biosensor's response (e.g., amperometric current) for each standard.
- Data Analysis: Plot the response versus analyte concentration. Perform linear regression on the data within the linear range. The slope of this line represents the sensitivity. The linear range is determined from the lowest concentration to the highest where the response maintains linearity (e.g., R² > 0.99).
- Limit of Detection (LOD): Calculate the LOD based on the standard deviation of the response of the blank (or a low-concentration sample) and the slope of the calibration curve (e.g., LOD = 3.3 × σ/S, where σ is the standard deviation of the blank and S is the slope of the calibration curve).
- Repeatability and Reproducibility: Perform repeated measurements (n ≥ 3) at a specific concentration to calculate the relative standard deviation (RSD), quantifying repeatability. Fabricate multiple sensors (n ≥ 3) following the same optimized protocol to assess reproducibility.
Validation with Real Samples
To establish real-world applicability, the biosensor must be tested against complex, real-world samples and compared to a standard reference method.
- Objective: To evaluate the accuracy and robustness of the optimized biosensor in a relevant biological matrix, such as human serum.
- Methodology:
- Sample Preparation: Obtain real samples (e.g., serum). These may be spiked with known concentrations of the analyte to create samples with varying levels.
- Analysis: Measure the analyte concentration in the real samples using both the newly fabricated optimized biosensor and the established reference method (e.g., a standard clinical chemistry analyzer).
- Statistical Correlation: Use statistical tests (e.g., paired t-test, linear regression analysis between the two methods) to determine if there is a significant difference between the results obtained from the optimized biosensor and the reference method. A high correlation coefficient and a slope close to 1 indicate excellent agreement [12].
Quantitative Data Presentation and Comparison
The success of the optimization and validation is demonstrated by comparing the performance of the CCD-optimized biosensor against those developed using less rigorous methods.
Table 1: Comparison of Biosensor Performance Optimized by Different Methods
| Optimization Method | Linear Range (M) | Detection Limit (M) | Sensitivity (μA mM⁻¹ cm⁻²) | Key Advantage |
|---|---|---|---|---|
| One-Factor-at-a-Time (OFAT) | Not specified in detail | Not specified in detail | Not specified in detail | Simple, intuitive; serves as a baseline [71]. |
| 2² Factorial Design | Not specified in detail | Not specified in detail | Not specified in detail | Captures interactions between two factors [12]. |
| Central Composite Design (CCD) | 2.0 × 10⁻⁵ to 1.9 × 10⁻³ | 2.1 × 10⁻⁶ | 168.5 | Captures complex nonlinear relationships and interactions, leading to superior overall performance [12]. |
The quantitative superiority of the CCD approach is further highlighted by its application in other bioprocesses, such as enzyme production.
Table 2: Performance Enhancement via CCD in Bioprocess Optimization
| Process | Performance Metric | OFAT Result | CCD Result | Fold Improvement |
|---|---|---|---|---|
| L-asparaginase Production [71] | Enzyme Specific Activity (IU) | 85.7 | 295.6 | 3.4x |
| Glucose Biosensor Fabrication [12] | Sensitivity & Detection Limit | Baseline Performance | Enhanced Performance | Significant, quantifiable improvement reported |
The Scientist's Toolkit: Essential Research Reagents and Materials
The experimental validation relies on a suite of specialized materials and reagents. The following table details key items essential for biosensor fabrication and optimization based on the cited research.
Table 3: Key Research Reagent Solutions for Biosensor Optimization
| Research Reagent/Material | Function in Biosensor Fabrication | Exemplar Application |
|---|---|---|
| Carboxylated Multiwall Carbon Nanotubes (c-MWCNT) | Enhances electron transfer kinetics and provides a high-surface-area platform for biomolecule immobilization. | Used as a conductive nanomaterial in an amperometric glucose biosensor [12]. |
| Titanium Dioxide Nanoparticles (TiO₂NP) | Can act as a photocatalytic material, improve biocompatibility, and further increase the active surface area. | Co-immobilized with GOx and c-MWCNT to boost the performance of a glucose biosensor [12]. |
| Glucose Oxidase (GOx) | The biorecognition element that specifically catalyzes the oxidation of glucose, producing a measurable signal. | The model enzyme used in the optimization of a glucose biosensor via CCD [12]. |
| L-Asparagine | Substrate for the enzyme L-asparaginase; used to measure enzyme activity during optimization. | Served as the key substrate in the CCD-based optimization of L-asparaginase production [71]. |
| Wheat Bran | A low-cost, complex agro-industrial byproduct used as a solid substrate and nutrient source in fermentation. | Employed as an economical carbon source for the optimized production of L-asparaginase using SSF [71]. |
Visualizing the Validation Workflow
The entire pathway from the CCD model to the confirmation of optimal conditions involves a critical decision point based on experimental data. The following workflow delineates this validation logic and the subsequent actions.
The rigorous experimental validation of predicted optimal conditions is the definitive step that translates a statistical model into a reliable, high-performance biosensor. By following a structured protocol—fabricating the sensor at the specified optimum, quantitatively assessing its analytical figures of merit, and validating its accuracy against a reference method in real samples—researchers can conclusively quantify success. As demonstrated in case studies, employing CCD for optimization leads to significant performance enhancements over conventional methods. This validation framework provides the empirical evidence required for the confident integration of optimized biosensors into drug development pipelines and clinical diagnostics, ensuring they meet the stringent requirements for sensitivity, accuracy, and robustness.
In the field of biosensor research and development, optimizing performance parameters such as sensitivity, dynamic range, and specificity is paramount. The process of optimization, however, presents significant challenges due to the complex, multi-factor, and often non-linear nature of these systems. Traditional One-Variable-at-a-Time (OVAT) approaches have been widely used but possess inherent limitations for characterizing such complex interactions. Design of Experiments (DoE) provides a statistically rigorous, systematic framework for efficient process optimization by varying multiple factors simultaneously. Within the DoE methodology, various experimental designs exist, each with distinct advantages and applications. This review provides an in-depth technical comparison between Central Composite Design (CCD), OVAT, and other DoE approaches, specifically within the context of optimizing biosensor performance, with a focus on central composite design's role in advancing biosensor research.
Fundamental Methodologies and Comparative Framework
One-Variable-at-a-Time (OVAT) Approach
The OVAT approach is a traditional optimization method where a single factor is varied while all other variables are held constant until an optimal response is identified. This process is repeated sequentially for each factor suspected of influencing the response.
- Core Principle: Isolates the effect of a single independent variable by maintaining all other parameters at fixed levels.
- Typical Workflow: The process begins with initial baseline conditions. Factor X1 is varied, and the optimal setting for X1 is determined and fixed. Subsequently, Factor X2 is varied while X1 remains fixed at its new optimum, and this sequence continues for all relevant factors (Xn).
- Key Limitations: The methodology is inherently inefficient, requiring a large number of experimental runs. Crucially, it is incapable of detecting interactions between factors. For instance, the optimal level of a biorecognition element concentration may depend on the pH of the assay buffer. OVAT fails to capture these synergies or antagonisms, which are common in complex biosensing systems, and often results in the identification of local, rather than global, optima [2].
Design of Experiments (DoE) and Central Composite Design (CCD)
DoE is a statistical methodology that systematically varies multiple factors simultaneously according to a predefined experimental matrix to efficiently explore a experimental response space.
- Core Principle: Actively varies all factors together in a structured pattern to model main effects, interaction effects, and quadratic effects, thereby building a comprehensive mathematical representation of the process.
- Common DoE Types:
- Screening Designs (e.g., Definitive Screening Design - DSD): Used in early research phases to rapidly identify the most influential factors from a large set of potential variables with minimal experimental effort [72] [2].
- Full Factorial Designs: Test all possible combinations of factors and their levels, providing complete interaction data but becoming prohibitively large as factors increase.
- Response Surface Methodology (RSM) Designs: Employed after key factors are identified to model curvature and find the true optimum. Central Composite Design (CCD) is a premier RSM design [73] [6].
Central Composite Design (CCD) is a highly efficient RSM design used for building a second-order (quadratic) model for a response without requiring a complete three-level factorial experiment. A standard CCD comprises three distinct elements, making it particularly powerful for biosensor optimization, where response surfaces are often non-linear:
- Factorial Points: A two-level full or fractional factorial design that estimates linear and interaction effects.
- Center Points: Multiple replicates at the center of the design space to estimate pure experimental error and model curvature.
- Axial Points (Star Points): Points located at a distance α (alpha) from the center along each factor axis, which allows for the estimation of quadratic effects [6].
This structure makes CCD exceptionally suited for modeling the complex, non-linear relationships often encountered in biosensor development, such as the effect of reagent concentrations and buffer conditions on sensor signal output.
Head-to-Head Methodology Comparison
The table below provides a structured, quantitative comparison of the OVAT, CCD, and other relevant DoE approaches.
Table 1: Comparative Analysis of Experimental Optimization Methodologies
| Feature | OVAT | Full Factorial | Screening Designs (e.g., DSD) | Central Composite Design (CCD) |
|---|---|---|---|---|
| Primary Goal | Find improved conditions | Characterize all factor effects | Identify critical factors from many | Model curvature and find optimum |
| Factor Interactions | Not detectable | Fully characterized | Limited or confounded | Fully characterized for key factors |
| Experimental Efficiency | Low | Low for many factors | Very High | High for quadratic models |
| Modeling Capability | Linear, local | Linear + Interactions | Main effects, some interactions | Full Quadratic (Linear + Interaction + Curvature) |
| Optimal Output | Local optimum | Map of factorial space | List of significant factors | Global optimum with model equation |
| Best Use Case | Very simple systems | Few factors (<5), linear systems | Early-stage factor screening | Later-stage optimization of critical variables |
Table 2: Experimental Efficiency Comparison for a 3-Factor System
| Methodology | Estimated Number of Experiments | Information Gained |
|---|---|---|
| OVAT | ~15-20 runs (5 levels/factor + replicates) | Linear effect of each factor in isolation |
| Full Factorial (2 Levels) | 8 runs + replicates | Linear and interaction effects |
| Central Composite Design (CCD) | 8 factorial + 6 axial + ~6 center = 20 runs [6] | Linear, interaction, and quadratic effects |
The workflow difference between OVAT and DoE is fundamental. The following diagram illustrates the sequential, one-dimensional path of OVAT versus the multi-dimensional, iterative learning process of a DoE-based approach.
Central Composite Design in Biosensor Optimization
Application of CCD in Biosensor Development
CCD's ability to efficiently model non-linear relationships makes it exceptionally valuable in biosensor optimization, where response surfaces are rarely linear. For instance, the relationship between the concentration of a reporter protein and the dynamic range of a biosensor often follows a curve, increasing to a point before plateauing or decreasing due to effects like steric hindrance or self-quenching.
A study optimizing an RNA integrity biosensor effectively utilized an iterative DoE approach. An initial Definitive Screening Design (DSD) was employed to identify critical factors from a broad set of potential variables. This was followed by a subsequent optimization round, a strategy perfectly suited for a CCD, which led to a 4.1-fold increase in dynamic range and reduced the required RNA concentration by one-third [72]. This two-stage process—screening followed by optimization using a design like CCD—is a powerful paradigm for efficient biosensor development.
Another exemplary application is found in the development of alkali-activated materials (AAMs), where a CCD was used to optimize the compressive strength of mortar by modeling the effects of silica content, particle size, and replacement ratio of rice husk ash (RHA). The study used a CCD with 17 experimental runs (8 factorial points, 6 axial points, and 3 center points) to build a quadratic model. This model successfully identified the optimal combination of factors and confirmed the predicted optimum with a validation experiment, achieving a compressive strength of 49.8 MPa [6]. This directly mirrors the needs of biosensor optimization, where multiple material and chemical parameters must be balanced to maximize performance metrics like sensitivity.
Detailed Experimental Protocol for a CCD
The following is a generalized protocol for executing a Central Composite Design in a biosensor optimization context, based on methodologies from cited research [6] [2].
Define the Optimization Objective and Response: Clearly identify the primary response variable to be optimized (e.g., biosensor dynamic range, sensitivity, limit of detection, signal-to-noise ratio). Ensure a reliable and reproducible quantitative assay is in place to measure this response.
Select the Critical Factors and Their Ranges: Based on prior knowledge or initial screening designs (e.g., a DSD or fractional factorial), choose typically 2 to 4 key continuous factors for optimization (e.g., concentration of biorecognition element, pH of assay buffer, incubation temperature, concentration of a signaling reagent). Define realistic low and high levels for each factor based on practical constraints or previous experiments.
Construct the CCD Matrix: Using statistical software (e.g., Minitab, JMP, Design-Expert), generate the CCD experimental matrix. For three factors, this will typically consist of:
- 8 factorial points (a full 2³ factorial) covering all combinations of the low and high factor levels.
- 6 axial points (also called star points), where one factor is set to ±α (alpha) while all others are at their center points. The value of α is chosen to ensure rotatability; for three factors, it is often approximately 1.682.
- 4-6 center points, where all factors are set at their mid-points. These are crucial for estimating pure error and checking for curvature. The software will output a randomized run order listing the specific factor settings for each of the approximately 18-20 experimental runs.
Execute Experiments and Collect Data: Perform all experimental runs in the randomized order prescribed by the software to minimize the effects of confounding from external variables. Precisely measure the response for each run.
Statistical Analysis and Model Building: Input the response data into the statistical software. Perform multiple linear regression to fit a second-order polynomial model of the form:
Y = β₀ + ΣβᵢXᵢ + ΣβᵢᵢXᵢ² + ΣβᵢⱼXᵢXⱼwhere Y is the predicted response, β₀ is the constant, βᵢ are the linear coefficients, βᵢᵢ are the quadratic coefficients, and βᵢⱼ are the interaction coefficients.- Analyze the Analysis of Variance (ANOVA) to assess the significance and adequacy of the model.
- Check the coefficient of determination (R²) and the adjusted R² to evaluate how well the model explains the variability in the data.
- Examine diagnostic plots (e.g., residual vs. predicted plots) to validate model assumptions.
Visualize the Response Surface and Locate the Optimum: Use the software's optimization tools to generate contour plots and 3D response surface plots. These visualizations help identify the factor levels that produce the optimal response (maximum or minimum, as desired) and understand the interaction between factors.
Model Validation: Perform a final confirmation experiment using the optimal factor levels predicted by the model. Compare the experimentally observed response with the model's prediction to validate the model's accuracy and robustness.
The Scientist's Toolkit: Essential Reagents and Materials
The optimization of a biosensor using DoE relies on a foundation of specific biochemical reagents and materials. The following table details key components commonly featured in such research [74].
Table 3: Key Research Reagent Solutions for Biosensor Optimization
| Reagent/Material | Function in Biosensor Development & DoE | Application Example |
|---|---|---|
| Biorecognition Elements | Provides specificity by binding the target analyte. The type and concentration are often key factors in a DoE. | Antibodies, aptamers, enzymes, or whole cells [74] [75]. |
| Signaling Labels/Nanomaterials | Generates a detectable signal (optical, electrochemical) upon analyte binding. Concentration and type are critical DoE factors. | Gold nanoparticles, fluorescent dyes, enzymes, or quantum dots [74]. |
| Blocking Agents (e.g., BSA, casein) | Coats the sensor surface to minimize non-specific binding, a key source of noise. The choice and concentration are often optimized. | Added to assay buffer or used in sensor strip construction [74]. |
| Membranes/Substrates | The solid support for the assay (e.g., nitrocellulose). Pore size, flow rate, and protein holding capacity are vital material properties. | Used as the conjugate pad and reaction membrane in lateral flow assays [74]. |
| Conjugation Buffers | A stable pH environment is crucial for forming and stabilizing the bioconjugate between the biorecognition element and the label. pH and composition are DoE factors. | Used during the conjugation of antibodies to gold nanoparticles [74]. |
| Statistical Software | The essential tool for designing the experiment, randomizing runs, performing complex statistical analysis, and generating response surface models. | Minitab, JMP, Design-Expert, or R [6] [2]. |
The structure of a Central Composite Design for a two-factor system, which is easily visualized, demonstrates how the design efficiently spans the experimental space to fit a complex model.
The transition from OVAT to structured DoE approaches represents a fundamental shift in optimization strategy for complex systems like biosensors. While OVAT offers simplicity, its inefficiency and inability to detect factor interactions make it unsuitable for modern biosensor development. Central Composite Design stands out as a particularly powerful tool within the DoE arsenal for the critical optimization phase. Its ability to efficiently model non-linear effects and interactions with a manageable number of experiments provides researchers with a deep, predictive understanding of their biosensing system, ultimately leading to superior performance, accelerated development timelines, and more robust diagnostic tools. The integration of a sequential strategy—using screening designs to identify vital factors followed by CCD to model and optimize them—establishes a rigorous and highly effective framework for advancing biosensor research.
The limit of detection (LOD) and sensitivity are two of the most critical figures of merit in biosensor performance, determining the ability to identify minimal analyte concentrations and respond to minute changes in target binding events. Within the framework of central composite design (CCD) for biosensor optimization, a systematic approach to evaluating factors that influence these parameters is paramount. This whitepaper provides a comprehensive technical overview of demonstrated strategies that have successfully enhanced LOD and sensitivity, detailing the experimental methodologies, underlying signaling pathways, and key reagent solutions that underpin these advancements. The focus on CCD contextualizes these achievements as part of a structured, multi-factorial optimization process essential for developing next-generation biosensors for research and clinical applications.
Theoretical Foundations of Detection Limit and Sensitivity
The performance of a biosensor is fundamentally governed by the relationship between its sensitivity and the minimum detectable signal, which is influenced by various noise regimes.
Defining Limit of Detection and Sensitivity
In quantitative terms, the limit of detection (LOD) is typically defined as the lowest analyte concentration that can be consistently distinguished from a blank sample, often calculated as three times the standard deviation of the background signal divided by the sensitivity of the calibration curve. Sensitivity, in the context of photonic sensors like the Mach-Zehnder interferometer (MZI), refers to the magnitude of the sensor's output change per unit change in refractive index, expressed in nm/RIU (refractive index unit) or as a phase shift [76].
Noise Regimes and Their Impact on LOD
For photonic biosensors, the attainable LOD interacts with sensor design in distinct ways depending on the dominant noise source, which can be categorized into three regimes [76]:
- Regime A (Noise unrelated to the sensor): This includes factors like detector noise. In this regime, the LOD scales inversely with the sensing arm length ( Ls ) ( \left( \text{LOD} \propto \frac{1}{Ls} \right) ), allowing for straightforward improvement via design.
- Regime B (Noise affecting a single arm): Examples include inhomogeneity of the sample liquid affecting only the sensing arm. Here, the LOD is independent of the sensing length ( \left( \text{LOD} = 3\sigma_b \right) ) and cannot be improved through sensor design alone.
- Regime C (Noise affecting both arms): This encompasses factors like temperature fluctuations. The LOD depends on the difference in responsiveness of the two arms to the noise source. Designing the sensor for balanced arms can significantly minimize the LOD in this regime.
The following diagram illustrates the logical relationship between sensor design, noise regimes, and the resulting impact on the Limit of Detection, which is central to optimization strategies.
Quantified Advancements in Detection Limits
Recent research has demonstrated significant improvements in LOD across various biosensing platforms and target analytes. The following table summarizes key quantitative achievements.
Table 1: Demonstrated Limits of Detection in Recent Biosensor Research
| Target Analyte | Biosensor Platform | Signal Amplification/Strategy | Reported LOD | Linear Range | Reference |
|---|---|---|---|---|---|
| miR-21 & miR-31 (Colorectal Cancer) | SERS | 3D layered assembly clusters | 3.46 aM & 6.49 aM | Not Specified | [77] |
| miRNA (General) | Electrochemical | AuNP@MXenes nanocomposite | 0.138 fM (miR-141) | 0.5–5×10⁷ fM | [78] |
| piRNA-823 (Colorectal Cancer) | Photoelectrochemical (PEC) | Photosensitive hybridization & enzyme signal amplification | 0.016 fM | Not Specified | [77] |
| α-Fetoprotein (AFP) | SERS Immunoassay | Au-Ag Nanostars platform | 16.73 ng/mL | 500–0 ng/mL | [79] |
| Photonic Crystal Biosensor | Topographically selective functionalization | PNIPAM hydrogel nanoparticle mask | >1 order of magnitude improvement vs. non-selective | Not Specified | [80] |
| miR-92a-3p (Gastric Cancer) | Ratio Fluorescence Biosensor | Catalytic hairpin assembly on nano-platform | 0.047 pM | Not Specified | [77] |
| miRNA-204 (Colorectal Cancer) | Electrochemical Biosensor | Not Specified | High sensitivity (precise LOD not given) | Not Specified | [77] |
Detailed Experimental Protocols for Key Methodologies
Topographically-Selective Functionalization for Nanoscale Sensors
This protocol addresses the key challenge of target depletion due to non-specific functionalization of the large, inactive substrate area relative to the tiny active sensing region [80].
- Step 1: Surface Activation. Clean the sensor chip (e.g., Silicon-on-Insulator with a 2D Photonic Crystal structure) in piranha solution (3:1 v/v conc. sulfuric acid to 30% hydrogen peroxide) for 30 minutes. Caution: Piranha reacts vigorously with organics. Rinse with de-ionized water and dry under a nitrogen stream.
- Step 2: Aminosilanation. Incubate the chip with a 1% (v/v) solution of (3-aminopropyl)dimethylethoxysilane in anhydrous toluene for 20 minutes on an orbital shaker. Wash repeatedly with anhydrous toluene, dry with nitrogen, and bake at 110°C for 30 minutes.
- Step 3: Glutaraldehyde Functionalization. Apply a solution of 1.25% (v/v) glutaraldehyde in MPBS buffer (10mM NaH₂PO₄, 10 mM Na₂HPO₄, 150 mM NaCl, pH 7.2) to the chip for 60 minutes on a shaker. Wash with MPBS and water, then dry under nitrogen.
- Step 4: PNIPAM Nanoparticle Dip-Coating. Dilute synthesized PNIPAM hydrogel nanoparticles 1:100 v/v in double-distilled water. Using a syringe pump, dip the functionalized chip into the solution at 500 µm/min. Submerge half the chip, hold for 30 seconds for equilibration, then withdraw at 1000 µm/min. Allow the chip to dry completely.
- Step 5: Probe Immobilization and Assay. The PNIPAM particles act as a topographically-selective mask, blocking the non-sensing regions. Immobilize capture antibodies or other probes onto the exposed, activated sensing region only. Perform the target binding assay, leading to a significant reduction in non-specific depletion and an order-of-magnitude LOD improvement [80].
SERS-Based Immunoassay Using Au-Ag Nanostars
This protocol leverages the intense electromagnetic field enhancement at the sharp tips of nanostars for ultra-sensitive detection [79].
- Step 1: Nanostar Synthesis and Optimization. Synthesize Au-Ag nanostars via a surfactant-free, aqueous-phase method. Tune the final concentration and morphology of the nanostars via centrifugation time (e.g., 10, 30, 60 minutes). Characterize the SERS enhancement using probe molecules like Methylene Blue (MB).
- Step 2: Functionalization of the SERS Platform. Incubate the optimized nanostars with mercaptopropionic acid (MPA) to form a self-assembled monolayer. Activate the carboxyl groups of MPA using a mixture of 1-Ethyl-3-(3-dimethylaminopropyl) carbodiimide (EDC) and N-Hydroxysuccinimide (NHS) to create amine-reactive esters.
- Step 3: Antibody Conjugation. Covalently attach monoclonal anti-α-fetoprotein antibodies (AFP-Ab) to the activated NHS esters on the nanostar surface via incubation. Purify the Ab-conjugated nanostars to remove unbound antibodies.
- Step 4: Antigen Detection and SERS Readout. Incubate the functionalized nanostars with the sample containing the target AFP antigen. After binding and a washing step, perform SERS measurement. The assay exploits the intrinsic vibrational modes of the captured AFP protein, eliminating the need for an external Raman reporter. The LOD for this assay was determined to be 16.73 ng/mL [79].
The workflow for this SERS-based immunoassay is delineated in the following diagram, showing the sequence from nanostar preparation to final detection.
The Scientist's Toolkit: Key Research Reagent Solutions
The successful implementation of the aforementioned protocols relies on a suite of specialized reagents and materials. This table catalogs the essential components and their functions.
Table 2: Essential Research Reagents for Advanced Biosensor Development
| Reagent/Material | Function/Application | Key Characteristics |
|---|---|---|
| PNIPAM Hydrogel Nanoparticles | Topographically-selective masking agent | Self-assembling, size-exclusive, prevents non-specific probe immobilization [80]. |
| Gold-Silver (Au-Ag) Nanostars | SERS substrate | Sharp-tipped morphology provides intense plasmonic enhancement for Raman signals [79]. |
| Mercaptopropionic Acid (MPA) | Surface functionalization linker | Forms SAM on noble metals, provides carboxyl groups for biomolecule conjugation [79]. |
| EDC/NHS Crosslinker System | Bioconjugation chemistry | Activates carboxyl groups to form stable amide bonds with antibody amines [79]. |
| Aminopropylsilanes | Surface functionalization | Primes inorganic surfaces (e.g., SiO₂) with amine groups for subsequent chemistry [80]. |
| Glutaraldehyde | Homobifunctional crosslinker | Links surface amines to amine-containing biomolecules [80]. |
| Catalytic Hairpin Assembly (CHA) | Nucleic acid signal amplification | Isothermal, enzyme-free amplification for DNA/RNA targets [77]. |
| MXenes (e.g., Ti₃C₂) | Nanocomposite electrode material | High electrical conductivity, large surface area, enhances electrochemical signal [78] [77]. |
The demonstrated efficacy in enhancing biosensor LOD and sensitivity, as detailed in this whitepaper, relies on a multi-faceted approach. This includes innovative physical masking strategies, the deployment of advanced nanomaterials with superior plasmonic and electrochemical properties, and the implementation of sophisticated signal amplification pathways. When executed within the systematic framework of central composite design, these strategies enable researchers to efficiently navigate the complex interplay of factors influencing sensor performance. The continued refinement of these protocols and reagent toolkits is fundamental to achieving the ultra-sensitive, robust, and clinically actionable biosensing systems required for the future of diagnostics and biomedical research.
The optimization of biosensors through central composite design (CCD) requires meticulous consideration of how complex biological matrices influence analytical performance. Serum and plasma represent particularly challenging environments due to their variable protein content, autofluorescence, and diverse pH and ionic strength conditions that can distort detection signals. The core thesis of this work posits that a successful CCD framework for biosensors must systematically account for these matrix effects to ensure reliable real-world applicability. This technical guide examines the performance of various biosensing platforms within complex matrices, providing a foundation for robust experimental design and optimization in diagnostic development.
A critical challenge confounding many biosensor technologies is signal distortion that occurs in various matrices due to heterogeneity in ionic strength, pH, temperature, and autofluorescence [81]. While laboratory demonstrations often show exceptional sensitivity under ideal buffer conditions, applying such sensitive detection to biological samples in non-ideal situations presents a more substantial challenge. This matrix insensitivity becomes a crucial design parameter for biosensors intended for clinical diagnostics, where samples may include serum, plasma, urine, saliva, or cell lysates.
Performance Analysis Across Biological Matrices
Comparative Analysis of Serum, Plasma, and Whole Blood
Table 1: Comparison of Biosensor Performance Across Different Biological Matrices
| Matrix Type | Optimal Application | Key Advantages | Limitations/Considerations | Representative Performance |
|---|---|---|---|---|
| Serum | Neurofilament Light Chain (NfL) detection [82]; BPS and BPP detection [83] | High correlation with plasma results; stable for long-term storage (-20°C for 12 months) [82]; provides standardized data for specific biomarkers | Small positive bias (3.7%) compared to lithium-heparin plasma [82] | Strong correlation with CSF (Spearman's rho = 0.80) [82] |
| Plasma (Lithium-Heparin) | Interchangeable with serum for SARS-CoV-2 serology [84] | High correlation with serum; suitable for large-scale studies | Shows 3.7% positive bias versus serum for NfL [82] | Very strong correlation with serum (Spearman's rho = 0.98) [82] |
| Whole Blood | Detection of BPF, BPAF, and BPAP [83] | Excellent stability; highest concentration of ΣBPs [83] | Requires complex sample preparation | Ideal for reflecting systemic exposure [83] |
| Urine | Bisphenol A (BPA) detection [83] | Minimal matrix effects; highest sensitivity for certain biomarkers [83] | Primarily reflects recent exposure rather than long-term accumulation | Reliable for recent exposure assessment [83] |
| Cerebrospinal Fluid (CSF) | Neurological biomarkers like NfL [82] | Direct measurement of CNS biomarkers | Invasive collection procedure; shows considerable variability in ratio to serum (median 54.5, range 1.6-385.6) [82] | High correlation with serum NfL [82] |
Research demonstrates that while serum and plasma generally show strong correlations for many biomarkers, notable differences exist in certain applications. For neurofilament light chain (NfL), a sensitive biomarker of neuronal damage, serum and lithium-heparin plasma showed a very strong correlation (Spearman's rho = 0.98), though with a consistent 3.7% positive bias for plasma [82]. Similarly, for SARS-CoV-2 serological and neutralization assays, serum and plasma performed equivalently in antibody detection and quantification, confirming their interchangeability for large-scale seroprevalence studies [84].
The stability of biomarkers during storage also varies by matrix. Serum NfL demonstrated excellent stability when stored at -20°C for 12 months, with a mean change from baseline of -7.1% [82]. This stability information is crucial for designing biosensor validation experiments that may involve biobanked samples.
Advanced Biosensor Platforms with Enhanced Matrix Tolerance
Table 2: Performance Metrics of Advanced Biosensing Platforms in Complex Matrices
| Biosensor Technology | Detection Principle | Matrix Tolerance Features | Sensitivity Metrics | Applications in Complex Matrices |
|---|---|---|---|---|
| Magnetic Nanosensor | Giant magnetoresistive (GMR) sensors with magnetic nanoparticle tags [81] | Insensitive to pH (4-10), temperature changes, turbidity, and ionic strength [81] | Detection down to 50 attomolar with amplification; linear dynamic range >6 orders of magnitude [81] | Demonstrated for CEA, VEGF detection in serum, urine, saliva with minimal background [81] |
| Pedestal High-Contrast Grating (PHCG) | Refractometric sensing via guided-mode resonances [85] | Silicon-based structure with low optical losses prevents local heating-induced refractive index changes [85] | Bulk sensitivity: 536 nm/RIU; LoD for avidin: 2.1 ng/mL [85] | Functionalized with biotin for avidin detection in solution [85] |
| PCF-SPR with ML Optimization | Photonic crystal fiber surface plasmon resonance [86] [87] | Machine learning and explainable AI for design optimization to enhance performance in diverse media [86] [87] | Maximum wavelength sensitivity: 125,000 nm/RIU; amplitude sensitivity: -1422.34 RIU⁻¹ [86] [87] | Suitable for detection in blood and serum; cancer cell detection [86] [87] |
| Peptide-based SERS Biosensor | Surface-enhanced Raman spectroscopy with peptide-functionalized AuNPs [54] | High specificity in complex serum matrices; unaffected by nonspecific biofouling [54] | Detection limits of 0.43-8.04 ng/mL for SARS-CoV-2 antibodies; 100% sensitivity in serum [54] | Detection of SARS-CoV-2 antibodies in convalescent and control sera (n=104) [54] |
Innovative biosensing platforms have been developed specifically to address matrix challenges. Magnetic nanosensor technology represents a particularly matrix-insensitive approach, as the magnetic background signal in biological samples is negligible and doesn't interfere with the magnetic transduction mechanism [81]. This platform maintains consistent performance across varying pH (4-10), temperature fluctuations, and different ionic strength conditions that would typically compromise other sensing technologies.
Dielectric sensing structures like pedestal high-contrast gratings (PHCG) offer another matrix-resilient approach by leveraging narrow optical resonances and low absorption characteristics of silicon, which prevent local heating that could alter the local refractive index in measurements [85]. The pedestal design provides an 11.2% improvement in bulk refractive index sensitivity compared to conventional HCG designs, achieving 536 nm/RIU and a lower limit of detection for avidin (2.1 ng/mL) [85].
Experimental Protocols for Matrix-Specific Validation
Serum Stability Assessment Protocol
The evaluation of biomarker stability in serum during frozen storage follows a systematic protocol:
- Sample Collection: Obtain serum samples from relevant patient populations (e.g., neurology department for NfL studies) [82].
- Storage Conditions: Aliquot samples and store at -20°C for long-term stability assessment [82].
- Repeated Measurements: Perform measurements at predetermined intervals (e.g., over 12 months) using the same analytical platform [82].
- Data Analysis: Calculate percentage change from baseline with confidence intervals. In stability studies for NfL, the mean change after 12 months was -7.1% (95% CI: -1.7% to -12.6%) [82].
This protocol confirms that serum NfL remains stable during extended frozen storage, validating the use of biobanked samples for retrospective studies.
Functionalization for Specific Detection in Serum
For avidin detection in solution using pedestal HCG biosensors, the functionalization process involves:
- Surface Silanization: Treat the HCG surface with amino-propyltrimethoxysilane (APTMS) to create reactive amine groups [85].
- Biotin Functionalization: Covalently link biotin to the silanized surface, creating a selective capture layer for avidin [85].
- Sample Exposure: Introduce avidin solutions at various concentrations (in buffer or diluted serum) to the functionalized surface.
- Quantitative Measurement: Monitor resonance shifts to calculate limit of detection (LoD) and limit of quantification (LoQ). PHCG demonstrated a LoD of 2.1 ng/mL and LoQ of 85 ng/mL, significantly better than conventional HCG (LoD 3.2 ng/mL, LoQ 213 ng/mL) [85].
Magnetic Nanosensor Assay for Diverse Biological Fluids
The protocol for matrix-insensitive protein detection using magnetic nanosensors includes:
- Sensor Functionalization: Spot arrays of GMR sensors with capture antibodies specific to target proteins (e.g., CEA, VEGF, lactoferrin) [81].
- Sample Preparation: Present biological samples (serum, urine, saliva, cell lysates) with minimal or no pretreatment [81].
- Sandwich Assay Implementation: Incubate samples with biotinylated detection antibodies followed by streptavidin-coated magnetic nanoparticle tags [81].
- Magnetic Detection: Apply an external magnetic field and measure sensor resistance changes due to bound magnetic nanoparticles [81].
- Multiplexed Analysis: Analyze multiple targets simultaneously by functionalizing different sensors with specific antibodies within the same array [81].
This protocol has been successfully demonstrated for simultaneous detection of multiple tumor markers in PBS, mouse serum, lysis buffer, human urine, human saliva, and human serum with virtually identical signals across matrices [81].
Visualization of Experimental Workflows and Signaling Pathways
Biosensor Experimental Workflow for Complex Matrices
Biosensor Matrix Testing Workflow
Magnetic Nanosensor Signaling Pathway
Magnetic Nanosensor Detection Pathway
The Scientist's Toolkit: Essential Research Reagent Solutions
Table 3: Key Research Reagent Solutions for Biosensor Applications in Complex Matrices
| Reagent/Material | Function | Application Example | Performance Considerations |
|---|---|---|---|
| Gold Nanoparticles (~30 nm) | Signal amplification and plasmonic enhancement [54] | SERS-based biosensors for SARS-CoV-2 antibody detection [54] | Synthesized via Turkevich method; functionalized with peptides [54] |
| 4-Mercaptobenzoic Acid (MBA) | Raman reporter molecule [54] | SERS biosensors for serological analysis [54] | Serves as stabilizer and signal generator in AuNP-based platforms [54] |
| Streptavidin-Coated Magnetic Nanoparticles | Magnetic tags for GMR sensors [81] | Matrix-insensitive protein detection in serum, urine [81] | Enable detection down to attomolar concentrations with minimal background [81] |
| Biotin-Avidin System | High-affinity binding for surface functionalization [85] | PHCG biosensors for avidin detection [85] | Provides specific capture mechanism; LoD of 2.1 ng/mL in PHCG [85] |
| Amino-propyltrimethoxysilane (APTMS) | Surface silanization for functionalization [85] | Dielectric biosensors like PHCG [85] | Creates reactive amine groups for subsequent bioconjugation [85] |
| Photonic Crystal Fiber (PCF) | Waveguide for SPR biosensors [86] [87] | High-sensitivity refractive index detection [86] [87] | Machine learning optimization achieves 125,000 nm/RIU sensitivity [86] [87] |
| Synthetic Peptides (P44 series) | Biorecognition elements for specific antibodies [54] | COVID-19 serological testing [54] | Mimic immunodominant regions of viral proteins; enable variant-specific detection [54] |
The integration of matrix performance data into central composite design frameworks represents a critical advancement in biosensor optimization. The evidence presented demonstrates that successful biosensor deployment requires not only exceptional sensitivity in controlled buffers but maintained performance across diverse biological matrices. Key principles emerge for CCD frameworks: (1) matrix effects must be quantified early in the optimization process, (2) certain platforms like magnetic nanosensors offer inherent advantages for complex media, and (3) biomarker stability in specific matrices should inform sample handling protocols.
Future directions in biosensor development should prioritize matrix-insensitive designs that maintain performance across the spectrum of clinical samples. The incorporation of machine learning and explainable AI, as demonstrated in PCF-SPR biosensor optimization [86] [87], provides powerful tools for navigating the complex parameter space of biosensor design while accounting for matrix effects. As biosensor technologies continue to evolve, their real-world utility will be determined not by their ideal condition performance alone, but by their reliability in the complex, variable matrices encountered in clinical practice.
The development of high-performance biosensors is a complex multidisciplinary endeavor, crucial for advancements in medical diagnostics, environmental monitoring, and food safety. A significant obstacle limiting their widespread adoption, particularly as dependable point-of-care tests, is the challenge of systematic optimization [15] [4]. Traditional optimization methodologies, notably the one-factor-at-a-time (OFAT) approach, remain prevalent despite fundamental limitations. OFAT investigates factors serially while holding others constant, generating only localized knowledge of the optimization space and critically failing to detect interactions between factors [15] [4]. This often leads to suboptimal conditions being identified and a failure to achieve the true performance potential of the biosensing system.
The resource consumption associated with OFAT is substantial. As the number of variables increases, the required number of experimental runs grows linearly, making comprehensive optimization of multifactor systems prohibitively expensive and time-consuming [51] [71]. Furthermore, the data obtained lacks statistical robustness, providing no reliable information on factor significance or interaction effects. Within the specific context of a broader thesis on central composite design (CCD) in biosensor optimization research, this whitepaper provides a detailed cost-benefit analysis. It demonstrates how the adoption of CCD, a powerful Response Surface Methodology (RSM), systematically reduces experimental effort and resource consumption while yielding superior, statistically-validated biosensor performance.
Theoretical Foundations: Central Composite Design as an Efficient Optimization Tool
Central Composite Design is a second-order experimental design that is exceptionally well-suited for optimizing processes where the response is suspected to follow a nonlinear, quadratic function of the experimental variables [15] [51]. It provides a global view of the experimental domain, enabling the construction of a predictive model that maps the relationship between input factors and the output response.
Architectural Composition of a CCD
A CCD efficiently combines several types of experimental points to build a comprehensive model without requiring an exhaustive exploration of the entire experimental space [51]. The total number of experiments (N) in a CCD is calculated as:
N = 2^k + 2k + n
Where:
2^krepresents a two-level full or fractional factorial design.2krepresents the axial (or star) points.nrepresents the center points [51].
This structure is visualized in the following workflow diagram, which outlines the sequential stages of biosensor optimization using CCD:
The CCD Advantage: A Quantitative Comparison with OFAT
The fundamental benefit of CCD lies in its strategic allocation of experimental resources. The table below provides a comparative analysis of the experimental effort required by OFAT versus CCD for a different number of factors (k), assuming a typical OFAT structure (5 levels per factor, with 3 center points for CCD).
Table 1: Quantitative Comparison of Experimental Effort: OFAT vs. CCD
| Number of Factors (k) | OFAT Experiments (5 levels/factor) | CCD Experiments (N = 2^k + 2k + 3) | Experimental Reduction Factor |
|---|---|---|---|
| 2 | 25 | 11 | 2.3x |
| 3 | 125 | 17 | 7.4x |
| 4 | 625 | 27 | 23.1x |
| 5 | 3125 | 45 | 69.4x |
As illustrated, the efficiency gain becomes exponentially more significant as the complexity of the system increases. For a 5-factor optimization—common in biosensor development where variables may include biorecognition element concentration, pH, temperature, nanomaterial loading, and incubation time—CCD can achieve optimization with over 69 times fewer experiments than a comparable OFAT approach [51] [71]. This translates directly into massive savings in time, consumables, and labor.
Case Study: Optimization of a Glucose Biosensor
The practical application and benefits of CCD are effectively demonstrated in the optimization of an amperometric glucose biosensor based on a nanocomposite electrode [12].
Experimental Protocol and Methodology
1. Research Reagent Solutions: Table 2: Key Materials and Their Functions in Glucose Biosensor Fabrication
| Material/Reagent | Function in the Experiment |
|---|---|
| Carboxylated Multiwall Carbon Nanotubes (c-MWCNT) | Enhances electrical conductivity and provides a high surface area for enzyme immobilization. |
| Titanium Dioxide Nanoparticles (TiO₂NP) | Improves biocompatibility and potentially stabilizes the enzyme. |
| Glucose Oxidase (GOx) | The biorecognition element that catalyzes the oxidation of glucose, producing a measurable signal. |
| Phosphate Buffer Saline (PBS) | Provides a stable pH environment for the enzymatic reaction. |
| Glutaraldehyde | Serves as a crosslinking agent to immobilize the enzyme on the electrode surface (if used). |
2. Optimization Procedure:
- Factor Selection: The three critical factors identified for optimization were the amounts of c-MWCNT, TiO₂NP, and GOx [12].
- Experimental Design: A five-level, three-factorial CCD was employed. This design allowed the researchers to model the interactive and quadratic effects of these three components on the biosensor's response (e.g., sensitivity or current).
- Biosensor Fabrication and Testing: Electrodes were fabricated according to the CCD matrix. The amperometric response to glucose was measured for each electrode composition, and the data was fitted to a second-order polynomial model. Analysis of Variance (ANOVA) was used to validate the model's significance and identify the impact of each factor [12].
The following diagram details the specific interactions and relationships between the optimized components and the final biosensor performance:
Results and Cost-Benefit Outcome
The application of CCD led to the identification of an optimal electrode surface composition that would have been extremely difficult to locate using OFAT. The resulting biosensor exhibited excellent performance characteristics [12]. Crucially, the authors compared the CCD-optimized biosensor with those fabricated using OFAT and a simpler 2² factorial design. The biosensor born from the CCD optimization protocol demonstrated superior analytical performance, confirming that the identified optimum was not just resource-efficient to find, but also technically superior [12].
Expanded Applications and Broader Impact
The principles of CCD-driven optimization extend across various domains of biosensor research and development, consistently demonstrating significant resource savings and performance gains.
Table 3: Applications of CCD in Biosensor and Bioprocess Optimization
| Application Context | Optimized Factors | Key Benefit Achieved |
|---|---|---|
| L-Asparaginase Production [71] | pH, temperature, wheat bran, incubation time, yeast extract. | 3.4-fold increase in enzyme specific activity compared to OFAT; dramatic reduction in experimental runs. |
| Fenton Oxidation (Model for Biosensor Interface Chemistry) [51] | Initial dye concentration, Dye:Fe²⁺ ratio, H₂O₂:Fe²⁺ ratio, pH. | Achieved 99% decolorization efficiency; CCD model (R²=0.97) was more predictive than Taguchi method (R²=0.95). |
| Chitosan Spheres Synthesis (for biosensor encapsulation) [20] | Chitosan concentration, acetic acid concentration, NaOH concentration, needle diameter. | Systematically achieved spheres with a target 1 mm diameter and regular morphology for consistent sensor performance. |
Beyond direct biosensor fabrication, the cost-benefit philosophy of systematic experimental design is being applied to related fields. For instance, in Structural Health Monitoring (SHM), which shares with biosensing the challenge of optimizing sensor networks under budgetary constraints, methods are being developed to maximize the "information per unit cost" of deployed sensors [88]. This involves sophisticated optimization statements that either maximize the information gain given a fixed budget or maximize the information-cost ratio, directly mirroring the cost-benefit objectives of CCD in biosensor development [88].
The evidence presented firmly establishes that Central Composite Design is not merely a statistical tool but a pivotal resource-multiplication strategy in biosensor research. The core benefit is twofold: a drastic reduction in experimental effort and a simultaneous enhancement in final product performance. By replacing the inefficient and myopic OFAT approach with the structured, global perspective of CCD, researchers can:
- Accelerate Development Timelines: Reducing the number of experiments from hundreds or thousands to dozens directly shortens the R&D cycle.
- Lower Resource Consumption: Significant savings are achieved in costly reagents, nanomaterials, and specialized labor.
- Uncover Critical Interactions: The models generated reveal synergistic or antagonistic effects between factors, leading to more robust and optimized biosensor designs.
- Build Predictive, Statistically-Validated Models: The resulting empirical models provide deep insight into the system and allow for precise prediction of biosensor performance within the experimental domain.
For the biosensor community, the adoption of CCD and related Design of Experiment methodologies is a critical step toward overcoming the barrier of systematic optimization. It facilitates the sustainable and reliable integration of biosensing devices into point-of-care diagnostics and other applications, ensuring that these powerful tools can be developed both efficiently and effectively.
Conclusion
Central Composite Design stands as a cornerstone methodology for the systematic and efficient optimization of biosensors, decisively overcoming the inefficiencies and oversights of univariate approaches. By enabling the precise modeling of complex variable interactions and nonlinear effects, CCD facilitates the development of biosensors with superior analytical performance, including enhanced sensitivity, lower detection limits, and improved reproducibility. The integration of CCD with emerging technologies like machine learning and artificial intelligence promises to further accelerate the design of next-generation, ultra-sensitive diagnostic platforms. As the biosensor field advances toward point-of-care and liquid biopsy applications, the adoption of robust, data-driven optimization strategies like CCD is paramount. This will be instrumental in translating innovative biosensing concepts into reliable, clinically validated tools that can significantly impact personalized medicine, disease monitoring, and global health outcomes.
References
- 1. Chemometrics-Assisted Enhancement of Electrochemical ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC12019774/]
- 2. A Design of Experiments (DoE) Approach Accelerates the ... [https://www.nature.com/articles/s41598-019-47846-6]
- 3. Central composite design optimized fluorescent method ... [https://www.nature.com/articles/s41598-025-24799-7]
- 4. Perspectives on systematic optimization of ultrasensitive ... [https://pubs.rsc.org/en/content/articlehtml/2024/tc/d4tc02131b]
- 5. OVAT Analysis and Response Surface Methodology Based ... [https://www.mdpi.com/1660-3397/19/5/248]
- 6. Optimization Using Central Composite Design of the ... [https://www.mdpi.com/2673-7108/5/1/5]
- 7. The design, fabrication, and applications of flexible biosensing ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC6310145/]
- 8. Response Surface Methodology (RSM) in Design of ... [https://www.6sigma.us/six-sigma-in-focus/response-surface-methodology-rsm/]
- 9. Response surface methodology [https://en.wikipedia.org/wiki/Response_surface_methodology]
- 10. Response Surface Methodology - Overview & Applications [https://www.neuralconcept.com/post/response-surface-methodology-overview-applications]
- 11. What are response surface designs, central composite ... [https://support.minitab.com/en-us/minitab/help-and-how-to/statistical-modeling/doe/supporting-topics/response-surface-designs/response-surface-central-composite-and-box-behnken-designs/]
- 12. Application of central composite design for the optimization ... [https://pubmed.ncbi.nlm.nih.gov/30467766/]
- 13. 5.3.3.6.3. Comparisons of response surface designs [https://www.itl.nist.gov/div898/handbook/pri/section3/pri3363.htm]
- 14. Application of Central Composite Design for Optimization ... [https://www.mdpi.com/2073-4441/14/16/2539]
- 15. Perspectives on systematic optimization of ultrasensitive ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC11403992/]
- 16. A review of the use of central composite design in ... [https://www.sciencedirect.com/science/article/abs/pii/S0308814625011008]
- 17. Central Composite Design (CCD) [https://2025.help.altair.com/2025/hwdesktop/hst/topics/design_exploration/method_central_composite_design_r.htm]
- 18. Central Composite Design (CCD) [https://www.statease.com/docs/v23.1/designs/ccd/]
- 19. Designing a Central Composite Design [https://community.jmp.com/t5/Statistical-Thinking-for/Designing-a-Central-Composite-Design/ta-p/271998]
- 20. Optimization by Central Composite Experimental Design of ... [https://www.mdpi.com/2313-7673/5/4/63]
- 21. A Comparison of Methods for Estimating Quadratic Effects in ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC3481550/]
- 22. 5.3.3.6.1. Central Composite Designs (CCD) [https://www.itl.nist.gov/div898/handbook/pri/section3/pri3361.htm]
- 23. Central composite design [https://en.wikipedia.org/wiki/Central_composite_design]
- 24. Generate all quadratic interactions in a regression model [https://blogs.sas.com/content/iml/2021/02/15/quadratic-interactions-regression.html]
- 25. How should I model interactions between explanatory ... [https://stats.stackexchange.com/questions/124920/how-should-i-model-interactions-between-explanatory-variables-when-one-of-them-m]
- 26. Climbing the mountain: experimental design for the efficient ... [https://jbioleng.biomedcentral.com/articles/10.1186/s13036-017-0078-z]
- 27. Central composite design (CCD)-Response surface ... [https://www.sciencedirect.com/science/article/pii/S2214157X23007943]
- 28. Nanomaterials in Biosensors: Fundamentals and Applications [https://pmc.ncbi.nlm.nih.gov/articles/PMC7152100/]
- 29. Sensitivity studies and optimization of an impedance- ... [https://www.sciencedirect.com/science/article/pii/S2590137024000438]
- 30. A direct and simple immobilization route for ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC8724356/]
- 31. Electrochemical (bio)sensors based on carbon quantum ... [https://www.sciencedirect.com/science/article/abs/pii/S0003269722004626]
- 32. Nanostructured Materials in Glucose Biosensing [https://pmc.ncbi.nlm.nih.gov/articles/PMC12563017/]
- 33. Enhancing sensitivity towards electrochemical miRNA ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC11271339/]
- 34. An electrochemical biosensor for the detection of microRNA ... [https://jbioleng.biomedcentral.com/articles/10.1186/s13036-025-00492-1]
- 35. Conformational change-based electrochemical biosensor ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC12548363/]
- 36. Emerging electrochemical nanoscale architectures for ... [https://www.sciencedirect.com/science/article/pii/S1385894725017243]
- 37. Central Composite Design for Response Surface ... [https://www.intechopen.com/chapters/74955]
- 38. Artificial intelligence−powered electrochemical sensor [https://pmc.ncbi.nlm.nih.gov/articles/PMC11425101/]
- 39. Central composite design-assisted visual and non-invasive ... [https://www.sciencedirect.com/science/article/abs/pii/S0003267024005221]
- 40. Deep Learning-Enhanced Portable Chemiluminescence ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC11851613/]
- 41. Integration of Artificial Intelligence in Biosensors for Enhanced ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC12564411/]
- 42. Application of Biosensors for Detection of Pathogenic Food ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC7344754/]
- 43. Biosensors—Publication Trends and Knowledge Domain ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC6603684/]
- 44. Biosensors: the new wave in cancer diagnosis - PMC [https://pmc.ncbi.nlm.nih.gov/articles/PMC3781701/]
- 45. Artificial intelligence and biosensors: Transforming cancer ... [https://www.sciencedirect.com/science/article/pii/S2590093525000293]
- 46. Recent advances in biosensors: structure, principles ... [https://www.sciencedirect.com/science/article/abs/pii/S1046202325001665]
- 47. Response Surface Designs Part 1 — Types and Properties [https://www.chromatographyonline.com/view/response-surface-designs-part-1-types-and-properties]
- 48. Stat-Ease » v23.1 » Central Composite Design [https://www.statease.com/docs/v23.1/contents/response-surface-designs/central-composite-design/]
- 49. Challenges and Opportunities in Smart Biosensing for ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC12455640/]
- 50. Smartphone-Based Biosensors: Current Trends ... [https://www.mdpi.com/2673-4591/106/1/10]
- 51. A Comparison of Central Composite Design and Taguchi ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC4167450/]
- 52. Central composite design optimization of cobalt-copper ... [https://www.sciencedirect.com/science/article/abs/pii/S0013468625008333]
- 53. A step by step example of a Central Composite Design (CCD) [https://experimentaldesignhub.com/blog/doe_10/]
- 54. Peptide-based biosensors for variant-specific detection of ... [https://pubs.rsc.org/en/content/articlehtml/2025/ma/d5ma00485c]
- 55. Using Central Composite Design for Process Optimization [https://help.reliasoft.com/articles/content/hotwire/issue146/hottopics146.htm]
- 56. Combining biosensor and metabolic network optimization ... [https://biotechnologyforbiofuels.biomedcentral.com/articles/10.1186/s13068-025-02640-7]
- 57. Development of High-Performance Whole Cell Biosensors ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC7146887/]
- 58. Algorithm assisted comprehensive optimization of SPR ... [https://www.sciencedirect.com/science/article/abs/pii/S0956566325007225]
- 59. Rapid biosensor development using plant hormone ... [https://www.nature.com/articles/s41587-022-01364-5]
- 60. Designing the Future of Biosensing: Advances in Aptamer ... [https://www.mdpi.com/2079-6374/15/10/637]
- 61. Accelerating antibody discovery and optimization with high ... [https://jbiomedsci.biomedcentral.com/articles/10.1186/s12929-025-01141-x]
- 62. Model Validation Using Residuals [https://www.fuelcellstore.com/blog-section/fuel-cell-information/model-validation-using-residuals]
- 63. Model Adequacy in Assessing the Predictive Performance ... [https://www.mdpi.com/2218-0532/92/4/64]
- 64. Evaluation of Calibration Equations by Using Regression ... [https://www.mdpi.com/1424-8220/22/2/447]
- 65. Development of enzyme-based biosensor for residual ... [https://www.sciencedirect.com/science/article/abs/pii/S0026265X24019684]
- 66. Validation and verification framework and data integratio... [https://www.degruyterbrill.com/document/doi/10.1515/cclm-2023-1455/html?lang=en&srsltid=AfmBOoqYxewccf_FCaFJa5Z1SOCf9rb2q6vkeHFeoK5Cb0MdVBWO957_]
- 67. Multivariate Optimization of Electrochemical Biosensors for the ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC10377565/]
- 68. Synergistic optimization strategies for the development of ... [https://www.sciencedirect.com/science/article/abs/pii/S095656632500096X]
- 69. Optimal design of amperometric biosensors applying multi ... [https://www.sciencedirect.com/science/article/abs/pii/S0013468616314189]
- 70. Enhancement and optimization of a graphene-based ... [https://www.nature.com/articles/s41598-025-20570-0]
- 71. Statistical Optimization of Process Parameters by Central Composite Design (CCD) for an Enhanced Production of L-asparaginase by Myroides gitamensis BSH-3, a Novel Species - PMC [https://www.ncbi.nlm.nih.gov/pmc/articles/PMC6359705/]
- 72. Enhancing the performance of an in vitro RNA biosensor ... [https://pubmed.ncbi.nlm.nih.gov/40071731/]
- 73. Designing the design of experiments (DOE) [https://www.sciencedirect.com/science/article/pii/S037877882100582X]
- 74. Review Biosensors and lateral flow immunoassays [https://www.sciencedirect.com/science/article/abs/pii/S0009898125001512]
- 75. Biosensors, Volume 15, Issue 8 (August 2025) – 85 articles [https://www.mdpi.com/2079-6374/15/8]
- 76. Study on the limit of detection in MZI-based biosensor ... [https://www.nature.com/articles/s41598-019-42305-8]
- 77. Applications and development trend of biosensors in the ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC12131493/]
- 78. A concise overview of advancements in ultrasensitive ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC10715268/]
- 79. Biosensors, Volume 15, Issue 9 (September 2025) [https://www.mdpi.com/2079-6374/15/9]
- 80. Enhancing the Detection Limit of Nanoscale Biosensors via ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC3923412/]
- 81. Matrix-insensitive protein assays push the limits of ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC4165514/]
- 82. matrix comparison and long-term stability in frozen serum [https://www.sciencedirect.com/science/article/pii/S0009898125005169]
- 83. Comparison of bisphenols in human urine, whole blood ... [https://www.oaepublish.com/articles/jeea.2025.49]
- 84. Comparative performance of serum and plasma samples in ... [https://pubmed.ncbi.nlm.nih.gov/40374015/]
- 85. Pedestal High-Contrast Gratings for Biosensing [https://www.mdpi.com/2079-4991/12/10/1748]
- 86. Design optimization of high-sensitivity PCF-SPR biosensor ... [https://journals.plos.org/plosone/article?id=10.1371/journal.pone.0330944]
- 87. Design optimization of high-sensitivity PCF-SPR biosensor ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC12435684/]
- 88. Cost–Benefit Optimization of Structural Health Monitoring ... [https://www.mdpi.com/1424-8220/18/7/2174]