Zwitterionic Polymer Brushes: Revolutionizing Antifouling Biosensors for Biomedical Applications
This article comprehensively explores the application of zwitterionic polymer brushes in creating advanced antifouling biosensor surfaces.
Zwitterionic Polymer Brushes: Revolutionizing Antifouling Biosensors for Biomedical Applications
Abstract
This article comprehensively explores the application of zwitterionic polymer brushes in creating advanced antifouling biosensor surfaces. Tailored for researchers, scientists, and drug development professionals, it covers the fundamental antifouling mechanisms, synthesis methodologies, and optimization strategies for these materials. It further provides a critical validation of their performance against traditional coatings like PEG in complex biological matrices, including blood, serum, and food samples. The content synthesizes recent scientific advances to serve as a foundational guide for developing next-generation, robust biosensing platforms for clinical diagnostics, therapeutic drug monitoring, and point-of-care testing.
The Science of Superhydrophilicity: How Zwitterionic Brushes Achieve Ultimate Antifouling
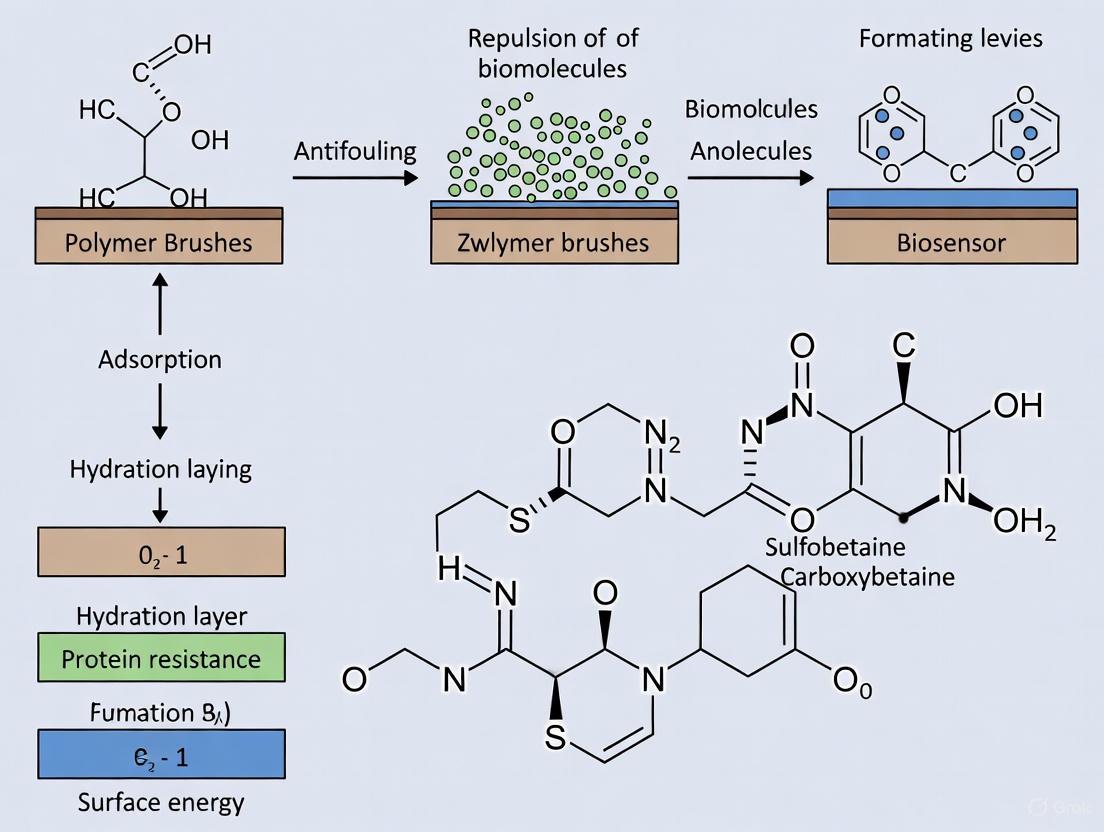
Biological fouling, the non-specific adsorption of proteins, cells, and other biomolecules, is a fundamental challenge compromising the sensitivity, stability, and longevity of biosensors [1]. Zwitterionic polymer brushes have emerged as a superior surface modification strategy to address this problem. These brushes are thin polymer films where chains are tethered by one end to a substrate at high density, creating a brush-like architecture [2]. Each polymer chain contains zwitterionic repeating units—molecular segments carrying a pair of oppositely charged groups, resulting in a net neutral charge [3] [1].
Their exceptional antifouling performance stems from the formation of a tightly bound hydration layer via electrostatic interactions. Water molecules bind so strongly to the charged groups that they form a physical and energetic barrier, preventing the approach and adhesion of foulants [3] [4]. This mechanism provides a more stable and effective antifouling effect than traditional poly(ethylene glycol) (PEG) coatings, which are prone to oxidative degradation and can trigger immune responses [1] [4]. For biosensors, this translates to enhanced reliability and a reduced rate of false positives caused by non-specific binding.
Structural Diversity and Chemical Classes of Zwitterionic Brushes
The antifouling performance and material properties of zwitterionic brushes are dictated by their chemical structure. The major classes of zwitterions are defined by the specific anionic and cationic groups they incorporate, each offering distinct advantages [3].
Table 1: Major Classes of Zwitterionic Polymers for Brush Fabrication
| Zwitterionic Class | Chemical Structure | Key Properties | Common Monomers |
|---|---|---|---|
| Sulfobetaine (SB) | Quaternary ammonium (+) linked to sulfonate (-) [3] | High hydrophilicity, strong resistance to protein/bacterial adhesion, tolerance to high salt levels [3] | Sulfobetaine methacrylate (SBMA) [3] |
| Carboxybetaine (CB) | Quaternary ammonium (+) linked to carboxylate (-) [3] | Excellent non-fouling; carboxylate group allows for facile secondary functionalization with biomolecules [3] | Carboxybetaine methacrylate (CBMA), Carboxybetaine acrylamide (CBAA) [3] |
| Phosphorylcholine (PC) | Phosphorylcholine group mimicking phospholipid headgroups in cell membranes [3] | Exceptional hemocompatibility, ideal for blood-contacting devices and implants [3] | 2-Methacryloyloxyethyl phosphorylcholine (MPC) [3] |
The following diagram illustrates the structural relationships between the different zwitterionic classes and their charged groups:
Diagram: Structural classification of major zwitterionic polymer types, showing their constituent charged groups.
Beyond these classic betaine structures, hybrid systems represent a frontier in material design. For instance, combining zwitterions with PEG can blend superior non-fouling abilities with desired mechanical properties [3]. Furthermore, incorporating zwitterionic polymers into hydrogel networks with nanocomposites like cellulose nanocrystals or Laponite clay can dramatically enhance mechanical strength, making them suitable for load-bearing applications in robust sensors or implants [4].
The Critical Role of Charge Balance and the Antifouling Mechanism
The defining feature of zwitterionic materials is their charge balance—the presence of an equal number of positively and negatively charged groups within the same molecular unit, resulting in overall electroneutrality [3] [1]. This charge balance is not merely a chemical detail but the foundation of their antifouling mechanism.
A balanced charge distribution enables the formation of a dense hydration layer through ionic solvation. Unlike PEG, which binds water via weaker hydrogen bonding, zwitterionic groups electrostatically attract and bind water molecules more strongly, creating a more stable physical barrier [3] [4]. This barrier is energetically unfavorable for biomolecules to displace, effectively resisting the initial, critical step of protein adsorption that leads to broader biofouling [1]. Charge neutrality also eliminates electrostatic driving forces for the non-specific adsorption of charged biomolecules [3].
Recent sensitive measurements, such as total internal reflection microscopy (TIRM), have revealed that even these "electroneutral" surfaces can exhibit significant long-range electrostatic interactions that influence contaminant distribution, challenging the previously overlooked role of long-range forces in their antifouling mechanism [2].
The Anti-Polyelectrolyte Effect
A unique phenomenon exhibited by many zwitterionic brushes is the "anti-polyelectrolyte effect." While typical polyelectrolyte brushes collapse in salt solution, certain zwitterionic brushes swell and stretch [5]. The added salt ions screen the attractive electrostatic interactions between the opposite charges within the brush, causing the polymer chains to expand [5]. This effect can be harnessed to create smart, salt-responsive surfaces with switchable properties, such as tunable friction and gating material uptake [5].
Experimental Protocols for Biosensor Surface Engineering
The successful application of zwitterionic brushes on biosensor surfaces depends on robust and reproducible coating methodologies. The following protocols detail two high-performance techniques.
Protocol: Surface-Initiated Atom Transfer Radical Polymerization (SI-ATRP)
SI-ATRP is a controlled radical polymerization technique that grows polymer chains directly from an initiator-modified substrate, enabling precise control over brush thickness and density [3] [6].
Workflow Overview:
Diagram: Key stages of the SI-ATRP process for growing zwitterionic polymer brushes.
Step 1: Substrate Functionalization. The biosensor substrate (e.g., porous silicon, gold) must be modified with an ATRP initiator. For a silicon surface, this involves:
- Cleaning and Etching: Clean the substrate and treat with a 2.5% aqueous HF solution for 90 seconds to create a hydrogen-terminated, oxide-free surface [6].
- Initiator Attachment: Perform thermal hydrosilylation by immersing the substrate in a purged vinylbenzyl chloride (VBC) solution at 85-100 °C to form a stable Si-C bond and present alkyl halide initiator groups [6].
Step 2: ARGET-ATRP of Zwitterionic Monomer. The "Activators Regenerated by Electron Transfer" (ARGET) method is used for its operational ease, as it tolerates some oxygen [6].
- Reaction Setup: In a schlenk line or glovebox, prepare the polymerization mixture. A typical solution for polySBMA includes:
- Monomer: Sulfobetaine methacrylate (SBMA), 75 mg.
- Solvent: A mixture of deionized water (8 mL) and methanol (7 mL).
- Catalyst: CuBr₂ and ligand Tris(2-pyridylmethyl)amine (TPMA) in methanol.
- Reducing Agent: L-ascorbic acid in methanol (added last to reactivate the catalyst) [6].
- Polymerization: Transfer the degassed solution to vials containing the initiator-functionalized substrates. Allow the reaction to proceed for 16 hours at room temperature to grow the polymer brush [6].
- Reaction Setup: In a schlenk line or glovebox, prepare the polymerization mixture. A typical solution for polySBMA includes:
Step 3: Post-Polymerization Processing. Remove the substrates and wash them extensively in a 1:1 water/methanol solution on a shaker table for 16 hours to remove any unreacted monomer and untethered polymer, then dry with N₂ [6].
Protocol: Photopolymerization Grafting
Photopolymerization is a highly versatile and rapid method for grafting zwitterionic brushes onto surfaces, suitable for patterning and complex geometries [3].
- Step 1: Surface Priming. The substrate is first coated with a photoinitiator. This can be achieved by depositing a thin layer of a benzophenone- or aryl azide-based initiator.
- Step 2: Coating Application. The substrate is coated with an aqueous solution containing the zwitterionic monomer (e.g., SBMA, CBMA).
- Step 3: UV Irradiation. Expose the coated substrate to UV light (e.g., 365 nm wavelength) for a defined period (seconds to minutes). The UV radiation generates radicals that initiate the polymerization of the monomer, grafting the brushes onto the surface.
- Step 4: Washing and Characterization. Thoroughly rinse the grafted surface with deionized water to remove physisorbed material and characterize the resulting brush layer.
Quantitative Performance and Material Data
The performance of zwitterionic brushes can be quantitatively evaluated against traditional coatings and benchmarked using key physicochemical measurements.
Table 2: Antifouling Performance and Key Properties of Coating Technologies
| Parameter | Zwitterionic Brushes (e.g., polySBMA) | PEG Coatings |
|---|---|---|
| Protein Adsorption | "Ultra-low" or "almost zero" from blood serum/plasma [1] [6] | Low, but susceptible to degradation and immune recognition [1] |
| Structural Stability | Chemically robust; stable Si-C bonds from hydrosilylation [6] | Prone to oxidative degradation [1] [4] |
| Hydration Mechanism | Ionic solvation (binds ~7-8 H₂O/unit) [4] | Hydrogen bonding (binds ~1 H₂O/unit) [4] |
| Immunogenicity | Low; biomimetic, does not typically elicit specific antibodies [1] | Can produce anti-PEG antibodies, leading to accelerated blood clearance [1] |
Table 3: Experimentally Measured Long-Range Interactions of polyCBMA Brushes
| NaCl Concentration (mM) | Measured Debye Length, κ⁻¹ (nm) | Theoretical Debye Length (nm) | Observation |
|---|---|---|---|
| 0.1 | 31.1 | 30.4 | Strong electrostatic repulsion observed >300 nm from surface [2] |
| 1.0 | 9.3 | 9.6 | Repulsion range decreases with increasing ionic strength [2] |
| 10.0 | 3.4 | 3.0 | Settling of probe particles occurs; long-range repulsion is suppressed [2] |
The Scientist's Toolkit: Essential Research Reagents and Materials
This table lists critical materials and their functions for developing zwitterionic brushes via SI-ATRP.
Table 4: Essential Research Reagents for SI-ATRP of Zwitterionic Brushes
| Reagent / Material | Function / Role | Example / Note |
|---|---|---|
| Zwitterionic Monomer | Polymer building block; defines antifouling and chemical properties | Sulfobetaine methacrylate (SBMA), Carboxybetaine acrylamide (CBAA) [3] [6] |
| ATRP Initiator | Tethers to substrate and initiates polymer chain growth | Vinylbenzyl chloride (VBC) for thermal hydrosilylation on silicon [6] |
| Catalyst System | Controls the radical polymerization process | Copper(II) bromide (CuBr₂) with Tris(2-pyridylmethyl)amine (TPMA) ligand [6] |
| Reducing Agent | Regenerates active catalyst; enables ARGET-ATRP | L-Ascorbic acid [6] |
| Solvent System | Dissolves monomer and catalyst; influences polymerization kinetics | Water/Methanol mixtures [6] |
The antifouling performance of zwitterionic polymer brushes is fundamentally governed by the mechanism of electrostatic-induced hydration. This phenomenon describes the powerful interaction between the balanced positive and negative charges on the polymer chain and water molecules, leading to the formation of a tightly bound hydration layer that acts as a physical and energetic barrier against fouling. Unlike poly(ethylene glycol) (PEG) which relies on hydrogen bonding for hydration, zwitterionic polymers bind water molecules through stronger, more stable ionic solvation [1] [4]. The simultaneous presence of cationic and anionic groups in a single repeating unit creates a large dipole moment, resulting in a highly polar and superhydrophilic surface that remains electrically neutral overall [7]. The strength of this hydration layer is such that zwitterionic polymers can bind at least 7 to 8 water molecules per repeating unit, forming a denser and more robust barrier than PEG-based materials [4]. This review delineates the quantitative data, experimental protocols, and material toolkits essential for leveraging this mechanism in the development of advanced antifouling biosensor surfaces.
Quantitative Data on Mechanism and Performance
The following tables summarize key quantitative data related to the properties and antifouling performance of zwitterionic polymers, providing a basis for material selection and comparison.
Table 1: Key Properties and Performance Metrics of Major Zwitterionic Polymers
| Zwitterionic Polymer Type | Chemical Traits | Hydration Capacity (H₂O molecules/unit) | Key Antifouling Performance Findings |
|---|---|---|---|
| Poly(sulfobetaine methacrylate) (PSBMA) | Quaternary ammonium with sulfonate group; Highly hydrophilic, salt-tolerant [3]. | Not explicitly quantified, but forms a dense hydration layer [1]. | In salt solution, its surface shows no notable change in potential; Na⁺ can reduce protein adhesion via competitive adsorption [8]. |
| Poly(carboxybetaine methacrylate) (PCBMA) | Quaternary ammonium with carboxylate group; Non-fouling and reactive [3]. | Not explicitly quantified, but forms a dense hydration layer [1]. | Na⁺ diminishes salt bridges between brushes and proteins, reducing adhesion [8]. |
| Poly(2-methacryloyloxyethyl phosphorylcholine) (PMPC) | Phosphorylcholine zwitterion mimicking cell membranes; Excellent hemocompatibility [3]. | Not explicitly quantified, but forms a dense hydration layer [1]. | In salt solution, exhibits significant cation accumulation and a positive shift in surface potential [8]. |
Table 2: Experimental Performance Data from Case Studies
| Study Focus | Material System | Key Performance Metric | Result |
|---|---|---|---|
| Biosensor Electrode Coating [9] | Zwitterionic Polypyrrole (ZiPPy) | Electrochemical Impedance | Lower impedance than bare or PPy-coated electrodes. |
| Non-specific Protein Adsorption | Significantly lower fouling than control electrodes. | ||
| Assay Simplicity | Detection of SARS-CoV-2 antibodies in native human saliva without purification. | ||
| Membrane Modification [10] | CaB NPs-modified PVDF Membrane | Pure Water Flux | Increased by 3.59-fold compared to pristine membrane. |
| Water Contact Angle | Reduced from 74.9° (pristine) to 39.4° (modified). | ||
| Total Fouling Resistance | Decreased by 20–30% for protein foulants. |
Experimental Protocols
Protocol 1: Fabrication of ZiPPy-Coated Biosensor Electrodes via Electropolymerization
This protocol describes the one-step electropolymerization method for creating a low-fouling, zwitterionic biosensor surface, adapted from a study demonstrating SARS-CoV-2 antibody detection in saliva [9].
- Primary Reagents: Zwitterionic pyrrole (ZiPy) monomer, appropriate electrolyte (e.g., phosphate buffer saline), and target affinity ligands (e.g., antibodies, viral proteins).
- Equipment: Electrochemical workstation, working electrode (e.g., carbon or gold), counter electrode, reference electrode.
Procedure:
- Monomer Preparation: Synthesize the ZiPy monomer and dissolve it in the selected electrolyte solution to create the monomer solution [9].
- Ligand Incorporation: Mix the affinity ligands of interest (e.g., SARS-CoV-2 spike protein) directly into the ZiPy monomer solution. This allows for co-immobilization during the subsequent polymerization step [9].
- Surface Preparation: Clean the working electrode (gold or carbon) according to standard protocols (e.g., oxygen plasma treatment for gold, polishing for carbon) to ensure a clean and reactive surface.
- Electropolymerization: Drop-cast the ZiPy monomer solution (with or without ligands) onto the electrode surface. Apply a controlled electrical potential to initiate the polymerization process. The polymerization is typically rapid, completing in under 7 minutes [9].
- Post-processing and Validation: Rinse the coated electrode gently to remove any unbound monomers or ligands. The resulting ZiPPy-coated electrode can be characterized using techniques such as Atomic Force Microscopy (AFM) to confirm coating morphology and Fourier-Transform Infrared Spectroscopy (FTIR) to verify the presence of zwitterionic functional groups [9].
Protocol 2: Investigating Ionic Response Using AFM and Molecular Dynamics
This protocol outlines a methodology for probing the atomic-level interactions between zwitterionic brushes, ions, and proteins, which is critical for understanding the electrostatic-induced hydration mechanism in various ionic environments [8].
- Primary Reagents: Substrates grafted with zwitterionic polymer brushes (PMPC, PSBMA, PCBMA), protein solutions (e.g., BSA), and salt solutions (e.g., NaCl, CaCl₂).
- Equipment: Atomic Force Microscope (AFM), Molecular Dynamics (MD) simulation software and computational resources.
Procedure:
- Sample Preparation: Prepare surfaces grafted with different types of zwitterionic polymer brushes (PMPC, PSBMA, PCBMA) using techniques like Surface-Initiated Atom Transfer Radical Polymerization (SI-ATRP) [3].
- Environmental Control: Immerse the prepared surfaces in salt solutions of varying ionic strengths and types (e.g., monovalent Na⁺ vs. divalent Ca²⁺).
- Surface Potential Measurement: Use AFM-based techniques to measure the surface potential of the zwitterionic brushes in the different salt solutions. This quantifies the accumulation of ions at the brush surface [8].
- Protein Adhesion Assay: Use AFM to quantify the adhesion forces between a protein-coated tip and the zwitterionic brush surface under the same set of ionic conditions [8].
- Molecular Dynamics Simulation: Complement experimental data with all-atom MD simulations. Model the polymer brushes, water molecules, ions, and proteins to observe at an atomic scale how different ions affect the structure of the hydration layer and interact with the charged groups on the polymer [8].
- Data Correlation: Correlate the experimental findings (surface potential shifts, protein adhesion forces) with the simulation data to elucidate the molecular-level mechanism, such as the role of Ca²⁺ in forming "cation bridges" or Na⁺ in competitive adsorption [8].
Mechanism Visualization
Diagram 1: Hydration-based antifouling mechanism.
Diagram 2: ZiPPy biosensor fabrication workflow.
The Scientist's Toolkit: Research Reagent Solutions
Table 3: Essential Research Reagents and Materials
| Reagent/Material | Function/Description | Key Application Note |
|---|---|---|
| SBMA Monomer | Serves as the primary building block for poly(sulfobetaine) brushes, providing quaternary ammonium and sulfonate groups [3]. | Used for creating surfaces with high hydrophilicity and salt tolerance; ideal for fundamental studies on hydration. |
| CBMA Monomer | The key monomer for poly(carboxybetaine), featuring a reactive carboxylate group alongside the quaternary ammonium [3]. | Allows for post-functionalization of the non-fouling surface with peptides or drugs, adding functionality. |
| MPC Monomer | Used to synthesize polymers mimicking the phosphorylcholine head groups of cell membranes [3]. | Excellent for applications requiring high hemocompatibility, such as blood-contacting sensors or implants. |
| Laponite XLG Nanosheets | Acts as a physical crosslinker and nanofiller in hydrogel composites [4]. | Enhances the mechanical strength of zwitterionic hydrogels, making them suitable for load-bearing applications. |
| ZiPy Monomer | A pyrrole derivative functionalized with zwitterionic groups [9]. | Enables rapid, one-step electropolymerization of antifouling coatings on biosensor electrodes. |
The performance and reliability of biosensors are critically dependent on their ability to resist fouling in complex biological environments. For decades, poly(ethylene glycol) (PEG) and its derivatives have served as the gold standard for antifouling coatings, forming a protective barrier through hydrogen bonding with water molecules. However, the emergence of zwitterionic materials, which utilize ionic solvation to create superior hydration layers, presents a paradigm shift in surface modification strategies. This application note details the molecular mechanisms, performance characteristics, and experimental protocols for implementing zwitterionic polymer brushes as advanced antifouling coatings for biosensor surfaces, moving beyond the limitations of traditional PEG-based systems.
The fundamental distinction between these two approaches lies in their hydration mechanisms. PEG surfaces bind water molecules via hydrogen bonding, whereas zwitterionic polymers contain both cationic and anionic groups within their molecular structure that interact with water molecules through stronger, more stable ionic solvation [4]. This difference in fundamental interaction strength translates directly to the enhanced antifouling performance observed in zwitterionic materials, particularly in demanding salt solution environments where biosensors must operate.
Molecular Hydration Mechanisms
Ionic Solvation in Zwitterionic Materials
Zwitterionic polymers feature covalently bound pairs of oppositely charged functional groups in their repeating units. This unique molecular structure facilitates intense ionic solvation, where both cationic and anionic groups undergo direct ion-dipole interactions with water molecules. This results in the formation of a dense, tightly bound hydration layer that acts as a physical and energetic barrier against foulant adhesion [4]. The strength of this interaction is evidenced by the fact that zwitterionic polymers can bind at least 7-8 water molecules per repeating unit, with lower free energies of hydration compared to PEG, making the hydration process more thermodynamically favorable [4].
Molecular dynamics simulations have revealed that the structural organization of this hydration layer varies significantly between different zwitterionic chemistries. For example, the surface of poly(2-methacryloyloxyethyl phosphorylcholine) (PMPC) and poly(sulfobetaine methacrylate) (PSBMA) zwitterionic polymer brushes in salt solution exhibits a significant accumulation of cations, which results in a positive shift in surface potential [8]. This ion-specific response directly influences the resultant antifouling performance through competitive adsorption mechanisms.
Hydrogen Bonding in PEG
Traditional PEG-based materials create hydration layers through hydrogen bonding between ether oxygen atoms in their backbone and water molecules. While effective in many applications, this interaction is generally weaker than ionic solvation, typically binding only one water molecule per ethylene glycol unit [4]. Furthermore, the PEG backbone is chemically unstable and prone to oxidative degradation, which can compromise long-term antifouling performance [4]. Perhaps more critically, PEGylated surfaces can elicit the production of PEG-specific antibodies that recognize PEGylated nanoparticles and liposomes, leading to complement activation, inflammatory immune responses, and rapid clearance from the bloodstream [4]. These inherent drawbacks severely restrict PEG's application in long-use and in vivo systems.
Quantitative Performance Comparison
Table 1: Comparative Analysis of Hydration and Antifouling Properties
| Property | Zwitterionic Materials | PEG-Based Materials |
|---|---|---|
| Hydration Mechanism | Ionic solvation (ion-dipole interactions) | Hydrogen bonding |
| Water Molecules Bound per Unit | ≥7-8 [4] | ~1 [4] |
| Free Energy of Hydration | Lower (more favorable) [4] | Higher (less favorable) [4] |
| Oxidative Stability | High | Low, prone to degradation [4] |
| Immunogenic Response | Low | Can elicit PEG-specific antibodies [4] |
| Salt Solution Performance | Anti-polyelectrolyte effect improves performance [4] | Performance can degrade |
Table 2: Specific Zwitterionic Polymer Performance Characteristics
| Zwitterionic Type | Surface Potential in Salt | Response to Divalent Cations | Protein Adhesion Reduction |
|---|---|---|---|
| PSBMA | Positive shift due to cation accumulation [8] | Enhanced protein adhesion via Ca²⁺ bridges [8] | Significant with monovalent ions [8] |
| PMPC | Positive shift due to cation accumulation [8] | Enhanced protein adhesion via Ca²⁺ bridges [8] | Significant with monovalent ions [8] |
| PCBMA | No notable change in potential [8] | Enhanced protein adhesion via Ca²⁺ bridges [8] | Significant with monovalent ions [8] |
The salt response behavior is particularly noteworthy for biosensor applications. Zwitterionic materials exhibit a unique anti-polyelectrolyte effect, where their performance actually improves in saline environments [4]. When placed in electrolyte solutions, electrostatic interactions between positive and negative charges within the zwitterionic polymer are partially shielded, leading to chain stretching and enhanced hydration [4]. Molecular dynamics simulations have further elucidated that monovalent ions like Na⁺ can diminish salt bridges between zwitterionic polymer brushes and proteins via competitive adsorption, thereby reducing protein adhesion [8].
Experimental Protocols
Molecular Dynamics Simulation for Mechanism Validation
Purpose: To investigate the ionic response mechanism, surface hydration, and protein adhesion behavior of zwitterionic polymer brushes at the atomic level.
Materials:
- Simulation Software: GROMACS, AMBER, or LAMMPS
- Force Fields: CHARMM36 or OPLS-AA
- Zwitterionic Polymer Models: PSBMA, PMPC, PCBMA
- Solution Ions: Na⁺, Ca²⁺, Cl⁻
- Protein Models: Lysozyme, BSA, or other relevant foulants
Procedure:
- System Setup: Construct models of zwitterionic polymer brushes grafted on a surface with sufficient grafting density (≥0.5 chains/nm²) in a simulation box.
- Solvation: Hydrate the system with explicit water models (TIP3P or SPC/E) and add ions to achieve physiological salt concentration (0.15M NaCl or specific ion combinations).
- Equilibration: Perform energy minimization followed by NVT and NPT equilibration runs (300K, 1 atm) for 10-20 ns until system properties stabilize.
- Production Run: Conduct extended molecular dynamics simulations (100-200 ns) with 2 fs time steps.
- Analysis:
- Calculate surface potential distribution normal to the interface
- Quantify water diffusion coefficients within the hydration layer
- Analyze ion distribution profiles (cation/anion accumulation)
- Perform steered molecular dynamics (SMD) to simulate protein approach and adhesion forces
- Monitor hydrogen bonding and ionic interactions between polymer brushes and foulants
Key Parameters:
- Grafting density of polymer brushes
- Ion type and concentration
- Hydration layer thickness and dynamics
- Interaction energy with model proteins
Fabrication of Y-Shaped Glycopeptide Antifouling Biosensors
Purpose: To create an electrochemical biosensor with enhanced antifouling performance through strategic molecular design.
Materials:
- Antifouling Peptides:
- Pep1 (NH₂-CPPPPEKEKEKE) - Linear peptide control
- Pep2 (CPPPPEK(KSRE)DER) - Y-shaped peptide
- Pep3 (CPPPPEK[KS(Glc)RE]DER) - Y-shaped glycopeptide [11]
- Electrode Substrates: Gold working electrodes (2mm diameter)
- Conductive Materials: Chloroplatinic acid (H₂PtCl₆) for Pt nanoparticle electrodeposition
- Binding Chemistry: AFB1-specific aptamer (5'-SH-(CH₂)₆-GTT GGG CAC GTG TTG TCT CTC TGT GTC TCG TGC CCT TCG CTA GGC CCA CA-3') [11]
- Characterization Tools: Zeta potential analyzer, electrochemical工作站
Procedure:
- Electrode Pretreatment:
- Polish gold electrodes with 0.3 and 0.05 μm alumina slurry sequentially
- Clean via sonication in ethanol and deionized water (5 minutes each)
- Dry under nitrogen stream
- Perform electrochemical cleaning in 0.5M H₂SO₄ by cyclic voltammetry scanning until stable gold oxide formation/reduction peaks appear
Platinum Nanoparticle Electrodeposition:
- Prepare electrodeposition solution: 1.0 mM H₂PtCl₆ in 0.1M K₂SO₄
- Use chronoamperometry at -0.25 V (vs. Ag/AgCl) for 60 seconds to deposit Pt NPs
- Rinse thoroughly with deionized water to remove loosely adsorbed ions
Glycopeptide and Aptamer Immobilization:
- Prepare peptide solution: 0.5 mM Y-shaped glycopeptide (Pep3) in PBS (pH 7.4)
- Incubate modified electrodes in peptide solution for 12 hours at 4°C to form stable Pt-S bonds
- Rinse with PBS to remove physically adsorbed peptides
- Activate surface with 2.0 mM 1-ethyl-3-(3-dimethylaminopropyl)carbodiimide (EDC) and 5.0 mM N-hydroxysuccinimide (NHS) for 30 minutes
- Incubate with 1.0 μM thiolated AFB1 aptamer for 2 hours at room temperature
- Block remaining active sites with 1.0 mM 6-mercapto-1-hexanol for 1 hour
Antifouling Performance Evaluation:
- Expose modified electrodes to complex matrices (soy sauce, milk powder, chestnut extracts) for 30 minutes
- Measure electrochemical impedance before and after exposure
- Calculate antifouling efficiency based on signal retention
- Characterize hydration layer properties via molecular dynamics simulations
Quality Control:
- Verify peptide immobilization via zeta potential measurement (target: ~0 mV)
- Confirm Pt NP deposition through increased electrochemical surface area
- Validate aptamer functionality with control measurements in AFB1 standards
Visualization 1: Molecular mechanisms of hydration and their performance outcomes for PEG and zwitterionic materials.
The Scientist's Toolkit: Essential Research Reagents
Table 3: Key Research Reagents for Zwitterionic Antifouling Research
| Reagent/Material | Function/Application | Key Characteristics |
|---|---|---|
| SBMA Monomer (Sulfobetaine methacrylate) | Synthesis of PSBMA zwitterionic polymers | Strong ionic solvation, anti-polyelectrolyte effect [4] |
| PCBMA Monomer (Carboxybetaine methacrylate) | Synthesis of PCBMA zwitterionic polymers | Charge-neutral surface, salt-responsive behavior [8] |
| PMPC Monomer (2-methacryloyloxyethyl phosphorylcholine) | Biomimetic zwitterionic polymer synthesis | Phosphorylcholine group, excellent biocompatibility [8] |
| Y-Shaped Glycopeptide (CPPPPEK[KS(Glc)RE]DER) | High-performance antifouling biosensors | Glucose-modified, enhanced hydration, steric hindrance [11] |
| Laponite XLG Nanosheets | Mechanical reinforcement of hydrogels | Physical crosslinker, enhances strength and toughness [4] |
| Cellulose Nanocrystals (CNCs) | Green reinforcement nanomaterial | Biocompatible, renewable, improves mechanical properties [4] |
| Chloroplatinic Acid (H₂PtCl₆) | Pt nanoparticle electrodeposition | Enhances conductivity, facilitates biomolecule immobilization [11] |
Visualization 2: Development workflow for zwitterionic antifouling biosensor surfaces.
The transition from PEG-based hydration via hydrogen bonding to zwitterionic ionic solvation represents a significant advancement in antifouling technology for biosensor applications. The robust scientific evidence demonstrates that zwitterionic materials offer superior hydration capacity, enhanced stability, and reduced immunogenic response compared to traditional PEG coatings. The strategic design of zwitterionic polymer brushes, particularly when incorporating advanced structural elements like Y-shaped glycopeptides and appropriate mechanical reinforcement strategies, enables the development of biosensor surfaces capable of maintaining performance even in the most challenging biological environments. As research continues to refine our understanding of structure-property relationships in these materials, zwitterionic polymers are poised to become the new benchmark for antifouling surface modifications in diagnostic and therapeutic applications.
Zwitterionic polymers, characterized by their repeating units bearing paired positive and negative charges, have emerged as a leading class of materials for creating antifouling surfaces on biomedical devices and biosensors. These materials outperform traditional poly(ethylene glycol) (PEG) by forming a denser hydration layer through electrostatic interactions with water molecules, resulting in superior stability and resistance to non-specific protein adsorption [1]. Within this class, three families have demonstrated exceptional promise: polycarboxybetaine (PCB), polysulfobetaine (PSB), and polymers based on a 2-methacryloyloxyethyl phosphorylcholine (MPC) motif, which mimics the phosphorylcholine (PMPC) groups found in cell membranes [12]. This application note details the key characteristics, performance data, and experimental protocols for utilizing these zwitterionic families in the development of antifouling biosensor surfaces, providing a practical toolkit for researchers and scientists in the field.
Comparative Analysis of Zwitterionic Families
Table 1: Key Characteristics of Zwitterionic Polymer Families
| Characteristic | Polycarboxybetaine (PCB) | Polysulfobetaine (PSB) | Phosphorylcholine (PMPC) |
|---|---|---|---|
| Chemical Motif | Quaternary ammonium cation + carboxylate anion [13] | Quaternary ammonium cation + sulfonate anion [14] | Phosphorylcholine group from phospholipids [12] |
| Primary Antifouling Mechanism | Superior hydration & strong electrostatic water binding [13] | Strong hydration layer from zwitterionic character [14] | Highly hydrated structure mimicking cell membrane [12] |
| Key Advantage | Enhanced stability & reduced immunogenicity vs. PEG; functionalizable carboxyl group [13] | Excellent resistance to proteins, cells, and bacteria [15] | High biocompatibility; provides lubricious surface [12] |
| Representative Material Forms | Micelles, hydrogels, nanoparticles, lipid coatings [13] [16] | Nanowires, hyperporous networks, nanofibers, mesoporous films [14] | Industrial-scale polymer production for surface treatment [12] |
| Protein Adsorption (Quantitative) | -- | 20–125 ng cm⁻² (from QCM studies); <20 ng cm⁻² for nanostructures against albumin/hemoglobin [14] | Ultra-low protein and lipid adsorption [12] |
| Hemocompatibility Performance | -- | Reduced complement activation (C3bc, sC5b-9) vs. acrylate glass [14] | Prevents thrombus formation; used in cardiovascular devices [12] |
Table 2: Application Performance in Advanced Systems
| Application / System | Polymer & Form | Key Performance Outcome | Reference |
|---|---|---|---|
| mRNA-LNP Delivery | PCB-lipid (2 kDa polymer, DMG chain) in LNPs | Higher mRNA transfection efficiency and functional protein expression vs. PEG-LNPs; mitigates accelerated blood clearance [16] [17] | [16] |
| Implantable Bioelectronics | PSBEDOT (sulfobetaine-functionalized PEDOT) | Low interfacial impedance & high stability; resistance to proteins, cells, and bacteria [15] | [15] |
| Hemocompatible Coatings | PSB Nanowires & Nanofibers | Albumin/Hemoglobin adsorption <20 ng cm⁻²; reduced complement activation and neutrophil response [14] | [14] |
| Medical Device Surface | PMPC-based Polymers | Stable immobilization on metals, ceramics, elastomers; prevents bacterial adhesion and biofilm formation [12] | [12] |
Experimental Protocols for Coating and Evaluation
Protocol: Fabrication of Polysulfobetaine Nanostructured Coatings
This protocol describes the creation of PSB hydrogel coatings with controlled nanostructures on sensor substrates, adapted from methods used to achieve superior hemocompatibility [14].
Materials:
- Substrates: Silicon wafers with SiO₂ layer, glass slides, or Au/quartz resonators.
- Monomer: [2-(Methacryloylamino)propyl]dimethyl(3-sulfopropyl)ammonium hydroxide (sulfobetaine methacrylate, SBMA).
- Crosslinker: 1,4-bis(acryloyl)piperazine (BAP).
- Initiator: 2,2′-azobis(2-methylpropionamidine) dihydrochloride (ABAH).
- Sacrificial Templates: Anodized aluminum oxide (AAO) membrane (for nanowires), latex beads (for hyperporous networks), or a liquid crystalline (LC) medium (for nanofibers).
- Surface Primer: [3-(Methacryloyloxy)propyl]trimethoxysilane.
- Solvents: Water, methanol, triethylamine, toluene, acetone.
Procedure:
- Substrate Functionalization: a. Clean substrates (e.g., SiO₂/Au/quartz) with oxygen plasma or piranha solution. b. Immerse the substrates in a 2% (v/v) solution of [3-(methacryloyloxy)propyl]trimethoxysilane in anhydrous toluene for 12 hours to form a reactive surface. c. Rinse thoroughly with toluene and acetone, then dry under a nitrogen stream.
Template-Polymer Mixture Preparation: a. For Nanowires (PSBAAO): Infiltrate an AAO membrane (200 nm pore diameter) with an aqueous solution containing 1.0 M SBMA, 1% (mol) BAP, and 10 mM ABAH. b. For Nanofibers (PSBLC): Prepare a lyotropic liquid crystal template by mixing 1.5 M AOT in p-xylene with an aqueous solution of 98% SBMA and 1% BAP. Adjust the molar ratio of water to AOT to 12-15.
Photopolymerization: a. Place the template-initiator-substrate assembly in a sealed glass chamber. b. Purge the chamber with nitrogen gas for 15 minutes to remove oxygen. c. Irradiate with UV light (e.g., 365 nm at 5 mW/cm²) for 2 hours to initiate polymerization.
Template Removal: a. Carefully dissolve the sacrificial template: use 1M NaOH for AAO membranes, tetrahydrofuran for latex beads, and a 1:1 water/acetone mixture for the LC medium. b. Rinse the resulting nanostructured PSB hydrogel coating extensively with deionized water and store in PBS.
Evaluation:
- Topography: Analyze surface morphology using Scanning Electron Microscopy (SEM).
- Protein Adsorption: Use Quartz Crystal Microbalance (QCM) to measure non-specific adsorption from solutions of albumin, fibrinogen, or human plasma. Target adsorption levels are <100 ng cm⁻² [14].
- Hemocompatibility: Incubate with human whole blood for 1 hour and analyze biomarkers for complement activation (C3bc, sC5b-9), neutrophil activation (MPO release), and platelet activation.
Protocol: Electrochemical Deposition of Zwitterionic Conducting Polymers
This protocol is for coating metallic biosensor electrodes with zwitterionic conducting polymers like PSBEDOT, which combine antifouling properties with electronic conductivity [15].
Materials:
- Monomer: Sulfobetaine 3,4-ethylenedioxythiophene (SBEDOT) or its derivatives (SBEDOT-4, SBEDOT-5).
- Electrolyte: 0.1 M Sodium nitrate (NaNO₃) in deionized water.
- Working Electrode: Gold or Indium Tin Oxide (ITO) electrode.
- Counter Electrode: Platinum wire.
- Reference Electrode: Ag/AgCl.
Procedure:
- Electrode Preparation: Clean the working electrode (e.g., gold) via cyclic voltammetry in 0.5 M H₂SO₄ or by sonication in isopropanol and water.
- Electropolymerization: a. Prepare an aqueous solution containing 40 mM SBEDOT monomer in 0.1 M NaNO₃ electrolyte. b. Assemble the three-electrode cell with the prepared working, counter, and reference electrodes. c. Use chronoamperometry or cyclic voltammetry to deposit the polymer film. For SBEDOT, apply a constant potential of +1.1 V (vs. Ag/AgCl) for 100-200 seconds.
- Post-processing: Rinse the coated electrode thoroughly with deionized water to remove any unreacted monomer or electrolyte.
Evaluation:
- Electrochemical Impedance Spectroscopy (EIS): Measure interfacial impedance at 1 Hz. PSBEDOT derivatives can achieve impedance ~20 times lower than unmodified PEDOT [15].
- Antifouling Test: Immerse the coated electrode in a 1 mg/mL fibrinogen solution for 1 hour. Protein adsorption should be minimal (3-5% of the level on PEDOT) [15].
- Surface Morphology: Characterize using SEM to observe the polymer morphology.
The Scientist's Toolkit: Essential Research Reagents
Table 3: Key Reagents for Zwitterionic Polymer Research
| Reagent / Material | Function / Role | Example Use Case |
|---|---|---|
| SBMA (Sulfobetaine Methacrylate) | Primary monomer for creating polysulfobetaine (PSB) hydrogels and polymers via radical polymerization [14]. | Fabrication of hemocompatible nanostructured coatings [14]. |
| MPC (2-Methacryloyloxyethyl Phosphorylcholine) | Primary monomer for creating PMPC-based biomimetic polymers [12]. | Surface treatment of medical devices and implants to prevent thrombus formation [12]. |
| Carboxybetaine Acrylate/Methacrylate | Monomer for synthesizing PCB polymers with functionalizable carboxyl groups [13]. | Modification of lipid nanoparticles (LNPs) for mRNA delivery [16]. |
| 1,4-bis(acryloyl)piperazine (BAP) | Crosslinking agent to form stable, three-dimensional hydrogel networks from linear polymers [14]. | Creating mechanically robust PSB hydrogel coatings [14]. |
| [3-(Methacryloyloxy)propyl]trimethoxysilane | Coupling agent that provides surface-tethered polymerizable groups on oxide surfaces (Si/SiO₂, ITO) [14]. | Priming biosensor substrates for robust grafting of polymer brushes [14]. |
| Azobis(2-methylpropionamidine) dihydrochloride (ABAH) | Water-soluble radical initiator for photochemically induced polymerization. | Initiating the polymerization of SBMA in aqueous solutions and templates [14]. |
| Anodized Aluminum Oxide (AAO) Membrane | Sacrificial template with nano-sized channels for creating polymer nanowire structures [14]. | Fabricating PSB nanowire coatings to enhance hydration and antifouling [14]. |
The Critical Role of Grafting Density and Molecular Weight in Fouling Resistance
Biofouling, the non-specific adsorption of proteins, cells, and microorganisms to surfaces, presents a significant challenge to the reliability and longevity of biomedical devices and biosensors [1]. Even minor biofilm formation can compromise the function of implantable sensors, diagnostic platforms, and drug delivery systems, leading to inaccurate readings, device failure, and adverse patient outcomes [18]. Surface modification with zwitterionic polymer brushes has emerged as a powerful strategy to combat biofouling due to their superior ability to form a tightly bound hydration layer that acts as a physical and energy barrier against foulant adhesion [19] [1].
While zwitterionic materials are recognized for their excellent antifouling properties, their performance is not intrinsic but is critically dependent on specific structural parameters. Among these, grafting density and molecular weight (which directly influences brush thickness) are two of the most crucial factors determining fouling resistance [20] [21]. Optimizing these parameters is essential for developing highly effective, stable antifouling surfaces for sensitive applications such as biosensors. This Application Note details the experimental protocols and analytical methods for fabricating and characterizing zwitterionic polymer brushes with controlled grafting density and molecular weight, and for evaluating their antifouling performance.
Theoretical Background: Why Grafting Density and Molecular Weight Matter
Zwitterionic polymers, such as poly(sulfobetaine methacrylate) (pSBMA), possess a molecular structure where each repeating unit carries both a positive and a negative charge, resulting in overall electrical neutrality [1]. Their exceptional antifouling properties stem from their superhydrophilicity, which enables them to bind water molecules more strongly than other hydrophilic materials like polyethylene glycol (PEG) [19]. This formation of a dense hydration layer through electrostatic interactions is the primary mechanism that prevents the approach and adsorption of biomolecules [1] [22].
The efficacy of this hydration barrier is profoundly affected by the physical conformation of the polymer brushes on the surface, which is controlled by grafting density and molecular weight:
- Grafting Density (σ): Defined as the number of polymer chains per unit area, it determines how closely the polymer chains are packed. At low densities, chains exist in a "mushroom" regime, offering poor surface coverage. At high densities, chains are forced to stretch away from the surface into a "brush" regime, creating a dense, uniform, and impenetrable antifouling barrier [20] [21].
- Molecular Weight (MW) and Brush Thickness: The molecular weight of the polymer chains dictates their length and the resulting dry thickness of the brush layer. Combined with grafting density, it determines the hydration and steric repulsion properties of the interface [23].
The relationship between these parameters for zwitterionic brushes is complex. Unlike some hydrophilic polymers, the antibiofouling performance of polyzwitterions does not always follow a simple polynomial curve with a clear maximum. Their unique dipolar structure can lead to interchain associations, which may compromise fouling resistance if the grafting density is not optimized to prevent these interactions [20]. Molecular dynamics simulations have confirmed that zwitterionic brushes exhibit stronger interactions with water and higher surface resistance to proteins than traditional PEG brushes, but this performance is contingent on their optimal packing structure [22].
Experimental Protocols
The following section provides a detailed, step-by-step methodology for the fabrication of zwitterionic polymer brushes with controlled parameters and the evaluation of their antifouling performance.
Fabrication of pSBMA Brushes via Surface-Initiated ATRP
Surface-Initiated Atom Transfer Radical Polymerization (SI-ATRP) is a robust controlled radical polymerization technique that allows for precise control over the molecular weight and grafting density of polymer brushes [19] [21].
Substrate Pre-Functionalization (Initiator Immobilization)
- Objective: To create a uniform layer of ATRP initiation sites on the substrate surface.
- Materials:
- Procedure:
- Surface Activation: Clean the substrate thoroughly (e.g., Soxhlet extraction for PET [21], oxygen plasma for silicon).
- Aminolysis (for PET): Immerse PET fabrics in a 40% (v/v) solution of ethylenediamine in ethanol at 55°C. Vary the reaction time (15 min to 3.5 hours) to control the density of introduced amine groups, which directly influences subsequent initiator density and polymer grafting density [21].
- Dopamine Coating (for PES): Immerse the substrate in a freshly prepared dopamine solution (2 mg/mL in Tris-HCl buffer, pH 8.5) for several hours to form a thin, adhesive polydopamine film. Rinse with deionized water [19].
- Initiator Immobilization: Transfer the amine-functionalized substrate to a Schlenk flask containing anhydrous THF. Add BIBB (1.1 mL, 9.5 mmol) and TEA (0.7 mL, 5 mmol) as an acid scavenger under a nitrogen atmosphere. React for 24 hours at room temperature with stirring.
- Washing: Remove the substrate (now
PET_BrorPES_Br) and wash extensively with THF, ethanol, and water to remove any physisorbed initiator. Dry under a stream of nitrogen.
SI-ATRP of Sulfobetaine Methacrylate (SBMA)
- Objective: To grow pSBMA brushes from the initiator-functionalized surface with controlled molecular weight and thickness.
- Materials:
- Initiator-functionalized substrate (
PET_Br,PES_Br) - Sulfobetaine methacrylate (SBMA) monomer
- Copper(I) bromide (CuBr) catalyst
- Copper(II) bromide (CuBr2) deactivator
- 2,2'-Bipyridyl (bpy) ligand
- Methanol and deionized water (4:1 v/v) as solvent
- Initiator-functionalized substrate (
- Procedure:
- Solution Preparation: In a nitrogen-purged Schlenk flask, add methanol/water (5 mL), SBMA monomer (10 g, 35 mmol), bipyridyl (0.312 g, 2 mmol), CuBr (0.144 g, 1 mmol), and CuBr2 (0.011 g, 0.05 mmol). The Cu(II) species is added to control the polymerization rate and improve livingness.
- Degassing: Purge the polymerization mixture with nitrogen or argon for 20-30 minutes to remove oxygen.
- Polymerization: Transfer the solution to a flask containing the initiator-functionalized substrate under a nitrogen atmosphere. Seal the flask and allow the polymerization to proceed at room temperature. Vary the polymerization time (1 to 24 hours) to control the molecular weight and dry thickness of the pSBMA brushes [23] [21].
- Termination: Remove the substrate from the solution and rinse copiously with ethanol and water to terminate the reaction and remove unreacted monomer and catalyst residues.
- Characterization: The successful grafting of pSBMA brushes can be confirmed by ATR-FTIR (characteristic peaks at ~1030 cm⁻¹ and ~1170 cm⁻¹ for the sulfonate group) [21]. Brush thickness can be measured using ellipsometry or atomic force microscopy (AFM).
Quantitative Assessment of Antifouling Performance
Protein Adsorption Test
- Objective: To quantify the non-specific adsorption of proteins onto the modified surfaces.
- Materials:
- pSBMA-grafted substrates and controls (pristine, initiator-only)
- Bovine Serum Albumin (BSA) solution (1 mg/mL in PBS)
- Phosphate Buffered Saline (PBS), pH 7.4
- Micro-BCA or Bradford Protein Assay Kit
- Procedure:
- Incubate each substrate in 1 mL of BSA solution for 1 hour at 37°C.
- Remove the substrate and gently rinse with PBS to remove loosely bound protein.
- Transfer the substrate to a fresh tube with 1 mL of 1% (w/v) sodium dodecyl sulfate (SDS) solution and incubate at 60°C for 1 hour to desorb the bound proteins.
- Measure the protein concentration in the SDS eluate using a standard micro-BCA or Bradford assay.
- Calculate the surface density of adsorbed BSA (µg/cm²). High-performing zwitterionic brushes should show a >90% reduction in BSA adsorption compared to the pristine substrate [19].
Dynamic Biofouling Assay
- Objective: To evaluate the resistance to bacterial adhesion and biofilm formation under dynamic conditions.
- Materials:
- pSBMA-grafted substrates and controls
- Escherichia coli or Staphylococcus epidermidis culture in a suitable growth medium (e.g., LB)
- Flow cell or parallel flow chamber
- Confocal Laser Scanning Microscope (CLSM)
- Live/Dead bacterial viability stain
- Procedure:
- Circulate a bacterial suspension (~10⁸ CFU/mL) over the substrate surfaces in a flow cell at a physiologically relevant shear rate for several hours.
- Stop the flow and gently rinse the channels with sterile PBS to remove non-adhered cells.
- Stain the substrates with a Live/Dead BacLight stain.
- Image the surfaces using CLSM to visualize and quantify the adhered bacterial biomass.
- Analyze the images to determine the percentage surface coverage and the ratio of live to dead cells. A successful coating can show a ~90% decrease in live biomass compared to controls [19] [23].
Data Presentation and Analysis
Table 1: Impact of Polymer Brush Parameters on Fouling Resistance and Material Properties
| Parameter Varied | Experimental Range | Observed Impact on Brush Properties | Effect on Fouling Resistance | Citation |
|---|---|---|---|---|
| Polymerization Time (MW/Thickness) | 1 to 24 hours | Dry thickness from ~6 nm to 180 nm | Great resistance against S. epidermidis and A. niger; effect is significant initially but plateaus at higher MW [23]. | [23] [21] |
| Aminolysis Time (Grafting Density) | 15 min to 3.5 hours | Increased water retention capacity; higher density of initiator sites. | Shorter times lead to insufficient coverage; longer times optimize density and maximize protein repellency [21]. | [21] |
| Alkyl Spacer Length (CSL) | CSL=3 vs. CSL=4 | CSL=3: hydrophilic regardless of MW. CSL=4: hydrophobicity increases with MW. | Both show great submerged antifouling, indicating CSL and MW are secondary to a well-formed brush layer [23]. | [23] |
The Scientist's Toolkit: Essential Research Reagents
Table 2: Key Reagents for SI-ATRP of Zwitterionic Brushes
| Reagent / Material | Function / Role | Critical Consideration |
|---|---|---|
| Sulfobetaine Methacrylate (SBMA) | The zwitterionic monomer that forms the antifouling brush. | Purify before use to remove inhibitors. The charged group influences hydration capacity [22]. |
| α-Bromoisobutyryl bromide (BIBB) | The ATRP initiator that is covalently anchored to the substrate. | Handle under inert, anhydrous conditions due to high reactivity with water and air. |
| Copper(I) Bromide (CuBr)/ Bipyridyl | Catalyst/Ligand system that controls the ATRP equilibrium. | Strict removal of oxygen is crucial to prevent catalyst deactivation. |
| Copper(II) Bromide (CuBr₂) | Deactivator agent that improves control over the polymerization. | Adding a small amount helps suppress unwanted chain termination, enabling higher MW. |
| Ethylenediamine (EDA) | Creates amine functional groups on inert polyester (PET) surfaces via aminolysis. | Reaction time is a key variable to tune the final grafting density of the polymer brush [21]. |
Workflow and Relationship Visualization
The following diagram summarizes the logical and experimental pathway for optimizing zwitterionic brushes, from parameter control to performance outcome.
For researchers and drug development professionals engineering advanced biosensor interfaces, the meticulous control over grafting density and molecular weight is not merely a synthetic detail but a fundamental requirement for achieving maximum fouling resistance. The protocols outlined herein provide a reliable roadmap for fabricating zwitterionic polySBMA brushes with tailored properties. The data consistently demonstrates that a high-density "brush" conformation, achieved through controlled pre-functionalization and SI-ATRP, is the primary determinant for forming a dense hydration layer that effectively repels proteins and microorganisms. By systematically optimizing these parameters, scientists can develop highly stable and sensitive biosensing platforms capable of operating reliably in complex biological fluids.
From Lab to Device: Synthesizing and Applying Brushes on Biosensor Interfaces
The precise engineering of surfaces, particularly through the synthesis of zwitterionic polymer brushes, is fundamental to developing advanced antifouling biosensor interfaces. Among the various polymer modification techniques, Surface-Initiated Atom Transfer Radical Polymerization (SI-ATRP) and 'Grafting-Through' methods have emerged as powerful synthetic tools for creating well-defined polymer architectures on solid substrates [24] [25]. These techniques enable researchers to tailor the physicochemical properties of surfaces, making them indispensable for constructing biosensor platforms that resist non-specific protein adsorption and maintain optimal performance in complex biological environments [26] [27]. The selection of an appropriate grafting methodology directly influences critical brush characteristics such as grafting density, chain conformation, and film thickness, which collectively determine the ultimate antifouling efficacy of the modified surface [24].
Polymer grafting encompasses three principal synthetic approaches: "grafting-to," where pre-synthesized polymer chains with reactive end-groups are covalently attached to a surface; "grafting-from," where polymer chains grow directly from initiator-functionalized surfaces; and "grafting-through," which involves the copolymerization of surface-attached macromonomers with free monomers in solution [24] [28]. Each method presents distinct advantages and limitations concerning grafting density, synthetic complexity, and structural control. For high-performance zwitterionic polymer brushes in antifouling biosensor applications, SI-ATRP (a "grafting-from" technique) and "grafting-through" methods offer complementary pathways to optimize brush properties for specific operational requirements [28].
Comparative Analysis: SI-ATRP vs. Grafting-Through Methods
The strategic selection between SI-ATRP and grafting-through methodologies requires a thorough understanding of their respective capabilities and limitations. The following table provides a systematic comparison of these techniques across critical parameters relevant to zwitterionic polymer brush synthesis for antifouling biosensors:
Table 1: Comparative analysis of SI-ATRP and Grafting-Through methods
| Parameter | SI-ATRP | Grafting-Through |
|---|---|---|
| Fundamental Approach | Polymer chains grow directly from surface-initiated sites via controlled radical polymerization [28] | Surface-attached macromonomers copolymerized with free monomers in solution [24] [28] |
| Typical Grafting Density | High grafting density achievable (0.067-0.122 chains/nm² reported for BaTiO₃ nanoparticles) [29] | Generally yields lower grafting density compared to grafting-from approaches [28] |
| Molecular Weight Control | Excellent control over molecular weight and dispersity (e.g., PDI of 1.259-1.263 achievable) [29] | Moderate control, influenced by macromonomer reactivity and ratio [24] |
| Structural Complexity | Enables precise architecture control (block copolymers, gradients) [30] [28] | Suitable for creating heterogeneous surface structures [24] |
| Catalyst Requirements | Requires transition metal catalysts (copper complexes); advanced methods use ppm levels [31] [29] | Varies based on polymerization mechanism; may not require metal catalysts |
| Application Advantages | Ideal for high-density zwitterionic brushes optimizing antifouling performance [27] | Useful for creating mosaic grafting surfaces with multiple functionalities [24] |
This comparative analysis reveals that SI-ATRP generally provides superior control over brush density and molecular parameters, which are critical factors for optimizing the antifouling performance of zwitterionic polymer brushes [27]. The grafting-through method, while typically yielding lower grafting densities, offers advantages for creating specialized surface architectures with heterogeneous compositions that may be beneficial for multifunctional biosensor platforms [24].
Surface-Initiated Atom Transfer Radical Polymerization (SI-ATRP)
Fundamental Principles and Reaction Mechanism
SI-ATRP is an advanced controlled radical polymerization technique that extends the conventional ATRP process to surface-bound initiators [30]. The method operates through a reversible redox mechanism mediated by a transition metal catalyst, typically copper complexes with nitrogen-based ligands [28]. This catalytic system establishes a dynamic equilibrium between active radical species and dormant alkyl halides, ensuring controlled polymer chain growth from surface-bound initiators while minimizing termination reactions [30] [28]. The SI-ATRP process begins with the reduction of Cu⁺I to Cu⁺I by the surface-initiated alkyl halide, generating radicals that propagate through monomer addition. The growing polymer chains are subsequently deactivated back to the dormant state through oxidation by Cu⁺II, maintaining the critical balance between active and dormant species necessary for controlled polymerization [28].
The robustness of SI-ATRP stems from its ability to precisely control key brush parameters including molecular weight, dispersity, composition, and architecture [28]. This precision is particularly valuable for zwitterionic polymer brushes in biosensor applications, where optimal antifouling performance requires careful tuning of brush density and thickness [27]. Through systematic manipulation of reaction parameters including catalyst concentration, monomer-to-initiator ratio, and reaction time, researchers can fine-tune the structural characteristics of the resulting polymer brushes to meet specific biosensor requirements [30].
Advanced SI-ATRP Techniques and Protocol
Recent advancements in SI-ATRP methodology have addressed several limitations of conventional approaches, particularly regarding catalyst removal and oxygen sensitivity [31]. Techniques such as Activator Regenerated by Electron Transfer (ARGET) ATRP have significantly improved the practical implementation of SI-ATRP by dramatically reducing required catalyst concentrations to parts-per-million (ppm) levels while tolerating limited amounts of oxygen [31] [29]. In ARGET ATRP, reducing agents including tin(II) 2-ethylhexanoate (Sn(EH)₂), ascorbic acid, or glucose continuously regenerate the active Cu(I) catalyst from the Cu(II) deactivator species formed during termination events [31]. This approach not only simplifies purification procedures but also enhances the environmental compatibility of the process [31].
Table 2: Key research reagents for SI-ATRP of zwitterionic polymer brushes
| Reagent Category | Specific Examples | Function in Polymerization |
|---|---|---|
| Catalyst Complex | CuBr/PMDETA, CuBr₂/TPMA, CuBr₂/Me₆TREN [31] [32] | Mediates reversible activation/deactivation cycle |
| Reducing Agents (for ARGET) | Sn(EH)₂, ascorbic acid, glucose [31] | Regenerates Cu(I) activator from Cu(II) deactivator |
| Zwitterionic Monomers | Sulfobetaine methacrylate, carboxybetaine acrylamide [27] | Forms hydrated antifouling polymer layer |
| Surface Initiators | BiB, BIBB, BPTS [30] | Covalently anchored to substrate to initiate growth |
| Solvents | Water, DMF, ethanol [31] [32] | Reaction medium; affects catalyst activity |
Comprehensive SI-ATRP Protocol for Zwitterionic Polymer Brushes:
Substrate Preparation and Initiator Immobilization: Begin with thorough cleaning of the substrate (e.g., gold sensor chips, silica nanoparticles) using appropriate methods (oxygen plasma, piranha solution, or UV-ozone treatment). Functionalize the cleaned surface with ATRP initiators, typically through silanization (for oxide surfaces) or thiol-gold self-assembled monolayer formation (for noble metal surfaces) [30] [28]. Common initiators include bromoisobutyryl bromide (BiBB) or (3-(2-bromoisobutyryl)oxypropyl)triethoxysilane [30]. Verify successful initiator attachment through surface characterization techniques such as X-ray photoelectron spectroscopy (XPS) or Fourier-transform infrared spectroscopy (FTIR) [29].
Reaction Mixture Preparation: In a Schlenk flask, combine the zwitterionic monomer (e.g., sulfobetaine methacrylate, 1.0 M final concentration) with an appropriate solvent (water or aqueous/organic mixtures). Add the ligand (PMDETA or Me₆TREN, 0.135 equiv relative to initiator) and the sacrificial initiator (ethyl α-bromoisobutyrate, 1.0 equiv relative to surface initiators) to improve control over the polymerization [31] [29]. Degas the mixture through freeze-pump-thaw cycles (3 cycles) or nitrogen sparging (30 minutes) to remove oxygen.
Catalyst Introduction and Polymerization Initiation: In a separate vessel, prepare the catalyst complex (CuBr₂, 0.03-0.10 equiv relative to initiator) under inert atmosphere [31] [29]. Transfer the degassed monomer solution to the catalyst under nitrogen flow, resulting in a color change indicating complex formation. Quickly introduce the initiator-functionalized substrates to the reaction mixture. Seal the system and maintain at the desired temperature (typically 25-70°C, depending on monomer reactivity) with constant agitation [31].
Polymerization and Monitoring: Allow the reaction to proceed for a predetermined time (2-24 hours) based on the target brush thickness. For precise kinetic control, periodically withdraw small aliquots to monitor monomer conversion via ¹H NMR or gravimetric analysis, and molecular weight characteristics of free polymer (from sacrificial initiator) via gel permeation chromatography (GPC) [31].
Termination and Purification: Terminate the polymerization by exposing the reaction to air and diluting with solvent. Carefully remove the functionalized substrates and rinse extensively with appropriate solvents (water, ethanol) to remove physisorbed monomers, catalyst residues, and untethered polymer chains [31] [29]. For rigorous purification, implement additional washing steps with chelating agents (e.g., EDTA solution) to ensure complete copper removal, which is critical for biomedical applications [31].
Diagram 1: SI-ATRP experimental workflow for zwitterionic polymer brush synthesis
Grafting-Through Polymerization Method
Fundamental Principles and Reaction Mechanism
The grafting-through approach, also referred to as the "macromonomer method," involves the copolymerization of surface-anchored monomeric species (macromonomers) with free monomers in solution [24] [28]. In this technique, the substrate is first functionalized with polymerizable groups that subsequently participate in copolymerization reactions with complementary monomers from the solution phase [28]. This method differs fundamentally from SI-ATRP as the surface-bound species act as macromonomers rather than initiation sites, resulting in the incorporation of grafted chains through copolymerization events [24]. The grafting density in this approach is primarily determined by the surface density of macromonomers and their relative reactivity compared to the free monomers in solution [28].
A significant advantage of the grafting-through method is its applicability for creating specialized surface architectures such as mosaic grafts, where different monomers are patterned adjacent to each other on the surface [24] [25]. This capability is particularly valuable for multifunctional biosensor platforms that may require spatially defined regions with distinct physicochemical properties [24]. Additionally, the grafting-through technique can be implemented using various polymerization mechanisms beyond ATRP, including free radical polymerization, ring-opening metathesis polymerization (ROMP), and other conventional methods [24].
Experimental Protocol for Grafting-Through Method
Comprehensive Grafting-Through Protocol for Zwitterionic Polymer Brushes:
Substrate Functionalization with Polymerizable Groups: Clean the substrate thoroughly (e.g., sensor chips, nanoparticles) and functionalize with polymerizable groups. For silicon or gold surfaces, this typically involves silanization with compounds like 3-(trimethoxysilyl)propyl methacrylate or formation of thiol-terminated self-assembled monolayers with vinyl or acrylate termini [24]. Confirm functional group density through water contact angle measurements, XPS, or FTIR spectroscopy.
Macromonomer Synthesis (Optional): For systems requiring specific brush characteristics, pre-synthesize zwitterionic macromonomers with polymerizable end-groups via controlled polymerization techniques (e.g., ATRP, RAFT) followed by end-group modification [24]. Purify and characterize the macromonomers using GPC and NMR spectroscopy to ensure structural fidelity.
Copolymerization Reaction Preparation: In a reaction vessel, combine the functionalized substrates with the zwitterionic monomer (e.g., sulfobetaine methacrylate) and optional free macromonomer in an appropriate solvent. Add the initiator (e.g., AIBN for conventional free radical polymerization) at typical concentrations of 1-5 mol% relative to total monomers [24]. For controlled architectures, utilize ATRP or RAFT agents in the solution phase.
Polymerization Execution: Degas the reaction mixture through nitrogen sparging or freeze-pump-thaw cycles to remove oxygen. Initiate polymerization by heating to the appropriate temperature (typically 60-80°C for thermal initiators) or through photoinitiation using UV light [24] [25]. Allow the reaction to proceed for 2-24 hours with constant agitation to ensure uniform exposure.
Post-Polymerization Processing: Terminate the reaction by cooling and exposing to air. Remove the grafted substrates and wash extensively with sequential solvents (water, ethanol, tetrahydrofuran) to remove physisorbed species [24]. Characterize the resulting brush layers using ellipsometry, atomic force microscopy, and contact angle goniometry.
Diagram 2: Grafting-through method workflow for surface functionalization
Application to Zwitterionic Polymer Brushes for Antifouling Biosensors
Structure-Property Relationships in Antifouling Performance
The application of SI-ATRP and grafting-through methods for fabricating zwitterionic polymer brushes has revolutionized the development of antifouling biosensor surfaces [27]. Zwitterionic polymers, containing both cationic and anionic groups along their backbone, demonstrate exceptional antifouling capabilities due to their ability to form a tight hydration layer through electrostatic interactions with water molecules [27]. This hydration layer creates a physical and energetic barrier that effectively repels protein adsorption and cell attachment, thereby maintaining biosensor sensitivity and functionality in complex biological environments [27].
Machine learning approaches have recently provided quantitative insights into the critical relationship between brush parameters and antifouling performance [27]. Analysis of zwitterionic polymer brushes reveals that grafting density exerts a more significant influence on protein adsorption than molecular weight [27]. High-density brushes create a more uniform and impenetrable barrier against foulants, highlighting the advantage of SI-ATRP for optimal antifouling applications where maximum grafting density is desirable [30] [27]. Additionally, studies have identified an optimal thickness range (typically 30-60 nm) that minimizes protein adsorption, providing specific targets for brush synthesis [27].
Advanced Considerations for Biosensor Applications
For biosensor applications operating across diverse biological environments, the responsive behavior of zwitterionic polymer brushes to ionic strength must be carefully considered [27]. These brushes exhibit an "antipolyelectrolyte effect," where they transition from a collapsed conformation at low ionic strength to an extended chain conformation at higher salt concentrations due to shielding of intrachain electrostatic interactions [27]. This unique behavior can be strategically exploited in biosensor design to create environment-responsive systems or mitigated through careful brush parameter optimization for specific operational conditions [27].
The selection between SI-ATRP and grafting-through methods for biosensor applications depends on specific performance requirements. SI-ATRP is preferable when high grafting density and predictable brush thickness are paramount, such as in implantable sensors or point-of-care diagnostic devices [30] [27]. Conversely, the grafting-through approach offers advantages for creating multiplexed sensor platforms with spatially defined regions of different functionalities or when incorporating specialized macromonomers that are difficult to polymerize directly from surfaces [24]. Both methods enable the incorporation of specific biorecognition elements within or above the antifouling brush layer, facilitating the development of highly specific biosensing platforms that maintain functionality in fouling-prone environments like blood, serum, or wastewater [27].
Within the development of modern biosensors, the creation of antifouling surfaces is a critical step to ensure analytical accuracy and sensitivity. Zwitterionic polymer brushes have emerged as a premier coating technology to prevent the non-specific adsorption of proteins and other biomolecules, thereby reducing background noise and false signals. A significant research challenge lies in effectively immobilizing molecular recognition elements, such as antibodies or enzymes, onto these non-fouling surfaces without compromising their inherent resistance to biofouling. This document outlines two principal functionalization strategies—inkjet printing and covalent immobilization—detailing their application on zwitterionic polymer brush substrates for the fabrication of robust and sensitive biosensors. These protocols are designed for researchers and scientists engaged in the development of point-of-care diagnostics and other advanced biosensing platforms.
Functionalization Strategy 1: Inkjet Printing on Hybrid Brushes
Rationale and Surface Engineering
While pristine zwitterionic polymer brushes like poly(sulfobetaine)methacrylate (PSBMA) exhibit exceptional antifouling properties, their extreme hydrophilicity makes them unsuitable for the non-covalent immobilization of proteins via inkjet printing [33]. Aqueous antibody solutions printed on such surfaces tend to bead up and spread uncontrollably, leading to poor spot morphology and inconsistent assay results. To circumvent this limitation, a strategy engineering hybrid zwitterionic-cationic polymer brush coatings has been developed [33]. These hybrid systems, such as those incorporating poly(sulfobetaine)methacrylate with cationic monomers, allow for the precise tuning of surface wettability. This tunability creates a surface that is sufficiently hydrophobic to enable high-fidelity inkjet printing and robust immobilization of capture antibodies (Abc) via mild desiccation, while still maintaining a low enough fouling background to ensure high analytical performance [33].
Detailed Protocol: Fabrication and Printing
Protocol: SI-ATRP of Hybrid Zwitterionic Brushes and Inkjet Printing of Antibodies
Step 1: Substrate Preparation and ATRP Initiator Immobilization
- Clean glass or silicon substrates (e.g., 1x1 cm chips) thoroughly.
- Immerse substrates in a 10% (v/v) solution of (3-aminopropyl)triethoxysilane (APTES) in ethanol for 4 hours [33].
- Rinse extensively with ethanol and deionized (DI) water, then centrifuge at 150 rcf for 6 minutes and cure overnight at 120°C [33].
- Incubate the APTES-functionalized substrates in a solution of 1% (v/v) α-bromoisobutyryl bromide (BiB) and 1% (v/v) triethylamine in dichloromethane for 30 minutes [33].
- Rinse consecutively with dichloromethane, ethanol, and DI water. Centrifuge and cure at 120°C for 2 hours to yield ATRP-initiator functionalized surfaces (APTES-BiB) [33].
Step 2: Surface-Initiated Atom Transfer Radical Polymerization (SI-ATRP)
- Prepare a degassed polymerization solution containing the desired molar ratio of zwitterionic monomer (e.g., sulfobetaine methacrylate, SBMA) and cationic monomer (e.g., 2-(dimethylamino)ethyl methacrylate, DMAEMA) in a solvent mixture of methanol and DI water [33]. Include catalysts Copper(I) bromide and Copper(II) bromide, and the ligand HMTETA.
- Transfer the solution to a reaction vessel under an inert atmosphere (Argon).
- Immerse the initiator-functionalized substrates (APTES-BiB) in the polymerization solution. The polymerization time will determine the brush thickness (e.g., 5-100 nm) [33].
- Terminate the reaction by removing the substrates and rinsing extensively with DI water. Spin dry and store under ambient conditions [33].
Step 3: Inkjet Printing of Capture Probes
- Prepare an ink solution containing the capture antibody (e.g., 0.1-1.0 mg/mL in a suitable buffer such as phosphate-buffered saline).
- Load the ink into a piezoelectric inkjet printer.
- Using computer-aided design (CAD) software, define the pattern for the microarray (e.g., discrete spots with 100-300 µm diameter).
- Print the antibody solution onto the hybrid polymer brush surface.
- Following printing, subject the array to a mild desiccation step (e.g., air-drying or incubation in a desiccator) to non-covalently immobilize the antibodies onto the brush [33].
Step 4: Assay Execution
- The fabricated microarray can now be used in a sandwich immunoassay format. After blocking with a protein like BSA, introduce the sample containing the analyte.
- Subsequently, introduce a fluorophore-labeled detection antibody (Abd), which can also be pre-printed on-chip for a fully integrated point-of-care test [33].
The following workflow diagram illustrates the key stages of this process:
Functionalization Strategy 2: Covalent Immobilization
Rationale and Chemical Coupling
Covalent immobilization offers a robust alternative to physical adsorption, providing enhanced stability for biosensors intended for long-term storage or use in demanding environments. This method involves the formation of stable covalent bonds between the surface and the biomolecular probe, significantly reducing leaching and maintaining activity over extended periods [34] [35]. A common and effective strategy involves activating carboxyl-functionalized surfaces—such as those on oxidized carbon nanotubes or polymer brushes—using carbodiimide chemistry. This approach crosslinks primary amines on the antibodies to surface carboxyl groups, creating an amide bond [34] [35]. The superior stability of covalent linkages is clearly demonstrated in Table 2.
Table 2: Comparative Performance of Covalent vs. Physical Adsorption Immobilization
| Immobilization Parameter | Covalent Immobilization (EDC/sulfo-NHS) | Physical Adsorption | Source |
|---|---|---|---|
| Long-term Stability (Residual Activity after 25 days) | ~40% | ~20% | [34] [35] |
| Thermostability (Response retained after 60°C incubation) | Higher | Lower | [34] [35] |
| Sensitivity (for a model lactate biosensor) | 5.8 μA/mM | 9.4 μA/mM | [34] [35] |
| Linearity (for a model lactate biosensor) | Up to 0.12 mM | Up to 0.18 mM | [34] [35] |
| Resistance to Leaching | High | Low | [34] [36] |
Detailed Protocol: EDC/sulfo-NHS Coupling
Protocol: Covalent Immobilization of Antibodies via Carbodiimide Chemistry
Step 1: Surface Activation
- If working with a surface that lacks inherent carboxyl groups (e.g., bare cellulose paper or a neutral polymer brush), first introduce them. This can be achieved via oxidation with NaIO4 (for cellulose) or by using carboxyl-bearing monomers during brush synthesis [36].
- For already carboxyl-functionalized surfaces (e.g., oxidized SWCNT or a brush with carboxylic acid moieties), activate the carboxyl groups by immersing the substrate in a solution of 1-ethyl-3-(3-dimethylaminopropyl)carbodiimide hydrochloride (EDC) and N-hydroxysulfosuccinimide (sulfo-NHS) (e.g., 2.5 mM EDC and 4.5 mM sulfo-NHS) in 0.1 M MES buffer (pH 6.5) for 15-60 minutes at room temperature [34] [35]. This step forms an active ester intermediate.
Step 2: Antibody Coupling
- Gently rinse the activated surface with MES buffer (pH 6.5) to remove excess EDC/sulfo-NHS.
- Immediately apply a solution of the capture antibody (e.g., 0.1-1.0 mg/mL in 0.1 M phosphate-buffered saline, PBS, pH 7.4) to the surface. Ensure complete coverage.
- Incubate the reaction for 2-4 hours at room temperature or overnight at 4°C to allow amide bond formation between the activated esters on the surface and primary amines (lysine residues) on the antibody.
Step 3: Quenching and Blocking
- After coupling, rinse the surface thoroughly with PBS to remove any non-covalently bound antibodies.
- Quench any remaining active esters by incubating with a blocking agent such as 1M ethanolamine pH 8.5 or a solution of 1% Bovine Serum Albumin (BSA) in PBS.
- The surface is now ready for use in assay assembly.
The chemical pathway for this coupling reaction is detailed below:
The Scientist's Toolkit: Essential Research Reagent Solutions
Successful implementation of the aforementioned protocols requires a set of key reagents and materials. The following table lists essential items, their core functions, and example applications relevant to functionalizing antifouling biosensor surfaces.
Table 1: Key Research Reagent Solutions for Surface Functionalization
| Reagent / Material | Primary Function / Purpose | Example Application in Protocols |
|---|---|---|
| APTES ((3-Aminopropyl)triethoxysilane) | Silane coupling agent; introduces reactive amine groups to oxide surfaces (glass, silicon). | Priming substrates for subsequent initiator immobilization in SI-ATRP [33]. |
| EDC & sulfo-NHS | Carbodiimide crosslinker & water-soluble stabilizer; activates carboxyl groups for amide bond formation with primary amines. | Covalent immobilization of antibodies onto carboxyl-functionalized surfaces (e.g., oxidized SWCNT, brushes) [34] [35] [36]. |
| Zwitterionic Monomers (e.g., SBMA) | Polymer brush building blocks; confer extreme resistance to non-specific protein adsorption (antifouling). | Synthesis of the non-fouling base layer for biosensors [33]. |
| SI-ATRP Initiator (e.g., BiB) | Surface-tethered initiator; kickstarts the controlled "grafting-from" polymerization. | Growing polymer brushes with controlled thickness and density from the substrate [33]. |
| Pyridyl Disulfide (PDS) Monomers | Polymer brush building block; provides reversible, thiol-reactive handles for bioconjugation via disulfide exchange. | Fabrication of "catch-and-release" brushes for selective biomolecule immobilization and on-demand release [37]. |
| Glutaraldehyde | Homobifunctional crosslinker; links amine groups on surfaces to amine groups on proteins. | Immobilizing antibodies on aminated surfaces (e.g., APTES-modified paper) for P-ELISA [36]. |
The development of ultra-low fouling biosensor surfaces is critical for accurate diagnostics and long-term in vivo monitoring. Zwitterionic polymer brushes, which exhibit exceptional antifouling properties due to their ability to bind water molecules and form a strong hydration layer, have emerged as a leading material for this purpose [1] [38]. However, their super-hydrophilicity presents a significant challenge for fabrication using solution-based printing techniques, as the high surface energy of aqueous inks often leads to poor resolution and film formation [38]. This application note details a protocol for designing hybrid zwitterionic-cationic brushes that overcome this limitation. The incorporation of cationic co-monomers modulates the solution behavior and interfacial properties of the polymer, enabling the creation of patterned, high-performance antifouling surfaces via printing technologies for biosensor applications.
Background and Rationale
Zwitterionic Polymers as Antifouling Materials
Zwitterionic polymers contain both positive and negative charges within the same repeating unit, resulting in a net neutral but highly polar structure [7]. This unique configuration confers super-hydrophilicity and promotes the formation of a tightly bound hydration layer via ionic solvation, which effectively prevents the non-specific adsorption of proteins, cells, and microorganisms [1] [39]. This makes them a superior alternative to poly(ethylene glycol) (PEG), which suffers from oxidative degradation and immunogenicity [1] [38]. Their performance is governed by the specific zwitterionic motif used, with the most common being sulfobetaine (SB), carboxybetaine (CB), and phosphorylcholine (PC) [39] [38].
The Hydrophilicity Challenge in Printing
While their extreme hydrophilicity is beneficial for antifouling, it is detrimental to printing processes. Aqueous solutions of zwitterionic polymers exhibit low contact angles and tend to spread uncontrollably on substrates, leading to a loss of printing fidelity and feature definition. Furthermore, their anti-polyelectrolyte effect—where the polymer chain expands in high ionic strength solutions—can alter ink viscosity and de-stabilize the ink formulation [7]. The hybrid approach introduces cationic monomers, which can:
- Modulate Ink Rheology: Improve ink-substrate interaction and reduce spreading.
- Introduce pH-Responsiveness: Provide a handle for post-printing structural control via changes in ionic crosslinking [7].
- Maintain Biofunctionality: Allow for the retention of antifouling properties from the zwitterionic component while adding new functionality.
Research Reagent Solutions
The following table details the essential materials for synthesizing and characterizing hybrid zwitterionic-cationic brushes.
Table 1: Key Research Reagents and Materials
| Reagent/Material | Function/Description | Key Characteristic |
|---|---|---|
| Sulfobetaine Methacrylate (SBMA) | Primary zwitterionic monomer; provides ultralow fouling background [38]. | Strong hydration via ionic solvation; anti-polyelectrolyte effect [7]. |
| 2-(Dimethylamino)ethyl methacrylate (DMAEMA) | Cationic co-monomer; modulates hydrophilicity and introduces pH-responsiveness. | Tertiary amine group becomes protonated (cationic) at neutral-to-acidic pH. |
| Carboxybetaine Acrylamide (CBAA) | Alternative zwitterionic monomer; allows for bio-conjugation [39]. | Carboxyl group with higher pKa enables covalent coupling with biomolecules. |
| Atom Transfer Radical Polymerization (ATRP) Initiator | Immobilized on substrate to initiate "graft-from" polymerization [1] [38]. | Creates dense, well-defined polymer brushes (e.g., α-bromoisobutyryl bromide). |
| Copper(I) Bromide / Ligand | Catalyst system for ATRP. | Enables controlled radical polymerization from the surface. |
| Silicon Wafer/Gold Sensor Chip | Model substrates for biosensor development. | Flat, easily functionalized surfaces for QCM-D, SPR, or ellipsometry. |
Quantitative Properties of Polymer Motifs
The selection of monomer motifs directly influences the physicochemical and functional properties of the resulting brush. The data below should guide the design of the hybrid system.
Table 2: Comparative Properties of Zwitterionic and Cationic Motifs
| Polymer Motif | Hydration Strength | Fouling Resistance | Stimuli-Responsiveness | Key Application Note |
|---|---|---|---|---|
| Poly(SBMA) | High (binds more water molecules) [39] | Excellent, stable across ion strength [38] | Conformational change with salt, pH [39] | Ideal for stable antifouling; may complicate ink formulation. |
| Poly(CBAA) | High (stronger individual water bonds) [39] | Excellent | pH-responsive (carboxyl group pKa) [39] | Allows post-printing bio-conjugation; responsive handle. |
| Poly(DMAEMA) | Moderate (hydrophilic when protonated) | Poor (can be fouling) | Strong pH-response (cationic at low pH) | Primary component for tuning ink behavior and printability. |
Experimental Protocol: Surface-Initiated Polymerization of Hybrid Brushes
This protocol describes the "graft-from" synthesis of a P(SBMA-co-DMAEMA) brush on a silicon substrate via ATRP.
Substrate Preparation and Initiator Immobilization
- Substrate Cleaning: Clean silicon wafers (2x2 cm) by sonication in acetone and ethanol for 15 minutes each. Treat with oxygen plasma for 5 minutes to generate surface hydroxyl groups.
- Silanation: Immerse the substrates in a 2% (v/v) solution of (3-aminopropyl)triethoxysilane (APTES) in anhydrous toluene for 12 hours at room temperature. Rinse thoroughly with toluene and ethanol, then dry under a stream of nitrogen.
- Initiator Attachment: Transfer the aminated substrates into a round-bottom flask containing 50 mL of anhydrous dichloromethane. Add 2 mL of triethylamine as an acid scavenger. Under a nitrogen atmosphere, add 1 mL of α-bromoisobutyryl bromide dropwise with constant stirring. React for 1 hour at 0°C and then for 2 hours at room temperature. Wash sequentially with dichloromethane, methanol, and ethanol, then dry under nitrogen. The substrate is now ready for polymerization.
Hybrid Brush Synthesis via ATRP
- Monomer Solution Preparation: In a Schlenk flask, dissolve SBMA (1.00 g, 3.58 mmol) and DMAEMA (0.20 g, 1.27 mmol) in a 1:1 (v/v) mixture of methanol and deionized water (20 mL total). This creates an 80:20 zwitterionic:cationic monomer feed ratio.
- Catalyst Addition: Degas the solution by bubbling with nitrogen for 30 minutes. Add the ATRP catalyst system: Copper(I) Bromide (25.7 mg, 0.18 mmol) and the ligand N,N,N',N'',N''-Pentamethyldiethylenetriamine (PMDETA, 37.2 µL, 0.18 mmol).
- Polymerization: Place the initiator-functionalized substrate into the Schlenk flask. Seal the flask and perform three freeze-pump-thaw cycles to ensure an oxygen-free environment. Backfill with nitrogen and place the flask in a 30°C water bath for 2-4 hours with mild agitation.
- Termination and Cleaning: Carefully remove the substrate and rinse it extensively with copious amounts of deionized water and methanol to remove any physisorbed polymer and catalyst residues. Dry the substrate under a stream of nitrogen.
The following workflow diagram illustrates the key steps of the protocol.
Characterization and Performance Evaluation
Physicochemical Characterization
- Ellipsometry: Measure dry brush thickness in air. A successful polymerization should yield a thickness of 20-50 nm after 2 hours.
- Contact Angle Goniometry: Measure static water contact angle. A successful hybrid brush should show a contact angle between 30° and 50°, indicating a balance between the super-hydrophilicity of P(SBMA) (<20°) and the more hydrophobic P(DMAEMA) (~60° in its neutral state).
- X-ray Photoelectron Spectroscopy (XPS): Confirm the presence of both zwitterionic (S 2p signal from SBMA) and cationic (N 1s signal from both monomers) elements in the surface composition.
Antifouling Performance Assessment
- Quartz Crystal Microbalance with Dissipation (QCM-D):
- Mount the coated sensor in the flow chamber and establish a stable baseline in phosphate-buffered saline (PBS).
- Flow 1 mg/mL fibrinogen solution in PBS for 30 minutes.
- Switch back to PBS buffer and monitor the frequency (ΔF) and energy dissipation (ΔD) shifts.
- Success Criterion: The hybrid brush should exhibit protein adsorption of <5 ng/cm², comparable to pure zwitterionic brushes [38].
- Surface Plasmon Resonance (SPR): Use a similar protocol with undiluted human blood plasma. The hybrid surface should resist non-specific protein adsorption with >95% efficiency compared to a bare gold surface.
Printing Application Notes
For ink formulation, dissolve the synthesized hybrid brush polymer (if using a "graft-to" approach) or the monomer mixture (for in-situ polymerization post-printing) in a water-alcohol co-solvent (e.g., 3:1 water:isopropanol). The addition of 10-30 mol% cationic DMAEMA to the zwitterionic monomer feed is expected to significantly increase the ink's contact angle on polar surfaces, thereby improving feature definition in techniques like inkjet or aerosol jet printing. Post-printing, the surface can be treated with a high ionic strength buffer to trigger the anti-polyelectrolyte effect of the zwitterionic segments, further enhancing the hydration and antifouling properties of the final patterned film [7].
The development of wearable microneedle (MN) patches represents a paradigm shift in therapeutic drug monitoring, enabling minimally invasive access to biomarkers in interstitial fluid (ISF). A significant challenge in this domain is biofouling—the nonspecific adsorption of proteins and other biomolecules onto sensor surfaces—which severely compromises analytical accuracy and long-term stability [1]. Zwitterionic polymer brushes, characterized by mixed anionic and cationic groups maintaining overall electroneutrality, have emerged as a third-generation antifouling material to address this limitation [1]. These polymers create an ultra-low fouling surface through strong electrostatic interactions with water molecules, forming a dense hydration layer that effectively prevents nonspecific protein adsorption [1] [9]. This application note details the integration of antifouling zwitterionic coatings within wearable MN patches, framing their development and implementation within a broader research thesis on advanced biosensor interfaces.
Antifouling Zwitterionic Coatings: A Primer for Biosensor Interfaces
The Role of Zwitterionic Polymers in Biofouling Prevention
Biofouling on medical devices begins with the nonspecific adsorption of proteins, which facilitates subsequent adhesion of cells and microorganisms [1]. Zwitterionic polymers combat this by creating a surface that is super-hydrophilic and electrically neutral. Each repeating unit carries a pair of opposite charges, resulting in a molecule that interacts strongly with water molecules through ionic solvation [1]. This interaction forms a tightly bound hydration layer that serves as a physical and energetic barrier against protein adsorption, outperforming traditional poly(ethylene glycol) (PEG) coatings which can suffer from structural instability and immunogenicity [1].
Coating Strategies for Sensor Functionalization
Several effective methods exist for applying zwitterionic polymer brushes to sensor surfaces:
- Electropolymerization: Enables rapid (e.g., <7 minutes), controlled deposition of zwitterionic polymers like ZiPPy (zwitterionic polypyrrole) directly onto electrode surfaces [9]. This one-step process can simultaneously incorporate affinity ligands for specific target capture.
- Surface-Initiated Polymerization: "Grafting-from" techniques, such as Atom Transfer Radical Polymerization (ATRP), grow polymer chains directly from initiator-functionalized surfaces, yielding high grafting density optimal for antifouling [1] [40].
- Cross-linking: As demonstrated in methotrexate-monitoring MN patches, biopolymers like chitosan can be cross-linked on electrode surfaces to create a barrier against biofouling while allowing analyte adsorption [41].
Table 1: Key Characteristics of Antifouling Coating Strategies
| Coating Method | Key Features | Polymer Examples | Compatibility with MNs |
|---|---|---|---|
| Electropolymerization | Rapid (<7 min), controllable, enables ligand co-immobilization | Zwitterionic polypyrrole (ZiPPy) [9] | Excellent for conductive MN sensors |
| ATRP | High grafting density, precise control over chain length | Poly(carboxybetaine), poly(sulfobetaine) [1] | Suitable for various MN materials |
| Cross-linking | Simple process, uses biocompatible natural polymers | Chitosan [41] | Demonstrated in hollow MNs |
Case Study: Closed-Loop Therapeutic Drug Monitoring for Methotrexate
A recent groundbreaking development is a wearable hollow MN array patch designed for continuous monitoring of methotrexate (MTX), a chemotherapeutic agent [41]. This system exemplifies the practical integration of antifouling materials within a functional drug monitoring platform. The MN-based voltammetric sensor incorporates a cross-linked chitosan coating that serves a dual purpose: it provides antifouling protection while simultaneously enabling the adsorption of MTX at the electrode surface for sensitive analysis [41]. The chitosan coating effectively minimizes biofouling interference in protein-enriched environments, allowing the sensor to maintain excellent analytical performance during continuous operation for more than two days [41].
Integration with Closed-Loop Therapy
This monitoring system was further integrated with an iontophoretic hollow MN array patch for on-demand drug delivery, creating a cohesive platform for closed-loop therapeutic drug management [41]. Such systems are particularly valuable for drugs like MTX that have a narrow therapeutic window and require precise dosing to maximize efficacy while minimizing toxic side effects.
Experimental Protocol: Fabrication and Validation of Antifouling MN Sensors
Fabrication of Zwitterionic Polymer-Coated Microneedle Electrodes
Materials Required:
- Hollow microneedle array (e.g., silicon, polymer)
- ZiPPy monomer solution (synthesized from pyrrole and zwitterionic precursors) [9]
- Chitosan (medium molecular weight, ≥75% deacetylated)
- Cross-linking agent (e.g., genipin or glutaraldehyde)
- Phosphate buffered saline (PBS), pH 7.4
- Three-electrode electrochemical setup (working, counter, and reference electrodes)
- Affinity ligands (e.g., antibodies, proteins) for specific analyte capture
Procedure:
- MN Array Preparation: Clean MN arrays thoroughly with ethanol and deionized water, then dry under nitrogen stream.
- Coating Solution Preparation:
- Option A (ZiPPy)_: Prepare 10 mM ZiPPy monomer solution in PBS with 0.1 M sodium perchlorate as supporting electrolyte [9].
- Option B (Chitosan)_: Prepare 2% (w/v) chitosan solution in 1% acetic acid, followed by addition of 0.5% cross-linking agent.
- Surface Coating Application:
- For ZiPPy: Use electropolymerization by applying electrical potential (e.g., cyclic voltammetry from -0.2 to 0.8 V for 10 cycles at 50 mV/s) to deposit polymer directly onto MN electrodes [9].
- For chitosan: Dip-coat MN electrodes in chitosan solution, then cure at 40°C for 2 hours to complete cross-linking [41].
- Functionalization: For targeted sensing, mix affinity ligands with coating solution prior to application for simultaneous incorporation during polymer deposition.
- Curing and Storage: Rinse coated MN arrays with deionized water and store in dry, sterile conditions until use.
Analytical Performance Validation
Objective: Validate the antifouling performance and sensing capability of zwitterionic-coated MN sensors.
Table 2: Quantitative Performance Metrics of Antifouling MN Sensors
| Parameter | ZiPPy-Coated Electrodes [9] | Chitosan-Coated MN for MTX [41] | Uncoated/Bare Electrodes |
|---|---|---|---|
| Non-specific Protein Adsorption | >70% reduction vs. bare | Significant reduction in biofouling | High (reference) |
| Linear Detection Range | N/A | 25-400 μM (covers MTX therapeutic range) | N/A |
| Stability (Continuous Operation) | >90% signal retention after 48h | >2 days continuous operation | Significant signal drift |
| Electrochemical Impedance | Lower than bare/PPy electrodes | Functional in protein-rich artificial ISF | High/non-specific |
| Hydration (Contact Angle) | ~30° (highly hydrophilic) | N/A | ~70° (moderately hydrophobic) |
Validation Procedure:
- Antifouling Assessment:
- Incubate coated MN sensors in 10% fetal bovine serum or artificial ISF for 2 hours at 37°C.
- Measure non-specific protein adsorption using fluorescence microscopy (with FITC-labeled albumin) or electrochemical impedance spectroscopy (EIS).
- Compare results with uncoated MN sensors as control.
Analytical Performance:
- Test sensors in standard solutions with known analyte concentrations.
- For MTX monitoring: Validate across therapeutic range (25-400 μM) using voltammetric techniques [41].
- Calculate limit of detection (LOD), sensitivity, and linearity.
Stability Testing:
- Continuously monitor sensor response in protein-enriched artificial ISF over 48+ hours.
- Assess signal drift and fouling-induced performance degradation.
The Scientist's Toolkit: Essential Research Reagents and Materials
Table 3: Key Research Reagent Solutions for Antifouling MN Development
| Reagent/Material | Function/Application | Example Specifications |
|---|---|---|
| Zwitterionic Monomers | Foundation for antifouling polymer brushes | Carboxybetaine acrylamide, sulfobetaine methacrylate [1] |
| Chitosan | Natural biopolymer for cross-linked antifouling coatings | Medium molecular weight, ≥75% deacetylated [41] |
| Poly(ethylene glycol) diacrylate | Cross-linker for hydrogel-forming MNs | Mn 700 Da [42] |
| Hyaluronic Acid | Biocompatible matrix for dissolvable MNs | Pharmaceutical grade, suitable for microneedle fabrication [43] |
| ATRP Initiators | Surface-initiated polymerization for dense brushes | 2-Bromo-2-methylpropionate-based initiators [40] |
| Pyrrole Monomer | Base for conductive polymer composites | ≥98% purity, electropolymerization grade [9] |
Future Directions and Research Opportunities
The convergence of zwitterionic antifouling strategies with MN technology opens several promising research avenues guided by the Three I Principles (Integrated-Intelligent-Individualized) [44]:
- Integrated Systems: Development of fully closed-loop systems that combine real-time biomarker monitoring with automated drug delivery in a single wearable device [41] [43].
- Intelligent Interfaces: Implementation of AI-driven adaptive dosing algorithms that respond to dynamic physiological conditions and patient-specific pharmacokinetics [44].
- Individualized Therapeutics: Creation of modular MN arrays that can be customized for specific drug regimens and patient populations, particularly for pediatric and geriatric applications [43].
Further innovation should focus on creating battery-free, self-powered systems that harvest energy from patient movements [43], enhancing patient compliance through extended wear times and eliminating maintenance requirements. Additionally, the exploration of stimuli-responsive zwitterionic polymers that modulate their antifouling properties in response to specific biomarkers could enable next-generation diagnostic-therapeutic combinations.
Wearable microneedle patches represent a transformative platform for therapeutic drug monitoring, with zwitterionic polymer brushes serving as a critical component to ensure measurement accuracy through superior antifouling properties. The protocols and applications detailed in this document provide a roadmap for researchers developing closed-loop drug delivery systems that maintain functionality in complex biological environments. As this field advances, the integration of intelligent materials with patient-centered design promises to revolutionize personalized medicine through non-invasive, continuous therapeutic monitoring.
Therapeutic drug monitoring (TDM) of vancomycin is a critical clinical practice due to the antibiotic's narrow therapeutic window. Subtherapeutic concentrations can lead to treatment failure and antimicrobial resistance, while supratherapeutic levels pose significant risks of nephrotoxicity and ototoxicity [45] [46]. Traditional detection methods, including chromatography and immunoassays, are often time-consuming, expensive, and lack the portability needed for rapid, point-of-care testing [45] [47]. Electrochemical aptamer-based (E-AB) sensors present a promising alternative, offering the potential for rapid, sensitive, and specific detection [47].
A paramount challenge for any biosensor intended for use with complex biological samples like blood serum or milk is biofouling—the non-specific adsorption of proteins, cells, and other biomolecules onto the sensor surface. This fouling layer can severely compromise sensor performance by attenuating the signal, increasing background noise, and reducing selectivity [1] [48]. Within the context of a broader thesis on advanced antifouling materials, this case study explores the integration of zwitterionic polymer brushes as a surface modification strategy to enhance the robustness and reliability of E-AB sensors for vancomycin detection. These polymers, characterized by their mixed cationic and anionic groups on a single monomer unit, create a super-hydrophilic surface that binds water molecules tightly via electrostatic interactions, forming a physical and energetic barrier against non-specific adsorption [1] [49].
Sensor Fabrication and Antifouling Integration
The fabrication of a fouling-resistant E-AB sensor for vancomycin involves a multi-step process focused on constructing a stable, selective, and non-fouling interface on an electrode surface. The following workflow outlines the key stages, from the initial surface functionalization to the final sensor ready for measurement.
Materials and Reagent Solutions
The following table details the key reagents and materials required for the fabrication of the antifouling E-AB sensor.
Table 1: Essential Research Reagents for Sensor Fabrication
| Reagent/Material | Function/Brief Explanation |
|---|---|
| Thiolated Vancomycin Aptamer [47] | Biorecognition element; binds vancomycin with high specificity and affinity, inducing a measurable conformational change. |
| Gold Nanostructures (AuNSs) [47] | Nanomaterial coating; increases electrode active surface area, enhances electron transfer, and provides sites for aptamer immobilization. |
| 6-Mercapto-1-hexanol (MCH) [47] | Surface blocking agent; creates a well-oriented aptamer monolayer and displaces non-specifically adsorbed strands. |
| Zwitterionic Monomers (e.g., PSBMA, PMPC) [1] [49] [8] | Antifouling polymer precursor; forms a hydrophilic, charge-balanced brush that resists non-specific protein adsorption via a strong hydration layer. |
| Tris(2-carboxyethyl)phosphine (TCEP) [47] | Reducing agent; cleaves disulfide bonds in thiolated aptamers to ensure efficient attachment to the gold surface. |
| Ferro/Ferricyanide Redox Probe [47] | Electrochemical reporter; its electron transfer efficiency, measured via EIS, changes upon target-induced aptamer conformational switch. |
| Copper-based Catalyst (e.g., CuBr/Bpy) [49] | Catalyst for SI-ATRP; enables controlled, surface-initiated growth of zwitterionic polymer brushes from the sensor surface. |
Detailed Experimental Protocol
Protocol 1: Fabrication of AuNS-Modified Electrode
- Step 1: Electrode Pretreatment. Clean the bare gold or carbon-based working electrode. For gold electrodes, perform electrochemical cycling in a 0.5 M H₂SO₄ solution. For lab-printed carbon electrodes (C-PEs), polish and rinse according to manufacturer guidelines [47].
- Step 2: AuNSs Electrodeposition. Prepare an aqueous solution of 0.5 mM HAuCl₄ in 0.1 M H₂SO₄. Using a standard three-electrode system, immerse the pre-treated working electrode into the solution. Perform chronoamperometry by applying a constant potential of -0.4 V (vs. Ag/AgCl) for a duration of 60-120 seconds. This process results in the formation of cauliflower-shaped gold nanostructures on the electrode surface, which should be confirmed by characterization techniques like SEM or AFM [47].
- Step 3: Rinsing. After electrodeposition, thoroughly rinse the modified electrode with deionized water to remove any unbound reagents and dry it under a gentle stream of nitrogen.
Protocol 2: Aptamer Immobilization and Zwitterionic Coating
- Step 1: Aptamer Preparation. Dilute the thiolated vancomycin-specific aptamer to a concentration of 1-5 µM in an appropriate buffer (e.g., Tris-HCl, pH 7.4). To activate the thiol groups, incubate the aptamer solution with 1 mM TCEP for a minimum of 1 hour [47].
- Step 2: Aptamer Immobilization. Drop-cast the activated aptamer solution onto the AuNS-modified electrode and incubate in a humidified chamber for 12-16 hours at room temperature. This allows for the formation of stable Au-S bonds, covalently anchoring the aptamer to the surface.
- Step 3: Surface Blocking with MCH. After immobilization, rinse the electrode to remove loosely bound aptamers. Then, incubate it in a 1 mM solution of 6-mercapto-1-hexanol (MCH) for 30-60 minutes. This step passivates any remaining exposed gold sites, creating a more organized and efficient aptamer monolayer [47].
- Step 4: Grafting Zwitterionic Polymer Brush.
- Surface Initiation: First, immobilize an ATRP initiator (e.g., α-bromoisobutyryl bromide) onto the sensor surface.
- Polymerization: Prepare an aqueous solution containing the zwitterionic monomer, such as sulfobetaine methacrylate (SBMA) or 2-methacryloyloxyethyl phosphorylcholine (MPC), along with the Cu(I) catalyst and ligand (e.g., CuBr/Bpy). Deoxygenate the solution by purging with nitrogen. Immerse the initiator-functionalized sensor into this solution to allow surface-initiated atom transfer radical polymerization (SI-ATRP) to proceed for several hours. This grows a dense, antifouling polymer brush from the surface [1] [49].
- Final Rinsing: After polymerization, rinse the sensor extensively with deionized water and store it in PBS buffer at 4°C until use.
Performance and Data Analysis
The performance of the fabricated E-AB sensor is evaluated using electrochemical impedance spectroscopy (EIS), which monitors the electron transfer resistance of the ferro/ferricyanide redox probe. Binding of vancomycin to its aptamer induces a conformational change, altering the interfacial properties at the electrode surface and increasing the measured electron transfer resistance (Rₑₜ) [47].
Table 2: Quantitative Performance Comparison of Vancomycin Sensing Platforms
| Sensor Platform | Detection Method | Linear Range | Limit of Detection (LOD) | Antifouling Strategy | Reference |
|---|---|---|---|---|---|
| Aptamer/AuNSs on C-PE (This Work) | EIS | 50 – 1000 nM | 1.72 nM | Zwitterionic Polymer Brush | [47] |
| Graphene/Glassy Carbon | SWV | Up to 50 µM | 0.2 µM | High surface wettability | [45] [46] |
| PAA-Cu-MOF/Glassy Carbon | DPV | Not Specified | 1 nM | Not Specified | [45] [46] |
| Gold Nanostructure-Graphene | DPV | Not Specified | Not Specified | Not Specified | [46] |
The mechanism of the zwitterionic polymer brush, such as poly(SBMA) or poly(MPC), involves forming a dense hydration layer via electrostatic interactions with water molecules. This layer creates a physical and energetic barrier that effectively repels the non-specific adsorption of proteins and other biomolecules, thereby preserving the sensor's sensitivity and specificity in complex media [1] [8]. The following diagram illustrates the antifouling mechanism at the molecular level.
This case study demonstrates a robust protocol for developing an electrochemical aptasensor for vancomycin, integrating zwitterionic polymer brushes as a highly effective antifouling interface. The combination of a specific aptamer bioreceptor with a nanostructured electrode and a biomimetic antifouling coating results in a sensor with high sensitivity and suitability for analysis in complex biological fluids like serum and milk [47].
Future research directions should focus on translating this platform into practical clinical and environmental monitoring tools. Key areas for development include the creation of fully integrated, flexible, and wearable form factors for non-invasive vancomycin monitoring in sweat, leveraging the correlation between sweat and serum concentrations [47]. Furthermore, the long-term stability and shelf-life of these sensors, particularly the durability of the zwitterionic coating under continuous flow or repeated use, require thorough investigation. Finally, testing the sensor with a wide range of real, unprocessed clinical samples from diverse patient populations will be essential to validate its performance and reliability for point-of-care therapeutic drug monitoring.
Optimizing Performance and Overcoming Environmental Challenges
The Impact of pH and Salt Concentration on Brush Stability and Neutrality
Zwitterionic polymer brushes have emerged as a leading platform for creating antifouling biosensor surfaces, owing to their ability to form a strong hydration barrier that resists non-specific protein adsorption. A critical factor determining their performance is the maintenance of brush stability and charge neutrality under varying environmental conditions. This Application Note details how pH and salt concentration directly impact these crucial parameters, providing researchers with structured data, validated protocols, and design principles to optimize brush performance for biosensing and drug development applications.
Key Data Summaries
Impact of pH on the Conformation and Stability of Zwitterionic Brushes
Table 1: pH-Dependent Conformational Changes and Stability of Various Zwitterionic Brushes
| Polymer Brush | Low pH Behavior | Neutral pH Behavior | High pH Behavior | Key Stability Findings |
|---|---|---|---|---|
| PCysMA (Poly(cysteine methacrylate)) | Extended (Cationic) [50] | Zwitterionic, Anti-fouling [50] | Extended (Anionic) [50] | Good long-term stability at physiological pH; complex degradation in alkaline solution [50]. |
| pCBMAA (Poly(carboxybetaine methacrylamide)) | Positively Charged (COOH protonated) [51] | Zwitterionic (Net Neutral) [51] | Zwitterionic (Net Neutral), negative Zeta Potential observed [51] | Excellent long-term antifouling stability; charge state tunable via copolymerization [51]. |
| pSBMA (Poly(sulfobetaine methacrylate)) | N/A | Zwitterionic (Net Neutral) [51] | Zwitterionic (Net Neutral), negative Zeta Potential observed [51] | High-density grafting enhances stability; 8x stability increase in pH 12 solutions [52] [53]. |
| pSBMAA (Poly(sulfobetaine methacrylamide)) | N/A | Zwitterionic (Net Neutral) [51] | Zwitterionic (Net Neutral) [51] | Exhibits minimal electrostatic interactions; resistant to fouling across pH range [51]. |
Impact of Salt Concentration and Ion Valency on Brush Performance
Table 2: Effects of Salt Concentration and Ion Type on Brush Properties and Fouling
| Parameter | Impact on Brush Conformation | Impact on Antifouling Performance | Key Research Findings |
|---|---|---|---|
| Increasing Monovalent Salt (e.g., Na⁺) | Reduced electrostatic repulsion within brush, leading to more collapsed conformation [54]. | Can reduce protein adhesion via competitive adsorption with brush functional groups [8]. | Dynamic response magnitude of brush thickness to electric fields is strongly reduced at high salt concentrations [54]. |
| Divalent Cations (e.g., Ca²⁺) | Can cause significant brush collapse due to strong electrostatic bridging [8]. | Enhances protein adhesion via Ca²⁺ bridging between brush and proteins, compromising antifouling [8]. | Accumulation of cations in salt solution can cause a positive shift in the surface potential of PMPC and PSBMA brushes [8]. |
| High Salt Concentration (General) | Induces rich electro-responsive behavior; an opposite electric field from ion distribution resists chain deformation [54]. | Varies by brush type; can influence non-specific interactions in complex media. | The "fall time" for brush conformational change is shorter for stiff brushes than flexible ones under electric fields in salty environments [54]. |
Experimental Protocols
Grafting Zwitterionic Brushes via SI-ARGET-AgTRP
This protocol describes the grafting of poly(sulfobetaine methacrylate) (pSBMA) brushes onto a ceramic substrate, optimized for high density and stability [52] [53].
Step 1: Surface Initiation Preparation
- Clean the substrate (e.g., alumina ceramic membrane) thoroughly with organic solvents and plasma treatment.
- Immerse the substrate in a solution of α-bromoisobutyryl bromide (BiBB) and triethylamine in tetrahydrofuran to immobilize the ATRP initiator.
- React for a controlled duration (e.g., 3.0 hours) to achieve a high density of initiator sites, which is critical for forming a dense brush.
- Rinse extensively with tetrahydrofuran and deionized water to remove physisorbed species.
Step 2: Surface-Initiated Polymerization
- Prepare the polymerization solution containing the zwitterionic monomer (e.g., Sulfobetaine methacrylate - SBMA), the catalyst (e.g., Cu(II)Br₂ with 2,2'-Bipyridyl), and the reducing agent (e.g., L-Ascorbic acid - L-AA) in a water/methanol mixture.
- Deoxygenate the solution by bubbling with nitrogen or argon for 20-30 minutes.
- Transfer the solution to the reaction vessel containing the initiator-functionalized substrate under an inert atmosphere.
- Allow the polymerization to proceed at room temperature for a predetermined time (e.g., 8 hours) to control brush thickness.
- Terminate the reaction by exposing the substrate to air. Rinse the modified substrate vigorously with deionized water and ethanol to remove any unreacted monomer and catalyst.
Assessing Brush Stability and Neutrality
This protocol outlines methods to characterize the pH stability and charge state of the grafted zwitterionic brushes.
Accelerated Stability Testing in Acidic/Alkaline Conditions
- Procedure: Immerse the brush-grafted substrates in solutions of pH 2 and pH 12 at room temperature or elevated temperatures for set periods. Rinse and dry the samples.
- Measurement: Use ellipsometry to measure the dry thickness of the brushes before and after exposure. A stable brush will show minimal thickness loss.
- Data Interpretation: Calculate the stability increase factor by comparing the degradation rate of optimized high-density brushes to control samples. Studies have shown stability increases of 15x at pH 2 and 8x at pH 12 for high-density pSBMA brushes [52].
Characterizing Surface Charge and Isoelectric Point (pI)
- Zeta Potential Measurement: Use a surface zeta potential analyzer to measure the potential of the brush-coated surface across a wide pH range (e.g., pH 3 to 10).
- Determining pI: Identify the pH at which the zeta potential is zero; this is the isoelectric point where the brush is neutrally charged.
- Data Interpretation: Note that a negative zeta potential at pH values above the pI is commonly observed for polybetaines (e.g., pCBMAA, pSBMA) and does not necessarily indicate a net anionic charge that promotes fouling [51].
Evaluating Antifouling Performance
- Protein Adsorption Test: Immerse the brush substrate in a solution of a model protein (e.g., Bovine Serum Albumin, BSA) at a specific concentration (e.g., 1 mg/mL) for 1-2 hours.
- Quantification: Use a quartz crystal microbalance with dissipation (QCM-D) or surface plasmon resonance (SPR) to quantify the mass of adsorbed protein in real-time. Alternatively, label the protein with a fluorescent dye and use fluorescence microscopy for quantification.
- Performance Metric: High-performing brushes should resist over 98% of protein adsorption compared to an unmodified surface [52].
The Scientist's Toolkit
Table 3: Essential Reagents and Materials for Zwitterionic Brush Research
| Reagent/Material | Function in Research | Example Application |
|---|---|---|
| Sulfobetaine Methacrylate (SBMA) | Zwitterionic monomer for creating ultra-low fouling surfaces. | Grafting of pSBMA brushes for biosensor surfaces and marine anti-fouling coatings [52] [55]. |
| Carboxybetaine Methacrylamide (CBMAA) | Zwitterionic monomer with a carboxyl group usable for biorecognition element immobilization. | Functionalizable antifouling platforms for biosensing applications [51]. |
| α-Bromoisobutyryl Bromide (BiBB) | ATRP initiator used to functionalize surfaces for "grafting-from" polymerization. | Immobilization of initiator sites on aminated surfaces (e.g., APTES-silica) [52] [50]. |
| Cu(II)Br₂ / Bipyridyl / L-Ascorbic Acid | Catalyst system (Cu source, ligand, reducing agent) for ARGET-ATRP. | Enables controlled radical polymerization with low catalyst concentration, suitable for grafting from surfaces [52] [53]. |
| Poly(carboxybetaine) Active Esters | Functional groups for covalent immobilization of biorecognition elements (BREs) like antibodies. | Converting non-fouling brush surfaces into specific biosensing interfaces [51]. |
Workflow and Mechanism Diagrams
Brush Fabrication and pH Response Workflow
Salt Ion Interaction Mechanism
Discussion and Design Principles
The data and mechanisms presented lead to several key design principles for fabricating stable, neutral zwitterionic brushes for biosensors:
- Prioritize High Grafting Density: A high-density brush structure is paramount, not only for superior antifouling via a dense hydration layer but also for dramatically enhancing the stability of the anchoring chemistry against hydrolytic degradation, especially under extreme pH conditions encountered during cleaning cycles [52].
- Select Brushes Based on Operational Environment:
- For biosensing in physiological buffers, PCysMA and pSBMAA offer excellent stability and neutrality at pH 7.4 [51] [50].
- If post-functionalization is required, pCBMAA is the material of choice, as its carboxyl groups can be used to covalently attach biorecognition elements while maintaining low fouling after careful optimization of charge balance [51].
- In environments with high divalent cation concentration (e.g., Ca²⁺), pSBMA brushes may be preferred, though all brushes are susceptible to bridging-fouling, indicating a need for environmental control or further material innovation [8].
- Interpret Zeta Potential Cautiously: A negative zeta potential measurement at physiological pH does not necessarily indicate a failure of brush neutrality or predict poor antifouling performance. This apparent charge may not translate into functional, fouling-inducing electrostatic interactions [51].
- Understand Salt-Specific Effects: Ionic strength and valency are critical design parameters. Monovalent ions can be beneficial, while divalent cations like Ca²⁺ pose a significant fouling risk through ionic bridging mechanisms that must be mitigated for robust biosensor operation in complex fluids [54] [8].
Surface preconditioning is a critical, often determinative step in the fabrication of robust antifouling surfaces for biomedical and biosensing applications. For zwitterionic polymer brushes, which are a cornerstone of modern antifouling strategies, the substrate's initial chemical and physical properties directly govern the density, stability, and ultimate performance of the grafted layer [1] [8]. Proper preconditioning ensures the formation of a uniform, high-coverage brush layer that can effectively resist the nonspecific adsorption of proteins, cells, and other biomolecules—a paramount requirement for the reliability and longevity of biosensor surfaces [1]. This protocol details surface preconditioning techniques tailored to enhance the antifouling performance of zwitterionic polymer brushes, framed within a research context aimed at minimizing biofouling on sensitive analytical devices.
The exceptional antifouling capacity of zwitterionic polymers stems from their ability to form a dense hydration layer via electrostatic interactions with water molecules [1]. This layer creates a physical and energetic barrier that prevents foulants from adhering. Realizing this theoretical performance in practice, however, requires a meticulously prepared surface that presents sufficient and appropriate active sites for polymer initiation or attachment [8].
Preconditioning Principles and Mechanisms
The primary objective of surface preconditioning is to engineer a substrate interface that facilitates the optimal grafting of zwitterionic polymers. The mechanism of biofouling begins with the instantaneous, non-specific adsorption of proteins onto a material's surface, which subsequently facilitates the adhesion of cells and microorganisms [1] [56]. Zwitterionic polymers, such as poly(sulfobetaine methacrylate) (PSBMA), poly(carboxybetaine methacrylate) (PCBMA), and poly(2-methacryloyloxyethyl phosphorylcholine) (PMPC), combat this by presenting a super-hydrophilic and electrically neutral surface that mimics the outer cell membrane [1].
The preconditioning process directly addresses the interfacial interactions that are the most critical factor in biofilm formation [1]. By creating a uniform layer of initiators or functional groups, preconditioning enables the growth of a dense brush conformation. This conformation is essential for forming the complete hydration layer that confers ultra-low fouling properties [8]. The diagram below outlines the logical pathway from surface preconditioning to enhanced antifouling performance.
Materials and Reagents
Table 1: Essential Research Reagent Solutions for Surface Preconditioning and Antifouling Evaluation
| Reagent/Material | Function/Application | Key Characteristics & Considerations |
|---|---|---|
| Zwitterionic Monomers (e.g., SBMA, CBMA) [1] [8] | Building blocks for polymer brush synthesis. | Ultra-low protein adsorption; forms a dense hydration layer via ionic solvation. |
| ATRP Initiator (e.g., Bromoalkylsilane for silica, Bromoalkanethiol for gold) [1] | Provides active sites for controlled surface-initiated polymerization. | High purity is critical for uniform initiator monolayer formation. |
| Piranha Solution (3:1 v/v Conc. H₂SO₄ : H₂O₂) | Caution: Highly corrosive and reactive. For rigorous cleaning of silica and glass substrates. | Removes organic residues; creates hydroxyl groups for subsequent silanization. |
| Oxygen Plasma Cleaner | Surface cleaning and activation for various substrates (polymers, metals, metal oxides). | Increases surface energy and creates reactive oxygen species for better initiator binding. |
| Buffers (e.g., Phosphate Buffered Saline (PBS), HEPES) [8] | Medium for antifouling performance tests (e.g., protein adsorption). | Ionic strength and composition (Ca²⁺ vs. Na⁺) can significantly affect fouling behavior [8]. |
| Model Foulants (e.g., Bovine Serum Albumin (BSA), Fibrinogen) [1] [57] | Quantitative evaluation of antifouling performance. | BSA is a standard model protein; Fibrinogen is relevant for blood-contacting devices. |
Preconditioning Protocols for Common Biosensor Substrates
The following protocols are designed for standard 1 cm x 1 cm substrate chips. All procedures should be performed in a clean, controlled environment, using appropriate personal protective equipment.
Protocol A: Preconditioning of Gold Surfaces
Gold is a prevalent substrate for surface plasmon resonance (SPR)-based biosensors. This protocol outlines the steps to form a self-assembled monolayer (SAM) of an ATRP initiator.
Step 1: Substrate Cleaning
- Immerse gold substrates in fresh Piranha solution for 30 minutes. (Note: Exercise extreme caution.)
- Rinse thoroughly with copious amounts of Millipore water (>18 MΩ·cm) and absolute ethanol.
- Dry under a stream of ultrapure nitrogen gas.
Step 2: Initiator Immobilization
- Prepare a 1 mM solution of an ATRP initiator (e.g., 2-bromo-2-methyl-N-(3-(trimethoxysilyl)propyl) propanamide for silica; 11-(2-bromo-2-methylpropanoyloxy) undecyl-1-thiol for gold) in anhydrous toluene.
- Immerse the cleaned substrates in the initiator solution under an inert atmosphere (e.g., N₂ or Ar) for 12-24 hours.
- Remove the substrates and sonicate sequentially in toluene, ethanol, and methanol for 2 minutes each to remove physisorbed initiator.
- Dry under a stream of nitrogen and store in a desiccator until use.
Table 2: Troubleshooting Guide for Gold Surface Preconditioning
| Problem | Potential Cause | Solution |
|---|---|---|
| Incomplete or patchy initiator coverage | Contaminated gold surface; moisture in toluene. | Ensure rigorous cleaning; use anhydrous, inhibitor-free solvents and store/store under inert gas. |
| Poor polymerization efficiency | Degraded initiator; inactive terminal bromine groups. | Source fresh initiator; confirm initiator synthesis protocol and storage conditions (-20°C, dark). |
| High non-specific adsorption on final surface | Low brush density or presence of defects. | Optimize initiator concentration and reaction time; characterize initiator layer with ellipsometry and XPS. |
General Protocol C: Surface Activation via Plasma Treatment
Plasma treatment is a versatile method for cleaning and functionalizing a wide range of substrates, including polymers and metal oxides.
- Step 1: Plasma Activation
- Place substrates in the plasma chamber.
- Evacuate the chamber to a base pressure below 10⁻² mbar.
- Introduce oxygen gas at a flow rate of 10-50 sccm to maintain a working pressure of 0.1-0.5 mbar.
- Apply an RF power (e.g., 50-100 W) for 1-5 minutes. Optimization Note: Excessive power/time can cause surface damage.
- Vent the chamber and proceed to initiator immobilization immediately (within 15 minutes) to prevent surface reorganization and deactivation.
The experimental workflow below integrates these preconditioning protocols with subsequent polymerization and evaluation stages.
Performance Evaluation and Data Analysis
After grafting zwitterionic polymer brushes onto the preconditioned surfaces, their antifouling performance must be quantitatively evaluated.
Method 1: Protein Adsorption Assay
- Procedure: Incubate the modified substrate in a solution of a model protein (e.g., 1 mg/mL BSA in PBS) for 1 hour at 37°C.
- Rinsing: Gently rinse the substrate with PBS to remove loosely attached proteins.
- Quantification: Use spectroscopic (e.g., Micro-BCA assay) or surface-sensitive techniques (e.g., Quartz Crystal Microbalance with Dissipation (QCM-D) or Surface Plasmon Resonance (SPR)) to quantify the amount of adsorbed protein.
- Analysis: Compare the adsorbed mass on the zwitterionic surface against a control surface (e.g., unmodified substrate). High-performing zwitterionic surfaces can achieve over 95% reduction in protein adsorption compared to controls [1].
Method 2: Flux Recovery Ratio (FRR) in Filtration This method is particularly relevant for membrane-based biosensors.
- Procedure: Measure the pure water flux (Jw1) of the modified membrane. Perform filtration with a foulant solution (e.g., 1000 ppm BSA or sodium alginate solution) for a set time. Clean the membrane and measure the recovered water flux (Jw2).
- Calculation: Calculate FRR = (Jw2 / Jw1) × 100%.
- Analysis: A high FRR indicates excellent antifouling and self-cleaning properties. State-of-the-art zwitterionic-modified membranes can achieve FRR values exceeding 95% [58] [57].
Table 3: Quantitative Antifouling Performance Metrics from Literature
| Material System | Test Method | Key Performance Metric | Reported Value |
|---|---|---|---|
| EDA-TPA CONs composite membrane [57] | BSA Filtration | Flux Recovery Ratio (FRR) | ~96% |
| LSMM-modified PVDF membrane [58] | Oily Wastewater Filtration | Flux Recovery Ratio (FRR) | 100% (over 5 cycles) |
| LSMM-modified PVDF membrane [58] | Pure Water Permeability | Water Flux Increase | 58% increase (880 L m⁻² h⁻¹) |
| Zwitterionic Polymer Brushes [1] [8] | Protein Adsorption | Reduction in Adsorption | >95% (vs. bare substrate) |
It is crucial to note that the ionic environment can significantly impact performance. Recent atomistic insights reveal that divalent cations like Ca²⁺ can enhance protein adhesion to certain zwitterionic brushes (e.g., PSBMA and PCBMA) via cation bridging, whereas monovalent ions like Na⁺ can reduce fouling through competitive adsorption [8]. This must be considered when designing evaluation experiments for biosensors that will operate in complex physiological fluids.
Effective surface preconditioning is the foundational step in fabricating high-performance, antifouling zwitterionic biosensor surfaces. The protocols detailed herein for gold, silica, and polymer substrates are designed to maximize the density and stability of the subsequent polymer brush layer. Key to success is the rigorous removal of contaminants and the uniform presentation of initiator molecules.
For researchers in drug development and biosensing, adopting these preconditioning techniques can significantly enhance the reliability of analytical results by minimizing background noise from non-specific binding. When integrating these protocols into a thesis project, it is recommended to consistently employ surface characterization tools (e.g., XPS, AFM, Ellipsometry) to correlate preconditioning parameters with initiator density and, ultimately, antifouling performance. Future work should focus on optimizing preconditioning for increasingly complex and miniaturized biosensor geometries and for operation in high-ionic-strength biological environments where fouling mechanisms are more complex [8].
The performance of biosensors, particularly in complex media such as whole blood, is fundamentally governed by the surface properties of the sensing interface. A critical challenge lies in balancing two competing requirements: achieving effective immobilization of biological recognition elements (e.g., aptamers, antibodies) while simultaneously conferring robust resistance to nonspecific fouling from proteins, cells, and other biomolecules. Zwitterionic polymer brushes have emerged as a leading platform to address this challenge, creating a surface that is both highly resistant to fouling and readily functionalizable. Their antifouling performance stems from their ability to form a dense hydration layer via electrostatic interactions with water molecules, creating a physical and energetic barrier that prevents the adsorption of foulants [1]. This application note details the protocols and underlying principles for fabricating and characterizing zwitterionic polymer brush-based biosensor surfaces with tuned wettability to optimize both probe immobilization and fouling resistance, specifically within the context of developing highly sensitive and reliable biomedical diagnostics.
Table 1: Key Performance Metrics of Zwitterionic Polymer Brush Surfaces from Literature
| Material/Coating | Lowest Friction Coefficient | Protein/Cell Adhesion Reduction | Coating Stability | Primary Application |
|---|---|---|---|---|
| pSBMA Brushes (WPU/PEGDA) | < 0.005 [59] | Excellent anti-protein/cell adhesion and thrombosis prevention [59] | Coating loss < 7.5 μg/cm² after 7 h sonication [59] | Medical device coatings |
| Zwitterionic Nanogel (NIPMAM-co-SBMA) | Not Specified | Efficient reduction of bacterial adhesion [60] | Superior in vitro stability vs. charged nanogels [60] | Implant surface coatings |
| DMAPS/pDA Anti-fouling Layer | Not Specified | Hinders nonspecific adsorption in whole blood [61] | Robust adhesion via polydopamine primer [61] | Electrochemical aptasensors |
The Scientist's Toolkit: Research Reagent Solutions
The following table outlines essential materials required for the fabrication of zwitterionic antifouling biosensor interfaces.
Table 2: Essential Reagents and Materials for Zwitterionic Biosensor Fabrication
| Item Name | Function/Description | Key Characteristics |
|---|---|---|
| Sulfobetaine Methacrylate (SBMA) | Zwitterionic monomer for polymer brush synthesis [59] | Contains both quaternary ammonium and sulfonate groups; forms strong hydration layer. |
| Dopamine Hydrochloride | Forms a polydopamine (pDA) adhesive primer layer [61] | Enables strong substrate-independent adhesion and provides a platform for secondary functionalization. |
| 3-(Dimethyl(methacryloyloxyethyl)ammonium) Propane Sulfonate (DMAPS) | Zwitterionic molecule for anti-fouling layer creation [61] | Can be anchored onto pDA layers to create a biomimetic, fouling-resistant biointerface. |
| Atom Transfer Radical Polymerization (ATRP) Initiator | Initiates surface-controlled polymer brush growth [62] | Typically a silane-based molecule (e.g., 3-(2-bromoisobutyramido)propyl(trimethoxy)silane) for surface immobilization. |
| Tris(2-(dimethylamino)ethyl)amine (Me6TREN) | Ligand in SI-ATRP catalyst system [62] | Forms a complex with copper catalysts, controlling the radical polymerization process. |
Core Principles and Signaling Pathways
The antifouling mechanism of zwitterionic surfaces is not a traditional biochemical signaling pathway but rather a physico-chemical process centered on the formation of a surface hydration layer. The following diagram visualizes this core principle and the subsequent strategy for biosensor development.
Diagram 1: Biosensor Surface Engineering Workflow. This illustrates the key stages in creating a functional biosensor interface, from surface priming with polydopamine to the final probe immobilization on the antifouling zwitterionic layer.
Experimental Protocols
Protocol: Fabrication of Robust Zwitterionic Polymer Brushes via Subsurface-Initiated Growth
This protocol, adapted from recent research, describes a rapid method for grafting sulfobetaine methacrylate (SBMA) polymer brushes using a porous hydrogel layer to enhance grafting efficiency and stability [59].
- Key Materials: Waterborne polyurethane/poly(ethylene glycol) diacrylate (WPU/PEGDA) hydrogel, Sulfobetaine methacrylate (SBMA) monomer, Photoinitiator.
- Equipment: UV light source, Plasma cleaner, Sonication bath, Spin coater, Contact angle goniometer.
Step-by-Step Procedure:
- Substrate Preparation: Clean the substrate (e.g., silicon wafer, gold sensor chip, ITO electrode) sequentially with acetone, ethanol, and deionized water in a sonicator for 15 minutes each. Dry under a stream of nitrogen.
- Hydrogel Layer Deposition: Prepare a WPU/PEGDA mixture with a photoinitiator. Deposit a thin layer of this mixture onto the clean substrate using spin coating. Cure the layer under UV light to form a cross-linked porous hydrogel.
- Surface-Initiated Polymerization: Prepare an aqueous solution of the SBMA monomer. Apply this solution to the hydrogel-modified surface. The porous structure of the hydrogel acts as a subsurface initiator, facilitating the rapid growth of pSBMA brushes. Allow the reaction to proceed for 40 minutes at room temperature.
- Post-processing and Validation: Rinse the modified surface thoroughly with copious amounts of deionized water to remove any unreacted monomer. Dry the surface with nitrogen. Validate coating stability by subjecting it to sonication in water for 1 hour and measuring mass loss (target: < 7.5 μg/cm² after 7 hours) [59].
Protocol: Constructing an Anti-fouling Aptasensor Biointerface for Whole Blood Detection
This protocol details the creation of a biomimetic biointerface on an indium tin oxide (ITO) electrode, integrating antifouling properties with specific biorecognition for direct detection in whole blood [61].
- Key Materials: Dopamine hydrochloride, 3-Dimethyl(methacryloyloxyethyl) ammonium propane sulfonate (DMAPS), CA72-4 specific aptamer, Tris-HCl buffer (10 mM, pH 8.5).
- Equipment: Electrochemical workstation, X-ray Photoelectron Spectrometer (XPS), OCA video-based optical contact-angle measuring system.
Step-by-Step Procedure:
- ITO Electrode Pretreatment: Sonicate the ITO electrode in acetone, ethanol, and ultrapure water for 30 minutes each. Dry under a nitrogen stream.
- Polydopamine Priming: Immerse the clean ITO electrode in 10 mL of Tris-HCl buffer (pH 8.5) containing 2 mg/mL of dopamine hydrochloride. Allow the oxidative self-polymerization of dopamine to proceed at room temperature for several hours to form a robust polydopamine (pDA) adhesive layer. Rinse with ultrapure water to remove unbounded PDA or dopamine.
- Zwitterionic Anti-fouling Layer Construction: Anchor the zwitterionic molecule DMAPS and the aptamer onto the pDA/ITO electrode surface via Michael addition reaction. Incubate the pDA-coated electrode in an aqueous solution of DMAPS and the aptamer for a defined period.
- Surface Characterization: Characterize the chemical modification using XPS, confirming the presence of sulfur (S2p peak) from DMAPS. Validate the antifouling performance by measuring the non-specific adsorption of proteins or cells from whole blood.
Data Interpretation & Performance Validation
Rigorous characterization is essential to confirm the successful fabrication and performance of the modified biosensor surfaces. The following diagram outlines the logical relationship between characterization methods and the key properties they validate.
Diagram 2: Surface Characterization Logic Map. This shows the connection between experimental techniques and the specific surface properties they are used to investigate.
Table 3: Key Validation Experiments and Expected Outcomes
| Validation Method | Experimental Details | Expected Outcome for a Successful Coating |
|---|---|---|
| X-ray Photoelectron Spectroscopy (XPS) | Analyze surface elemental composition. | Detection of sulfur (S2p) and nitrogen (N1s) peaks, confirming the presence of zwitterionic groups [61]. |
| Contact Angle Measurement | Measure static water contact angle with a 5 μL droplet. | Low contact angle (< 20°), indicating super-hydrophilicity and strong surface hydration [62] [1]. |
| Friction Test | Macroscopic friction test under lubrication. | Ultralow friction coefficient (< 0.005), indicating excellent lubricity and anti-adhesion [59]. |
| Antifouling Test | Expose surface to protein solutions (e.g., fibrinogen) or whole blood. | Ultra-low protein adsorption (< 5 ng/cm²) and minimal cell adhesion, demonstrating fouling resistance [59] [61] [1]. |
| Sensor Performance | Measure detection of target analyte (e.g., CA72-4) in buffer and whole blood. | High sensitivity and limit of detection (e.g., LOD of 7.11 U/mL for CA72-4 in blood), with reliable results in clinical samples [61]. |
The strategic tuning of surface wettability through the application of zwitterionic polymer brushes provides a powerful and robust solution to the critical challenge of balancing probe immobilization with fouling resistance in biosensors. The protocols outlined herein, centered on scalable and stable fabrication methods like subsurface-initiated polymerization and polydopamine-assisted surface chemistry, enable the creation of biointerfaces capable of operating directly in complex biological fluids such as whole blood. By leveraging the unique ability of zwitterionic materials to form a strong hydration layer, researchers can develop biosensors with enhanced sensitivity, reliability, and suitability for point-of-care clinical diagnostics, thereby advancing the translation of biosensing technologies from the laboratory to the clinic.
Ensuring Mechanical Stability and Long-Term Durability on Wearable Sensors
For wearable sensors to transition from laboratory proof-of-concept to reliable clinical and consumer devices, ensuring their mechanical stability and long-term durability is paramount. This challenge is accentuated when these sensors are integrated with advanced antifouling surfaces, such as zwitterionic polymer brushes, which are a key focus of contemporary biosensor research. Zwitterionic polymers, characterized by mixed anionic and cationic groups that confer overall electroneutrality, have emerged as a third-generation antifouling material due to their ultra-low protein adsorption and excellent biocompatibility [1]. Their super-hydrophilicity enables them to form a dense hydration layer through ionic solvation, effectively resisting non-specific protein adhesion—the first step in biological contamination [1]. However, the mechanical integrity of both the sensor substrate and this antifouling coating must be maintained under dynamic physiological conditions, including repeated stretching, bending, and moisture exposure, to ensure consistent sensor performance and user compliance over extended monitoring periods. This document provides detailed application notes and standardized protocols to address these critical challenges.
Key Challenges in Mechanical Durability
The mechanical failure of wearable sensors typically occurs due to a mismatch between the engineered device properties and the dynamic biological environment of human skin. Key challenges include:
- Skin-Device Mechanical Mismatch: Human skin is an anisotropic, viscoelastic structure with a Young's modulus ranging from 5 kPa to 140 MPa and can undergo deformations up to 30% on skin surfaces and over 100% on joints [63]. This creates significant stress on rigid or poorly matched sensor materials.
- Dynamic Physiological Environment: The skin undergoes constant mechanical stress from daily activities (e.g., over 20,000 breaths per day) and biological processes, such as epidermal turnover every 7-14 days and variable sweat production (14-54 glands per cm² depending on location) [63].
- Adhesive Failure: Pressure-sensitive adhesives (PSAs), commonly acrylic-based for long-term wear, must balance interfacial adhesion and cohesive strength. Over time, hydrogen and van der Waals bonds increase adhesive tack, but material selection critically impacts durability [63].
- Coating Delamination: Zwitterionic polymer brushes, while excellent for antifouling, require stable covalent grafting to the substrate to maintain integrity under mechanical stress and hydration.
Quantitative Material Characterization and Testing Protocols
Mechanical Property Assessment
A multi-faceted approach is required to characterize the mechanical properties of sensor substrates and coatings.
Protocol 1: Atomic Force Microscopy (AFM) for Soft Material Characterization
This protocol is adapted for characterizing the local mechanical properties of soft hydrogels and polymer coatings, relevant to zwitterionic surfaces [64].
- Sample Preparation: Synthesize poly(N-isopropylacrylamide) (PNIPAM) hydrogels in different methanol-water mixtures to create a calibration set with Young's modulus ranging from 100 Pa to 10 kPa. For zwitterionic polymer brushes, graft them onto representative substrate samples (e.g., SIS, TPU).
- AFM Setup: Use a commercial AFM (e.g., Bruker Dimension Icon) with both sharp pyramidal probes (MLCT, cone semi-angle 17°) for high spatial resolution and spherical colloidal probes (radius ~2550 nm) for superior performance on soft samples. Calibrate the elastic constant of each cantilever using the thermal tune method.
- Measurement Parameters: Immerse samples in deionized water at 23°C. Set ramp length between 5-10 μm, maximum force between 5-30 nN (depending on sample softness), and indentation velocity at ~20 μm/s.
- Data Acquisition: Perform Force Volume (FV) measurements with a 64x64 resolution at 10-12 different macroscopic positions on each sample to ensure statistical reliability.
- Data Analysis:
- Preprocess raw data to obtain force (F) vs. indentation (δ) curves. Determine the contact point using a histogram method to distinguish the non-contact Gaussian distribution.
- Fit the indentation curves using the Hertz model for spherical probes:
F(δ) = (4/3) * (E/(1-ν²)) * √R * δ^(3/2) - For sharp pyramidal probes, use the Sneddon model (with Bilodeau approximation):
F(δ) = (E/(1-ν²)) * tan(α) * δ²where E is the Young's modulus, ν is the Poisson ratio (0.5 for gels), R is the probe radius, and α is the half-opening angle.
Table 1: Key Parameters for AFM Characterization of Soft Materials
| Parameter | Spherical Probes | Sharp Pyramidal Probes |
|---|---|---|
| Typical Young's Modulus Range | 100 Pa - 10 kPa [64] | 1 MPa and above [64] |
| Spatial Resolution | Mesoscopic (lower) | Nanometric (higher) |
| Advantages | Reduced local pressure, well-defined geometry, validity of linear elastic models | High local pressure for significant indentation on stiffer samples, high resolution |
| Recommended Use | Homogeneous soft materials, zwitterionic hydrogels | Heterogeneous samples, thin or stiff coatings |
Protocol 2: In-Situ Evaluation of Sensor-Skin Interface Durability
This protocol assesses the long-term performance of the complete sensor device on human subjects [63].
- Subject Selection and On-Body Location: Recruit a cohort of volunteers (e.g., n=30) with high activity levels. Select test sites based on the intended application (e.g., lower torso, forearm, joints).
- Material Application: Apply at least five different candidate non-woven medical tapes or sensor patches to the selected sites. Ensure the materials vary in key properties like elasticity, Moisture Vapor Transmission Rate (MVTR), and adhesive thickness.
- Study Duration and Monitoring: Conduct tests over a minimum 7-day period, aiming for eventual 14-21 day continuous wear [63].
- Data Collection:
- User Perception: Administer Likert-scale questionnaires at regular intervals (e.g., daily) to quantify comfort, irritation, and overall perception.
- Skin Interaction: Capture high-resolution images of the material-skin interface daily to document irritation, maceration, or contact dermatitis.
- Sensor Functionality: For active sensor patches, simultaneously record signal quality (e.g., signal-to-noise ratio, motion artifacts) to correlate mechanical integrity with performance.
Critical Material Properties and Performance Data
The table below summarizes quantitative data and key properties identified from recent studies as critical for long-term durability.
Table 2: Critical Material Properties for Long-Term Wearable Sensor Durability
| Material Property | Target Value / Optimal Characteristic | Impact on Durability & Comfort |
|---|---|---|
| Substrate Elastic Modulus | Tunable to match skin (5 kPa - 140 MPa) [63] | Reduces mechanical mismatch, minimizes stress at the interface, prevents delamination. |
| Moisture Vapor Transmission Rate (MVTR) | Sufficiently high, but very high MVTR may not improve perception [63] | Prevents maceration, maintains skin health; however, the relationship with user perception is non-linear. |
| Adhesive Type | Acrylic-based Pressure-Sensitive Adhesives (PSAs) with tunable viscoelasticity [63] | Provides a balance of adhesive strength (for bonding) and cohesive strength (for clean removal and movement). |
| Substrate Stretchability | Capable of withstanding 30% strain on skin, >100% on joints [63] | Maintains electrical and mechanical integrity during body movement. |
| Hydration Layer Stability | Dense hydration via ionic solvation (Zwitterionic) [1] | Provides superior antifouling and stability compared to PEG, which undergoes depolymerization. |
Integration of Zwitterionic Antifouling Coatings
The application of zwitterionic polymer brushes to sensor surfaces must be performed in a way that does not compromise mechanical flexibility and adhesion.
Protocol 3: Grafting Zwitterionic Polymer Brushes for Mechanical Stability
- Substrate Activation: Clean the sensor substrate (e.g., SIS, TPU, or gold electrode) thoroughly. Activate the surface using oxygen plasma treatment (e.g., Diener FEMTO system) to generate reactive hydroxyl or peroxide groups.
- Surface-Initiated Polymerization: Employ Atom Transfer Radical Polymerization (ATRP) or Reversible Addition-Fragmentation Chain Transfer (RAFT) polymerization for high polymer coverage and stable coatings [1].
- ATRP Method: Immerse the activated substrate in a degassed solution containing zwitterionic monomer (e.g., sulfobetaine methacrylate), ATRP initiator, catalyst (e.g., CuBr), and ligand. Allow polymerization to proceed for 2-24 hours at controlled temperature (e.g., 30-60°C) to control brush thickness.
- Post-Processing and Validation: Rinse the grafted substrate thoroughly with deionized water to remove physisorbed polymer. Characterize the coating using AFM (Protocol 1) to confirm uniform thickness and mechanical properties. Validate antifouling performance via protein adsorption assays (e.g., using fluorescence-labeled fibrinogen).
The Scientist's Toolkit: Research Reagent Solutions
Table 3: Essential Materials for Fabricating Durable and Antifouling Wearable Sensors
| Reagent / Material | Function / Application | Specific Examples & Notes |
|---|---|---|
| Polystyrene-block-polyisoprene-block-polystyrene (SIS) | A soft, stretchable thermoplastic elastomer used as the primary substrate for conformable sensors [65]. | Styrene 22 wt% (Sigma-Aldrich CAS: 25038-32-8). Can be dissolved in toluene [65]. |
| Silver Nanowires (Ag NWs) | Form conductive, stretchable electrodes within the polymer matrix [65]. | Synthesized from PVP, EG, and AgNO₃; can be spray-coated and annealed [65]. |
| Thermoplastic Polyurethane (TPU) | A flexible and scalable substrate for screen-printed flexible electronics [63]. | Offers a good balance of flexibility, durability, and biocompatibility. |
| Zwitterionic Monomers | The building blocks for creating ultra-low fouling polymer brush coatings [1]. | e.g., Sulfobetaine methacrylate (SBMA) or carboxybetaine methacrylate (CBMA). |
| ATRP Initiator | Covalently binds to the substrate and initiates the growth of polymer brushes [1]. | e.g., 2-Bromoisobutyryl bromide (BiBB). Requires surface pre-activation. |
| Pressure-Sensitive Adhesives (PSAs) | Provide secure attachment to the skin for longer-term wear [63]. | Acrylic-based PSAs are preferred for their tunable viscoelasticity and water permeability. |
Experimental Workflow and Logical Relationships
The following diagram outlines the comprehensive workflow for developing and validating mechanically stable wearable sensors, integrating both material synthesis and testing phases.
Sensor Development Workflow
This workflow illustrates the iterative process of fabricating sensor components, applying antifouling coatings, and rigorously testing mechanical and biological performance to achieve a successful prototype.
Concluding Remarks
Achieving mechanical stability and long-term durability in wearable sensors is a multifaceted challenge that requires careful consideration of material properties, skin-device interactions, and the integration of functional antifouling coatings like zwitterionic polymer brushes. By adhering to the standardized protocols for mechanical characterization (AFM), in-situ evaluation, and robust coating application outlined in this document, researchers can systematically develop sensor devices that maintain performance and user comfort over extended periods. The future of wearable sensors in clinical applications depends on such rigorous, user-centered design and validation processes.
The application of antifouling coatings, particularly zwitterionic polymer brushes, is a critical step in developing reliable electrochemical biosensors for use in complex biological fluids. While these coatings effectively mitigate biofouling—the non-specific adsorption of proteins, cells, and other biomolecules—they can simultaneously introduce significant signal interference and diminish electrochemical sensitivity. This challenge arises from the fundamental conflict between a coating's inherent electrical insulation and the sensor's requirement for efficient electron transfer. This Application Note provides a detailed framework of protocols and analytical methods for designing and characterizing zwitterionic polymer brush coatings that achieve superior antifouling without compromising the critical signal-to-noise ratio of electrochemical biosensors.
Experimental Protocols
Fabrication of Zwitterionic Polymer Brushes via Surface Wettability Modulation
This protocol describes a rapid and robust method for grafting zwitterionic poly(sulfobetaine methacrylate) (PSBMA) brushes onto medical-grade polymer substrates (e.g., thermoplastic polyurethane, TPU), leveraging a subsurface-initiated approach to enhance grafting speed and stability [66].
Materials:
- Substrates: TPU, PVC, or POM.
- Photoinitiator: Benzophenone (BP).
- Hydrogel Precursors: Waterborne polyurethane (WPU) and Poly(ethylene glycol) diacrylate (PEGDA).
- Zwitterionic Monomer: Sulfobetaine methacrylate (SBMA).
- Solvents: Anhydrous ethanol.
Procedure:
- Substrate Pre-treatment and Initiator Embedding:
- Clean substrate surfaces successively using acetone and ethanol in an ultrasonic bath.
- Immerse the substrate in an anhydrous ethanol solution of Benzophenone (BP) to allow the hydrophobic photoinitiator to permeate the substrate interior.
- WPU/PEGDA Hydrogel Layer Formation:
- Immerse the initiator-embedded substrate in a mixed aqueous solution of WPU and PEGDA.
- Expose the system to UV irradiation. This initiates the formation of a loosely packed, interpenetrating WPU/PEGDA hydrogel network within the substrate, significantly enhancing surface wettability.
- Surface-Initiated Grafting of PSBMA Brushes:
- Immerse the substrate with the grown WPU/PEGDA layer into an aqueous solution containing the SBMA monomer.
- Perform a second UV irradiation step. The enhanced wettability facilitates the rapid diffusion of hydrophilic SBMA monomers to the initiation sites, leading to the uniform growth of PSBMA polymer brushes.
Validation: This method drastically reduces the polymer brush grafting time from several hours to 40 minutes. The coating demonstrates exceptional stability, with a mass loss of < 7.5 µg/cm² after 7 hours of sonication [66].
The following workflow illustrates this fabrication process:
Nozzle-Printing of Thick Porous Nanocomposite Coatings
For applications requiring enhanced durability and mass transport, a micrometer-thick, porous nanocomposite coating can be applied via nozzle printing. This method allows for precise localization on the working electrode [67].
Materials:
- Emulsion Components: Bovine Serum Albumin (BSA), Gold Nanowires (AuNWs), hexadecane (oil phase), phosphate buffer saline (PBS, aqueous phase).
- Cross-linker: Glutaraldehyde (GA).
Procedure:
- Oil-in-Water Emulsion Preparation:
- Create an emulsion by ultrasonicating a mixture of hexadecane and a PBS solution containing BSA and AuNWs.
- Optimize sonication time (e.g., 25 minutes) to achieve a stable emulsion with a narrow oil droplet size distribution (~325 nm) and a high zeta potential (~ -75.5 mV), which prevents phase separation.
- Nozzle Printing and Cross-linking:
- Add glutaraldehyde to the emulsion immediately before printing to initiate cross-linking.
- Use a nozzle printer to deposit the emulsion specifically onto the working electrode(s) of a multiplexed sensor.
- Heat the printed electrode to evaporate the oil and complete the cross-linking of the BSA matrix, resulting in a ~1 µm thick coating with interconnected pores and integrated conductive AuNWs.
Advantages: This localized printing prevents compromising the reference and counter electrodes. The porous structure facilitates analyte diffusion, addressing the mass transport barrier common to thicker coatings, and maintains direct electron transfer via the AuNWs [67].
Optimizing Electrochemical Impedance Spectroscopy (EIS) Measurements in Interference-Prone Environments
Accurate EIS assessment of high-impedance coated surfaces in the field is vulnerable to ambient electromagnetic interference, which causes stochastic data scatter. The following protocol ensures data accuracy [68].
Procedure:
- Equipment Setup: Use a modern, high-end EIS instrument capable of accurately measuring impedances up to ~10 GΩ·cm².
- Signal Amplitude Selection:
- Avoid low signal amplitudes (10-50 mV), as these are highly susceptible to interference and instrument imprecision, boosting data spread to ~5%.
- Apply a higher signal amplitude ≥150 mV. Amplitudes between 190 mV and 250 mV mitigate interference effects to <1% and achieve an accuracy of the impedance logarithm of 97.8%.
- Validation: Systematically investigate the effects of AC interference using dummy cells and calibration foils that mimic intact industrial coatings before field deployment.
Table 1: Impact of EIS Signal Amplitude on Measurement Accuracy and Precision under AC Interference [68]
| EIS Signal Amplitude | Interference Amplitude | Estimated Data Spread | Key Observation |
|---|---|---|---|
| Low (10 - 50 mV) | ≤ 250 mV | ~5% | Significant stochastic scatter in phase angle; limited by instrument imprecision. |
| High (≥ 150 mV) | ≤ 250 mV | <1% | Robust interference suppression; accurate and precise measurements. |
| Very High (≥ 190 mV) | ≤ 250 mV | <1% | Accuracy of impedance logarithm reaches 97.8%. |
The Scientist's Toolkit: Essential Research Reagents
Table 2: Key Reagents for Fabricating and Testing Zwitterionic Antifouling Coatings
| Reagent/Solution | Function/Description | Application Context |
|---|---|---|
| Sulfobetaine Methacrylate (SBMA) | Zwitterionic monomer conferring ultra-low fouling via a strong hydration layer. | Grafting of polymer brushes like PSBMA for antifouling surfaces [66]. |
| Benzophenone (BP) | Hydrophobic photoinitiator that permeates polymer substrates for subsurface initiation. | Enables UV-induced grafting from within substrates like TPU [66]. |
| Waterborne Polyurethane (WPU) / PEGDA Blend | Forms an interpenetrating hydrogel layer to modulate surface wettability. | Serves as an intermediate layer to facilitate rapid grafting of zwitterionic brushes [66]. |
| BSA-AuNW Emulsion | Forms a conductive, porous, and antifouling nanocomposite matrix. | Nozzle-printing of thick (~1 µm), localized coatings on working electrodes [67]. |
| Zwitterionic EK Peptide (e.g., EKEKEKEKEKGGC) | Short, sequence-defined peptide for surface passivation. | Covalent immobilization on porous substrates like PSi for broad-spectrum antifouling [69]. |
| Si-MEG-OH (Monoethylene glycol silane) | An ultrathin silane polymer layer forming a covalent antifouling network. | Creates a hydrophilic barrier on hydroxylated surfaces (e.g., gold modified with β-mercaptoethanol) [70]. |
Analytical and Validation Methods
Assessing Antifouling Performance
- Protein Adsorption Test: Immerse coated substrates in complex biofluids (e.g., undiluted serum, gastrointestinal fluid, bacterial lysate) for a defined period. Quantify non-specific adsorption using optical methods (e.g., fluorescence microscopy for labeled proteins) or quartz crystal microbalance with dissipation (QCM-D) monitoring. Effective zwitterionic coatings can reduce fouling from undiluted serum by ≥75% and up to 90% [69] [70].
- Cell Adhesion Assay: Incubate coated surfaces with model bacterial (e.g., E. coli, S. aureus) or mammalian cells. Assess adhesion via microscopy and cell counting. High-performing coatings show a marked reduction in adhered cells compared to unmodified surfaces [69].
Quantifying Electrochemical Sensitivity
The ultimate validation of a coated biosensor is its analytical performance in detecting a target analyte within a fouling environment.
- Lactoferrin Detection in GI Fluid: An aptasensor passivated with the zwitterionic peptide EKEKEKEKEKGGC demonstrated a more than one order of magnitude improvement in both the limit of detection (LOD) and signal-to-noise ratio compared to PEG-passivated sensors [69].
- ErbB2 Detection in Human Serum: A biosensor with an antifouling peptide interface assembled via robust Pt-S interactions successfully discriminated between serum from breast cancer patients and healthy individuals, showcasing high sensitivity and specificity in a complex medium [71].
Understanding the Ionic Response Mechanism
The antifouling performance of zwitterionic brushes can be modulated by the ionic environment. Atomistic insights reveal:
- Salt Solution Behavior: The surfaces of PSBMA and PMPC brushes in salt solution can exhibit a significant accumulation of cations, leading to a positive shift in surface potential [8].
- Protein Adhesion Mechanism:
- Divalent Cations (e.g., Ca²⁺): Enhance protein adhesion by forming strong "Ca²⁺ bridges" between the brush and proteins.
- Monovalent Cations (e.g., Na⁺): Can diminish protein adhesion by competitively adsorbing to the brush, reducing the number of direct salt bridges with proteins [8].
This understanding is critical for designing sensors intended for specific physiological fluids with known ion compositions. The diagram below summarizes this ionic response mechanism.
Proof in Performance: Validating Zwitterionic Brushes Against PEG and in Complex Media
The performance of biosensors is critically dependent on the interface between the synthetic material and the complex biological environment. Non-specific adsorption of proteins, cells, and other biomolecules—a process known as fouling—onto biosensor surfaces diminishes signal-to-noise ratios, reduces specificity, and compromises long-term stability [72]. For researchers and drug development professionals, this fouling presents a significant barrier to obtaining reliable, reproducible data in assays, diagnostics, and in vivo monitoring applications.
For years, poly(ethylene glycol) (PEG)-based coatings have been the gold standard for mitigating biofouling. Their effectiveness is attributed to a combination of factors including steric hindrance, hydrophilicity, and the formation of a hydration layer that creates a physical and thermodynamic barrier to biomolecular adsorption [73]. However, the search for more robust and performative surfaces has led to the emergence of zwitterionic polymer brushes as a powerful alternative. These brushes, characterized by their dense structure and balanced charge, create a super-hydrophilic surface that binds water molecules even more strongly than PEG, presenting a potentially insurmountable energy barrier for adsorbing proteins [74] [72].
This Application Note provides a direct, quantitative comparison of these two coating strategies. It details experimental protocols for their fabrication and evaluation, equipping scientists with the necessary tools to make an informed selection for their specific biosensor applications, framed within the broader research context of developing advanced antifouling surfaces.
Quantitative Performance Comparison
The selection of an antifouling coating is a trade-off between performance, stability, and fabrication complexity. The following tables summarize key characteristics and analytical performance metrics of zwitterionic polymer brushes and PEG-based coatings, based on current literature.
Table 1: Characteristics of Zwitterionic vs. PEG-Based Coatings
| Characteristic | Zwitterionic Polymer Brushes | PEG-Based Coatings |
|---|---|---|
| Antifouling Mechanism | Electrostatic hydration; forms a dense, stable water layer via charged groups [74] [72] | Steric hindrance & thermodynamic barrier; forms a hydration layer via hydrogen bonding [73] |
| Common Grafting Methods | Surface-Initiated Atom Transfer Radical Polymerization (SI-ATRP), "graft-from" [72] | "Graft-to" (e.g., PEG-DOPA), "graft-from" ATRP, polydopamine-assisted coating [73] |
| Hydrolytic Stability | Generally high; but can be susceptible to degradation depending on the chemical bond (e.g., ester linkers) [72] | Moderate; PEG can undergo oxidative degradation in biological environments [73] |
| Functionalization | Excellent; 3D-functionalization via copolymerization (e.g., with azide-monomers) for subsequent "click" chemistry [72] | Good; functional end-groups (e.g., amine, thiol) available for bioconjugation [73] [75] |
| Relative Cost & Complexity | Higher; requires controlled polymerization and often anhydrous conditions [72] | Moderate; wide availability of functionalized PEGs simplifies grafting [73] |
Table 2: Analytical Performance Metrics in Antifouling Assays
| Performance Metric | Zwitterionic Polymer Brushes | PEG-Based Coatings | Experimental Context |
|---|---|---|---|
| Non-specific Protein Adsorption | < 1 ng/cm² [72] | < 1 ng/cm² (High-density grafts) [73] | Serum or single-protein solution (e.g., fibrinogen); measured by OWLS, ellipsometry. |
| Cell Attachment Resistance | > 90% reduction [72] | > 90% reduction [73] | Fibroblast or macrophage culture on coated surfaces; quantification via microscopy or flow cytometry. |
| Phagocytosis by Macrophages | Strongly reduced (unless specifically targeted) [72] | Data not available in search results | In vitro assays with murine macrophages. |
| Performance in Complex Media | Excellent resistance in 10% serum; minimal non-specific binding in IP-MS [72] | Effective, but may be compromised by oxidative degradation over time [73] | Immunoprecipitation-mass spectrometry (IP-MS); biosensing in serum/plasma. |
| Marine Fouling Resistance | Data not available in search results | Effective against diatoms and algae; performance enhances with PEG content [73] | Exposure to Navicula perminuta (diatom) and Ulva linza (green algae). |
Experimental Protocols
Protocol 1: Grafting Zwitterionic Sulfobetaine Brushes via SI-ATRP
This protocol details the formation of antifouling zwitterionic brushes via Surface-Initiated Atom Transfer Radical Polymerization (SI-ATRP), a "graft-from" technique that yields dense, high-performance brushes [72].
3.1.1 Materials
- Substrate: Gold-coated glass slides or silicon wafers.
- Initiator: ATRP-initiator thiol (e.g., 11-(2-bromo-2-methylpropanoyloxy)undecyl-1-thiol) for gold surfaces.
- Monomer: Sulfobetaine methacrylate (e.g., [2-(Methacryloyloxy)ethyl]dimethyl-(3-sulfopropyl)ammonium hydroxide).
- Catalyst System: Copper(I) bromide (CuBr) and ligand (e.g., N,N,N',N'',N''-Pentamethyldiethylenetriamine, PMDETA).
- Solvent: Methanol/water mixture (typically 2:1 v/v), degassed.
- Azide Monomer: Azide-functionalized sulfobetaine (e.g., for subsequent functionalization via click chemistry).
3.1.2 Procedure
- Substrate Preparation and Initiator Immobilization:
- Clean gold substrates thoroughly with Piranha solution (Caution: Extremely corrosive), followed by rinsing with ethanol and drying under a stream of N₂.
- Immerse the clean substrates in a 1 mM ethanolic solution of the ATRP-initiator thiol for 12-24 hours to form a self-assembled monolayer (SAM).
- Rinse the substrates copiously with ethanol and dry under N₂.
Polymerization Solution Preparation:
- In a Schlenk flask, dissolve the sulfobetaine methacrylate monomer in the degassed methanol/water mixture to a final concentration of 0.5 - 2 M.
- Add the PMDETA ligand to the solution.
- Seal the flask with a rubber septum and purge the solution with N₂ or Ar for 30 minutes to remove oxygen.
Surface-Initiated ATRP:
- Under an inert atmosphere, add the CuBr catalyst to the Schlenk flask.
- Immediately transfer the polymerization solution to a reaction vessel containing the initiator-functionalized substrates.
- Allow the polymerization to proceed for 1-8 hours at room temperature or slightly elevated temperature (e.g., 30°C). The brush thickness increases with time.
- Terminate the reaction by opening the vessel and exposing it to air.
Post-Polymerization Processing:
- Rinse the coated substrates thoroughly with Milli-Q water and methanol to remove physisorbed catalyst and monomer.
- Soak the substrates in a 10 mM EDTA solution (pH ~8) for 1 hour to chelate and remove any residual copper ions.
- Rinse again with water and methanol, and dry under N₂. Store the brushes in a clean, dry environment.
3.1.3 Characterization
- Ellipsometry: Measure dry and hydrated brush thickness on silicon substrates.
- FT-IR and XPS: Confirm the chemical composition of the brush layer.
- Contact Angle Goniometry: Verify high hydrophilicity (very low water contact angle).
Protocol 2: Functionalization of Zwitterionic Brushes via "Click" Chemistry
This protocol enables the site-specific coupling of biomolecules (e.g., antibodies, peptides) to the azide-functionalized zwitterionic brushes, creating a "romantic surface" that resists fouling while allowing specific binding [72].
- Preparation of Alkyne-Functionalized Ligand: Prepare the targeting ligand (e.g., an antibody, biotin, or mannose) with an alkyne group using standard bioconjugation techniques.
- "Click" Reaction Setup: Prepare a degassed PBS or aqueous buffer solution containing the alkyne-functionalized ligand, a Cu(I) catalyst (e.g., from CuSO₄ and sodium ascorbate), and a stabilizing ligand like THPTA.
- Incubation: Immerse the azide-functionalized zwitterionic brush in the reaction solution. Incubate for 2-12 hours at room temperature with gentle agitation.
- Rinsing: After incubation, rinse the substrate thoroughly with PBS, a 10 mM EDTA solution, and finally Milli-Q water to terminate the reaction and remove all catalysts.
Protocol 3: Biomimetic PEG Grafting via Polydopamine Adlayer
This "graft-to" method is a versatile and simple technique for applying antifouling PEG coatings to a wide variety of material surfaces (metals, polymers, ceramics) [73].
3.3.1 Materials
- Substrate: Any material (glass, TiO₂, Au, PTFE, PS, PU, etc.).
- PEG Polymer: Amine-terminated or thiol-terminated PEG (mPEG-NH₂ or mPEG-SH). Molecular weight can be selected based on the desired balance between steric hindrance and grafting density.
- Dopamine Hydrochloride.
- Buffer: 10 mM Tris-HCl buffer, pH 8.5.
3.3.2 Procedure
- Polydopamine Primer Deposition:
- Prepare a 2 mg/mL solution of dopamine hydrochloride in the Tris buffer. The solution will gradually turn dark brown.
- Immerse the clean substrates in the dopamine solution for 30 minutes to 24 hours with gentle agitation. A thin polydopamine (PDA) film will form on all surfaces.
- Remove the substrates and rinse with Milli-Q water to remove loosely bound PDA.
- PEG Grafting:
- Prepare a 1-10 mM solution of mPEG-NH₂ (or mPEG-SH) in a mild buffer or water.
- Immerse the PDA-coated substrates in the PEG solution for 4-12 hours. The amine/thiol groups of PEG will react with the quinone groups of the PDA adlayer via Michael addition or Schiff base formation.
- Rinse the substrates thoroughly with water to remove unreacted PEG and dry under N₂.
3.3.3 Characterization
- Water Contact Angle: A significant decrease compared to the bare substrate indicates successful PEG grafting.
- XPS: Look for the characteristic nitrogen signal from the PDA layer and the increased C-O/C-N signal from PEG.
- Antifouling Validation: Use cell attachment assays (e.g., with fibroblasts) or protein adsorption tests (e.g., with serum) to confirm performance.
Visualizing the Antifouling Coating Workflows
The following diagrams illustrate the core chemical processes and experimental workflows for creating the two types of antifouling surfaces.
Diagram 1: Antifouling Surface Fabrication Workflows. Two primary methods for creating antifouling surfaces are shown: (Left) A multi-step "graft-from" process for growing zwitterionic polymer brushes, allowing for high-density and functionalizable coatings. (Right) A simpler, biomimetic "graft-to" approach for applying PEG coatings to virtually any material surface.
Diagram 2: Molecular Antifouling Mechanisms. While both PEG and zwitterionic coatings prevent fouling by creating a hydration layer, their underlying molecular mechanisms differ. PEG relies on steric hindrance and hydrogen bonding, whereas zwitterionic brushes use strong electrostatic interactions to bind water, potentially leading to a more robust barrier.
The Scientist's Toolkit: Essential Research Reagents
The following table lists key materials required for the fabrication and evaluation of antifouling coatings as described in these protocols.
Table 3: Essential Reagents for Antifouling Coating Research
| Reagent / Material | Function / Application | Example / Key Property |
|---|---|---|
| Sulfobetaine Methacrylate Monomer | The primary monomer for forming zwitterionic polymer brushes via SI-ATRP [72]. | e.g., [2-(Methacryloyloxy)ethyl]dimethyl-(3-sulfopropyl)ammonium hydroxide. |
| ATRP Initiator Thiol | Forms a self-assembled monolayer (SAM) on gold surfaces to initiate brush growth [72]. | e.g., 11-(2-bromo-2-methylpropanoyloxy)undecyl-1-thiol. |
| Amine-Terminated PEG (mPEG-NH₂) | The active polymer for "graft-to" coating; reacts with polydopamine quinones [73]. | Molecular weight: 1k - 10k Da; high purity. |
| Dopamine Hydrochloride | Forms a universal, adherent polydopamine primer layer on virtually any substrate [73]. | Allows subsequent grafting of amines/thiols. |
| Copper(I) Bromide (CuBr) & Ligand (PMDETA) | Catalyst system for ATRP and Copper-Catalyzed Azide-Alkyne Cycloaddition (CuAAC) [72]. | Must be handled under inert atmosphere for ATRP. |
| Azide-Functionalized Sulfobetaine | Co-monomer for introducing click-chemistry handles into zwitterionic brushes for bioconjugation [72]. | Enables creation of "romantic surfaces". |
This direct analytical showdown demonstrates that both zwitterionic polymer brushes and PEG-based coatings provide exceptional resistance to biofouling, achieving non-specific protein adsorption below 1 ng/cm² under optimal conditions [73] [72]. The choice between them, therefore, depends on the specific demands of the biosensing application.
For maximum analytical performance and stability in complex, long-term deployments, zwitterionic brushes present a superior option. Their robust electrostatic hydration mechanism and the ability to create a highly dense, brush-like architecture via "graft-from" methods offer potentially longer-lasting antifouling. Furthermore, the straightforward 3D-functionalization via "click" chemistry makes them the premier choice for creating sophisticated "romantic surfaces" where specific capture is required amidst a challenging background [72].
For versatility, simplicity, and cost-effectiveness, PEG-based coatings, particularly those applied via the biomimetic polydopamine method, are highly advantageous. The ability to coat almost any material with a single, straightforward process is a significant benefit for prototyping or for applications involving complex device geometries [73].
The future of antifouling biosensor surfaces will likely involve further refinement of these materials, including the development of novel zwitterionic monomers with enhanced stability [72] and the synthesis of monodisperse, highly pure PEG derivatives to improve coating consistency and performance [75]. The insights and protocols provided here serve as a foundation for researchers to select, implement, and advance these critical coating technologies for more reliable and sensitive biosensors.
The deployment of biosensors in complex biological fluids like blood, serum, and plasma is fundamentally challenged by biofouling, leading to signal drift and a subsequent loss of measurement accuracy and longevity [76]. This application note details standardized protocols for quantifying this fouling resistance, specifically within the context of a research thesis focused on zwitterionic polymer brushes for antifouling biosensor surfaces. We provide a framework for researchers to quantitatively compare the performance of various surface modifications, enabling the rational design of more stable and reliable biosensors for clinical diagnostics and drug development.
The core challenge is that nonspecific adsorption of proteins, lipids, and cells onto the sensor's active interface creates a fouling layer [77] [76]. This layer can perturb the transducer signal (e.g., by increasing impedance or mass) and sterically hinder the binding of target analytes to capture probes. Signal drift, the temporal decay of the sensor's baseline or output, is a direct consequence of this fouling process. This document outlines methods to deconstruct and measure the components of this drift.
The following tables consolidate key quantitative findings from the literature on antifouling performance and signal drift in relevant biological media.
Table 1: Quantified Signal Drift and Fouling in Complex Media
| Sensor Platform | Surface Chemistry | Biofluid | Key Fouling/Drift Metric | Performance Summary | Reference |
|---|---|---|---|---|---|
| Electrochemical Aptamer | Alkanethiol SAM | Undiluted Whole Blood, 37°C | Signal loss over time | Biphasic signal loss: ~50% rapid drop in 1.5 h (fouling), then slow linear decrease (SAM desorption) | [77] |
| Imaging SPR (iSPR) | Zwitterionic (SBMA) Polymer | Undiluted Blood Plasma | Non-specific adsorption resistance | Superior antifouling vs. PEG; minimal non-specific binding from plasma | [78] |
| Imaging SPR (iSPR) | PEG 3500 | Undiluted Blood Plasma | Non-specific adsorption resistance | Excellent antifouling properties, slightly outperformed by zwitterionic SBMA | [78] |
| QCM | PEDOT-PC Zwitterionic Copolymer | Protein Solutions | Mass adsorption from fouling | Fouling reduced with higher zwitterionic content, but specific capture efficiency also diminished | [79] |
Table 2: Antifouling Material Performance Comparison
| Material Type | Example | Proposed Antifouling Mechanism | Key Strengths | Reported Limitations |
|---|---|---|---|---|
| Zwitterionic Polymers | Poly(SBMA), Poly(CBMA) | Electrostatic-induced hydration; steric repulsion; long-range electrostatic repulsion [2] | Excellent stability; high hydration capacity; resistance to oxidation | May reduce specific capture efficiency [79]; requires controlled grafting |
| PEG-based Polymers | PEG, POEGMA | Steric hindrance; formation of hydration layer | Well-established history; effective antifouling | Susceptible to oxidative degradation [78] [61] |
| Hydrogel-like Brushes | Poly(HPMAA) | Strong hydration; steric repulsion | Recommended for high-quality antifouling layers [80] | Requires optimized thickness (20-30 nm) and high grafting density |
Experimental Protocols for Quantifying Fouling Resistance
Protocol: Quartz Crystal Microbalance with Dissipation (QCM-D) for Protein Fouling Kinetics
1. Objective: To quantitatively monitor the real-time mass adsorption of fouling agents from serum or plasma onto sensor surfaces.
2. Materials:
- QCM-D instrument (e.g., Biolin Scientific)
- Gold-coated QCM-D sensors (QSX 301)
- Poly(EDOT-MI-co-EDOT-PC) solution for electropolymerization [79]
- Running buffer: Phosphate Buffered Saline (PBS), pH 7.4
- Challenge solutions: 100% blood plasma, 50% serum in PBS, or 1 mg/mL BSA in PBS
- Peristaltic pump
3. Methodology: 1. Sensor Modification: Electropolymerize the zwitterionic copolymer or other polymer brushes onto the gold sensor surface. A common protocol involves applying a positive constant potential (e.g., 1.1 V vs. Ag/Ag+) for 5 seconds, followed by a reduction step (-0.5 V for 3 s) in a monomer solution [79]. 2. Baseline Establishment: Place the modified sensor in the QCM-D flow chamber. Flow running buffer at a constant rate (e.g., 30 µL/min) until a stable frequency (Δf < 2 Hz over 30 min) is achieved. 3. Fouling Challenge: Switch the flow to the challenge biofluid (e.g., 100% plasma). Monitor the shifts in resonance frequency (Δf) and energy dissipation (ΔD) in real-time. A decrease in frequency indicates mass adsorption. 4. Buffer Rinse: Switch back to running buffer to remove loosely adsorbed material and assess the irreversible fouling component. 5. Data Analysis: Use the Sauerbrey equation to correlate the frequency shift with the adsorbed mass. The dissipation factor provides insights into the viscoelastic properties of the adsorbed layer.
Protocol: Electrochemical Measurement of Signal Drift in Whole Blood
1. Objective: To characterize the signal stability and identify mechanisms of drift for electrochemical biosensors in whole blood.
2. Materials:
- Potentiostat (e.g., Autolab PGSTAT128N)
- Gold working electrode, Pt counter electrode, Ag/AgCl reference electrode
- Self-assembled monolayer (SAM) formation reagents (e.g., alkanethiols)
- Methylene blue (MB)-modified DNA or enzyme-resistant RNA probe [77]
- Undiluted, heparinized whole blood, maintained at 37°C
3. Methodology: 1. Sensor Fabrication: Clean the gold electrode. Immerse it in a solution of thiolated molecular probes to form a dense SAM. Backfill with a short-chain mercaptan to minimize defects. 2. Initial Signal Measurement: In a buffer solution, perform square-wave voltammetry (SWV) to record the initial peak current of the redox reporter (e.g., Methylene Blue). 3. Drift Measurement in Blood: Immerse the sensor in undiluted whole blood at 37°C. Continuously or intermittently perform SWV scans over several hours. 4. Mechanism Deconstruction: * Fouling Test: After a period of interrogation (e.g., 2.5 h), wash the sensor with a solubilizing agent like concentrated urea. Signal recovery indicates reversible biofouling is a major contributor [77]. * Electrochemical Stress Test: In PBS, vary the SWV potential window. A strong dependence of drift rate on the applied potential indicates electrochemically driven desorption of the SAM is a key mechanism [77]. 5. Data Analysis: Plot normalized signal vs. time. The drift profile can often be deconvoluted into a rapid, exponential phase (dominated by fouling) and a slower, linear phase (dominated by SAM desorption).
Protocol: Surface Plasmon Resonance (SPR) for Specific vs. Non-Specific Binding
1. Objective: To simultaneously measure the specific capture of a target analyte and the non-specific adsorption of interferents from complex media.
2. Materials:
- SPR or imaging SPR (SPRi) instrument
- SPR gold chips
- Poly(carboxybetaine methacrylate) (PCBMA) or poly(oligo(ethylene glycol) methyl ether methacrylate) (POEGMA) grafting solutions [78] [2]
- Biomolecular receptors (antibodies, aptamers)
- Target analyte in buffer and in spiked biofluid (e.g., serum)
3. Methodology: 1. Surface Functionalization: Graft a zwitterionic polymer brush (e.g., PCBMA via ATRP) onto the SPR chip. Subsequently, immobilize the biorecognition element (e.g., an antibody) using bio-orthogonal chemistry. 2. Antifouling Test: Flow the pure biofluid (e.g., serum) over the functionalized surface. The resulting change in resonance units (RU) is a direct measure of non-specific adsorption. 3. Specific Binding Test: Flow a solution of the target analyte prepared in the same biofluid over the surface. The total RU change is a combination of specific binding and non-specific adsorption. 4. Data Analysis: The specific binding signal is isolated by subtracting the response from a control channel (lacking the receptor) or from the initial antifouling baseline. Compare the signal-to-noise ratio between different polymer brush coatings.
Visualization of Experimental Workflows and Interactions
Sensor Fouling and Signal Drift Experimental Workflow
The following diagram illustrates the core experimental process for evaluating sensor fouling, from surface preparation to data analysis.
Interaction Mechanisms at the Functionalized Sensor Interface
This diagram visualizes the competing interactions at the sensor interface that determine the observed signal drift.
The Scientist's Toolkit: Research Reagent Solutions
Table 3: Essential Materials for Fouling Resistance Experiments
| Category | Item | Function/Description | Key Consideration |
|---|---|---|---|
| Surface Chemistry | EDOT-MI and EDOT-PC Monomers | Electropolymerizable monomers to create a surface with tunable maleimide (for probe attachment) and phosphorylcholine (antifouling) group ratios [79]. | Enables quantitative study of the trade-off between fouling resistance and capture efficiency. |
| Carboxybetaine Methacrylate (CBMA) | A zwitterionic monomer for grafting antifouling polymer brushes via ATRP [2] [80]. | Forms a strong hydration layer via electrostatic interactions; recommended for high-quality brushes. | |
| Sulfobetaine Methacrylate (SBMA) | A zwitterionic monomer for creating highly protein-resistant surfaces [78]. | Shows superior antifouling in SPR compared to PEG coatings. | |
| Biorecognition | Thiol- or Cysteine-terminated Peptide Aptamers | Allows for specific, oriented immobilization on gold or maleimide-functionalized surfaces via thiol-gold or thiol-maleimide "click" chemistry [61] [79]. | Provides high specificity and stability compared to some antibodies. |
| Experimental Tools | QCM-D Gold Sensors (QSX 301) | Standardized sensor chips for mass-sensitive adsorption measurements in liquid. | Ensure consistent surface roughness and chemistry for reproducible grafting. |
| Piranha Solution (H₂SO₄:H₂O₂) | A powerful oxidizing agent for cleaning organic residues from gold sensor surfaces prior to modification. | Handle with extreme caution; requires specialized safety protocols. | |
| Challenge Media | Undiluted Whole Blood (Heparinized) | The most challenging biofluid for testing sensor stability, containing the full spectrum of fouling agents (cells, proteins, lipids) [77]. | Must be used fresh or properly stored; heparin prevents clotting without chelating calcium like EDTA. |
| Blood Plasma or Serum | Used to test fouling from soluble proteins and lipids without the complication of cells [78] [76]. | Serum lacks clotting factors, which may be present in plasma and could adsorb to surfaces. |
Polyethylene glycol (PEG) has long been regarded as the "gold standard" for imparting stealth properties to biomaterials, drugs, and biosensors due to its excellent biocompatibility and ability to reduce nonspecific protein adsorption [1]. However, extensive clinical use of PEGylated therapeutics has revealed a significant limitation: PEG can trigger immune responses leading to the production of anti-PEG antibodies (APAs) [81] [82]. These antibodies can accelerate blood clearance of PEGylated agents, reduce therapeutic efficacy, and potentially cause hypersensitivity reactions [81] [83]. This immunogenicity challenge is particularly critical for biosensor applications, where fouling and immune recognition compromise device functionality and longevity.
The issue of PEG immunogenicity has moved from theoretical concern to clinical reality. Pre-existing anti-PEG antibodies have been detected in approximately 72% of the population with no prior exposure to PEGylated drugs, likely due to widespread presence of PEG in consumer products [81]. This high prevalence, combined with the immune response triggered by repeated administration of PEGylated agents, creates a substantial challenge for implanted biomedical devices including biosensors [81] [82].
Understanding Anti-PEG Antibody Generation and Consequences
Mechanisms of Anti-PEG Antibody Production
Anti-PEG antibody generation occurs through two primary immunological pathways with distinct characteristics and implications for biosensor performance.
Table 1: Mechanisms of Anti-PEG Antibody Generation
| Pathway Type | Antigen Trigger | Immune Cells Involved | Antibody Output | Clinical Impact |
|---|---|---|---|---|
| Thymus-Dependent (TD) | PEGylated proteins/peptides [81] | B-cells, CD4+ T-cells (TFH) [81] | High-affinity IgG [81] | Long-term immunologic memory, reduced drug efficacy [81] |
| Thymus-Independent (TI-2) | Multivalent PEG structures (nanoparticles, liposomes) [81] | Marginal zone B-cells [81] | Primarily IgM, some IgG [81] | Accelerated blood clearance, complement activation [81] |
The following diagram illustrates these two distinct immunological pathways responsible for anti-PEG antibody production:
Biological Consequences of Anti-PEG Antibodies
The presence of anti-PEG antibodies triggers several biological effects that directly impact biosensor performance and therapeutic efficacy:
- Accelerated Blood Clearance (ABC) Phenomenon: Anti-PEG antibodies bind to PEGylated surfaces, promoting opsonization and rapid clearance by the mononuclear phagocyte system, substantially reducing circulation time [81] [84].
- Complement Activation: Antibody binding can trigger complement cascade activation, potentially leading to hypersensitivity reactions and inflammatory responses at the implant site [84].
- Reduced Efficacy: Antibody binding can sterically hinder the functional components of biosensors or drug delivery systems, impairing their intended activity [81] [83].
Zwitterionic Polymers as Next-Generation Antifouling Solutions
Advantages of Zwitterionic Polymer Brushes
Zwitterionic polymers represent a promising alternative to PEG, offering superior antifouling properties without the immunogenic concerns associated with PEG. These polymers contain both positive and negative charged groups while maintaining overall electrical neutrality, creating a robust hydration layer through electrostatic interactions with water molecules [1].
Table 2: Comparison of Antifouling Polymer Systems
| Characteristic | PEG-Based Materials | Zwitterionic Polymers | Advantage |
|---|---|---|---|
| Hydration Mechanism | Hydrogen bonding [1] | Electrostatic interactions [1] | Denser hydration layer [1] |
| Stability | Susceptible to oxidation and degradation [1] | High chemical and physical stability [1] | Longer functional lifetime |
| Immunogenicity | Induces anti-PEG antibodies [81] [82] | Minimal immunogenicity reported [1] | Reduced immune recognition |
| Protein Adsorption | Low (~5-10% surface coverage) [1] | Ultra-low (<1% surface coverage) [1] | Superior fouling resistance |
Zwitterionic Polymer Variants and Characteristics
Three main classes of zwitterionic polymers have demonstrated exceptional antifouling properties for biomedical applications:
- Poly(carboxybetaine methacrylate) (PCBMA): Contains carboxylate and quaternary ammonium groups, exhibits excellent resistance to non-specific protein adsorption and cell adhesion [85].
- Poly(sulfobetaine methacrylate) (PSBMA): Features sulfonate groups that provide strong hydration and stability across various pH conditions [8].
- Poly(2-methacryloyloxyethyl phosphorylcholine) (PMPC): Mimics the outer surface of cell membranes, demonstrating exceptional biocompatibility and protein resistance [8].
Experimental Protocols for Zwitterionic Polymer Brush Evaluation
Protocol 1: Surface-Initiated Atom Transfer Radical Polymerization (SI-ATRP) of Zwitterionic Brushes
Principle: SI-ATRP allows controlled growth of polymer brushes from surfaces with precise control over brush thickness and density, critical for optimizing antifouling performance [1].
Materials:
- Silicon wafers or sensor substrates
- ATRP initiator (e.g., 2-bromoisobutyryl bromide)
- Zwitterionic monomer (PCBMA, PSBMA, or PMPC)
- Catalyst system (Cu(I)Br, bipyridyl ligand)
- Deoxygenated solvent (water/methanol mixture)
Procedure:
- Surface Preparation: Clean substrates with oxygen plasma treatment for 30 minutes at 100W.
- Initiator Immobilization: Immerse substrates in 1mM initiator solution in anhydrous toluene for 12 hours at room temperature.
- Polymerization Solution Preparation: Prepare monomer solution (2M zwitterionic monomer in 1:1 water:methanol). Add Cu(I)Br and ligand (1:2 molar ratio to monomer). Degas with nitrogen for 30 minutes.
- Polymerization: Transfer initiator-functionalized substrates to polymerization solution. React for 2-24 hours at room temperature under nitrogen atmosphere.
- Termination: Remove substrates and rinse extensively with deionized water and ethanol to remove catalyst and unreacted monomer.
- Characterization: Measure brush thickness by ellipsometry, composition by XPS, and morphology by AFM.
Protocol 2: Quantitative Protein Adsorption Assay Using Quartz Crystal Microbalance with Dissipation (QCM-D)
Principle: QCM-D measures mass changes on surfaces by monitoring resonance frequency shifts of a quartz crystal, enabling real-time quantification of protein adsorption on zwitterionic brushes [85].
Materials:
- QCM-D sensors with grafted zwitterionic brushes
- Reference surfaces (PEGylated and bare gold)
- Protein solutions (fibrinogen, albumin, serum)
- Phosphate buffered saline (PBS), pH 7.4
- QCM-D flow system
Procedure:
- Baseline Establishment: Mount sensors in QCM-D chambers and establish stable baseline with PBS flow at 0.1 mL/min for 30 minutes.
- Protein Exposure: Introduce 1 mg/mL protein solution in PBS for 60 minutes at 0.05 mL/min flow rate.
- Rinsing Phase: Return to pure PBS flow for additional 60 minutes to remove loosely adsorbed proteins.
- Data Analysis: Calculate adsorbed mass using Sauerbrey equation: Δm = -C·Δf/n, where C=17.7 ng·cm⁻²·Hz⁻¹ for 5 MHz crystals, n=overtone number.
- Comparative Analysis: Compare protein adsorption on zwitterionic surfaces versus PEGylated and bare gold controls.
Protocol 3: Anti-PEG Antibody Binding Assay Using Surface Plasmon Resonance (SPR)
Principle: SPR measures binding kinetics between anti-PEG antibodies and polymer surfaces, quantifying the immunogenic advantage of zwitterionic materials.
Materials:
- SPR chips with grafted PEG and zwitterionic polymers
- Anti-PEG IgG and IgM antibodies (commercial sources)
- Human serum samples with known APA levels
- Running buffer (HEPES-buffered saline with surfactant)
- SPR instrument with flow system
Procedure:
- Surface Preparation: Functionalize SPR chips with PEG brushes (MW 2k-5k Da) and zwitterionic brushes (PCBMA, PSBMA) using protocols in Section 4.1.
- Baseline Establishment: Prime SPR system with running buffer until stable baseline achieved.
- Antibody Injection: Inject anti-PEG antibody solutions (0.1-100 μg/mL) over sensor surfaces for 5 minutes association time.
- Dissociation Phase: Monitor dissociation in running buffer for 15 minutes.
- Regeneration: Regenerate surfaces with mild acid (10mM glycine, pH 2.5) between measurements.
- Data Analysis: Calculate binding response (RU) and kinetic parameters (ka, kd, KD) for each surface-antibody combination.
Research Reagent Solutions for Antifouling Surface Development
Table 3: Essential Research Reagents for Zwitterionic Polymer Brush Studies
| Reagent/Category | Specific Examples | Function/Application | Key Characteristics |
|---|---|---|---|
| Zwitterionic Monomers | Carboxybetaine methacrylate (CBMA) [85], Sulfobetaine methacrylate (SBMA) [8] | Building blocks for antifouling polymer brushes | Ultra-low protein adsorption, high hydrophilicity, chemical stability |
| Polymerization Initiators | 2-Bromoisobutyryl bromide [85], Trichlorosilane-based ATRP initiators | Surface priming for brush growth | Reactive toward surface OH groups, compatible with ATRP |
| Catalyst Systems | Cu(I)Br/PMDETA [85], Cu(I)Br/bipyridyl | Controlled radical polymerization | Oxygen-sensitive, determine polymerization rate and control |
| Characterization Standards | Fibrinogen, albumin, human serum [85] | Fouling resistance evaluation | Representative protein challenges for biofouling studies |
| Reference Materials | mPEG-DSPE, mPEG-DMG [84] | PEG-based comparator surfaces | Established stealth materials with known immunogenicity issues |
Implementation Strategy and Technical Considerations
The following workflow outlines a comprehensive approach for developing and evaluating zwitterionic polymer brush coatings for biosensor applications:
Key Technical Considerations for Implementation
- Surface Preparation: Optimal results require scrupulously clean substrates with uniform initiator deposition [85].
- Polymerization Control: Brush density and thickness significantly impact antifouling performance - target 20-100nm thickness with high grafting density [85].
- Environmental Factors: Zwitterionic polymer performance can vary with ionic strength and pH - test under physiologically relevant conditions [8].
- Stability Assessment: Evaluate long-term stability in biological media through accelerated aging studies [1].
The emerging challenge of PEG immunogenicity necessitates the development of next-generation antifouling materials for biomedical devices and biosensors. Zwitterionic polymer brushes represent a promising alternative, offering superior antifouling performance without triggering anti-polymer antibody responses. Through careful selection of zwitterionic chemistry, controlled surface grafting, and comprehensive evaluation using the protocols outlined herein, researchers can develop biosensor surfaces with enhanced longevity and reliability in complex biological environments. The experimental frameworks and technical considerations presented provide a roadmap for transitioning from PEG-based systems to zwitterionic alternatives that circumvent the limitations of anti-PEG antibody responses.
Zwitterionic polymer brushes have emerged as a leading class of materials for constructing antifouling surfaces on biosensors and medical devices. Their exceptional performance stems from their ability to form a strong hydration layer via electrostatic interactions, which effectively minimizes the non-specific adsorption of proteins, cells, and other biological foulants [1]. For biosensors, where signal accuracy is paramount, maintaining a pristine, non-fouling surface is critical for long-term functionality and reliability. A key determinant of the long-term efficacy of these coatings is their stability against hydrolytic and oxidative degradation in the biological milieu. This Application Note provides a quantitative summary of the stability data for various zwitterionic polymers and details experimental protocols for assessing their resistance, providing researchers with a framework for developing durable antifouling biosensor interfaces.
Quantitative Stability Data
The long-term stability of antifouling polymer brushes is a prerequisite for their application in implantable biosensors and long-term monitoring devices. The following tables summarize key stability data for different polymer classes under various conditions.
Table 1. Hydrolytic Stability of Zwitterionic and Non-Ionic Polymers in Aqueous Solutions [86]
| Polymer Class | Specific Polymer | Condition (1 Year at RT) | Observed Stability |
|---|---|---|---|
| Polymethacrylates | Poly(oligoethylene glycol methylether methacrylate) (POEGMA) | PBS, pH 7.4 | No degradation |
| Poly(sulfobetaine methacrylate) (P-SPE) | PBS, pH 7.4 | No degradation | |
| Poly(sulfobetaine methacrylate) (P-SPE) | 1 M HCl | No degradation | |
| Poly(sulfobetaine methacrylate) (P-SPE) | Sodium Carbonate, pH 10 | No degradation | |
| Polymethacrylamides | Poly(sulfobetaine methacrylamide) (P-SPP) | PBS, pH 7.4 | No degradation |
| Poly(sulfobetaine methacrylamide) (P-SPP) | 1 M HCl | No degradation | |
| Poly(sulfobetaine methacrylamide) (P-SPP) | Sodium Carbonate, pH 10 | No degradation | |
| Poly(sulfobetaine methacrylamide) (P-SPP) | 1 M NaOH | No degradation | |
| Polysulfabetaines | Various (with hemisulfate group) | PBS, pH 7.4 | Partially labile |
Table 2. Stability of PMPC Brushes with Enhanced Anchoring Chemistry [87]
| Brush System | Substrate | Anchoring Chemistry | Test Condition | Stability (Dry Thickness Loss) |
|---|---|---|---|---|
| Poly(2-methacryloyloxyethyl phosphorylcholine) (PMPC) | Silicon | PGMA-linked initiator | 4 weeks in saline solution | ~1% |
| 4 weeks in artificial sea water | ~1% | |||
| 100,000 ppm-hours in NaOCl | Retained hydrophilicity |
Table 3. Impact of Brush Properties on Antifouling Performance [27]
| Factor | Impact on Protein Adsorption | Key Finding |
|---|---|---|
| Polymer Density | High influence | Machine learning analysis identified density as a more critical factor than molecular weight for minimizing protein adsorption. |
| Molecular Weight | Moderate influence | An optimal brush thickness (often 30-60 nm in dry state) exists for minimizing fouling. |
Experimental Protocols
Protocol for Assessing Long-Term Hydrolytic Stability
This protocol is adapted from long-term stability studies on zwitterionic polymers to evaluate their resistance to hydrolysis under accelerated and physiologically relevant conditions [86].
Objective: To evaluate the stability of zwitterionic polymer brushes or bulk polymers against hydrolytic degradation in buffers of varying pH over an extended period.
Materials:
- Test Polymers: Zwitterionic polymer brushes synthesized on substrates or as bulk powder.
- Deuterated Buffers: PBS (pH 7.4) in D₂O, DCl in D₂O (pH ~0), Na₂CO₃/NaHCO₃ in D₂O (pH 10), NaOD in D₂O (pH ~14).
- NMR Tubes
- Spectrometer: NMR spectrometer.
Procedure:
- Sample Preparation: Place polymer samples (approx. 5-10 mg) into NMR tubes. For brush samples, substrate chips can be placed in the tube.
- Solution Addition: Add 0.5-0.7 mL of the desired deuterated buffer solution to each tube. For ionic zwitterionic polymers, add a defined amount of NaCl (e.g., 0.1-0.5 M) to suppress chain collapse and ensure the polymer is in solution [86].
- Baseline Measurement: Acquire a ¹H NMR spectrum of each sample at time zero (t=0).
- Incubation: Seal the tubes and store them at ambient temperature (e.g., 22-25 °C) for the duration of the study.
- Periodic Monitoring: At predetermined time intervals (e.g., 1, 3, 6, and 12 months), re-acquire the ¹H NMR spectrum for each sample without disturbing the incubation.
- Data Analysis: Compare the spectra to the t=0 baseline. Look for the appearance of new peaks or the disappearance of characteristic peaks (e.g., ester -OCH₃ for polymethacrylates, amide -CONH- for polymethacrylamides) to identify degradation products or side-chain cleavage.
Protocol for Enhancing Brush Stability via Anchoring Chemistry
This protocol details a method to substantially improve the stability of polymer brushes against degrafting from surfaces, a common failure mode in aqueous applications [87].
Objective: To graft zwitterionic brushes from a poly(glycidyl methacrylate) (PGMA) linker layer to enhance long-term stability in aqueous and saline environments.
Materials:
- Substrates: Silicon wafers, gold sensors, or other relevant biosensor substrates.
- Reagents: Glycidyl methacrylate (GMA), 2-bromoisobutyryl bromide, 2-methacryloyloxyethyl phosphorylcholine (MPC) monomer, catalyst (e.g., triethylamine), and ATRP initiator/catalyst system (e.g., CuBr/PMDETA).
- Solvents: Toluene, anisole, methanol.
Procedure:
- Substrate Cleaning: Clean the substrate thoroughly with oxygen plasma or piranha solution to generate surface hydroxyl groups.
- PGMA Linker Attachment:
- Immerse the substrate in a 0.5-1.0% (w/v) solution of PGMA in toluene for 20-30 minutes.
- Withdraw the substrate slowly to ensure a uniform film.
- Bake the substrate at 100-110 °C for 10-20 minutes to covalently anchor the PGMA epoxy groups to the surface hydroxyls.
- Initiator Immobilization:
- Expose the PGMA-coated substrate to a solution of 2-bromoisobutyryl bromide (0.1 M) and triethylamine (0.1 M) in toluene for 30-60 minutes at room temperature.
- Rinse thoroughly with toluene and ethanol to remove unbound initiator.
- Surface-Initiated ATRP:
- Place the initiator-functionalized substrate in a Schlenk flask containing a deoxygenated solution of MPC monomer (e.g., 1 M) in a methanol/water mixture.
- Add the ATRP catalyst (e.g., CuBr/PMDETA).
- Purge the reaction mixture with nitrogen and allow polymerization to proceed for a predetermined time (e.g., 1-4 hours) to achieve the desired brush thickness.
- Remove the substrate and rinse extensively with Milli-Q water to terminate the reaction and remove physisorbed polymer.
Visualizations
Stability Testing and Optimization Workflow
Structure-Property Relationships in Zwitterionic Brushes
The Scientist's Toolkit: Research Reagent Solutions
Table 4. Essential Materials for Zwitterionic Brush Synthesis and Stability Assessment
| Reagent / Material | Function / Explanation | Key Consideration |
|---|---|---|
| Sulfobetaine Methacrylamide (SPP) Monomer | Forms polyzwitterions with exceptional hydrolytic stability, even in strong base [86]. | Preferred over methacrylates for applications requiring extreme pH resistance. |
| 2-Methacryloyloxyethyl Phosphorylcholine (MPC) Monomer | Forms biocompatible PMPC brushes, widely used for antifouling coatings [87]. | |
| Poly(Glycidyl Methacrylate) (PGMA) | A polymeric anchoring layer that provides a multivalent, robust linkage for initiators, drastically reducing degrafting [87]. | Superior to monofunctional silane-based initiator layers. |
| ATRP Initiator (e.g., 2-Bromoisobutyryl bromide) | The surface-bound species that initiates the controlled growth of the polymer brush via ATRP [87]. | Must be coupled to the anchoring layer under anhydrous conditions. |
| Deuterated Buffers (PBS-D, DCl, NaOD in D₂O) | Allows for long-term stability monitoring of polymers in solution via ¹H NMR without interfering solvent signals [86]. | Required for direct, molecular-level assessment of hydrolytic degradation. |
| Quartz Crystal Microbalance with Dissipation (QCM-D) | A key analytical instrument for real-time, label-free measurement of protein adsorption onto brush-coated sensors [27]. | Directly quantifies antifouling performance. |
The reliable detection of specific biomarkers in complex biological fluids (e.g., blood, serum, plasma) is a cornerstone of modern diagnostics and therapeutic drug monitoring. However, the nonspecific adsorption of proteins, cells, and other biomolecules onto sensor surfaces—a phenomenon known as biofouling—severely compromises the sensitivity, accuracy, and longevity of biosensors. Zwitterionic polymer brushes, composed of monomers containing pairs of oppositely charged groups, have emerged as a powerful solution to this challenge. These brushes form a strong surface hydration layer via ionic solvation, creating a physical and energetic barrier that effectively resists fouling [3] [4]. This application note provides a consolidated overview of the validation of zwitterionic brush-modified surfaces across four major biosensing platforms: Optical Fibre Long-Period Grating (LPG), Electrochemical Aptamer-Based (E-AB), Quartz Crystal Microbalance (QCM), and Porous Silicon (PSi) sensors. The included protocols and data serve as a guide for implementing these advanced antifouling strategies in diverse biosensing applications.
The table below summarizes the key performance metrics of zwitterionic surfaces validated on different biosensor platforms, demonstrating their broad-spectrum antifouling efficacy and enhanced sensing capabilities.
Table 1: Validation of Zwitterionic Coatings Across Biosensing Platforms
| Sensing Platform | Zwitterionic Coating | Target Analyte | Test Medium | Key Antifouling/Sensing Performance |
|---|---|---|---|---|
| Optical Fibre (LPG) [88] | Antifouling Terpolymer Brush (ATB) of CBMAA, SBMAA, and HPMAA | IgG (Immunoglobulin G) | Buffer & Diluted Blood Plasma | Enabled specific detection of IgG in diluted plasma; demonstrated antifouling against plasma proteins. |
| Electrochemical (E-AB) [89] | Poly-sulfobetaine methacrylate (pSBMA) & Polydopamine (PDA) hybrid | Vancomycin | Blood, Serum, Milk, Artificial Interstitial Fluid | Signal drift < 8.5% over 24 hours; >90% signal retention in tissue-mimicking gels and ex vivo porcine skin. |
| Quartz Crystal Microbalance (QCM) [90] | Poly(MADMPAO) (pTMAO) brushes | Bovine Serum Albumin (BSA) | Salt solutions (NaCl, Na2SO4, MgCl2) | Marked decrease in frequency shift (Δf) indicating great inhibition of BSA adsorption. |
| Porous Silicon (PSi) [69] | Zwitterionic Peptide (EKEKEKEKEKGGC) | Lactoferrin | Gastrointestinal (GI) Fluid, Bacterial Lysate | >1 order of magnitude improvement in Limit of Detection (LOD) and Signal-to-Noise ratio over PEG-coated sensors. |
Experimental Protocols & Methodologies
Protocol A: Surface-Initiated Atom Transfer Radical Polymerization (SI-ATRP) on Optical Fibre LPG Sensors
This protocol details the grafting of an antifouling terpolymer brush (ATB) onto the sensitive region of an optical fibre Long-Period Grating (LPG) sensor, as described in the foundational work by Vrabcová et al. [88].
Principle: SI-ATRP is a "grafting from" technique that grows polymer chains directly from an initiator-modified surface, allowing for the formation of dense, well-defined polymer brushes.
Materials:
- Sensor Substrate: Photosensitive boron–germanium co-doped optical fibre (e.g., Fibercore PS1250/1500).
- Initiator: 11-mercaptoundecyl-2-bromo-2-methylpropanoate (for gold coating) or (MeO)3-Si-(CH2)11-Br (for silica surfaces).
- Monomers: Carboxybetaine methacrylamide (CBMAA), Sulfobetaine methacrylamide (SBMAA), N-(2-hydroxypropyl)methacrylamide (HPMAA).
- Catalyst System: Copper(I) chloride (CuCl), Copper(II) chloride (CuCl2), 1,4,8,11-tetramethyl-1,4,8,11-tetraazacyclotetradecane (Me4cyclam).
- Solvents: Anhydrous heptane, methanol, ultrapure water (18.0 MΩ·cm).
Procedure:
- Surface Preparation: Clean the LPG fibre sensor sequentially with acetone, isopropanol, and ethanol. Dry under a stream of inert gas (e.g., N2).
- Initiator Immobilization:
- For silica fibres, immerse the sensing region in an anhydrous heptane solution containing 1 mM bromo-silane initiator for 12 hours at room temperature under an inert atmosphere.
- For gold-coated fibres, immerse in a 1 mM ethanolic solution of the thiol-based initiator for the same duration.
- Rinsing: After immobilization, thoroughly rinse the fibre with the respective solvent (heptane or ethanol) to remove physisorbed initiator.
- Polymerization Solution Preparation: In a Schlenk flask, degas a mixture of the monomers (CBMAA, SBMAA, HPMAA), CuCl, CuCl2, and Me4cyclam catalyst in a methanol/water solvent system. Purge with an inert gas to remove oxygen.
- ATRP Polymerization: Transfer the initiator-functionalized fibre to the reaction flask, ensuring the sensitive region is fully immersed in the polymerization solution. Seal the flask and place it in a temperature-controlled bath at a defined temperature (e.g., 25-40°C) for a set period (e.g., 1-2 hours) to allow polymer brush growth.
- Termination and Cleaning: Remove the fibre from the solution and rinse extensively with methanol and ultrapure water to terminate the reaction and remove any unreacted monomers or catalyst.
Diagram 1: Workflow for SI-ATRP on an optical fibre LPG sensor.
Protocol B: Dopamine-Assisted Grafting of Zwitterionic Brushes for QCM Sensors
This protocol outlines a versatile method for modifying sensor surfaces using polydopamine (pDA) as a universal adhesive layer, followed by the grafting of zwitterionic polymer brushes, as applied to QCM chips [90].
Principle: Dopamine undergoes self-polymerization under mild alkaline conditions to form a thin, adherent pDA film on virtually any substrate. This film presents functional groups for subsequent immobilization of an ATRP initiator, enabling the "grafting from" of polymer brushes.
Materials:
- Sensor Substrate: Bare gold QCM chips (e.g., QSX301).
- Dopamine hydrochloride.
- Tris(hydroxymethyl)aminomethane (Tris) buffer (10 mM, pH 8.5).
- Initiator: e.g., Bromo-silanes or a bromo-functionalized ATRP initiator.
- Zwitterionic Monomer: e.g., synthesized MADMPAO [90] or commercial SBMA/CBMA.
- Catalyst System: Copper(I) bromide (CuBr), Tris(2-dimethylaminoethyl)amine (Me6TREN).
Procedure:
- pDA Coating:
- Prepare a dopamine solution (2 mg/mL) in 10 mM Tris buffer (pH 8.5).
- Immerse the clean gold QCM chip in the dopamine solution with gentle shaking for several hours (e.g., 4-8 hours) until a uniform pDA film is deposited.
- Rinse the chip thoroughly with ultrapure water and dry under N2.
- Initiator Attachment: Immerse the pDA-coated chip in a solution of the ATRP initiator to covalently anchor initiator molecules to the pDA layer.
- SI-ATRP: Follow a procedure similar to Protocol A, Step 5, using the desired zwitterionic monomer (e.g., MADMPAO, SBMA) to grow the polymer brush from the initiator-functionalized surface.
- Validation: The modified QCM chip can be validated by monitoring frequency (Δf) and dissipation (ΔD) shifts upon exposure to protein solutions like BSA. A minimal frequency shift indicates successful antifouling [90].
Protocol C: Functionalization with Zwitterionic Peptides for Porous Silicon Biosensors
This protocol describes the covalent immobilization of short, sequence-defined zwitterionic peptides onto a Porous Silicon (PSi) surface for ultralow fouling in complex biofluids [69].
Principle: Zwitterionic peptides with alternating charged amino acids (e.g., Glutamic Acid 'E' and Lysine 'K') form a stable, charge-neutral hydration layer. A terminal cysteine residue allows for directional conjugation onto silanized surfaces.
Materials:
- Sensor Substrate: Porous Silicon (PSi) thin films.
- Zwitterionic Peptide: e.g., EKEKEKEKEKGGC, synthesized commercially.
- (3-Mercaptopropyl)trimethoxysilane (MPTMS).
- Anhydrous toluene.
- Buffers: Phosphate Buffered Saline (PBS), etc.
Procedure:
- PSi Hydrosilylation: Thermally hydrosilylate the PSi film to create a stable, hydrogen-terminated Si-H surface.
- Silanization:
- Functionalize the PSi surface with MPTMS by immersing it in a 1-2% (v/v) solution of MPTMS in anhydrous toluene for 2-4 hours. This introduces thiol (-SH) groups onto the surface.
- Rinse with toluene and ethanol to remove excess silane.
- Peptide Conjugation:
- Immerse the thiol-functionalized PSi chip in a PBS solution (pH ~7.4) containing the zwitterionic peptide (e.g., 0.1-1.0 mM).
- Allow the reaction to proceed for 12-24 hours at room temperature. The thiol group of the peptide's terminal cysteine will form a disulfide bond with the thiolated surface.
- Rinsing and Storage: Rinse the chip extensively with PBS and ultrapure water to remove unbound peptide. The sensor is now ready for use or can be stored in buffer at 4°C.
The Scientist's Toolkit: Essential Reagents and Materials
Table 2: Key Research Reagent Solutions for Zwitterionic Biosensor Development
| Reagent / Material | Function / Role | Specific Examples & Notes |
|---|---|---|
| Zwitterionic Monomers | Building blocks of the antifouling polymer brush. | Carboxybetaine methacrylamide (CBMAA), Sulfobetaine methacrylamide (SBMAA) [88], Sulfobetaine methacrylate (SBMA) [89], MADMPAO (a TMAO analogue) [90]. |
| ATRP Initiators | Anchor points for surface-initiated polymer growth. | Thiol-based (e.g., 11-mercaptoundecyl-2-bromo-2-methylpropanoate) for gold; Silane-based (e.g., (MeO)3-Si-(CH2)11-Br) for silica/glass [88]. |
| ATRP Catalyst System | Controls the radical polymerization process. | Copper(I) Halide (CuCl, CuBr) + Ligand (Me4cyclam, Me6TREN) [88] [90]. |
| Polydopamine (pDA) | Universal bio-adhesive for surface priming. | Provides a versatile platform for initiator attachment on diverse substrates (metals, polymers, ceramics) [90] [89]. |
| Zwitterionic Peptides | Sequence-defined, short antifouling layer. | EK-repeat peptides (e.g., EKEKEKEKEKGGC) with a terminal cysteine for conjugation [69]. |
| Functional Silanes | Coupling agents for surface chemistry. | (3-Mercaptopropyl)trimethoxysilane (MPTMS) for introducing thiol groups on silicon/glass surfaces [69]. |
The consistent validation of zwitterionic coatings across SPR-relevant optical fibres, electrochemical, QCM, and PSi biosensors underscores their role as a universal antifouling strategy. The protocols detailed herein provide a reproducible framework for modifying diverse sensor platforms. By effectively mitigating biofouling, these coatings enhance signal-to-noise ratios, improve detection limits, and extend sensor operational lifetime in complex media, thereby accelerating the translation of biosensors from laboratory settings to real-world clinical and environmental applications.
Conclusion
Zwitterionic polymer brushes represent a paradigm shift in the development of antifouling biosensor surfaces, offering a potent combination of superior hydrophilicity, robust stability, and exceptional biocompatibility. Their ability to form a tight hydration layer via ionic solvation provides a more effective and durable barrier against biofouling than traditional PEG coatings. The successful application of these brushes in complex media—from undiluted blood plasma to food matrices—along with their immunity to anti-PEG antibodies, positions them as the leading material for the next generation of clinical and point-of-care diagnostics. Future research should focus on the discovery of novel zwitterionic monomers, the refinement of large-scale and substrate-agnostic fabrication techniques, and the execution of comprehensive in vivo studies to fully translate this promising technology from the laboratory to the clinic, ultimately enabling real-time, accurate health monitoring and personalized medicine.
References
- 1. Recent advances in zwitterionic polymers-based non- ... [https://www.sciencedirect.com/science/article/abs/pii/S2468519424003380]
- 2. Direct Measurements of Overlooked Long-Range ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC12004931/]
- 3. Hydrophilic Zwitterionic Coatings: Advanced Biomedical ... [https://hydromer.com/hydrophilic-zwitterionic-coatings-biomedical/]
- 4. Recent Advances in High-Strength Zwitterionic Polymer ... [https://link.springer.com/article/10.1007/s12209-025-00440-6]
- 5. Salt-responsive zwitterionic polymer brushes with anti- ... [https://www.sciencedirect.com/science/article/abs/pii/S2211339817300540]
- 6. Baseline Stability of Thermally Hydrosilated Porous Silicon ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC12311861/]
- 7. Structures, properties, and applications of zwitterionic ... [https://www.sciencedirect.com/science/article/pii/S2772571522000237]
- 8. Atomistic Insights into the Ionic Response and Mechanism ... [https://ui.adsabs.harvard.edu/abs/2025Small..2106233S/abstract]
- 9. Zwitterionic polymer electroplating facilitates the ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC8881349/]
- 10. Insights from the Dual-mechanism fouling model and ... [https://www.sciencedirect.com/science/article/abs/pii/S0013935125025125]
- 11. High-performance antifouling electrochemical aptsensor ... [https://www.sciencedirect.com/science/article/abs/pii/S0308814625038075]
- 12. Biomimetic polymers with phosphorylcholine groups as ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC11704457/]
- 13. Polycarboxybetaine in advanced drug delivery systems [https://www.sciencedirect.com/science/article/pii/S2590156725000143]
- 14. The impact of nanostructuring on the hemocompatibility ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC12150285/]
- 15. Structure-function study of poly(sulfobetaine 3,4- ... [https://www.sciencedirect.com/science/article/abs/pii/S1742706118303386]
- 16. Poly(carboxybetaine) lipids enhance mRNA therapeutics ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC12354250/]
- 17. (78d) Zwitterionic Poly(carboxybetaine) Containing Lipid ... [https://proceedings.aiche.org/conferences/aiche-annual-meeting/2024/proceeding/paper/78d-zwitterionic-polycarboxybetaine-containing-lipid-nanoparticles-enable-efficient-mrna]
- 18. Romantic Surfaces: A Systematic Overview of Stable ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC6365910/]
- 19. Zwitterionic polymer brush functionalized graphene oxide ... [https://www.sciencedirect.com/science/article/abs/pii/S0376738823006889]
- 20. The interplay between grafting density and protein ... [https://www.sciencedirect.com/science/article/pii/S3050562324000059]
- 21. Tuning the Density of Zwitterionic Polymer Brushes on PET ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC7023513/]
- 22. Molecular simulations and understanding of antifouling ... [https://pubs.rsc.org/en/content/articlelanding/2020/tb/d0tb00520g]
- 23. Fouling Resistance and Release Properties of Poly ... [https://pubmed.ncbi.nlm.nih.gov/36533943/]
- 24. Polymer Grafting and its chemical reactions - PMC [https://pmc.ncbi.nlm.nih.gov/articles/PMC9874337/]
- 25. Grafting: a versatile means to modify polymers: Techniques ... [https://www.sciencedirect.com/science/article/abs/pii/S007967000400067X]
- 26. Quantitative insights into electrostatics and structure of ... [https://www.sciencedirect.com/science/article/abs/pii/S1359029422000292]
- 27. Machine-Learning-Aided Understanding of Protein ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC11103666/]
- 28. Surface-Initiated Atom Transfer Radical Polymerization for ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC6766320/]
- 29. Comparative Study of Polymer-Grafted BaTiO3 Nanoparticles ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC10254319/]
- 30. Surface-initiated atom transfer radical polymerization for ... [https://www.sciencedirect.com/science/article/pii/S2949944524000169]
- 31. A comparative study on grafting polymers from cellulose ... [https://www.sciencedirect.com/science/article/abs/pii/S0144861718312396]
- 32. Photoinitiated ATRP - Matyjaszewski Polymer Group [https://www.cmu.edu/maty/atrp-how/procedures-for-initiation-of-ATRP/photoinitiated-atrp.html]
- 33. Engineering the surface properties of a zwitterionic polymer ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC6825806/]
- 34. A comparative study of different protein immobilization ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC4254293/]
- 35. A comparative study of different protein immobilization ... [https://www.sciencedirect.com/science/article/abs/pii/S095656631400668X]
- 36. Comparison of protein immobilization methods with ... [https://link.springer.com/article/10.1007/s00216-024-05575-4]
- 37. Orthogonally Functionalizable Redox-Responsive Polymer ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC12272546/]
- 38. Zwitterionic polymer grafted surfaces with significant ... [https://www.sciencedirect.com/science/article/abs/pii/S0001868620300464]
- 39. Engineering principles of zwitterionic hydrogels [https://pmc.ncbi.nlm.nih.gov/articles/PMC12284511/]
- 40. Polymer brush: a promising grafting approach to scaffolds ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC5346310/]
- 41. Toward a Closed-Loop System for Methotrexate Treatment [https://pubmed.ncbi.nlm.nih.gov/37856156/]
- 42. Engineering microneedles for biosensing and drug delivery [https://www.sciencedirect.com/science/article/pii/S2452199X2500221X]
- 43. A self-powered hydration-monitoring and drug-delivery ... [https://www.nature.com/articles/s41378-025-01000-7]
- 44. Recent advances in electric-stimulus assisted microneedle ... [https://pubmed.ncbi.nlm.nih.gov/41272674/]
- 45. Electrochemical biosensors for vancomycin monitoring in blood [https://pubs.rsc.org/en/content/articlehtml/2025/ra/d5ra07262j]
- 46. Electrochemical biosensors for vancomycin monitoring in ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC12570155/]
- 47. Electrochemical aptasensor for the selective detection of ... [https://link.springer.com/article/10.1007/s00604-025-06952-1]
- 48. Recent Advances in Antifouling Materials for Surface ... [https://www.mdpi.com/2073-4360/13/12/1929]
- 49. Systematic Comparison of Zwitterionic and Non- ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC6366122/]
- 50. Zwitterionic Poly(amino acid methacrylate) Brushes - PMC [https://pmc.ncbi.nlm.nih.gov/articles/PMC4195379/]
- 51. The Effective Charge of Low-Fouling Polybetaine Brushes [https://pmc.ncbi.nlm.nih.gov/articles/PMC12199473/]
- 52. Enhanced antifouling durability of zwitterionic polymer ... [https://www.sciencedirect.com/science/article/abs/pii/S138358662500560X]
- 53. A Stable Zwitterionic Polymer Brush Grafted Ceramic ... [https://papers.ssrn.com/sol3/papers.cfm?abstract_id=5023464]
- 54. Effects of chain stiffness and salt concentration on ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC3368832/]
- 55. Zwitterionic Polymer Brushes For Anti-Fouling [https://www.waterandwastewater.com/zwitterionic-polymer-brushes-for-anti-fouling/]
- 56. Design and Application of Antifouling Bio-Coatings [https://www.mdpi.com/2073-4360/17/6/793]
- 57. Adjustable structures and anti-fouling covalent organic ... [https://www.sciencedirect.com/science/article/abs/pii/S138358662404228X]
- 58. A scalable and chemical-free strategy for antifouling ... [https://link.springer.com/article/10.1007/s10853-025-11677-w]
- 59. Fabrication of robust zwitterionic polymer brushes based ... [https://ui.adsabs.harvard.edu/abs/2025ChEnJ.52166551S/abstract]
- 60. Tailoring antifouling performance of zwitterionic nanogels ... [https://www.sciencedirect.com/science/article/pii/S2468023025013367]
- 61. An anti-fouling aptasensor of biomimetic biointerface for ... [https://www.sciencedirect.com/science/article/abs/pii/S0925400525012031]
- 62. Wetting Behavior and Tribological Properties of Polymer ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC6630773/]
- 63. Critical Design Considerations for Longer-Term Wear and ... [https://www.mdpi.com/2306-5354/11/11/1058]
- 64. Space-resolved quantitative mechanical measurements of ... [https://www.nature.com/articles/am2016170]
- 65. Protocol to fabricate wearable stretchable microneedle ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC10709397/]
- 66. Fabrication of robust zwitterionic polymer brushes based ... [https://www.sciencedirect.com/science/article/abs/pii/S1385894725073899]
- 67. Micrometer-thick and porous nanocomposite coating for ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC10853525/]
- 68. AC interference impact on EIS assessment of organic ... [https://www.sciencedirect.com/science/article/abs/pii/S0300944022000649]
- 69. Porous silicon biosensors meet zwitterionic peptides [https://pubs.rsc.org/en/content/articlehtml/2025/nh/d5nh00478k]
- 70. Antifouling Modification of Gold Surfaces for Acoustic Wave ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC12191305/]
- 71. An electrochemical biosensor with enhanced antifouling ... [https://www.sciencedirect.com/science/article/abs/pii/S0925400524010517]
- 72. Zwitterionic polymer brushes for biomedical applications [https://research.wur.nl/en/publications/romantic-surfaces-zwitterionic-polymer-brushes-for-biomedical-app]
- 73. Fouling Resistant PEG Based Grafted Polymer Coatings [https://www.sigmaaldrich.com/US/en/technical-documents/technical-article/materials-science-and-engineering/nanoparticle-and-microparticle-synthesis/fouling-resistant?srsltid=AfmBOorpB2VCqlJh0vGkyHy64Dtg1dk8uaWzDCUUHOOF3OTIRk4TXsfp]
- 74. Surface charge control for zwitterionic polymer brushes [https://www.semanticscholar.org/paper/Surface-charge-control-for-zwitterionic-polymer-to-Guo-Ja%C5%84czewski/4185948673fb2d4f2244816da81b7a46077eea0f]
- 75. Monodisperse Polyethylene Glycol Derivativesme 2025- ... [https://www.archivemarketresearch.com/reports/monodisperse-polyethylene-glycol-derivativesme-79901]
- 76. Low-Fouling Substrates for Plasmonic Sensing of ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC7345924/]
- 77. Elucidating the Mechanisms Underlying the Signal Drift of ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC12038169/]
- 78. Surface characterization and antifouling properties of ... [https://www.sciencedirect.com/science/article/abs/pii/S0925400514015287]
- 79. Quantitatively Elucidating the Trade-Off between ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC11636210/]
- 80. Functionalized Antifouling Polymer Brushes for Biospecific ... [https://research.utwente.nl/en/publications/functionalized-antifouling-polymer-brushes-for-biospecific-surfac]
- 81. Anti-PEG Antibodies and Their Biological Impact on ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC12388889/]
- 82. PEG: Will It Come Back to You? Polyethelyne Glycol ... [https://www.frontiersin.org/journals/bioengineering-and-biotechnology/articles/10.3389/fbioe.2022.879988/full]
- 83. The benefits and risks of PEGylation in nanomedicine [https://www.nature.com/articles/s41565-025-01951-y]
- 84. Next-Generation mRNA-LNPs: Strategies to Reduce Anti ... [https://www.biochempeg.com/article/453.html]
- 85. Protein-resistant surface coatings formed by mixed polymer ... [https://www.sciencedirect.com/science/article/abs/pii/S0300944025005090]
- 86. Exploring the Long-Term Hydrolytic Behavior of ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC6403559/]
- 87. Substantially enhanced stability against degrafting of ... [https://www.sciencedirect.com/science/article/pii/S0014305717300137]
- 88. Optical fibre long-period grating sensors modified with ... [https://pubs.rsc.org/en/content/articlehtml/2025/bm/d4bm01447b]
- 89. Boosted Drug Sensor Stability With Zwitterionic Coating [https://www.azonano.com/news.aspx?newsID=41545]
- 90. Bioinspired Dopamine and N-Oxide-Based Zwitterionic ... [https://www.mdpi.com/2073-4360/16/12/1634]